Abstract
Background
Current guidelines recommend that anticoagulation is reasonable and less robust to prevent stroke among AF patients with an intermediate risk of stroke, suggesting the need for more evidence in this population. Therefore, our current systematic review and meta-analysis aimed to evaluate the effectiveness and safety outcomes of oral anticoagulation (OAC) therapy in AF patients with a single additional stroke risk factor (CHA2DS2-VASc score = 1 in males and 2 in females).
Methods
We performed the literature searches to identify relevant articles by systematically researching the PubMed and Embase databases. In the pooled analysis by the random-effects model, the comparative treatment indicators to present outcomes of OAC therapy compared with no OAC were adjusted hazard ratios (HRs) and 95% confidence intervals (CIs).
Results
A total of 6 studies were finally included. In the pooled analysis by the random-effects model, compared with no OAC therapy, OAC use was associated with increased risks of major bleeding (HR = 1.29, 95%CI: 1.06-1.57) and gastrointestinal bleeding (HR = 1.38, 95%CI: 1.04-1.82). There were no differences in stroke or systemic embolism (SSE) (HR = 0.84, 95%CI: 0.66-1.08), ischemic stroke (HR = 0.79, 95%CI: 0.27-2.33), and intracranial bleeding (HR = 1.17, 95%CI: 0.78-1.76) between the two studied groups
Conclusion
In patients with AF patients at an intermediate risk of stroke, OAC use was not associated with reductions in thromboembolic events, but might increase the bleeding risks compared with no treatment.
Introduction
Atrial fibrillation (AF) is a well-documented and independent risk factor for stroke or systemic embolism (SSE), 1 associated with increased rates of cardiovascular diseases, mortality and disability.2,3 Preventing thromboembolic events by anticoagulation therapy remains an urgent clinical issue in AF management.4,5 The use of vitamin-K antagonists (VKAs) such as warfarin is considered the conventional anticoagulation method for preventing thromboembolic events. In recent years, non-vitamin K oral anticoagulants (NOACs) have been demonstrated to be noninferior to VKAs due to their advantages of convenient administration, flexible dose adjustment, and superior net clinical benefit. As such, NOACs have been recommended as the first-line anticoagulants for AF-related stroke prevention therapy.6,7
Clinicians prescribe anticoagulation therapy for stroke prevention in AF patients based on the stroke risk stratification schemes. AF guidelines recommend the CHA2DS2-VASc score 8 (1 point each for congestive heart failure [C], hypertension [H], diabetes mellitus [D], vascular diseases [V], age of 65-74 years old, female sex [Sc]; 2 points each for age of ≥ 75 years old [A2] and history of stroke/transient ischemic attack/thromboembolism [S2]) as the criteria for stroke risk assessment (characterized as low [0 for males, 1 for females], moderate [1 for males, 2 for females], and high [≥ 2 for males, ≥ 3 for females] risk).6,7,9 Oral anticoagulation (OAC) has consistently been recommended for AF patients in the high-risk strata. However, whether patients with AF at moderate stroke risk (ie, CHA2DS2-VASc score of 1 in males or 2 in females) can benefit from OAC remains uncertain.
Several recent studies have explored the effectiveness and safety outcomes of OAC (NOACs or VKAs) in AF patients with a single non-sex-related risk factor.10–19 In a national cohort by Lip et al, 12 39,400 AF patients from the Danish Civil Registration System were defined as having low and intermediate stroke risks, suggesting that in patients with CHA2DS2-VASc score of 1, OAC offered a higher risk of major bleeding compared with no OAC intervention, whereas no difference in SSE was found between the two groups. A secondary analysis of the AVERROES study 11 found that apixaban showed a lower risk of ischemic stroke but no difference in bleeding risk compared with controls for the low- and intermediate- risk patients with AF. Herein, our current systematic review and meta-analysis aimed to evaluate the effectiveness and safety outcomes of OAC in AF patients at an intermediate risk of stroke.
Methods
Our current meta-analysis was performed according to the guidance from the Cochrane Handbook for Systematic Reviews, and we presented the findings based on the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-analyses) items.
