Abstract
New orally administered anticoagulants will simplify stroke-prevention strategies in patients with atrial fibrillation (AF). Novel anticoagulants, such as dabigatran etexilate, a direct thrombin inhibitor, and rivaroxaban, a direct factor Xa inhibitor, have been approved by the US Food and Drug Administration for the prevention of stroke and systemic embolism in patients with nonvalvular AF. In addition, the factor Xa inhibitor apixaban has been reported to be as effective as warfarin in a large, randomized clinical trial, and the efficacy of edoxaban is being assessed in a phase III warfarin comparison trial. This review discusses the limitations of vitamin K antagonist therapy for patients with AF and establishes the need for alternative, effective anticoagulation with an improved benefit–risk ratio for the prevention of stroke. Novel anticoagulants have the potential to provide convenient, effective stroke prophylaxis without many of the issues inherent in the use of traditional agents.
Introduction
Valvular rheumatic heart disease (rheumatic or valvular atrial fibrillation [AF]) and nonvalvular AF (NVAF) carry an increased risk of arterial thromboembolism and ischemic stroke,1,2 resulting from thrombi that form within the left atrium, particularly within the left atrial appendage. 3 The prevalence of diagnosed AF (valvular and nonvalvular) has been reported as 0.95% in a large, ethnically diverse US population. 4 Interestingly, only 4.9% of these patients had known valvular heart disease, 4 reflecting a declining incidence of rheumatic heart disease. The prevalence of AF increases with age, rising from 0.1% among those <55 years of age to 9.0% in those ≥80 years of age. 4
Since the 1960s, clinicians have employed long-term anticoagulation to prevent stroke in patients with rheumatic AF. 5 However, it was not until the 1990s that a series of prospective randomized clinical trials established a role for long-term anticoagulation in the prevention of NVAF-related stroke. A meta-analysis of these trials demonstrated that adjusted-dose warfarin reduced the relative risk (RR) of stroke by 62% compared with placebo. 6 When only ischemic stroke was analyzed, the RR reduction with adjusted-dose warfarin was 65%. 6
Until recently, vitamin K antagonists (VKAs) were the only form of oral anticoagulant medication approved for the long-term prevention of stroke in patients with AF, and they continue to be widely used for this purpose. 7 However, while VKAs are highly effective for the prevention of thrombotic disease, 8 they have well-known limitations, including a variable and unpredictable dose response and multiple drug–drug and food–drug interactions. 9 For these reasons, pharmaceutical companies have invested in the development of alternative anticoagulants, leading to the recent approvals by the US Food and Drug Administration (FDA) of dabigatran and rivaroxaban to reduce the risk of stroke and systemic embolism in patients with NVAF and to the approval of apixaban by the European Union for the AF indication.10–12 Despite the availability of these oral anticoagulants, the majority of patients managed with chronic oral anticoagulation continues to receive VKAs.
This review will discuss the limitations of VKA therapy for patients with AF and establish the ongoing need for alternative, effective anticoagulation with improved benefit–risk ratios for the prevention of stroke.
