Abstract
Background
Acute coronary syndrome (ACS), a life-threatening cardiovascular emergency, faces challenges in early diagnosis and risk stratification. MicroRNAs (miRNAs) have emerged as key regulators in cardiovascular diseases.
Objectives
To investigate the diagnostic/predictive significance of miR-1225-5p in ACS and its mechanism of regulating human coronary artery endothelial cells (HCAECs) injury induced by ox-LDL via targeting FAS.
Methods
miR-1225-5p levels in serum were determined by RT-qPCR in 99 ACS patients and controls, with correlation analysis to clinical indicators. HCAECs were used to assess viability, apoptosis, and inflammation/oxidative stress (IL-6, TNF-α, MDA, SOD) via CCK-8, flow cytometry, and ELISA. Target binding between FAS and miR-1225-5p was validated by dual-luciferase reporter and RIP assays, with functional validation experiments.
Results
miR-1225-5p was significantly downregulated in ACS (P < 0.001), with subtype-specific patterns (UA > NSTEMI > STEMI). ROC analysis showed high diagnostic accuracy (AUC = 0.885 for ACS; 0.790-0.964 for subtypes). miR-1225-5p (HR = 5.409, 95% CI = 1.281–22.837) and Gensini score (HR = 3.524, 95% CI = 1.066–11.646) acted as a predictive factor in ACS, and downregulated miR-1225-5p is linked to MACEs in ACS patients. Overexpression of miR-1225-5p alleviated ox-LDL-induced HCAECs injury (enhanced viability, suppressed apoptosis, reduced inflammation/oxidative stress). FAS acted as a specific target of miR-1225-5p and is negatively associated with miR-1225-5p (r = -0.721). Overexpression of FAS reversed miR-1225-5p-mediated protective effects on HCAECs.
Conclusion
miR-1225-5p is a potential diagnostic indicator for ACS and alleviates HCAECs injury by targeting FAS.
Introduction
Acute Coronary Syndrome (ACS) is a life-threatening condition often resulting from acute thrombosis following atherosclerotic plaque rupture. 1 Its sudden onset and the non-specific symptoms in many patients pose significant diagnostic challenges. 2 ACS, encompassing ST-elevation myocardial infarction (STEMI), non-ST-elevation myocardial infarction (NSTEMI), and unstable angina (UA), 3 is a critical manifestation of coronary artery disease and a leading global cause of mortality. 4 While cardiac troponin remains a cornerstone for diagnosis, its delayed release can hinder early intervention. 5 Similarly, the invasive nature of coronary angiography (CAG) limits its routine use. 6 Therefore, identifying novel biomarkers for the early diagnosis and timely intervention of ACS holds critical clinical significance.
Recent research has firmly established that dysregulated expression of microRNAs (miRNAs) plays a critical role in cardiovascular diseases and shows great promise as sensitive and stable circulating biomarkers. 7 For instance, Gaber MA et al demonstrated that miRNA-208 and miRNA-499 could act as novel biomarkers for the early detection of ACS. 8 Research findings reveal that miR-1225-5p shows significantly decreased in the plasma of older UA patients compared to stable angina (SA) patients, suggesting its potential as a diagnostic biomarker for UA. 9 Further strengthening its candidacy, a prospective study identified miR-1225-5p as a serological marker predictive of future ischemic stroke, showing a significant fold-change in baseline samples from affected individuals. 10 Thus, miR-1225-5p emerges from these studies as a promising biomarker broadly relevant to acute ischemic syndromes, including ACS. This study supports the notion that miR-1225-5p could function as a non-invasive biomarker for ACS. Furthermore, in a study investigating molecular players correlated with immune cell infiltration into atherosclerosis plaques, miR-1225-5p also exhibited dysregulated expression and played a critical regulatory role. 11 Therefore, we hypothesize that altered expression of miR-1225-5p may play a role in the pathogenesis of ACS and potentially influence the progression of atherosclerosis and cardiovascular diseases. However, the primary functions and the molecular mechanisms underlying miR-1225-5p in ACS remain poorly understood.
The FAS receptor (CD95/APO-1), which is encoded by the FAS gene, is a member of the tumor necrosis factor receptor superfamily (TNFRSF). Its primary role is to initiate apoptosis, a form of programed cell death essential for eliminating dysfunctional or abnormal cells. 12 Bossowska A et al observed upregulated FAS expression in lymphocytes from patients with acute myocardial infarction (AMI) and UA. 13 Additionally, Li Q et al found significantly elevated FAS expression in Tregs cells from AMI and UA patients, which may disrupt the balance between Th17 and Tregs cells, potentially contributing to the onset of ACS. 14 Therefore, we speculate that FAS plays a role in ACS, but the precise mechanism remains unclear.