Data Sources and Literature Searches
We performed the literature searches to identify relevant articles via systematically researching the PubMed and Embase databases. These studies explored the effectiveness and safety outcomes of OAC therapy in AF patients with a single non-sex stroke risk factor (CHA2DS2-VASc score of 1 for males and 2 for females) compared with no OAC therapy. We used the following search terms: (1) “atrial fibrillation” OR “atrial flutter”, (2) “oral anticoagulants” OR “vitamin K antagonists” OR “warfarin” OR “non-vitamin K antagonist oral anticoagulants” OR “direct oral anticoagulants” OR “novel oral anticoagulants” OR “dabigatran” OR “rivaroxaban” OR “apixaban” OR “edoxaban”, (3) “low risk” OR “non-sex” OR “single” OR “unique” OR “one additional”, and (4) “CHA2DS2-VASc”. These four categories of search terms were combined using the Boolean operator “and”. In addition, the reference lists of the retrieved articles and prior reviews20,21 were searched manually for additional eligible studies.
Inclusion and Exclusion Criteria
We included studies based on the following items: (1) observational cohort studies, post hoc analyses of randomized clinical trials (RCTs), and original RCTs reporting at least one of the effectiveness and/or safety outcomes between OAC versus no OAC therapy in non-valvular AF patients with a moderate stroke risk (ie, equivalent to CHA2DS2-VASc of 1 for males or 2 for females); (2) the OAC therapy included VKAs and NOACs (apixaban, rivaroxaban, edoxaban, and dabigatran), whereas no OAC therapy included antiplatelet agents or no treatment; and (3) the effectiveness outcomes were SSE, ischemic stroke, and all-cause death, whereas the safety outcomes included major bleeding, intracranial bleeding, and gastrointestinal bleeding. The definitions of these outcomes were applied according to the originally included studies.
We excluded studies according to the following criteria: (1) study types were animal experiments, cross-sectional studies, case reports, reviews, meta-analyses, comments, and conference abstracts; (2) the stroke risk was only assessed by the CHADS2 score that has been rarely used at present; (3) although several studies provided subgroup analyses stratify by stroke risk such as CHA2DS2-VASc score of 1 or 2, we excluded those that did not stratified by sex; (4) no effectiveness or safety outcomes could not be extracted; and (5) when facing overlapping articles from the same data sources, we included the study with the longest follow-up or highest sample size.
Study Selection and Data Extraction
Two reviewers independently screened the titles and abstracts of the retrieved studies from the electronic databases. Subsequently, we selected eligible studies after full-text screening based on pre-determined inclusion criteria. Disagreements were resolved by discussion between two reviewers or consultation with the corresponding author. The following data were independently extracted, mainly including authors and publication years, study interventions and controls, study design, baseline characteristics of included participants, inclusion and exclusion criteria, effectiveness and safety outcomes, and follow-up time.
Quality Assessment
We used the Newcastle-Ottawa Scale (NOS) tool to assess the quality of included studies. Three domains with a total of 9 items were included in this tool: the selection of cohorts (0-4 points), the comparability of cohorts (0-2 points), and the assessment of the outcome (0-3 points). We defined studies with a NOS of ≥6 points as moderate- to high-quality, whereas studies with a NOS of <6 points were regarded as low-quality.22–24
Statistical Analysis
All the analyses were performed using the Review Manager version 5.4 software (the Cochrane Collaboration 2014, Nordic Cochrane Centre Copenhagen, Denmark). The statistical heterogeneity across the included studies was usually dealt with by the Cochrane Q test and I² index. A P value of <0.1 in the Cochrane Q test or an I² value of >50% indicated significant heterogeneity between studies.
In the pooled analysis, the comparative treatment indicators to present outcomes of OAC therapy compared with no OAC were adjusted hazard ratios (HRs) and 95% confidence intervals (CIs). In addition, we also compared the effect of NOACs versus VKAs. The natural logarithm of the HR and its corresponding standard error was calculated. Then, the natural logarithms were pooled by a DerSimonian and Laird random-effects model with an inverse variance method. The subgroup analysis was not performed due to the limited data. Publication bias was evaluated by employing the funnel plots for visual inspection of asymmetry. However, we did not perform the publication bias analysis in this study because of the limited number of included studies for the studied outcomes. A value of P < .05 was considered statistically significant.