Limitations of VKAs
Difficulties in the management of VKAs are clinically important. A reduction in thromboembolism with VKAs is balanced by an increased risk of bleeding events. A meta-analysis by Hart and colleagues found that, compared with placebo, adjusted-dose warfarin was associated with a 130% increase in the RR for major extracranial hemorrhage. 13 With warfarin, the most effective stroke prophylaxis with the lowest risk of bleeding is achieved by maintaining the international normalized ratio (INR) within the American College of Chest Physicians (ACCP)-recommended range of 2.0 to 3.0. 14 However, even with good management, many patients are within the target range less than 60% of the time, 15 reflecting the unpredictable pharmacology, multiple drug and food interactions, and patient-specific factors such as genetic polymorphisms associated with these agents. 9
Antiplatelet agents, such as aspirin and clopidogrel, are not subject to many of the issues outlined above, but they are considerably less efficacious than warfarin.6,16 Current ACCP guidelines recommend that patients with AF who are at intermediate or high risk of stroke receive oral anticoagulants rather than aspirin or combination therapy with aspirin and clopidogrel. 14
Stroke Risk Stratification
Current risk stratification schemes are largely based on risk factors identified in randomized clinical trials. Some “weaker” risk factors, such as female gender and vascular diseases (including myocardial infarction [MI] and peripheral vascular disease), are not included. 17 However, the Birmingham 2009 scheme, a refinement of the CHADS2 scoring system known as CHA2DS2-VASc, incorporates these weaker factors. 17 The new scheme assigns 2 points each for stroke/transient ischemic attack (TIA)/systemic embolism, and age ≥75 years, and 1 point each for congestive heart failure, hypertension, diabetes mellitus, vascular disease (previous MI, peripheral arterial disease, or aortic plaque), age 65 to 74 years, and female gender. 17 A score of 0 defines low risk, 1 is moderate risk, and ≥2 is high risk. 17
The proportion of patients stratified to the individual risk categories (low, moderate, or high) differs substantially among existing schemes. 18 The Stroke Risk in Atrial Fibrillation Working Group compared 12 schemes and found that, in a representative cohort of 1000 patients with NVAF, the number of patients stratified as low risk varied between 7% and 42%. 18 In a similar analysis of 8 schemes, Lip and colleagues found that, in 1084 patients with AF from the European Heart Survey, as few as 9% to as many as 48% of the patients were classified as low risk. 17 Interestingly, the Birmingham 2009 scheme generated the 9% figure, and this corresponds with patients described in the publication as being at “truly low risk” (CHA2DS2-VASc score = 0 and no recorded thromboembolism events at 1 year). 17
Although schemes vary, studies suggest that as many as 90% of patients with AF are at moderate-to-high risk of stroke, warranting prophylaxis with anticoagulants. 18
Extending the Benefits of Anticoagulation
Prolonged monitoring (eg, via a Holter monitor or occasional event loop recorders) of patients hospitalized with ischemic stroke detected an additional 5% to 8% of patients with AF that was not present on the initial electrocardiograph (ECG), 19 a relatively low yield. Identification of risk factors in this population may help to limit the number of patients who require prolonged monitoring and increase the percentage found to have intermittent AF.
A study of the medical records of 827 patients with ischemic stroke found that hypertension was present in 86.7% of patients with AF and in 77.5% of patients without documented AF. 20 In addition, in a cohort of 633 stroke patients with hypertension, the incidence of echocardiographic abnormalities was significantly higher in those with documented AF: left atrial enlargement ≥4.0 cm, odds ratio (OR) 5.85; left ventricle ≥5.6 cm, OR 2.21; and ejection fraction <50%, OR 2.63. There was also more congestive heart failure (OR 3.89) and more coronary disease (OR 1.65). 21 Adding the presence of echocardiographic abnormalities to clinical risk factors should enhance the yield of prolonged event monitoring in patients who have had a stroke/TIA but no recorded AF as well as increase the proportion of patients with stroke who are diagnosed as having occult paroxysmal AF. This will allow the use of appropriate prophylaxis against recurrent strokes in this vulnerable patient population. 21
A retrospective study of 128 patients with implanted pacemakers or with automatic implantable cardioverters/defibrillators (AICD) who had ischemic strokes found that 71.1% had a diagnosis of intermittent or permanent AF. 22 The increased diagnosis of AF may be due to the permanent “Holter” monitoring in most current pacing and AICD devices and the frequent ECGs that these patients have; 98.9% of patients with strokes and AF had CHADS2 scores ≥2 or abnormal echocardiographic findings.22,23 Ziegler et al, studying a similar group of patients with implanted rhythm devices, found newly detected intermittent AF in 28% of patients with prior cryptogenic thromboembolic events. 24 Taking into account the number of patients with a documented history of AF, warfarin use, or antiarrhythmic drug use (on the presumption that they had prior AF), 63% of the study population would have had AF. 24 These studies of patients with stroke have shown a high incidence of AF or of risk factors that go along with intermittent AF, suggesting that AF may have been a substantial contributing factor in many ischemic strokes. It is likely that more ischemic strokes are caused by AF than previously thought. Risk factors associated with stroke (hypertension, congestive heart failure, advanced age, diabetes, and coronary artery disease) are also risk factors for AF, suggesting that AF may be the common mechanism leading to stroke.22,23 All these risk factors cause left atrial wall stress via systolic or, more commonly, diastolic dysfunction, promoting AF and subsequent arterial emboli in the fibrillating left atrium.