The present study investigated the dysregulated expression and clinical significance of miR-1225-5p in ACS patients across diverse subtypes, and elucidated the modulatory mechanism of miR-1225-5p in human coronary artery endothelial cells (HCAECs) injury induced by ox-LDL.
Materials and Methods
Research Object
A total of 99 patients undergoing ACS with percutaneous coronary intervention (PCI) at Xianyang Hospital of Yan'an University were categorized into UA (n = 30), NSTEMI (n = 40), and STEMI (n = 29) subgroups based on clinical diagnosis. The control group included 61 age- and sex-matched healthy volunteers who underwent routine health examinations at the same hospital during the same enrollment period. This research received approval from the Ethics Committee of Xianyang Hospital of Yan'an University. Inclusion criteria: (1) The patients were diagnosed as ACS by coronary angiography, and all of them were the first onset. (2) The clinical medical records were complete. (3) All participants voluntarily signed the informed consent form. Exclusion criteria: (1) Patients with dysfunctional liver and kidney function. (2) Patients with congenital cardiovascular diseases. (3) Patients with malignant tumors, infectious diseases and immune system diseases. (4) pregnant women.
Sample Collection
10 mL of fasting venous blood specimens were obtained from all enrolled participants. Following centrifugation at 3500 r/min for 10 min, the supernatant was divided into aliquots and stored at −80 °C for later analysis. Biochemical indicators were as follows: low-density lipoprotein (LDL), high-density lipoprotein (HDL), total cholesterol (TC), triglycerides (TG), white blood cell count (WBC), high sensitivity C-reactive protein (hs-CRP), cardiac troponin I (cTnI), and creatine kinase-MB (CK-MB), along with Gensini scores, were quantified using standardized protocols in the clinical laboratory.
Follow-up
All patients with ACS underwent prospective follow-up for 15 months starting from the date of hospital discharge through the 15th month post-discharge. Follow-up was conducted via a combination of scheduled outpatient visits, telephone contacts, and review of medical records including emergency department visits and readmissions. The follow-up endpoint was defined as the termination of the follow-up period or the occurrence of major adverse cardiovascular events (MACEs) among patients. MACEs were defined as all-cause mortality, recurrent angina pectoris, non-fatal myocardial infarction, vascular reconstruction and reinfarction.
Cell Culture
HCAECs were obtained from ATCC (USA) and cultured in DMEM containing 100 U/mL penicillin, 100 μg/mL streptomycin, and 10% fetal bovine serum at 37 °C within a humidified 5% CO₂ incubator. To mimic the pathological conditions of ACS, HCAECs were exposed to 25 μg/mL oxidized low-density lipoprotein (ox-LDL) for a duration of 48 h to induce endothelial dysfunction and inflammatory responses. The control group was untreated with ox-LDL to serve as a baseline for comparison.
Cell Transfection
The overexpression vectors for FAS (oe-FAS), oe-NC, miR-1225-5p mimic, and miR-NC were developed and produced by GenePharma (Shanghai, China). HCAECs were transfected 24 h after plating. Cells were enumerated and adjusted to a concentration of 2 × 105/mL. Upon reaching 70%-80% cell confluency, transfection was performed using Lipofectamine 3000 (Invitrogen, USA) as per the manufacturer's protocol. Briefly, 10 μL Lipofectamine 3000 and 10 μL nucleic acid vectors (oe-FAS/oe-NC/miR-1225-5p mimic/miR-NC) were separately diluted in 115 μL antibiotic-free medium, mixed after 5-min room temperature incubation, and added to 6-well plates. After 20-min complex formation under ambient conditions, the transfection mixture was introduced to cells, followed by incubation at 37 °C in 5% CO₂ for 48 h.
Cell Proliferation Assay
Cell proliferation was assessed using the CCK-8 assay kit (Beyotime, Shanghai) with a plate reader (Thermo Scientific, USA) via continuous monitoring of the optical density at 450 nm. A density of 5 × 103 cells per well was applied to plate cells into 96-well plates. Following transfection, 10 μL of CCK-8 reagent was administered to each well at 24, 48, and 72 h. After 2 h incubation at 37 °C in the dark, OD values at 450 nm were assayed to determine cell viability.
Flow Cytometry Analysis
Apoptosis in HCAECs was assessed with the Annexin V-FITC/PI apoptosis detection kit (BD Biosciences, USA). Following collection, two washes with phosphate-buffered saline (PBS), and resuspension within a binding buffer solution, the cells proceeded to subsequent processing. Following 15-min incubation with Annexin V-FITC and PI under dark conditions at 37 °C, apoptotic rates were quantified by FACSCalibur flow cytometer (BD Biosciences, USA).
