Abstract
Background:
Although utilization of anticoagulation in patients with atrial fibrillation (AF) has increased in recent years, contemporary data regarding thromboembolism and mortality incidence rates are limited outside of clinical trials. This study aimed to investigate the impact of the direct oral anticoagulants (DOACs) on the clinical outcomes of patients with AF included in the Tasmanian Atrial Fibrillation Study.
Methods:
The medical records of all patients with a primary or secondary diagnosis of AF who presented to public hospitals in Tasmania, Australia, between 2011 and 2015, were retrospectively reviewed. We investigated overall thromboembolic events (TEs), ischemic stroke/transient ischemic attack (IS/TIA), and mortality incidence rates in patients admitted to the Royal Hobart Hospital, the main teaching hospital in the state. We compared outcomes in 2 time periods: prior to the availability of DOACs (pre-DOAC; 2011 to mid-2013) and following their general availability after government subsidization (post-DOAC; mid-2013 to 2015).
Results:
Of the 2390 patients with AF admitted during the overall study period, 942 patients newly prescribed an antithrombotic medication (465 and 477 from the pre-DOAC and post-DOAC time periods, respectively) were followed. We observed a significant decrease in the incidence rates of overall TE (3.2 vs 1.7 per 100 patient-years [PY]; P < .001) and IS/TIA (2.1 vs 1.3 per 100 PY; P = .022) in the post-DOAC compared to the pre-DOAC period. All-cause mortality was significantly lower in the post-DOAC period (2.9 vs 2.2 per 100 PY, P = .028). Increasing age, prior stroke, and admission in the pre-DOAC era were all risk factors for TE, IS/TIA, and mortality in this study population. The risk of IS/TIA was more than doubled (hazard ratio: 2.54; 95% confidence interval: 1.17-5.52) in current smokers compared to ex- and nonsmokers.
Conclusion:
Thromboembolic event and all-cause mortality rates were lower following the widespread availability of DOACs in this population.
Introduction
Atrial fibrillation (AF) increases the risk of thromboembolic events (TEs) including ischemic stroke (IS), transient ischemic attack (TIA), pulmonary embolism (PE), and myocardial infarction (MI) and significantly increases the risk of morbidity and mortality. 1 Epidemiological research shows that the prevalence of AF is rising globally. 2,3 This is likely to result in a substantial increase in hospital admissions and mortality due to AF-related cardiovascular complications in the future. Encouragingly, observational data suggested that the proportion of patients with AF receiving oral anticoagulants (OACs) for stroke prevention is also increasing. 4,5 The major reasons for the growing rates of anticoagulation include the recent availability of direct-acting oral anticoagulants (DOACs), 4 updated AF guidelines, 6,7 and improved awareness about the importance of stroke prevention in AF. 2,8
Direct-acting oral anticoagulants were subsidized for stroke prevention in nonvalvular AF in Australia in August 2013 in the hope that they would improve the clinical outcomes of patients and reduce the burden of AF-related stroke in the community. Direct-acting oral anticoagulants were rapidly adopted by prescribers, and by mid-2015, almost 70% of anticoagulated patients with AF presenting to the Royal Hobart Hospital (RHH) received a DOAC. 9,10 The Tasmanian AF study was established in 2012 to monitor prescribing trends and patient outcomes in Tasmania, Australia, over time. 4,11,12 In this analysis, we aimed to investigate the impact of the availability of DOACs on the TE-related readmissions of patients with AF. Our specific objectives were: (1) to compare TE and mortality rates in patients commenced on antithrombotic therapy prior to and following the availability of DOACs and (2) to identify factors that influenced the risk of incident thromboembolism and all-cause mortality in patients with AF.