By identifying patients with these clinical and echocardiographic risk factors for stroke and AF, it may be possible to detect patients with occult paroxysmal AF before their first thromboembolic event. 21 Prolonged monitoring could be expanded to patients who have not experienced a stroke/TIA but who are at high risk so that appropriate preventive measures (eg, anticoagulation) can be taken. 21
New Oral Anticoagulants in the Development for Stroke Prevention in AF
Building on advances in the scientific understanding of the coagulation cascade, researchers have developed new oral anticoagulants with predictable responses and limited or no drug and food interactions that can be given at fixed doses without the need for routine coagulation monitoring. 25 The most successful efforts have focused on specific direct inhibition of single coagulation factors, in particular factor IIa (thrombin) and factor Xa. 26
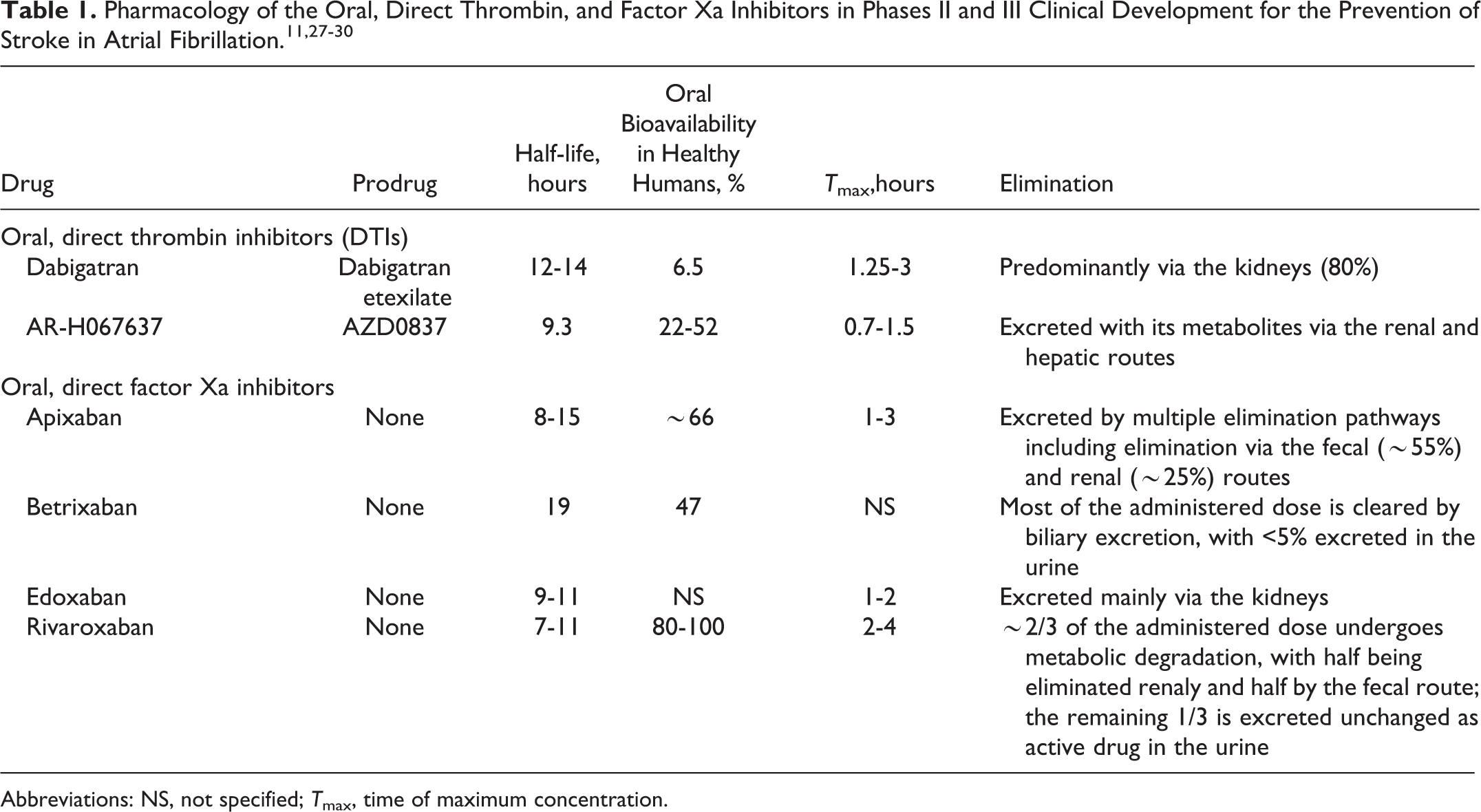
Table 1 summarizes the pharmacologic properties of the new oral anticoagulants.11,27–30 Table 2 lists those drugs in advanced clinical development for the prevention of stroke in patients with AF,31 –34 with an overview of their respective phase III trials.