Enzyme-Linked Immunosorbent Assay
Collected HCAECs, washed them three times with cold PBS, then resuspended 1 × 106 cells in 150-200 μL PBS to prepare a cell suspension. Lysed the cells by repeated freeze-thaw cycles, centrifuged samples at 2000 rpm for 10 min, then harvested the supernatant. The levels of inflammatory cytokines in HCAECs were quantified using Human TNF-α ELISA Kit and Human IL-6 ELISA Kit (Sigma-Aldrich, USA). Lipid peroxidation (MDA) levels and superoxide dismutase (SOD) activity were determined using corresponding assay kits (Sigma-Aldrich). Every experiment was conducted in strict adherence to the manufacturer's protocols.
Reverse Transcriptase-Quantitative PCR
Isolation of total RNA was performed using TRIzol reagent (Invitrogen, USA), followed by cDNA generation via the PrimeScript RT reagent Kit (TaKaRa, Japan). Quantitative real-time PCR was performed on a 7300 Real-Time PCR System (Applied Biosystems) following the subsequent cycling protocol: 95 °C for 10 min (pre-denaturation), followed by 40 cycles of 95 °C for 20 s, 60 °C for 15 s, and 72 °C for 20 s. Primer sequences were as follows: miR-1225-5p, forward 5’-GCCGAGGTGGGTACGGCCCA-3’, reverse 5’-CTCAACTGGTGTCGTGGA-3’; FAS, forward 5’-GGAGTACACAGACAAAGCCCA-3’, reverse 5’-CTTGGTATTCTGGGTCCGGG-3’; U6, forward 5’-CTCGCTTCGGCAGCACA-3’, reverse 5’-AACGCTTCACGAATTTGCGT-3’ and GAPDH, forward 5’-CCACTCCTCCACCTTTGAC-3’, reverse 5’-ACCCTGTTGCTGTAGCCA-3’. The relative expression levels were calculated using the 2–ΔΔCT method, with miR-1225-5p normalized to U6 and FAS mRNA normalized to GAPDH.
Dual-Luciferase Reporter Assay
Screening was performed for the target genes of miR-1225-5p using the databases TargetScan7.2 (https://www.targetscan.org/vert_72/), miRDB (https://mirdb.org/), and miRWalk (http://mirwalk.umm.uni-heidelberg.de/), then the core target genes with significant overlap in the prediction results of the three databases were screened. To construct the wild-type (WT) FAS plasmid, the WT-FAS 3’UTR sequence was inserted into the pGL3 luciferase reporter vector (Promega, USA). Site-directed mutagenesis was performed to generate the mutant FAS 3’UTR (MUT-FAS). HCAECs underwent co-transfected with WT-FAS/MUT-FAS plasmids and miR-1225-5p mimic (GenePharma, China) by Lipofectamine 3000 (Invitrogen, USA). Following transfection, cellular luciferase activity was measured via the Dual-Luciferase Reporter Assay System (Promega, USA).
RNA Immunoprecipitation (RIP)
RNA-protein complexes were enriched using anti-Ago2 antibody (Proteintech, USA) and anti-IgG antibody (Millipore, Merck, Germany) via the Magna RIP RNA-binding protein immunoprecipitation kit (Sigma-Aldrich, USA), following the manufacturer's protocol. Co-precipitated RNAs were extracted and quantified by RT-qPCR to analyze mRNA enrichment levels.
Statistical Analysis
An post hoc power analysis was performed using G*Power software. With an effect size (d) of 0.5 and a two-tailed alpha level of 0.05, the achieved power for the comparison between the two groups (99 vs 61 participants) was 0.86, which exceeds the conventional threshold of 0.80, indicating adequate statistical power. Chi-square teat was used to analyze the clinicopathological characteristics of enrolled patients. Data were expressed as mean ± SD, with statistical analyses and visualizations conducted using GraphPad Prism 9.0 and SPSS 26.0. Normality was assessed using the Kolmogorov-Smirnov test. Group comparisons were performed via independent samples t-test or one-way ANOVA, with Tukey's post-hoc test applied for multiple comparisons. ROC curves were plotted to evaluate the diagnostic performance of miR-1225-5p in ACS. Cox proportional hazards models analyzed variables associated with MACEs occurrence. Kaplan-Meier survival analysis was performed to visualize event-free survival rate, and Pearson correlation coefficients quantified pairwise variable relationships. Statistical significance was defined as P < 0.05.
Results
Characteristics of the Participants
Regarding age, gender, BMI, history of hypertension, and diabetes, there were no significant differences among all enrolled participants, so the data of the two groups were comparable. However, compared with the control group, patients with ACS exhibited significantly higher levels of LDL-C (P = 0.019), TC (P = 0.008), WBC (P < 0.001), hs-CRP (P < 0.001), cTnI (P < 0.001), and CK-MB (P < 0.001). Besides, the Gensini score of 38.53 ± 21.43 in ACS patients indicated extensive and severe coronary artery stenosis (Table 1).