Methods
Study Design and Participants
This was a retrospective observational study involving consecutive patients with AF admitted between 2011 and 2015 to the RHH, Tasmania, Australia. Patients were identified using the Australian Refined Diagnosis Related Groups, AR-DRG code-I48, for AF or flutter (referred to as AF hereafter). The Tasmanian Health and Medical Human Research Ethics Committee approved the study. Patients’ demographics, confirmation of the diagnosis of AF comorbidities, and prescribed medications were obtained by reviewing digital medical records. Baseline stroke and bleeding risk scores were calculated using the CHA2DS2-VASc (one point each for Congestive heart failure, Hypertension, Diabetes mellitus, Vascular disease, Sex category-females, Age 65-74 years, and two points for Age ≥ 75 years and prior Stroke/TIA or TE) and HAS-BLED (Hypertension, Abnormal renal/liver function, Stroke, Bleeding predisposition, Labile international normalized ratios (INRs, if documented on medical records), Age >65 years, Drugs (antiplatelet agents or non-steroidal anti-inflammatory drugs) or alcohol use > 8 units/week, previous stroke). 13,14 The Charlson comorbidity index (CCI) was used to quantify and compare comorbid conditions among groups. 15 The CCI, CHA2DS2-VASc, and HAS-BLED scores were estimated based on demographic variables and comorbidities diagnosed before or during index admissions. An index admission was defined as the participants’ first hospitalization to our study setting with AF as a primary or secondary diagnosis. Readmissions were subsequent hospitalizations after the index admission during the study period.
Follow-Up and Outcomes
To compare baseline characteristics, identify risk factors, and quantify incidence rates, patients were grouped according to the presence or absence of thromboembolism diagnosed at index admission or readmissions during follow-up. A TE was defined as a composite outcome including IS/TIA, other systemic embolic events (PE or deep vein thrombosis [DVT]), and MI. Hospitalizations due to TEs were ascertained by reviewing the medical records where these comorbidities were documented as a primary or secondary diagnosis during the study period. Only the first TE was used in the analyses.
Analysis of TE and mortality incidence rates involved patients with AF newly initiated on an antithrombotic therapy at their index admissions. Exposure to an antithrombotic therapy at the discharge of index admissions was stratified according to the prescription of warfarin, DOACs, and antiplatelet (APT) agents (prescribed without an anticoagulant). For each patient, follow-up began immediately after treatment initiation and continued to the first TE, treatment change, treatment discontinuation, in-hospital mortality, or end of the study period, whichever came first. For comparative purposes, we grouped study participants based on their index admission dates as follows: (1) pre-DOAC period—admissions between January 2011 and July 2013 and (2) post-DOAC period—admissions between August 2013 and July 2015. Finally, TE and all-cause in-hospital mortality incidence rates were also compared by treatment category initiated at the discharge of index hospitalizations.
Definitions of Antithrombotic Exposure, Switch or Discontinuation
Exposure to an antithrombotic (new initiation) was defined as discharge with an antithrombotic medication in patients with AF who did not have a documented history of being prescribed an antithrombotic prior to their index hospitalization. We considered treatment to be discontinued when patients previously receiving thromboprophylaxis were discharged without any antithrombotic medication at subsequent readmissions, because of an emergence of contraindications, or without documented reasons, for an unspecified period. An antithrombotic switch was defined as a change from one type of antithrombotic to another that occurred during a readmission.
Statistical Analysis
All statistical analyses were executed using R, version 3.2.5. Continuous variables were expressed as means (standard deviations), and group differences were compared using analysis of variance or independent samples Student t tests. Categorical variables were summarized as frequencies and proportions, and differences were tested using χ2 or Fisher exact tests, as appropriate.
We pooled IS/TIA, other systemic embolic events, and MI into a composite end point as overall TEs. Further, we pooled IS/TIA and other systemic embolic events as another composite end point and also completed an additional analysis for IS/TIA events only. The all-cause mortality incidence rate was estimated by following patients from the date of index hospitalization to death or the end of the study period. For all outcomes, the first event after AF diagnosis was included regardless of whether the patient had a prior history of thromboembolism. Incidence rates and corresponding 95% confidence intervals (CIs) were calculated as the number of the first event in each category divided by the total patient-years (PY) at risk for a particular outcome within the observation period. All of the events were expressed as rates/100 PY of follow-up. Cox proportional hazards regression analyses adjusted for baseline covariates were performed to identify risk factors for overall TE, IS/TIA, and all-cause mortality. The proportional hazards assumption was tested using scaled Schoenfeld residuals and was found appropriate for all the models. Risk relationships were presented as hazard ratios (HRs) and 95% CIs. A P value of <.05 was considered statistically significant for all analyses.
