Abstract
Direct oral anticoagulants (DOACs) are valid alternative options to vitamin K antagonists due to their limited interactions with drugs or food and the fact that they do not require regular coagulation monitoring. To this regard, recent practice guidelines recommend that DOACs should be considered as first-line anticoagulant therapy for stroke prevention in patients with nonvalvular atrial fibrillation (NVAF). This review (1) outlines current international guidelines for the management of DOACs to prevent stroke in patients with NVAF, (2) outlines indications for elderly patients as well as specific settings including acute coronary syndromes and intracranial hemorrhage, and (3) offers a practical guide for the use of DOACs in neurological settings.
Introduction
Direct oral anticoagulants (DOACs) are new molecules that block the activity of one single step in the anticoagulation cascade, unlike vitamin K antagonists (VKAs) which inhibit the formation of several vitamin K-dependent coagulation factors (factors II, VII, IX, and X). Direct oral anticoagulants fall into 2 classes: the oral direct thrombin inhibitors (dabigatran) 1 and oral direct factor Xa inhibitors (rivaroxaban, apixaban, and edoxaban). 2 –4
Four large clinical trials have reported that all DOACs are at least as effective as warfarin (international normalized ratio [INR]: 2.0-3.0) for stroke prevention and similarly safe in terms of bleeding risk in patients with nonvalvular atrial fibrillation (NVAF). 1 –4 Meta-analyses of these trials showed that DOACs significantly decrease overall mortality, cardiovascular mortality, and intracranial hemorrhages (ICHs) compared to warfarin, with a trend toward less overall bleeding. 5,6 Important practical advantages of DOACs over VKAs include fixed once- or twice-daily oral dosing and periodic coagulation monitoring not required; they have few known drug interactions based on metabolism alone and substantially no food interactions. Drawbacks of DOAC use include a risk of bleeding, especially in patients older than 75 years. In fact, a clinical trial has reported that 150 mg of dabigatran twice daily can significantly increase major gastrointestinal bleeding, when compared to warfarin use. 1 Dyspepsia has also been more commonly observed with dabigatran (both 110 mg and 150 mg), resulting in high discontinuation rates. 1 Other potential drawbacks for DOACs are the lack of routine laboratory test to measure their anticoagulant effects and, for factor Xa inhibitors, the lack of an approved antidote for reversal. 7
Direct oral anticoagulants are noninferior, with better safety due to a lower associated incidence rate of ICH, when compared to VKAs. Being so, the European Society of Cardiology guidelines recommend them as been broadly preferable to VKA in the clear majority of patients with NVAF. 8 The assessment of renal function by creatinine clearance is mandatory for all DOACs, especially for patients taking dabigatran due to its high renal clearance. Therefore, renal functioning should be assessed at least annually in patients with normal renal function (creatinine clearance [CrCl] ≥ 80 mL/min) or mild (CrCl 50-79 mL/min) renal impairment, and sensibly 2 to 3 times per year in patients with moderate (ie, creatinine clearance: 30-49 mL/min) renal impairment. 8 Compliance and adherence to treatment are mandatory to achieve good outcomes, since these drugs have a relatively short half-life; patients are left without any anticoagulation protection when more than 1 dose is missed. 9 Main pharmacological properties of DOACs are summarized in Table 1.
Clinical Pharmacology of DOACs.
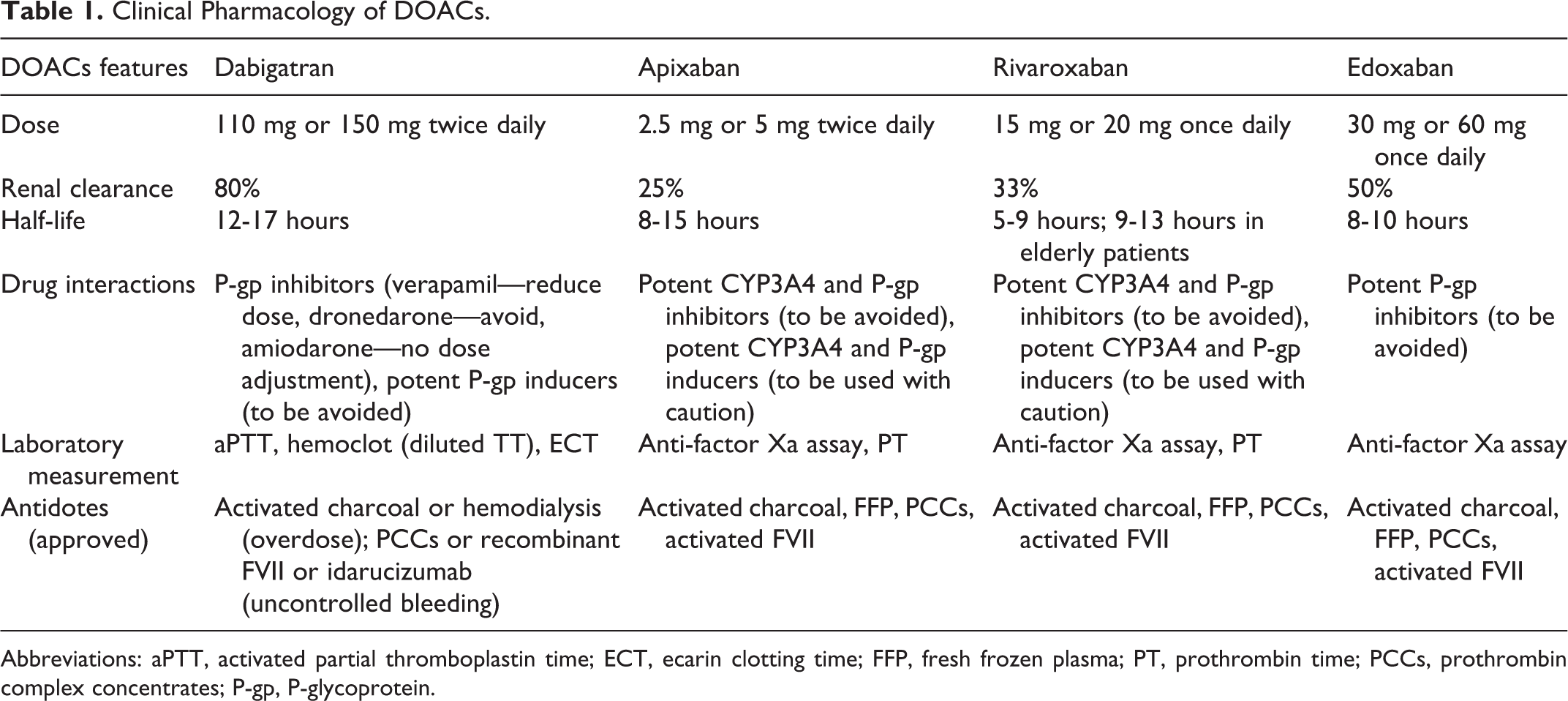
Abbreviations: aPTT, activated partial thromboplastin time; ECT, ecarin clotting time; FFP, fresh frozen plasma; PT, prothrombin time; PCCs, prothrombin complex concentrates; P-gp, P-glycoprotein.