General Information of the Enrolled Participants.

Notes: ACS: acute coronary syndrome; BMI: body mass index; LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol; TC: total cholesterol; TG: triglyceride; WBC: white blood cells; hs-CRP: hypersensitive C-reactive protein; cTnI: cardiac troponin I; CK-MB: creatine kinase-MB. Date was presented as mean ± SD, or N (%). P < 0.05 means a significant difference.
Differential Expression of miR-1225-5p in Different Subtypes of ACS and its Clinical Value
Results showed that miR-1225-5p expression was notably decreased in ACS patients compared to the control group (P < 0.001, Figure 1A). Additionally, miR-1225-5p expression was notably higher in UA patients than in NSTEMI patients (P = 0.008) and STEMI patients (P < 0.001), with the NSTEMI group exhibiting a significantly higher level compared to the STEMI group (P = 0.007, Figure 1B). ROC curve analysis demonstrated that miR-1225-5p could effectively identify ACS patients (AUC = 0.885; 81.82% sensitivity, 83.61% specificity), and it could effectively differentiate UA (AUC = 0.790; 63.33% sensitivity, 83.61% specificity), NSTEMI (AUC = 0.898; 80.00% sensitivity, 90.16% specificity), and STEMI (AUC = 0.964; 96.55% sensitivity, 85.25% specificity, Figure 1C). A correlation analysis was performed to investigate the relationship between miR-1225-5p and various clinical indicators in ACS patients. The findings revealed that miR-1225-5p exhibited a significant correlation with WBC (r = -0.545), hs-CRP (r = -0.616), cTnI (r = -0.612), CK-MB (r = -503), and the Gensini score (r = -0.531, Table 2).

Differential expression and its diagnostic significance of miR-1225-5p. A. The expression level of miR-1225-5p was markedly decreased in ACS patients. B. miR-1225-5p expression was notably reduced across all ACS subtypes. C. ROC curve shows that miR-1225-5p has diagnostic value and could distinguish different subtypes of ACS. **P < 0.01, ***P < 0.001.
Correlation Between miR-1225-5p Expression and Clinical Indicators.
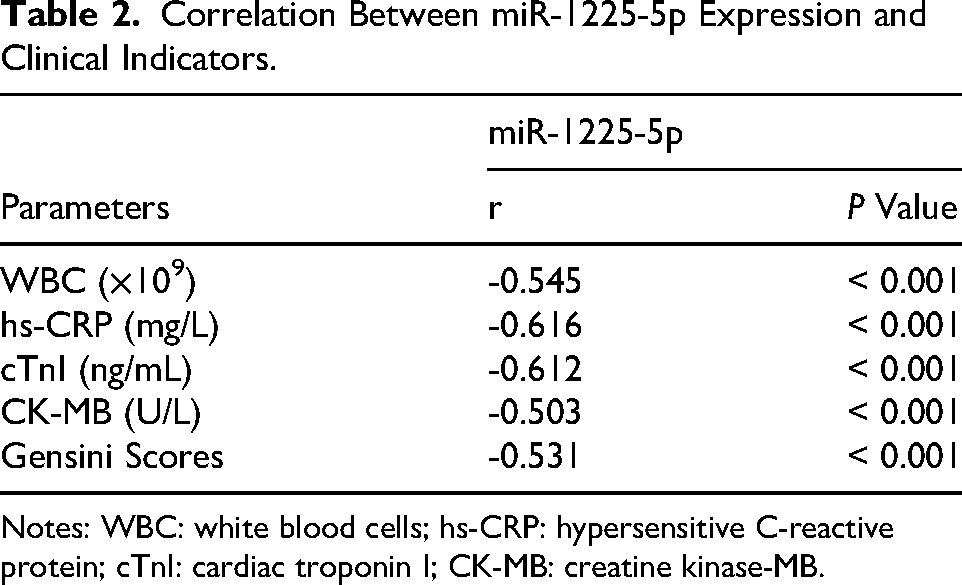
Notes: WBC: white blood cells; hs-CRP: hypersensitive C-reactive protein; cTnI: cardiac troponin I; CK-MB: creatine kinase-MB.
The Predictive Value of miR-1225-5p for MACEs
Cox regression analysis findings indicated that miR-1225-5p expression level (HR = 5.409, 95% CI = 1.281-22.837) and Gensini scores (HR = 3.524, 95% CI = 1.066–11.646) served as independent predictors for MACEs occurrence among ACS patients (Figure 2A). Patients were classified into low-expression and high-expression groups using the median expression level of miR-1225-5p as the cutoff, and Kaplan-Meier survival curves were generated. The findings showed that reduced miR-1225-5p expression correlated with higher odds of MACEs (P = 0.008, Figure 2B).