Results
Study Selection
The flow diagram of the researching strategies of this meta-analysis is presented in Figure 1. Overall, a total of 814 studies from the electronic databases were identified. During the title and abstract screenings, 785 studies were excluded that did not meet the pre-defined inclusion criteria. Subsequently, 29 remaining studies were assessed during the full-text screenings, and 23 studies were excluded because (1) studies included AF patients with CHA2DS2-VASc score of 1 or 2 but not stratified by sex (n = 8),10,11,25–31 (2) studies only included AF patients with a low stroke risk (ie, CHA2DS2-VASc score of 0, n = 3),32–34 or non-gender CHA2DS2-VASc score of 0-1 (n = 4), 35 (3) studies had the overlapping data (n = 4),12,13,36,37 (4) studies did not provide the subgroups based on the anticoagulation treatments (n = 2)38,39 or adjusted data (n = 1); 40 and (5) the stroke risk was only assessed by the CHADS2 score (n = 1). 41 Finally, a total of 6 observational cohort studies14–19 were included in this meta-analysis.
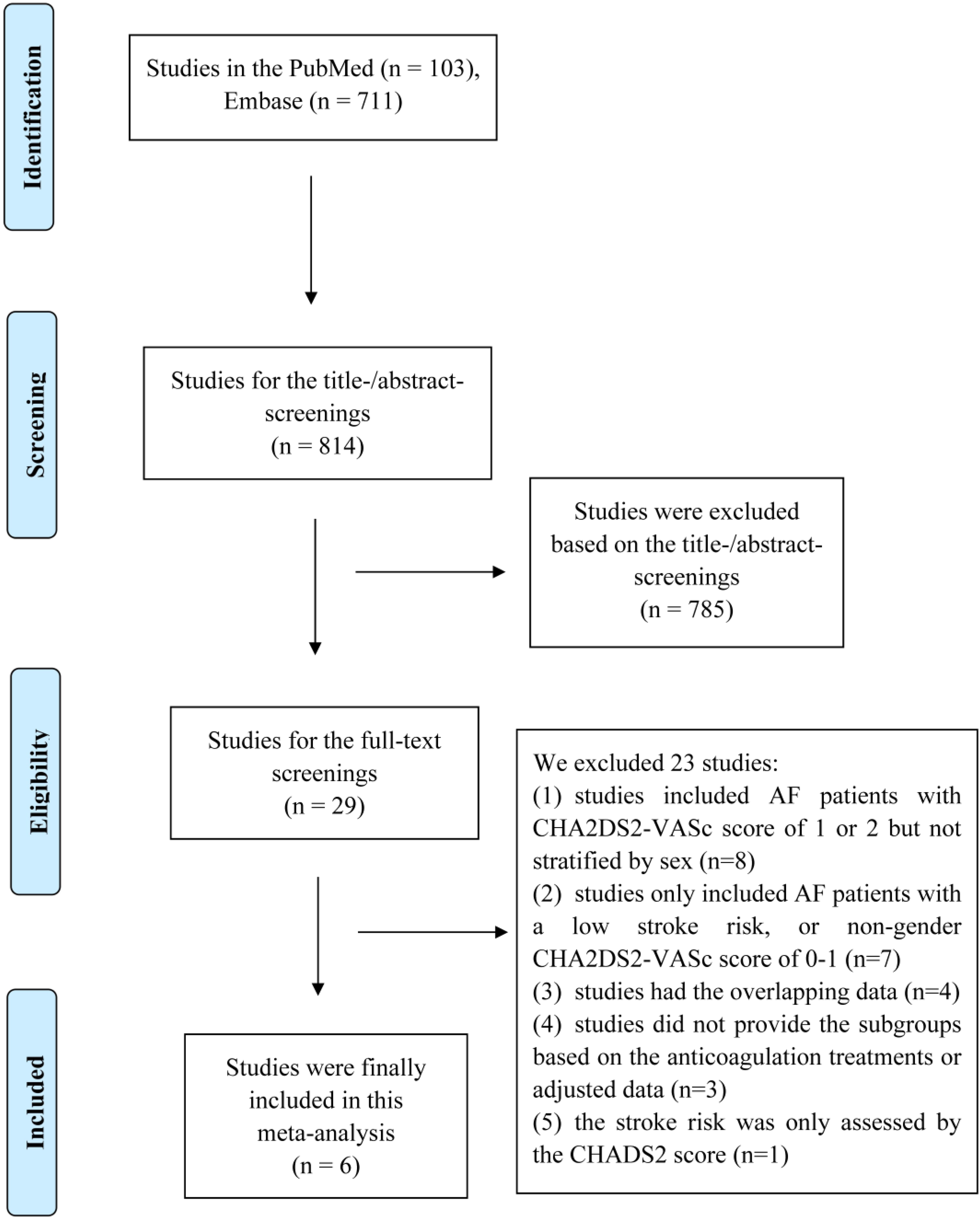
Diagram of Study Selection in this Systematic Review and Meta-Analysis.
