Abstract
Background:
Patients on dual antiplatelet therapy following percutaneous coronary intervention often have indications for concomitant oral anticoagulation, known as triple antithrombotic therapy. Majority of literature evaluating triple antithrombotic therapy fails to adequately represent patients with ST-elevation myocardial infarction and those prescribed potent P2Y12 inhibitors, ticagrelor or prasugrel. The purpose of this study was to evaluate the safety and efficacy of triple antithrombotic regimens containing ticagrelor or prasugrel versus clopidogrel after percutaneous coronary intervention in the setting of ST-elevation myocardial infarction.
Methods:
This was a single-center, retrospective cohort trial. The primary endpoint was net adverse clinical event, defined as the primary efficacy endpoint of death, myocardial infarction, or cerebrovascular accident and the primary safety endpoint of any bleeding event.
Results:
Between October 2017 and October 2019, a total of 65 patients with ST-elevation myocardial infarction were initiated on triple therapy. Forty-six patients were included in the primary analysis, of which 26 were discharged on triple antithrombotic therapy with clopidogrel and 20 discharged on potent P2Y12 inhibitors (ticagrelor or prasugrel). The primary endpoint occurred in 27% of the clopidogrel group and 40% of the potent P2Y12 inhibitor group (
Conclusions:
This small cohort study suggests, in patients with ST-elevation myocardial infarction undergoing percutaneous coronary intervention, the net adverse clinical event rate does not differ between clopidogrel and potent P2Y12 inhibitors in the setting of triple antithrombotic therapy. The results of this exploratory analysis warrant confirmation in a larger, randomized study.
Introduction
Patients on dual antiplatelet therapy (DAPT) following percutaneous coronary intervention (PCI) with stent implantation often have indications for concomitant oral anticoagulation (OAC), including atrial fibrillation (AF) or venous thromboembolism. 1 Up to 10% of patients undergoing PCI have a clinical indication for triple antithrombotic therapy (TAT). 2 Despite these indications, TAT increases the risk of bleeding indicating the need to define appropriate antithrombotic strategies associated with lower bleeding risks while maintaining efficacy. 3,4
The majority of data evaluating ischemic and bleeding outcomes of TAT strategies to date has evaluated clopidogrel as the P2Y12 inhibitor of choice with minimal representation of more potent P2Y12 inhibitors, ticagrelor and prasugrel. Further, the majority of patients evaluated had non-ST segment elevation myocardial infarction (NSTEMI) or unstable angina as the primary coronary event, or underwent elective PCI. 3,4 The availability of more potent P2Y12 inhibitors with proven efficacy in reducing ischemic outcomes in patients with acute coronary syndromes (ACS) has resulted in increased use of ticagrelor and prasugrel, particularly in the setting of ST-elevation myocardial infarction (STEMI). 5 -7 The TRITON-TIMI 38 trial showed patients with STEMI are at a higher risk of stent thrombosis regardless of stent type and demonstrated that prasugrel reduced that risk by 50%. 5 Despite improved efficacy, ticagrelor and prasugrel are known to increase the risk of major bleeding when compared to clopidogrel in the setting of ACS. The risk of bleeding with potent P2Y12 inhibitors in the setting of TAT is currently unknown. The purpose of this exploratory analysis was to evaluate the safety and efficacy of TAT regimens containing ticagrelor or prasugrel versus clopidogrel after PCI with stent deployment in the setting of STEMI.
Methods
This was a single-center, retrospective, cohort trial approved by the institutional review board at a large academic medical center. Patients were identified via the institution’s electronic medical record utilizing ICD 10 codes for STEMI and discharged on TAT between October 1, 2017, and October 1, 2019. Patients were evaluated for the duration of their admission and followed for 90 days after discharge.
Patients were included if they were
The primary endpoint was net adverse clinical event (NACE), defined as the primary efficacy endpoint of death, MI, or cerebrovascular accident and the primary safety endpoint of any bleeding event. Bleeding events were further classified by the Bleeding Academic Research Consortium (BARC) bleeding criteria Types 2-5, excluding Type 4 (CABG-related bleeding). BARC Type 1 events were also excluded given no actionable treatment is required; major bleeding was further defined by Types 3B-5. 8
Secondary endpoints included the individual components of NACE as described above, prescribing patterns in the setting of triple therapy for STEMI including the duration of aspirin therapy, choice of anticoagulant, and choice of P2Y12 inhibitor, and patient-specific factors influencing the choice of P2Y12 inhibitor including the type, location, and number of stent(s) placed.