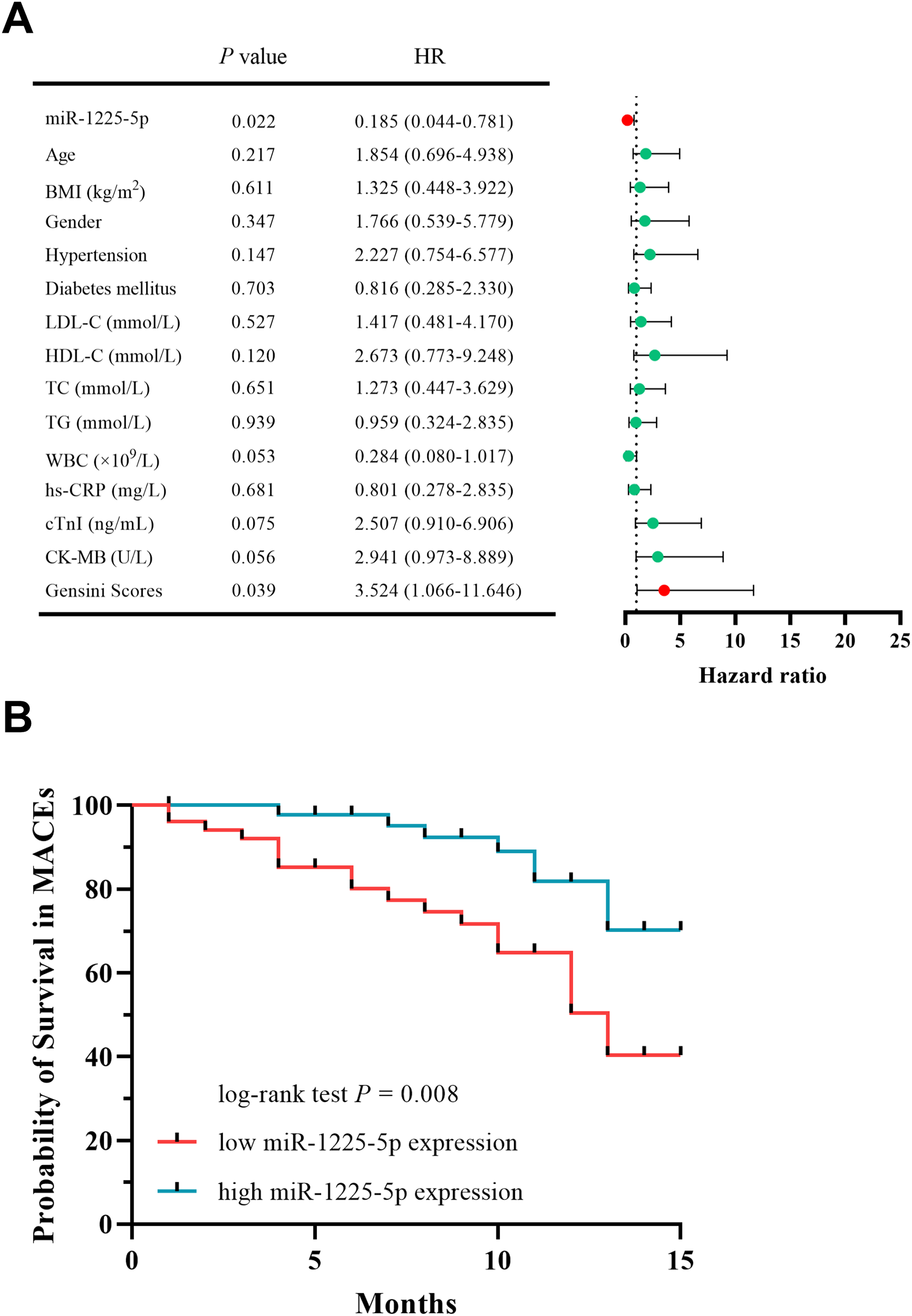
The clinical relevance of miR-1225-5p in forecasting MACEs among ACS patients. A. Cox regression analysis indicated that miR-1225-5p served as an independent predictor of MACEs. B. Kaplan-Meier survival curves indicated that lower levels of miR-1225-5p were associated with MACE in patients with ACS.