Table 1 shows the baseline characteristics of included studies. Among these included studies, the stroke risk was assessed by the CHA2DS2-VASc score, and non-gender CHA2DS2-VASc score of 1 (ie, CHA2DS2-VASc of 1 for males or 2 for females) suggested that AF patients had an intermediate risk of stroke. We noted that Komen et al enrolled patients from four Western European databases including Sweden, Denmark, Norway, and Scotland, Lip et al used the Danish nationwide databases, and Anjum et al applied the population database from Norway. Lip et al and Anjum et al might have certain overlapping data compared with the included study of Komen et al After discussion, for the studies by Lip et al and Anjum et al, we only included the effectiveness and safety outcomes that were not provided in the study of Komen et al The NOS tool was used to assess the quality of included studies, suggesting moderate-to-high quality.
Baseline Characteristics of Involved Studies in This Systematic Review and meta-Analysis.
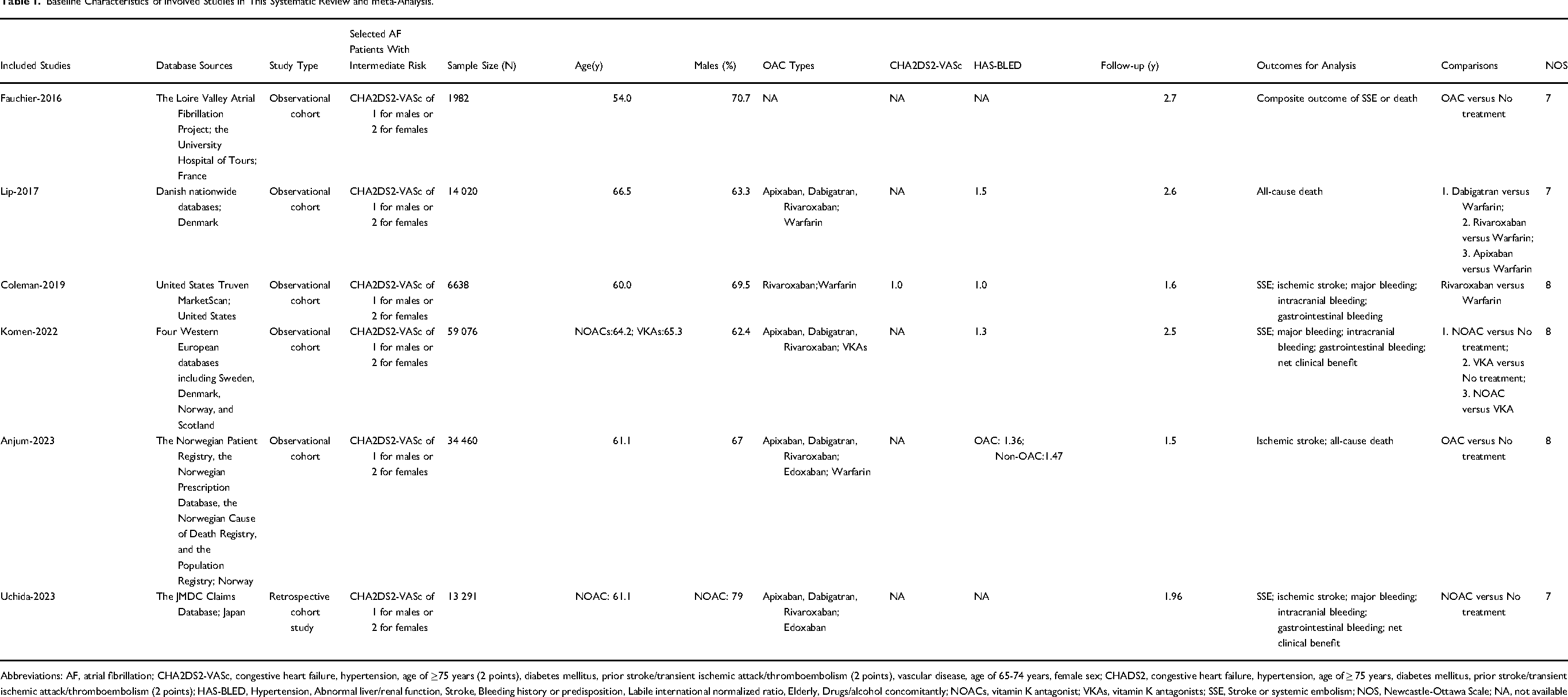
Abbreviations: AF, atrial fibrillation; CHA2DS2-VASc, congestive heart failure, hypertension, age of ≥75 years (2 points), diabetes mellitus, prior stroke/transient ischemic attack/thromboembolism (2 points), vascular disease, age of 65-74 years, female sex; CHADS2, congestive heart failure, hypertension, age of ≥ 75 years, diabetes mellitus, prior stroke/transient ischemic attack/thromboembolism (2 points); HAS-BLED, Hypertension, Abnormal liver/renal function, Stroke, Bleeding history or predisposition, Labile international normalized ratio, Elderly, Drugs/alcohol concomitantly; NOACs, vitamin K antagonist; VKAs, vitamin K antagonists; SSE, Stroke or systemic embolism; NOS, Newcastle-Ottawa Scale; NA, not available