Statistical Analysis
Percentages were reported for categorical variables, and chi-square tests were run for comparing rates between groups, clopidogrel and potent P2Y12 inhibitors, defined as ticagrelor and prasugrel. No power calculation was conducted given the small size of this exploratory analysis. For normally distributed continuous variables, means and standard deviations (SD) were reported, and t-tests were used to test for differences in means between groups. For skewed continuous variables, medians and interquartile ranges (IQR) were reported, and the distributions of the variables were compared between the two groups using the Wilcoxon rank-sum test. An alpha level of 0.05 was used to determine statistical significance. All analyses were performed using SAS version 9.4 software (SAS Institute, Cary, NC).
Results
Between October 2017 and October 2019, 665 patients with STEMI were admitted to the institution, of which a total of 65 were initiated on TAT. Of the 65 patients, 19 were excluded: 8 were not discharged on TAT, 6 had an index event misclassified as a STEMI (non-ST segment elevation myocardial infarction or acute decompensated heart failure), 4 patients underwent a CABG, and 1 patient was discharged on a parenteral anticoagulant (enoxaparin). Forty-six patients were included in the primary analysis, of which 26 were discharged on TAT with clopidogrel and 20 were discharged on TAT with a potent P2Y12 inhibitor. Baseline characteristics are summarized in Table 1. The clopidogrel and potent P2Y12 inhibitor groups differed in regard to baseline chronic kidney disease (50% vs. 80% respectively,
Baseline Characteristics.a

a Results are in the form n (%) unless otherwise noted.
b Clopidogrel n = 15 and potent P2Y12 inhibitors n = 10.
The primary endpoint was observed in 27% of patients receiving clopidogrel and 45% of patients receiving potent P2Y12 inhibitors (
Primary Endpoints.a
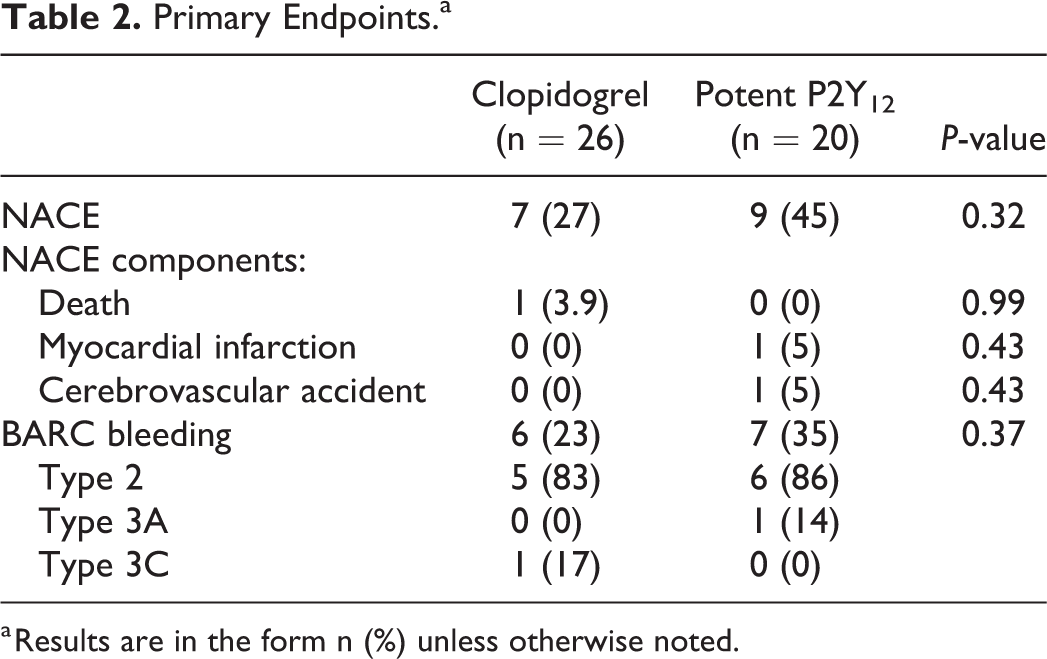
a Results are in the form n (%) unless otherwise noted.