Regulatory Role of miR-1225-5p in ox-LDL-Induced Injury of HCAECs
RT-qPCR data showed that miR-1225-5p was significantly suppressed in HCAECs induced by ox-LDL, which was counteracted by its mimic (P < 0.001, Figure 3A). Further studies revealed that ox-LDL induced cellular injury by suppressing viability and promoting apoptosis in HCAECs, while miR-1225-5p overexpression effectively counteracted these effects, restoring cell viability (P < 0.001, Figure 3B) and inhibiting apoptosis (P < 0.001, Figure 3C). Additionally, ELISA assays demonstrated that induction of oxidized ox-LDL led to elevated the levels of inflammatory cytokines (IL-6, TNF-α) and the oxidative stress marker (MDA), while reducing SOD activity in HCAECs. Notably, miR-1225-5p overexpression mitigated these oxidative and inflammatory damages by regulating the aforementioned biomarkers (P < 0.001, Figure 3D-G), thereby exerting protective effects against ox-LDL-induced injury in HCAECs.

The regulatory impact of miR-1225-5p in ox-LDL-induced HCAECs injury A. The expression of miR-1225-5p on ox-LDL induced HCAECs. B-C: miR-1225-5p enhances the viability of HCAECs injured by ox-LDL(B) and inhibits apoptosis (C). D-E: miR-1225-5p suppressed the levels of inflammatory cytokines IL-6 (D) and TNF-α (E). F-G: miR-1225-5p suppressed the levels of MDA (F) and enhanced SOD level (G). *P < 0.05, ***P < 0.001.
Screening of miR-1225-5p Target Genes
Nine candidate target genes were identified through the intersection of Venn diagrams (Figure 4A). RT-qPCR was used to quantify expression levels in ACS patients, revealing that FAS, PEG10, and ANKRD1 were aberrantly expressed in ACS (P < 0.05, Figure 4B). Subsequent validation assays showed that miR-1225-5p notably downregulated FAS expression (P < 0.001, Figure 4C), but no significant effects were detected for PEG10 or ANKRD1 (Figures 4D-E). Therefore, FAS was characterized as a key target gene of miR-1225-5p for further experiments.

The screening of miR-1225-5p target genes. A. 9 target genes of miR-1225-5p were screened by Venn diagram. B. FAS, PEG10, and ANKRD1 were aberrantly expressed in ACS. C-E: miR-1225-5p significantly downregulated FAS expression (C), whereas no significant effect was observed for PEG10 (D) or ANKRD1 (E). *P < 0.05, ***P < 0.001.
Validation of the Targeting Binding Interaction Between FAS and miR-1225-5p
Compared with the control group, FAS expression exhibited a significant upregulation in ACS patients (P < 0.001, Figure 5A). Correlation analysis demonstrated a significant negative correlation between FAS and miR-1225-5p (r = -0.721, P < 0.001, Figure 5B).

Validation of the targeting binding between FAS and miR-1225-5p. A. FAS expression was notably increased in ACS patients. B. FAS and miR-1225-5p demonstrated a significant inverse correlation C. Target interaction sites between FAS and miR-1225-5p. D-E: Dual-luciferase reporter assays (D) and RIP assay (E) validated the targeted binding between FAS and miR-1225-5p. F-G: Upregulation of FAS significantly downregulates miR-1225-5p (F), while miR-1225-5p significantly suppresses FAS expression (G). ***P < 0.001.
Database predictions identified the binding sites between FAS and miR-1225-5p (Figure 5C). Dual-luciferase reporter assays demonstrated that miR-1225-5p mimics notably decreased the luciferase activity of wild-type FAS (P < 0.001, Figure 5D). RIP assays confirmed specific binding of both FAS and miR-1225-5p to the anti-Ago2 antibody, with significant differences compared with the IgG control group (P < 0.001, Figure 5E), further validating their targeting interaction. Ox-LDL induction significantly downregulated miR-1225-5p expression and upregulated FAS expression in HCAECs. Notably, miR-1225-5p overexpression reversed these effects by restoring its own expression level and suppressing FAS expression in the injured HCAECs. Furthermore, FAS overexpression in the damaged cells counteracted this regulatory axis, leading to the recovery of both miR-1225-5p and FAS expression levels (P < 0.001, Figure 5F-G).
Mechanism of miR-1225-5p in ox-LDL Induced HCAECs Injury by Targeting FAS
miR-1225-5p exerted significant protective effects against ox-LDL-induced injury in HCAECs: on one hand, it enhanced HCAECs viability and inhibits cell apoptosis; on the other hand, it alleviated oxidative damage and inflammatory responses by downregulating the concentrations of pro-inflammatory cytokines IL-6 and TNF-α, reducing the oxidative stress marker MDA, and upregulating the activity of the antioxidant enzyme SOD. Notably, overexpressed FAS significantly reversed the miR-1225-5p-mediated recovery of ox-LDL induced HCAECs viability (P < 0.001, Figure 6A) and promoted apoptosis in these cells (P < 0.001, Figure 6B). Additionally, the regulatory effects of miR-1225-5p on inflammatory cytokines and oxidative stress markers were reversed by FAS overexpression: it notably elevated IL-6 (P < 0.001, Figure 6C), TNF-α (P < 0.001, Figure 6D), and MDA (P < 0.001, Figure 6E) levels, while reducing SOD levels (P < 0.001, Figure 6F).