Abbreviations: NS, not specified; T max, time of maximum concentration.
Oral, Direct Thrombin and Factor Xa Inhibitors in Late-Stage Clinical Development for the Prevention of Stroke in AF: Phase III Study Designs in Order of Study-Completion Date.
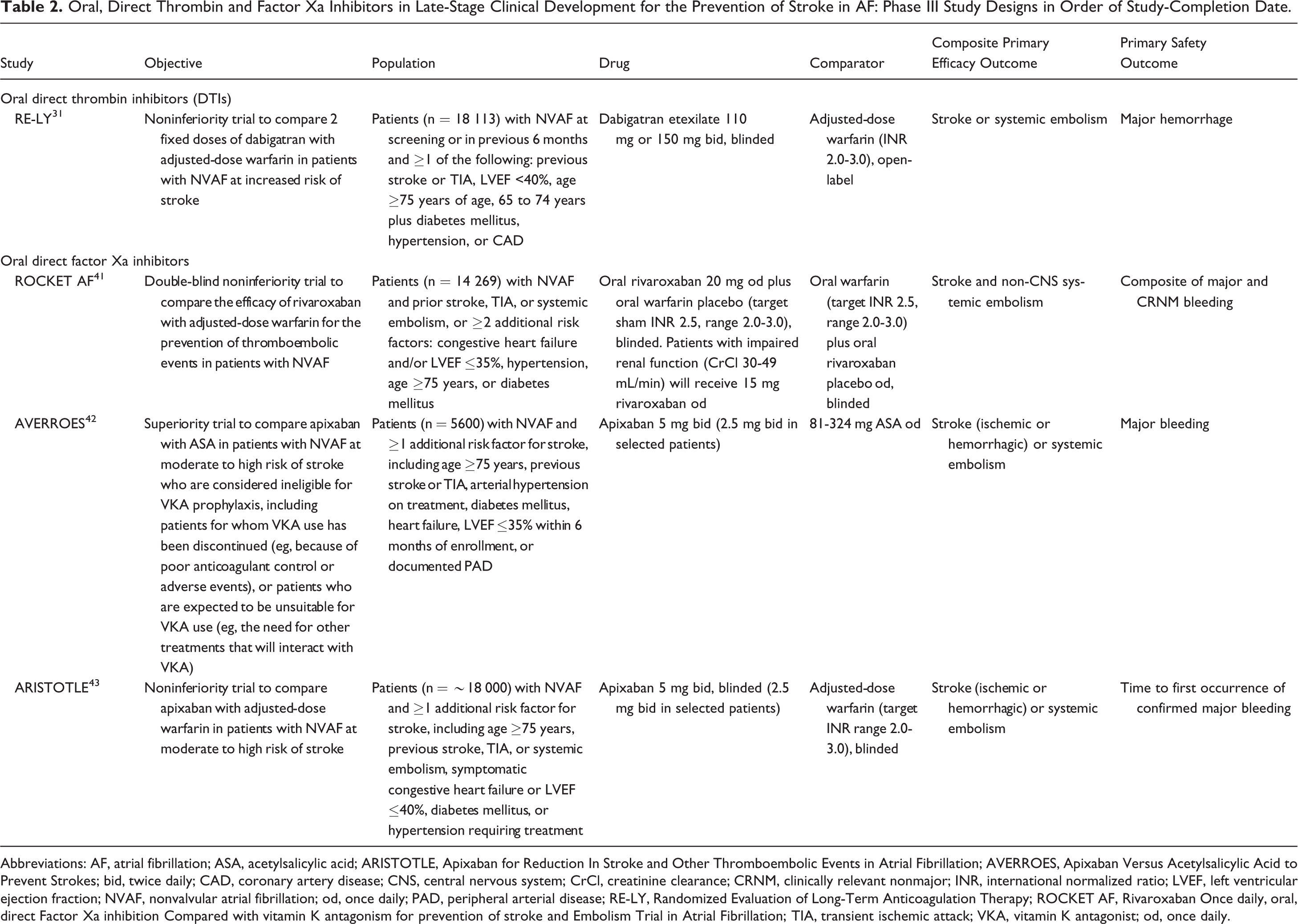
Abbreviations: AF, atrial fibrillation; ASA, acetylsalicylic acid; ARISTOTLE, Apixaban for Reduction In Stroke and Other Thromboembolic Events in Atrial Fibrillation; AVERROES, Apixaban Versus Acetylsalicylic Acid to Prevent Strokes; bid, twice daily; CAD, coronary artery disease; CNS, central nervous system; CrCl, creatinine clearance; CRNM, clinically relevant nonmajor; INR, international normalized ratio; LVEF, left ventricular ejection fraction; NVAF, nonvalvular atrial fibrillation; od, once daily; PAD, peripheral arterial disease; RE-LY, Randomized Evaluation of Long-Term Anticoagulation Therapy; ROCKET AF, Rivaroxaban Once daily, oral, direct Factor Xa inhibition Compared with vitamin K antagonism for prevention of stroke and Embolism Trial in Atrial Fibrillation; TIA, transient ischemic attack; VKA, vitamin K antagonist; od, once daily.
Oral, Direct Thrombin Inhibitors
Dabigatran
In October 2010, the FDA approved dabigatran, an oral direct thrombin inhibitor (DTI), as an agent to reduce the risk of stroke and systemic embolism in patients with NVAF. Approval was based on data from the Randomized Evaluation of Long-term anticoagulant therapY (RE-LY) study. 10