Results
During the study period (January 2011 to July 2015), we identified 2390 patients with AF as a primary or secondary diagnosis. In total, 384 (16.1%) patients experienced thromboembolic complications during their index hospitalization or at readmissions during follow-up; 264 (68.8%) had IS/TIA, 90 (23.4%) had MI, and 30 (7.8%) had other systemic embolic events including PE and DVT. Baseline characteristics of the overall study population and those admitted with thromboembolism are summarized in Table 1. Current smoking, hypertension, ischemic heart disease, and a history of prior stroke were significantly more frequent in patients admitted with TEs than those admitted due to other causes. Moreover, patients who experienced TEs were older and had a higher stroke and bleeding risk scores than patients who did not experience TEs.
Baseline Characteristics (Overall and by TE Diagnosed During Follow-Up).

Abbreviations: CCI, Charlson comorbidity index; SD, standard deviation; TE, thromboembolic event.
CHA2DS2-VASc, Congestive heart failure; Hypertension, Diabetes, Vascular disease (prior myocardial infarction, peripheral artery disease or aortic plaque), Age 65-74 years, Sex category-females, Age ≥ 75 years and Stroke or transient ischemic attack. HAS-BLED, Hypertension, Abnormal renal/liver function, Stroke, Bleeding predisposition, Labile international normalized ratios, Age >65 years, Drugs (antiplatelet agents or non-steroidal anti-inflammatory drugs) or alcohol use > 8 units/week, previous stroke. CCI, Charlson Comorbidity Index
aIncludes thromboembolic event (stroke/transient ischemic attack, systemic embolism, and myocardial infarction) diagnosed at index admission or readmissions.
bIncludes current and ex-smokers.
cIncludes patients with a history of deep venous thrombosis and pulmonary embolism.
Thromboembolic and Mortality Incidence Rates
Thromboembolic and mortality incidence rates categorized by admission eras and treatment groups are presented in Tables 2 and 3, respectively. A total of 942 patients (465 and 477 from the pre- and post-DOAC eras, respectively) newly initiated on antithrombotic therapy at their index admission were included for this analysis. Approximately half (52.2%) of the patients with AF admitted during the post-DOAC era received DOACs; 26.2% received warfarin and 22.6 received APT agents. As detailed in Table 3, a total of 258 patients were newly initiated on DOACs; most (88.7%) of these patients received factor Xa inhibitors, mainly rivaroxaban (142; 55.1%), followed by apixaban (87; 33.7%), while dabigatran was prescribed for a relatively small number of patients (29; 11.2%). Edoxaban was not approved in Australia during our study period. The mean follow-up duration for the pre- and post-DOAC cohorts was 1204 and 863 PY, respectively. Overall, 54 patients (39 from the pre-DOAC and 15 from the post-DOAC time periods) experienced TE during follow-up.
Thromboembolic Event and All-Cause Mortality Incidence Rates in Patients with AF by Admission Era.

Abbreviations: AF, atrial fibrillation; CI, confidence interval; DOAC, direct oral anticoagulant; IS, ischemic stroke; MI, myocardial infarction; PY, patient-years; SE, systemic embolic event; TEs, thromboembolic events; TIA, transient ischemic attack.
aIncludes ischemic stroke/transient ischemic attack, systemic embolism, and myocardial infarction.
bIncludes other systemic embolic diseases such as deep venous thrombosis and pulmonary embolism.
Thromboembolic Event and All-Cause Mortality Incidence Rates by Antithrombotic Therapy at Discharge of the Index Admission.