Mechanism of miR-1225-5p in HCAECs injury induced by ox-LDL through targeting FAS.A-B: Overexpressed FAS reverses the restorative effect of miR-1225-5p on the viability of ox-LDL induced HCAECs (A) and promotes apoptosis in these cells (B). C-F: Overexpressed FAS reverses the regulatory effects of miR-1225-5p by significantly upregulating the levels of inflammatory cytokines IL-6 (C) and TNF-α (D) as well as the oxidative stress marker MDA (E), while downregulating SOD (F) activity. ***P < 0.001.
Discussion
ACS is a common and critical condition in cardiology, characterized by rapid onset and a high likelihood of leading to severe consequences. 15 In China, cardiovascular disease ranks first among the leading causes of death for urban and rural residents. 16 The dual burden of its high incidence and high mortality has emerged as a key public health issue constraining social and economic development. 17 Current domestic and international research on diagnostic indicators for ACS primarily focuses on biomarkers such as cTnI and CRP, etc 18 However, these indicators are susceptible to interference from multiple disease states in the body, with complex influencing factors, making it difficult to achieve specific early prediction for ACS. 19 Thus, the early detection and clinical management of ACS is particularly important. Over the past few years, a growing body of research has indicated that miRNAs exhibit substantial potential in the diagnosis and treatment of ACS, and are promising candidates for its early diagnostic biomarkers. 20 Gaber MA et al found that miR-208 and miR-499 may function as biomarkers in the early diagnosis of ACS as early as 3 h before symptom onset. 8 Previous studies have demonstrated that miR-1225-5p exerts regulatory effects across different types of cancer, including hepatocellular carcinoma, 21 non-small cell lung cancer, 22 and colorectal cancer. 23 Additionally, Cui Y et al reported that miR-1225-5p is abnormally expressed in the plasma of patients with UA and SA, 9 which aligns well with the findings of the present study. More importantly, the research findings indicate that miR-1225-5p exhibits significant predictive efficacy for ACS and can effectively differentiate between patients with UA, NSTEMI, and STEMI. This result lays a solid foundation for the clinical translation of miR-1225-5p in ACS diagnosis.
A particularly intriguing finding in this study is that miR-1225-5p expression decreases progressively from patients with UA to NSTEMI and further to STEMI. This expression pattern is unlikely to be random; rather, it may mirror the underlying pathophysiological continuum of ACS. We hypothesize that this gradient is likely tied to several key clinical and molecular mechanisms. First, the declining expression may mirror the ascending severity of myocardial injury, with STEMI representing the most severe form involving complete occlusion and extensive necrosis, and UA/NSTEMI representing lesser degrees of ischemia. 24 The close alignment of the miR-1225-5p expression gradient with this clinical pathological spectrum positions it as a potential biomarker for the injury continuum in ACS. Second, miR-1225-5p expression differences may stem from varying inflammation and oxidative stress levels across ACS subtypes. Compared with UA, culprit plaques in acute MI (especially STEMI) exhibit more intense inflammation and oxidative stress. 25 This high pro-inflammatory factor/ROS microenvironment may stronger suppress miR-1225-5p expression, shaping the gradient from UA to STEMI. Collectively, the expression gradient of miR-1225-5p serves as both a mirror of ACS severity and a biomarker of considerable clinical relevance.
This investigation indicates that miR-1225-5p expression levels are significantly correlated with multiple major clinical indicators in ACS patient cohorts. From a pathological perspective, the core features of ACS involve vascular endothelial injury and systemic inflammatory response. 26 WBC and hs-CRP are commonly used indicators to reflect inflammatory responses 27 ; cTnI and CK-MB are specific biomarkers for myocardial injury 28 ; and the Gensini score, a scoring system for assessing coronary artery stenosis, reflects the severity of coronary lesions. 29 These indicators are closely related to disease severity, and miR-1225-5p is significantly correlated with them, suggesting that miR-1225-5p may serve as an important parameter for assessing ACS severity. Notably, this study further analyzed the correlation between miR-1225-5p expression and MACEs incidence in ACS patients. The results indicate that reduced expression of miR-1225-5p was correlated with an elevated risk of MACEs in patients with ACS, and could serve as an independent predictive biomarker for ACS.