Effectiveness and Safety Outcomes Between OAC and no OAC
A total of 4 studies assessed the effectiveness and safety outcomes between OAC and no OAC in AF patients who had an intermediate risk of stroke. In the study performed by Fauchier et al, the primary adverse outcome was a composite of SSE or death. They enrolled AF patients from the Loire Valley Atrial Fibrillation Project, and found that OAC was independently associated with a reduced risk of SSE or death (HR = 0.59, 95%CI: 0.40-0.86) in patients with non-gender CHA2DS2-VASc score of 1 after adjustments. In the pooled analysis by the random-effects model, OAC use was associated with increased risks of major bleeding (HR = 1.29, 95%CI: 1.06-1.57) and gastrointestinal bleeding (HR = 1.38, 95%CI: 1.04-1.82). There were no differences in SSE (HR = 0.84, 95%CI: 0.66-1.08), ischemic stroke (HR = 0.79, 95%CI: 0.27-2.33), and intracranial bleeding (HR = 1.17, 95%CI: 0.78-1.76) between the two studied groups (Figure 2).
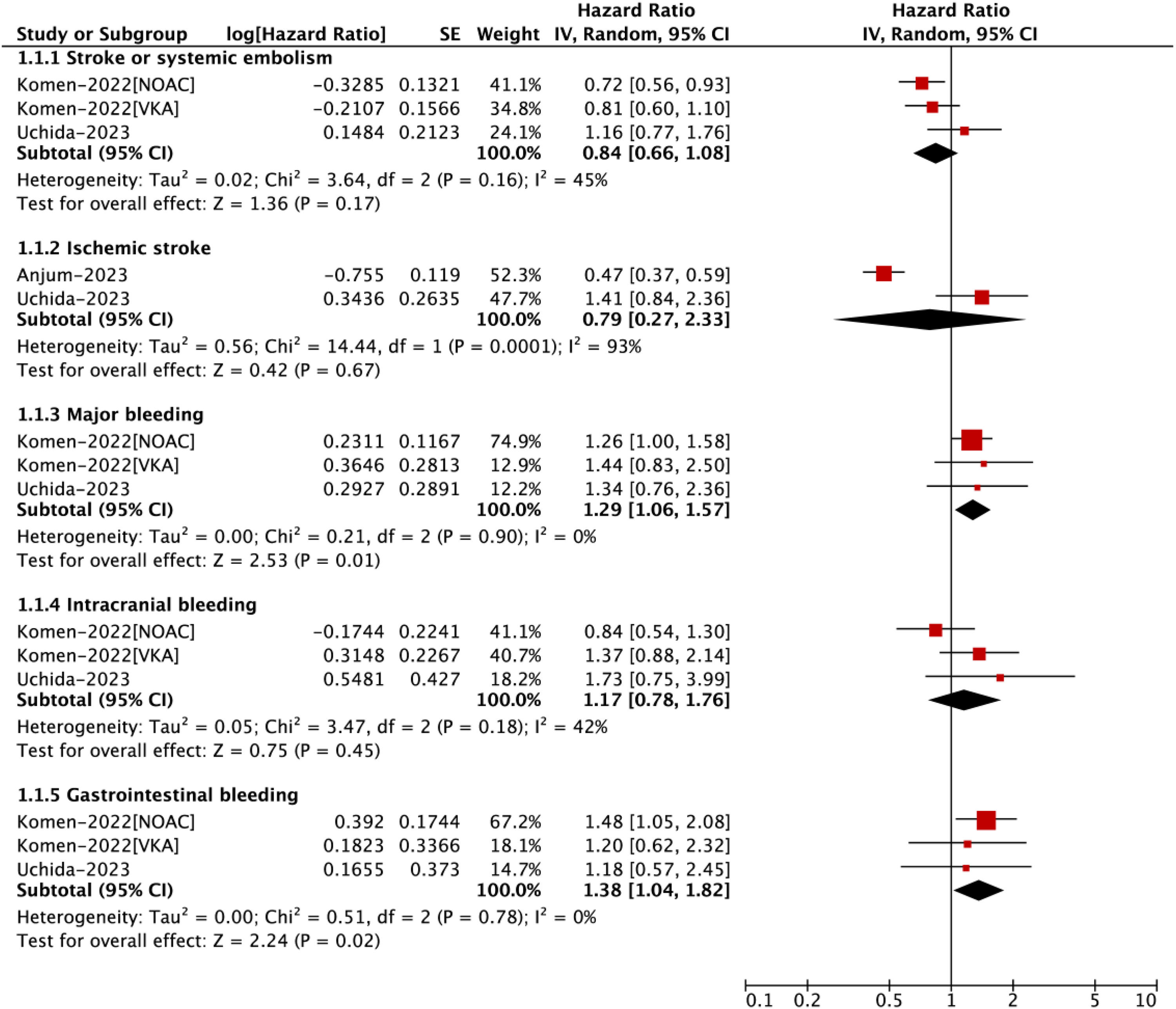
Effectiveness and Safety Outcomes Between OAC and no OAC in AF Patients with an Intermediate Risk of Stroke.
Effectiveness and Safety Outcomes Between NOACs and VKAs
A total of 3 studies assessed the effectiveness and safety outcomes between NOACs and VKAs in the studied population. Our pooled analysis by the random-effects model suggested that the use of NOACs was associated with a reduced risk of all-cause death (HR = 0.54, 95%CI: 0.43-0.68) compared