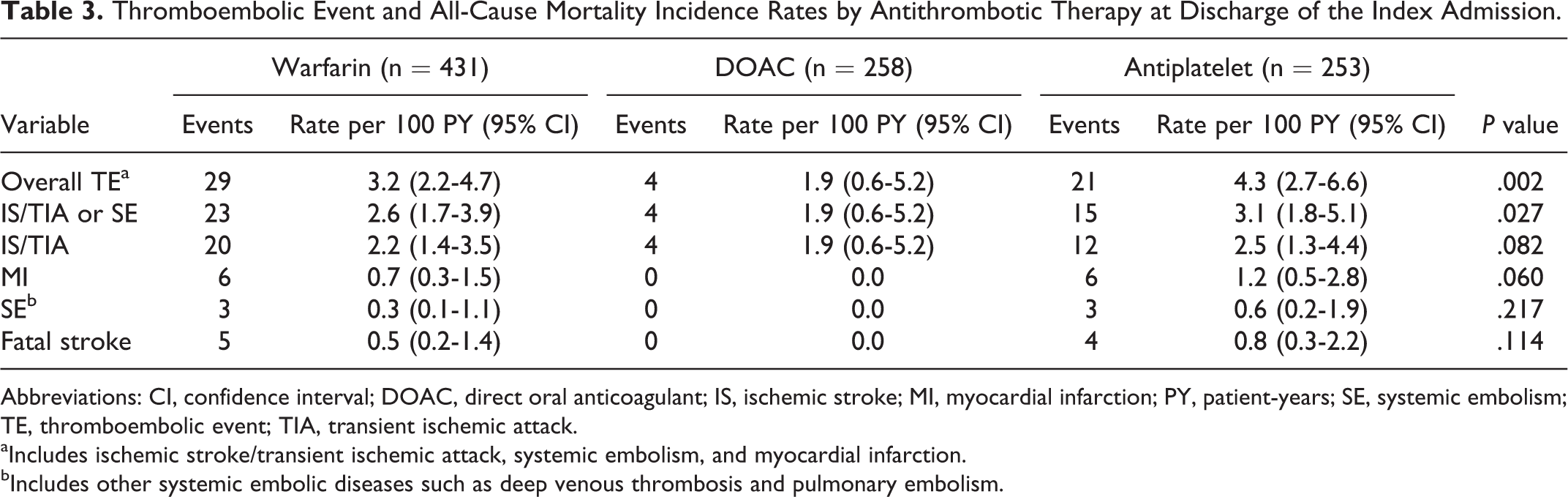
Abbreviations: CI, confidence interval; DOAC, direct oral anticoagulant; IS, ischemic stroke; MI, myocardial infarction; PY, patient-years; SE, systemic embolism; TE, thromboembolic event; TIA, transient ischemic attack.
aIncludes ischemic stroke/transient ischemic attack, systemic embolism, and myocardial infarction.
bIncludes other systemic embolic diseases such as deep venous thrombosis and pulmonary embolism.
A significant reduction was observed in the incidence rates of overall TEs, TE/TIA or systemic embolic events, IS/TIA events, and all-cause mortality in the post-DOAC compared to the pre-DOAC era. During follow-up, TE occurred in 39 patients in the pre-DOAC group and 15 patients in the post-DOAC group (3.2 vs 1.7 per 100 PY; P < .001). Combined IS/TIA or other systemic embolic events occurred in 29 patients in the former and 13 patients in the later period (2.4 vs 1.5 per 100 PY; P = .014). Similarly, a significant decline in the rate of IS/TIA was observed in the post-DOAC era (2.1 vs 1.3 per 100 PY; P = .022). Furthermore, the incidence rate of all-cause mortality was significantly lower in the post-DOAC era than in the pre-DOAC era (2.9 vs 2.2 per 100 PY; P = .028). The majority of all-cause mortalities occurred in warfarin-treated patients followed by those receiving APT agents. Of the 54 deaths observed, 28 (51.9%) and 16 (29.6%) events occurred in warfarin- and APT-treated patients, respectively; 10 (18.5%) events occurred in patients receiving warfarin-APT combination therapy. The remaining 10 (18.5%) deaths occurred in the DOAC cohort (7 in patients receiving rivaroxaban and 3 in those receiving apixaban).