Current research generally holds that coronary arterial endothelial injury serves as the core initiating step in the formation of atherosclerosis. 30 Among these, ox-LDL, which acts as the most critical atherogenic factor, serves as a key mediator of endothelial cell and smooth muscle cell damage. 31 The mechanisms underlying ox-LDL induced endothelial cell injury have long been a major focus of current research. A widely accepted view is that ox-LDL initiates the inflammatory response by inducing endothelial cells to release pro-inflammatory cytokines, including adhesion molecules and chemokines, 32 thereby leading to dysfunction or apoptosis of HCAECs. HCAECs serving as the first line of defense in the wall of coronary arteries, play a critical role in maintaining vascular homeostasis. Their aberrant apoptosis disrupts this endothelial equilibrium, accelerates the progression of atherosclerosis through multiple pathological mechanisms, and directly triggers severe cardiovascular events such as ACS. 33 IL-6 and TNF-α are the most representative pro-inflammatory cytokines in ACS, and their abnormal elevation is closely associated with plaque instability, thrombosis, and systemic inflammatory responses. 34 Additionally, oxidative stress, a key pathological mechanism of ACS, induces oxidative damage to lipids, proteins, and DNA, with MDA and SOD serving as critical markers for assessing this oxidative imbalance. 35 Previously, Wu J et al reported that miR-223-3p and miR-2909 could inhibit the expression of lipopolysaccharide-induced inflammatory cytokines IL-6 and TNF-α in human adipose-derived stem cells. 36 Studies by Zhao L et al have demonstrated that reducing miR-140-5p expression alleviates doxorubicin-induced myocardial oxidative damage in vivo and enhances SOD activity. 37 Previously, in research focusing on discovering candidate genes associated with immune cell infiltration in atherosclerosis, abnormal expression of miR-1225-5p was observed, 11 leading to the speculation that it may be linked to atherosclerosis. However, its specific underlying mechanism remains unclear. This study focused on investigating the regulatory function of miR-1225-5p within HCAECs injury induced by ox-LDL, based on the aforementioned multiple indicators, and revealed that increased expression of miR-1225-5p can alleviate HCAECs injury and inflammatory response.
FAS binds to its ligand FasL to activate downstream apoptotic signaling pathways, inducing programed cell death in target cells. The FAS/FasL pathway has been demonstrated to be implicated in the pathogenesis of multiple cancer types. 38 Zhang BY et al found that miR-98 regulates cardiomyocyte apoptosis by mediating FAS. 39 Previous studies have reported that FAS is upregulated in lymphocytes of ACS patients. 13 This study also observed that FAS was overexpressed in the serum of ACS patients, consistent with previous findings. More critically, this study experimentally verified the targeted binding relationship between FAS and miR-1225-5p. Research indicated that Fas/FasL-mediated vascular endothelial cell apoptosis suppresses angiogenesis in intervertebral discs, highlighting this pathway as a potential key target for endothelial cell regulation. 40 This study further functional experiments showed that miR-1225-5p overexpression alleviated the injury in HCAECs and alleviated damage through its targeting of FAS, and participated in the progression of ACS.
However, this study has several limitations that should be considered. Firstly, the single-center design and relatively limited sample size represent a notable constraint. While this approach ensured stringent and consistent patient phenotyping within our cohort, it inevitably restricts the generalizability of our findings to broader, more diverse populations. Furthermore, the sample size may have been underpowered to detect subtle but potentially important clinical correlations or subgroup effects. Consequently, a primary objective for our future research will be to initiate a large-scale, prospective, multi-center study to robustly validate the clinical significance of the miR-1225-5p/FAS pathway. In this validation, we will incorporate the measurement of early biomarkers such as H-FABP to complement the diagnostic perspective of this study. Secondly, our experimental findings are primarily based on in vitro cell culture systems, which cannot fully recapitulate the complex pathological microenvironment of ACS in vivo. Finally, although we validated the direct targeting of FAS by miR-1225-5p, the complete downstream signaling pathways remain to be fully elucidated. Addressing these mechanistic and translational gaps will be crucial to provide a more robust foundation for the precise diagnosis and targeted therapy of ACS.
Conclusions
In summary, miR-1225-5p is significantly decreased in ACS and its subtypes. miR-1225-5p can be regarded as a diagnostic biomarker of ACS and has predictive value for MACEs. miR-1225-5p attenuates HCAECs injury induced by ox-LDL via targeting FAS.
Footnotes
Ethical Considerations
This research received approval from the Ethics Committee of Xianyang Hospital of Yan'an University.
Consent to Participate
All participants voluntarily signed the informed consent form.
Consent for Publication
Not Applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed during this study are included in this article. Further enquiries can be directed to the corresponding author.