In terms of institutional prescribing patterns, the average duration of aspirin therapy prescribed on discharge was longer in the clopidogrel group versus the potent P2Y12 inhibitor group (4.9 versus 2.3 months, respectively). The type of OAC prescribed in the clopidogrel group included 58% on a direct-acting oral anticoagulant (DOAC) and 42% on warfarin as compared to the potent P2Y12 inhibitor group which included 45% on a DOAC and 55% on warfarin. Stent characteristics are summarized in Table 3. Drug eluting stents were the only type of stent placed in both groups. The highest risk artery stented was the left anterior descending artery in 69% of the clopidogrel group and 65% of the potent P2Y12 inhibitor group. More than one stent was placed in 50% of the clopidogrel group and 45% of the potent P2Y12 inhibitor group. Notably, 14 patients were originally prescribed a potent P2Y12 inhibitor and were subsequently discharged on clopidogrel due to cost (n = 12), perceived reduction in bleed risk (n = 1), and an adverse drug reaction (headache) to ticagrelor (n = 1).
Stent Characteristics.a
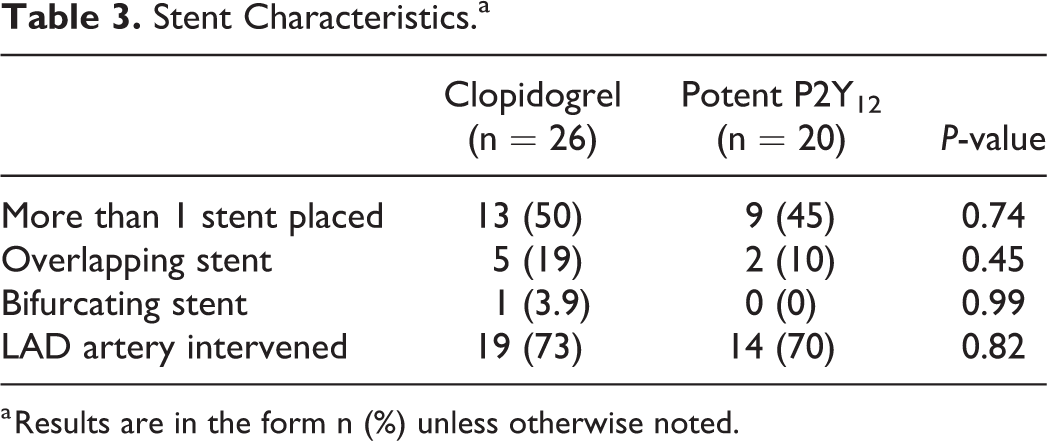
a Results are in the form n (%) unless otherwise noted.
Discussion
This small cohort study assessed the safety and efficacy of TAT utilizing potent P2Y12 inhibitors versus clopidogrel in patients with STEMI status-post PCI requiring stent deployment. The primary endpoint of NACE was similar between the potent P2Y12 inhibitor and clopidogrel groups at 90-days after discharge from the index hospitalization. All individual ischemic outcomes and BARC bleeding events were similar without any significant differences between groups.