When TE incidence rates were analyzed by antithrombotic prescribing at the discharge of the index hospitalizations, lower rates of overall TE and combined IS/TIA or other systemic embolic events were observed in patients initiated on DOACs than in the warfarin- and APT-only cohorts. Mean follow-up durations by treatment category were 894 PY for warfarin, 208 PY for DOACs, and 488 PY for APT agents. Most of the TE events (93%) were observed in the warfarin and APT cohorts, while only 4 TEs occurred in the DOAC cohort. Of the 4 TEs in the DOAC cohort, 1 IS occurred in each of the rivaroxaban, apixaban, and dabigatran groups; 1 TIA was also observed in an apixaban-treated patient. The overall TE incidence rates per 100 PY were 3.2 in the warfarin, 1.9 in the DOAC, and 4.3 in the APT groups (P = .002 for the group comparison). Moreover, IS/TIA or other systemic embolism incidence rates per 100 PY were 2.6 in the warfarin, 1.9 in the DOAC, and 3.1 in the APT agent groups (P = .027 for the group comparison).
Factors That Influenced the Risk of Thromboembolism and Mortality
Table 4 summarizes Cox regression analysis results for the risk factors associated with overall TEs (IS/TIA, other systemic embolic events, and MI), IS/TIA, and all-cause mortality. After adjusting for baseline covariates, independent risk factors associated with the incidence of overall TEs were increasing age, hypertension, history of prior stroke, and MI. Furthermore, admission in the pre-DOAC era (relative to admission in the post-DOAC era) was also significantly associated with an incident risk of TE (HR: 1.21; 95% CI: 1.02-1.84).
Factors That Influenced the Risk of Overall TEs, IS/TIA, and All-Cause Mortality.

Abbreviations: CI, confidence interval; DOAC, direct oral anticoagulant; HR, hazard ratio; IS, ischemic stroke; TEs, thromboembolic events; TIA, transient ischemic attack.
aIncludes current and ex-smokers.
bPulmonary embolism and deep venous thrombosis.
Independent risk factors for incident IS/TIA included increasing age, current smoking, hypertension, prior stroke, and hospitalization in the pre-DOAC era. Finally, risk factors independently associated with the incidence of all-cause mortality included increasing age, chronic renal diseases, prior stroke, MI, and admission in the pre-DOAC era. Sex category, alcohol use (>8 units/week, as documented on the patients’ medical records), a history of congestive heart failure, diabetes mellitus, chronic respiratory disease, ischemic heart disease, valvular heart diseases, and other systemic embolic events were not significantly associated with the risk of overall TEs, IS/TIA, and all-cause mortality in this study population.
Discussion
This study provides a comprehensive description of patients with AF admitted with TEs, identifies risk factors, and compares TE and all-cause mortality incidence rates prior to and following the introduction of DOACs into clinical practice. We observed a significant reduction in the incidence rates of TEs and all-cause mortality in patients with AF following the government subsidization of DOACs. The TE incidence rates were also lower in patients initiated on DOACs compared to those initiated on warfarin and APT agents at index admissions. Furthermore, hospitalization post-DOAC availability was associated with a decreased risk of overall TEs, IS/TIA, and all-cause mortality. This was despite the similarity in the baseline characteristics of the study participants newly initiated on antithrombotic therapies, particularly when we compared baseline stroke risk scores between the 2 time periods (Supplemental Table 5). The declining rates of TEs, IS/TIA, and total mortality in this analysis were consistent with findings that showed a declining trend of stroke and mortality incidence rates in AF reported from other countries. 16 –18