with VKA use. However, there were no significant differences in the risks of SSE (HR = 0.70, 95%CI: 0.36-1.36), ischemic stroke (HR = 0.88, 95%CI: 0.68-1.14), major bleeding (HR = 0.80, 95%CI: 0.64-1.00), intracranial bleeding (HR = 0.42, 95%CI: 0.11-1.55), and gastrointestinal bleeding (HR = 0.92, 95%CI: 0.69-1.21) in AF patients had an intermediate risk of stroke (Figure 3).

Effectiveness and Safety Outcomes Between NOACs and VKAs in AF Patients with an Intermediate Risk of Stroke.
Discussion
In patients with AF who present with a single additional stroke risk factor (defined as a CHA2DS2-VASc score of 1 in males and 2 in females), the prognostic benefit of OAC remains uncertain. Current AF guidelines consider anticoagulation as a reasonable but not strongly supported strategy for stroke prevention in this intermediate-risk population, reflecting the need for more robust evidence. Although several studies have explored the efficacy and safety of OAC compared with no anticoagulation in this subgroup, many of them rely on overlapping datasets, limiting the generalizability of their findings.
To address this evidence gap, we conducted a systematic review and meta-analysis summarizing the available data. Our results indicated that OAC use was not significantly associated with a reduction in thromboembolic events. However, OAC use was linked to a higher risk of major bleeding and gastrointestinal bleeding compared with no anticoagulation. Given the limited number of included studies, these findings should be interpreted with caution, and further high-quality data are required to validate these results.