Patients requiring TAT are at high risk for cardiovascular mortality and morbidity from both ischemic and bleeding events. The 2021 North American Consensus on the Management of Antithrombotic Therapy in Patients with Atrial Fibrillation Undergoing PCI recommends a short duration of TAT (up to 1 month) based on ischemic and bleeding risk and the use of clopidogrel plus OAC as an alternative to TAT in patients whose bleeding risk outweighs ischemic risk. 9 The 2019 American Heart Association/American College of Cardiology/Heart Rhythm Society AF, the European Society of Cardiology DAPT guidelines, and the 2020 ACC Expert Consensus Pathway for Anticoagulant and Antiplatelet Therapy recommend clopidogrel as the preferred P2Y12 inhibitor when used in TAT. 10 -12
Several trials have evaluated the ischemic benefits and bleeding risk with TAT versus dual therapy consisting of an antiplatelet plus OAC. The WOEST trial was the first to demonstrate fewer bleeding events with clopidogrel and warfarin compared to TAT; however, only clopidogrel was used and the number of patients with STEMI was not reported. 3 Both the PIONEER AF-PCI and RE-DUAL PCI trials demonstrated lower bleed rates in TAT with a DOAC versus TAT with warfarin, however both trials utilized clopidogrel as the main P2Y12 inhibitor and included a minimal number of patients with STEMI. 7,13 Optimal duration of TAT was evaluated in the ISAR-TRIPLE study and found no significant difference between 6 weeks of TAT and 6 months of TAT in a composite of ischemic or bleeding events. The number of patients with STEMI included was minimal at 0.8% and all patients received clopidogrel. 4 The AUGUSTUS trial comparing apixaban versus VKA and aspirin versus placebo resulted in less bleeding events with apixaban plus an antiplatelet without significant differences in ischemic events compared to regimens containing VKA, aspirin or both. 14 Similar to the previous trials, the most common P2Y12 inhibitor used was clopidogrel (92.6%) and the number of patients with STEMI was not reported (only 37.3% of the population presented with an unspecified acute coronary syndrome treated with PCI).
The trials evaluating TAT indicate that the use of dual antithrombotic therapy versus TAT (i.e. reducing the duration/use of aspirin) and the use of DOACs over warfarin reduces bleeding risk. 15,16 The minimal representation of patients with STEMI and use of ticagrelor and prasugrel limits applicability of these findings to this higher risk patient population and more potent antiplatelet options. Recent systematic reviews and meta-analyses comparing the safety and efficacy of ticagrelor and/or prasugrel versus clopidogrel with OAC with or without aspirin in patients with AF undergoing PCI found increased rates of bleeding with ticagrelor and prasugrel and mixed results in rates of MACE compared to clopidogrel. 17,18 We sought to explore the utilization of these potent P2Y12 inhibitors in STEMI, a population at high thrombotic risk.
Approximately half of the patients discharged on TAT in the present study were prescribed a potent P2Y12 inhibitor and the average duration of aspirin use was greater than 1 month in both groups. This finding reflects a more aggressive approach to TAT is being used in patients with STEMI. Patients were more likely to be discharged on a potent P2Y12 inhibitor if at a higher thrombotic risk (evidenced by the CHA2DS2-VASc score) despite also having a higher bleed risk (evidenced by the HAS-BLED score). Patients with a history of chronic kidney disease, myocardial infarction, and/or venous thromboembolism were also more likely to be discharged on a potent P2Y12 inhibitor indicating prescriber preference for a potent P2Y12 inhibitor in patients at higher thrombotic risk. Interestingly, stent characteristics including the number of stents placed, overlapping or bifurcating stents, and type of artery intervened did not appear to play a role in antiplatelet agent selection upon discharge. The rate of NACE was numerically higher in the potent P2Y12 inhibitor group. This hypothesis generating observation may be a result of the higher baseline thrombotic and bleeding risks in this group and warrants further investigation.
There are several limitations to our trial. The retrospective nature of this study could have led to bias or misclassification and unattainable key measures. Given the small sample size and lack of power analysis, the results of this study can only be considered exploratory as they may be impacted by type I error. Another limitation was some of the prescribing practices observed in this setting. Despite previous literature favoring DOACs in the setting of TAT, warfarin was prescribed in about 50% of patients in each group. Further, the duration of aspirin therapy was longer than the current guideline recommendations in this setting. Lastly, the indication for anticoagulation was not uniform and included indications where limited ischemic benefit was to be gained, such as left ventricular thrombus prophylaxis. 19 -22
In conclusion, this study demonstrated the use of clopidogrel versus a more potent P2Y12 inhibitor did not affect bleeding nor ischemic outcomes in patients on TAT status-post STEMI with PCI. The numerically higher event rate seen in the potent P2Y12 inhibitor group could be a result of the higher baseline thrombotic and bleed risks in this group, though this should be investigated further. Future prospective, randomized trials with a larger population are needed to confirm the findings of this exploratory analysis.
Footnotes
Author Contributions
KG, NS, JF, and KF contributed to the design and implementation of the research, to the analysis of the results, and to the writing of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