The RE-LY study evaluated the efficacy and safety of 2 different doses of dabigatran relative to warfarin in patients with AF. In total, 18 113 patients with AF were randomized to 1 of 3 arms: (1) adjusted dose warfarin, (2) dabigatran 110 mg twice daily (bid), or (3) dabigatran 150 mg bid. 31 The warfarin arm was open label but adverse events were adjudicated by reviewers blinded to treatment. Dabigatran 150 mg bid showed superiority to open-label warfarin for the primary efficacy end point of reduction of stroke or systemic embolism (1.11% vs 1.69% per year; P < .001), with a rate of major bleeding events similar to warfarin (3.11% vs 3.36% per year; P = .31).
Dabigatran 110 mg bid showed noninferiority to open-label warfarin with respect to the primary efficacy end point (1.53% vs 1.69% per year; P < .001), with a significantly lower rate of major bleeding events (2.71% vs 3.36% per year; P = .003). 31 No major toxicity was observed, although dyspepsia was more frequent with dabigatran than with warfarin (P < .001). 31 Dabigatran 150 mg was also associated with a higher rate of gastrointestinal bleeding and a significantly higher rate of MI than warfarin (0.74% vs 0.53% per year; P = .048). However, the rates of intracranial hemorrhage were significantly reduced with both doses of dabigatran compared with warfarin (0.23% and 0.30% per year for 110 mg and 150 mg dabigatran, respectively, vs 0.74% per year for warfarin; P < .001 for both). 31 The FDA has approved dabigatran to reduce the risk of stroke in patients with NVAF, using 150 mg bid in patients with creatinine clearance (CrCl) >30 mL/min and 75 mg bid in patients with CrCl 15 to 30 mL/min. There is no recommendation for dabigatran in patients with CrCl <15 mL/min or in patients on dialysis.