The explanations for these findings are likely multifactorial. First, changes in clinical practice such as increasing rates of OAC prescribing in recent years, driven mainly by the availability of DOACs, and updates in AF management guidelines could have improved patient outcomes. A recent analysis of OAC prescribing in AF in the same population showed the proportion of patients with AF receiving OACs increased significantly over the study period. 11,19 Second, DOACs are increasingly used in patients with nonvalvular AF in preference to warfarin. 4,10 Comparative analysis involving warfarin-treated patients in this study also indicated no significant difference in the overall TE, IS/TIA, and all-cause mortality incidence rates between the 2 eras (data not shown). This suggests the observed improvements in clinical outcomes were potentially due to the increasing utilization of DOACs in patients with AF rather than improvements in warfarin management. Based on trial results, this is expected to result in fewer complications such as intracranial hemorrhage or other major bleeding events. Clinical trials demonstrated lower rates of major bleeding and mortality in patients treated using DOACs compared to those receiving warfarin. 20,21 In addition, observational data have shown that all of the DOACs reduced intracerebral bleeds, the most feared complication of OAC therapy in AF, by at least 50%. 22 Meta-analysis studies also reported a significant decline in IS/systemic embolism and total mortality that appeared to be driven by a reduction in fatal bleeding in DOAC-treated patients compared to warfarin-treated patients. 23 –25
Although randomized clinical trials and the majority of observational data showed comparable efficacy between DOACs and warfarin in terms of stroke prevention, few studies indicated lower rates of thromboembolism in patients treated with DOACs than those treated with warfarin. 26 A real-world practice study among Asians with nonvalvular AF reported a lower risk of IS/systemic embolism and all-cause mortality in patients with AF receiving dabigatran and rivaroxaban compared with patients receiving warfarin. 27 Moreover, other factors such as improved management of chronic diseases including hypertension, congestive heart failure, and diabetes mellitus could also play significant roles in the reduction of TE and mortality incidence rates in AF.
The AF-related TE rates in cohort studies are highly heterogeneous and have ranged from 0.45 to 9.28 per 100 PY owing to variabilities in the study design, study setting, and the data source. 28 While low overall TE and IS/TIA incidence rates were observed in patients initiated on DOACs in this study, IS/TIA incidence rates in patients initiated on warfarin in this study were broadly similar to rates reported from large observational studies in Europe and Japan. 29 –31 Our analysis showed comparable rates of IS/TIA and other systemic embolic events, but a lower rate of all-cause mortality than rates reported from a large nationwide observational study in Denmark. 29 Furthermore, no MIs were observed in patients initiated on DOACs, while the incidence rate of MI in patients initiated on warfarin in this analysis corresponded with a 0.7 per 100 PY MI rate in patients with AF who received warfarin in the United Kingdom. 30
The risk of stroke in patients with AF is high and increases in the presence of other risk factors, particularly those described in the validated CHA2DS2-VASc stroke risk score. 13 However, the contribution of some of these factors may underestimate the true risk of stroke in the real-world patient management. For instance, a history of prior stroke is the most prominent risk factor for IS/TIA in AF, and the contribution of prior stroke in predicting IS/TIA recurrence is considered to be higher than that described in the CHA2DS2-VASc score. 32,33 Although existing evidence is not robust, other comorbidities and demographic factors not included in the CHA2DS2-VASc score such as chronic renal diseases and smoking could be important risk factors for thromboembolism in AF. Our study builds on the available data by comparing previously reported risk factors and identifying concomitant comorbidities as additional risk factors for TE and mortality in contemporary patients with AF.
The strongest risk factors for the overall TE and IS/TIA outcome used in this study were a history of stroke, followed by hypertension, admission in the pre-DOAC era, and increasing age. Patients with a history of prior IS/TIA had a more than 2-fold increase in the risk of overall TE and all-cause mortality and more than 3-fold increase in the risk of recurrent IS/TIA than patients without a history of prior IS/TIA. Previous studies also showed a history of prior stroke as the most powerful and consistent risk factor for recurring cerebrovascular events in AF. 33,34 Current smoking was also identified as an important risk factor for IS/TIA in our analysis; the risk of having an IS/TIA was more than doubled (HR: 2.54; 95% CI: 1.17-5.52) in current smokers over nonsmokers and former smokers.