In a multicentre observational study by Komen et al, 17 59,076 AF patients with CHA2DS2-VASc score of 1 were enrolled from four North European countries (Sweden, Denmark, Norway, and Scotland). No differences in SSE, intracranial bleeding, and major bleeding between OAC and no treatment, but a lower risk of all-cause death was presented in the OAC than that in no OAC group. Lip et al 12 revealed a decreased all-cause death risk, and increased risks of intracranial bleeding, and major bleeding, but no difference in SSE and ischemic stroke in VKA users than no VKA group. In addition, Coleman et al 15 compared the clinical benefits of standard-dose rivaroxaban with those of VKAs in patients with a non-sex-related CHA2DS2-VASc score of 1, demonstrating no difference in ischemic stroke, intracranial bleeding and major bleeding, but a significantly decreased risk of SSE in the rivaroxaban group compared with the VKA group. Thus, rivaroxaban might be superior to VKA for SSE prevention. A sub-analysis of Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation (ARISTOTLE) trial performed by Lopes et al 31 stratified AF patients with low, intermediate, and high stroke risk groups. After randomization of apixaban and warfarin, no differences in SSE, all-cause death, intracranial bleeding, and major bleeding were presented in low- and intermediate- risk patients between the two groups. In addition, Lip et al 14 enrolled patients with CHA2DS2-VASc of 1, and compared the effectiveness and safety outcomes of apixaban, rivaroxaban, and dabigatran with warfarin, respectively. Although no significant difference in SSE between NOACs versus VKAs, NOAC users showed reduced risks of all-cause death and major bleeding compared with VKAs in clinical practice. However, the evidence remains inconclusive and somewhat inconsistent.
VKAs have long been the standard therapy for thromboembolism prevention in AF, especially in patients at high stroke risk. In the intermediate-risk population, the observed reduction in all-cause mortality with VKAs was not accompanied by a significant reduction in thromboembolic events. Rather, VKAs were associated with an increased risk of bleeding, suggesting that warfarin may not offer a favorable benefit-risk profile in this subgroup. In contrast, NOACs—comprising direct thrombin inhibitors and factor Xa inhibitors—were developed to address the limitations of VKAs and are generally associated with a more favorable bleeding profile. Among NOACs, heterogeneity in clinical benefits has been observed. This suggests that individual NOACs may differ in their balance between thromboembolic prevention and bleeding risk. Nevertheless, due to limited data and the small number of comparative studies for each NOAC subtype, we were unable to conduct a subgroup analysis to determine the comparative net clinical benefit of each agent. This remains a critical area for future research.
Patient age also appears to influence the effectiveness and safety of OAC therapy in this group. In patients aged <75 years with a single non-sex-related stroke risk factor, ischemic stroke rates increased across ascending age strata. These data suggest that age is a modifier of thromboembolic risk even among patients classified as intermediate-risk, and support a more nuanced, age-stratified approach to anticoagulation decision-making in such patients.
Taken together, the effectiveness and safety of OAC in patients with intermediate stroke risk are heterogeneous, influenced by age, specific risk factors, and the type of anticoagulant used. Therefore, treatment decisions should be individualized using a comprehensive benefit-risk assessment rather than relying solely on the CHA2DS2-VASc score threshold.
Limitations
This systematic review has several limitations. First, all included studies were observational in nature; no RCTs were available, which limits the strength of the evidence and increases susceptibility to residual confounding. Second, selection bias and clinical heterogeneity across studies—particularly regarding baseline characteristics, definitions of endpoints, and study settings—could not be fully accounted for. Additionally, variations in follow-up duration, timing of anticoagulation initiation, and treatment adherence were not uniformly reported or adjusted for. Third, the limited number of included studies reduced the statistical power of our meta-analysis and precluded more detailed subgroup analyses. Finally, the classification of stroke risk based on the CHA₂DS₂-VASc score includes sex as a criterion, but we were unable to perform a sex-stratified analysis due to lack of disaggregated data, which restricts interpretation of sex-specific treatment effects.
Conclusions
In patients with AF at an intermediate risk of stroke, the available evidence from a limited number of studies suggests that OAC use may not significantly reduce thromboembolic events but could be associated with increased risks of major bleeding and gastrointestinal bleeding compared with no treatment. However, these findings should be interpreted with caution due to the limited number of studies and potential overlapping datasets. Further large-scale, high-quality studies are warranted to confirm these observations.
Footnotes
Author Contributions
ZL-Y and X-C were responsible for the study design, conducting the literature search, performing the systematic review and data collection, carrying out the statistical analysis, interpreting the results, and drafting the manuscript. WG-Z served as the principal investigator, contributing to the study design, assessing all results, and revising the manuscript for typographical and grammatical accuracy.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Sanming City Science and Technology Joint Funding Project (2022- S-65).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