Since its approval, there have been a number of reports of serious bleeding events in patients taking dabigatran. The issue is under close review by regulatory authorities from several countries, including the FDA, the Japanese Ministry of Health, Labour and Welfare, and the European Medicines Agency and has led to recommended changes to the prescribing information. Throughout the treatment period all patients taking dabigatran should be evaluated for any signs or symptoms of bleeding or anemia, especially those with risk factors, such as moderate renal impairment, concomitant antiplatelet therapy, and age >75 years. In addition, pre- and on-treatment renal assessment at least once a year is recommended in patients >75 years of age and in patients of any age when a decline in renal function is suspected.
Data from the RE-LY study have been used to assess the cost-effectiveness of dabigatran versus warfarin.35 –38 Dabigatran was shown to be more cost effective than warfarin in patients ≥65 years of age who were at a high risk of stroke or intracranial hemorrhage but only when the INR control of warfarin was poor. However, because of the current high drug price of dabigatran compared with warfarin, a study conducted in the United Kingdom, has suggested that warfarin remains suitable for the majority of patients with AF. 39
Further clinical data on dabigatran will be generated in the RELY-ABLE trial, 40 an extension study in patients with AF who completed RE-LY. The study is expected to be completed in April 2013.
Oral, Direct Factor Xa Inhibitors
Rivaroxaban
In December 2011, rivaroxaban was approved as an agent to reduce the risk of stroke and systemic embolism in patients with NVAF. 11
Rivaroxaban Once daily, oral, direct Factor Xa inhibition Compared with vitamin K antagonism for prevention of stroke and Embolism Trial in Atrial Fibrillation (ROCKET AF) was a randomized, phase III, double-blind, double-dummy study designed to test the noninferiority of rivaroxaban versus adjusted-dose warfarin for the prevention of thromboembolic events in 14 264 patients with NVAF. 41 Patients must have had a prior stroke, TIA, or systemic embolism or have had ≥2 additional risk factors for stroke. Compared with RE-LY, this patient population was at a higher risk of stroke.31,41 To further increase the risk profile, the number of patients with only 2 risk factors was capped at 10%. Remaining patients required ≥3 risk factors or a prior stroke, TIA, or systemic embolism.
In the primary analysis, the primary end point of stroke or systemic embolism occurred in 188 patients in the rivaroxaban group (1.7% per year) and in 241 patients in the warfarin group (2.2% per year), establishing rivaroxaban as noninferior to warfarin (P < .001). 41 Rates of major and clinically relevant nonmajor bleeding events were similar among patients treated with rivaroxaban or warfarin (14.9% per year vs 14.5% per year, respectively), although there was a significant reduction in the rate of intracranial hemorrhage in the rivaroxaban group (0.5% per year vs 0.7% per year; P = .02). 41
Analysis of events occurring during the end-of-study transition to open-label treatment with conventional anticoagulants showed a significantly higher incidence of primary events during the first month after termination of randomized treatment in patients transitioning from rivaroxaban compared with those transitioning from warfarin (22 vs 7; P = .008). 41 During transition, the median time to reach therapeutic INR was 13 days for those previously assigned to rivaroxaban. In contrast, therapeutic INR was reached in a median of 3 days in the warfarin group, which may be because many of these patients already had a therapeutic INR during the study period. 41 Therefore, these end-of-study events may have been related to the increased difficulty in achieving the transition from blinded trial therapy to the open-label use of a VKA in the rivaroxaban arm. 41
In patients with CrCl >50 mL/min, the recommended dose of rivaroxaban is 20 mg once daily (od); for patients with CrCl 15 to 50 mL/min, the recommended dose is 15 mg od. Rivaroxaban is not recommended for use in patients with a CrCl of <15 mL/min or severe hepatic impairment (Child-Pugh C). 11
Apixaban
Two phase III clinical trials have demonstrated the efficacy of apixaban for stroke prevention in patients with NVAF. 42 ,43 Based on the results of these studies, apixaban has filed for approval for the prevention of stroke and systemic embolism in patients with NVAF in the United States. Apixaban Versus Acetylsalicylic Acid to Prevent Strokes (AVERROES) was a randomized, double-blind, superiority trial comparing apixaban with acetylsalicylic acid (ASA) in 5600 patients with AF at moderate-to-high risk of stroke and considered ineligible for VKA prophylaxis. 42 The study was stopped early because a predefined interim analysis by the independent data monitoring committee found clear evidence of a clinically important reduction in stroke and systemic embolism in patients with AF who received apixaban compared with ASA. 44
Apixaban for Reduction In STroke and Other ThromboemboLic Events in Atrial Fibrillation (ARISTOTLE) was a randomized, double-blind, noninferiority trial comparing apixaban (5 mg bid) with adjusted-dose warfarin (INR 2.0-3.0) in 18 201 patients. 43 Apixaban was superior to warfarin for the primary outcome, ischemic or hemorrhagic stroke or systemic embolism, with events occurring at a rate of 1.27% per year and 1.60% per year, respectively (P = .01). 43 Major bleeding events occurred at a rate of 2.13% per year in the apixaban group and 3.09% per year in the warfarin group (P < .001). Rates of intracranial bleeding were significantly lower in the apixaban group (0.33% per year vs 0.80% per year; P < .001).