Similarly, an observational study involving optimally anticoagulated patients with AF found an independent association between smoking and IS/TIA. 32 The Framingham Heart Study also showed the risk of stroke in heavy smokers (>40 cigarettes/day) was twice that of light smokers (<10 cigarettes/day), whereas stroke risk decreased significantly 2 years after smoking cessation. 35 Evidence from pathological data and clinical research established that smoking causes mainly atherothrombotic strokes, as opposed to cardioembolic strokes due to AF. 36,37 Such strokes are shown to be effectively treated by APT therapy in preference to OAC therapy. 38,39 Further studies should investigate the differential relationship of smoking with the risk of cardioembolic stroke and outcomes of OAC versus APT therapy in smoking patients with AF.
Patients with advanced age, chronic renal diseases, prior IS/TIA, MI, and hospitalization in the pre-DOAC era (relative to the later period) were significant risk factors for all-cause mortality in this study. Similarly, a meta-analysis study by Gomez-Outes et al revealed that IS/TIA and bleeding accounted for 6% of all deaths, with aging and decreased creatinine clearance as significant risk factors for mortality in patients with AF. Unlike our data, however, congestive heart failure, diabetes mellitus, and male sex were also significant risk factors for mortality in their study. 40 In conclusion, our data suggest that, in addition to optimal anticoagulation of patients with AF, effective control of modifiable risk factors such as smoking and management of comorbidities is essential to further reducing thrombosis and mortality in patients with AF.
Strengths and Limitations
The major strength of our study is that we include a relatively large number of participants over an extended observation period comprising admissions from both the pre- and post-DOAC time periods. However, given the observational nature of our data, there are a number of limitations. First, there is a potential for selection bias inherent to retrospective observational studies; exposure to antithrombotic therapy at index admissions was not randomized. Second, we were not able to capture TEs resulting in readmission to other clinical settings, minor events managed in local general practices, severe thromboembolic complications leading to prehospital death, or death occurring outside our study setting. This may underestimate the overall TE and mortality incidence rates observed in this study. Third, as the study population was hospitalized patients with AF, incidence rates may not reflect clinical outcomes of AF populations managed in community settings. Fourth, despite statistical adjustments, there is always a possibility of residual confounding limiting our ability to draw causal inferences. Moreover, adherence to OAC therapy and its impact on patient outcome were not assessed in this study population. Lastly, the number of TEs in the DOAC cohort was insufficient to permit evaluation of the relative efficacy of each agent. Further investigations involving more patients with extended follow-up are required to assess comparative efficacy and safety of individual DOACs.
Conclusion
In this contemporary cohort of hospitalized patients with AF, the incidence rates of TE and all-cause mortality improved significantly following the introduction of DOACs. These improved clinical outcomes are likely to be multifactorial, potentially including the recent increase in OAC prescribing and the use of DOACs in preference to warfarin. Patients with a history of stroke, increasing age, and admissions in the pre-DOAC era were most vulnerable to incident TEs, IS/TIA, and all-cause mortality in this population. Furthermore, the risk of IS/TIA was more than doubled in current smokers compared to ex- and nonsmokers. The results suggest there are further opportunities to improve patient outcomes in AF through judicious use of OACs, effective management of comorbidities, and controlling modifiable stroke risk factors.
Supplemental Material
Supplementary_Material - Thromboembolism and Mortality in the Tasmanian Atrial Fibrillation Study
Supplementary_Material for Thromboembolism and Mortality in the Tasmanian Atrial Fibrillation Study by Endalkachew Admassie, Leanne Chalmers, and Luke R. Bereznicki in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Author Contributions
Admassie contributed to conception and design, acquisition and analysis, drafted the manuscript, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Chalmers contributed to conception, contributed to interpretation, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Bereznicki contributed to conception, contributed to interpretation, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was solely funded by the Division of Pharmacy, School of Medicine, University of Tasmania. Luke Bereznicki and Leanne Chalmers report receiving a grant from Aspen Pharmacare Australia, and Luke Bereznicki reports receiving personal fees for the provision of expert advice from Boehringer Ingelheim Pty Ltd. unrelated to this study.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