There are currently no individual cost-effectiveness analyses for rivaroxaban or apixaban. Despite this, a recent US study evaluated the medical cost savings associated with the use of these agents and dabigatran, based on data from their respective phase III trials. 45 Excluding drug costs, the use of dabigatran, rivaroxaban, and apixaban may be associated with lower medical costs compared with warfarin, with reductions of $179, $89, and $485 per patient year, respectively. 45
Edoxaban (DU-176b)
The Effective Anticoagulation with Factor Xa Next Generation in Atrial Fibrillation–Thrombolysis In Myocardial Infarction 48 (ENGAGE-AF TIMI 48) study is a randomized, double-blind, double-dummy trial in 20 500 patients with AF at moderate-to-high risk of stroke. 46 The trial, which is due to complete in March 2012, will compare the efficacy and safety of edoxaban (30 or 60 mg od) with warfarin for a period of 24 months. 46
Other Agents
A number of oral agents are in phase II clinical development for the prevention of stroke in AF. These include the DTI AZD0837 and the direct factor Xa inhibitors betrixaban (NCT00375609) and YM150 (NCT00448214; NCT00938730).28,29,47,48
Conclusions
The association between stroke and AF, including NVAF, has a long history. Although VKAs are effective for the prevention of stroke in patients with AF, they are difficult to manage in clinical practice. In light of this, antiplatelet agents have been studied as alternatives, but they are considerably less effective for stroke prophylaxis in patients with AF.
Because as many as 90% of patients with AF could benefit from anticoagulant stroke prophylaxis, the development of agents that provide safe, effective, and well-tolerated anticoagulation is important. The novel oral anticoagulants offer an improved benefit–risk ratio and, given in fixed doses without routine monitoring, they have the potential to simplify patient care, enhance adherence, and improve outcomes.
Although dabigatran and rivaroxaban are approved for the prevention of stroke and systemic embolism in patients with NVAF, there are some concerns over their use. First, there is, as yet, no easily accessible test for the level of anticoagulation afforded by these new agents in the individual patient. Prothrombin time is not accurate and partial thromboplastin times have not proved useful with these newer agents.49,50 Second, it is important that patients do not skip doses because 1 or 2 days without these agents will theoretically remove all stroke protection. Third, whereas vitamin K and fresh frozen plasma can be used to reverse VKA anticoagulation, 51 there are no definite antidotes for the new oral anticoagulants. Prothrombin complex concentrates have been used to reverse rivaroxaban, and dialysis has been used to treat dabigatran overdose,52,53 but in most instances supportive care while waiting for drug excretion or metabolism is sufficient. In spite of these concerns, new oral anticoagulants are likely to become more important in protecting patients with paroxysmal, persistent, or permanent AF and may play a key role in extending prophylaxis to patients with stroke risk factors who are found to have occult intermittent AF on long-term monitoring. New oral anticoagulants also have the potential to extend effective prophylaxis to those currently considered ineligible for VKAs.
Footnotes
Acknowledgment
The author would like to acknowledge Richard Dobson, PhD, who provided editorial support.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Janssen Scientific Affairs, LLC, Raritan, New Jersey, funded editorial support for this article. The author received no financial support for the research, authorship, and publication of this article.