Stroke in Patients With NVAF
When Should DOACs Be Initiated?
In case of secondary prevention of stroke in patients with NVAF, DOACs lead to a more favorable net clinical benefit than warfarin in reducing recurrence and risk of bleeding. In the case of ischemic stroke, clinical study data regarding the optimal time for initiating anticoagulation are lacking, since the pivotal studies excluded patients in the early poststroke phase. However, in the last few months, interesting data have been published, supporting the safety and efficacy of early initiation of DOACs after acute stroke. 10,11 In the absence of precise evidence-based guidelines, timing of DOACs initiation is currently determined by neurological severity (which is usually related to stroke size). Specifically, DOACs should be started as soon as possible in patients with transient ischemic attack (TIA), after few days in patients with small, nondisabling infarct, after about 1 week in those with a moderate stroke, while severe strokes with large infarcts should be treated after 2 weeks (Figure 1). This time schedule, with minor variants, was initially advocated as the “1-3-6-12-day” rule of thumb. 9 Their fast onset of action makes DOACs capable of avoiding the bridging with low-molecular-weight heparin. 8 Current knowledge about intervention strategies in early secondary prevention of ischemic stroke could be summarized and translated into a practical algorithm as in Figure 1.
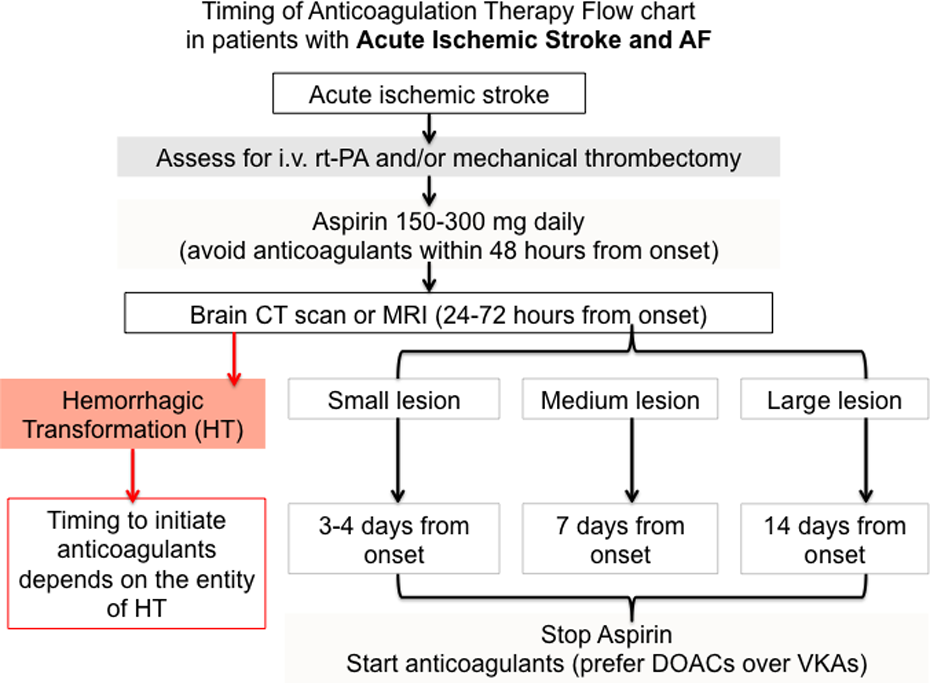
Flowchart of timing of anticoagulation therapy in patients with acute ischemic stroke and atrial fibrillation. Lesion size can be defined as follows: small, if maximum width ≤1.5 cm; medium, if involving partial arterial or border zone territories; large, if involving 1 or more complete arterial territories. Modified from Paciaroni et al. 12
Which Scores Should Be Considered?
Treatment with DOACs should be based upon thromboembolic or hemorrhagic risk assessment scores (Table 2). The CHA2DS2-VASc (see Table 2 for details and acronym expansion) score identifies patients having “truly low” stroke risk and predicts as good as and possibly better than the previous CHADS2 score for thromboembolism. 13
The CHA2DS2-VASc and the HAS-BLED Scores for the Thromboembolic and Hemorrhagic Risk Assessment, Respectively.
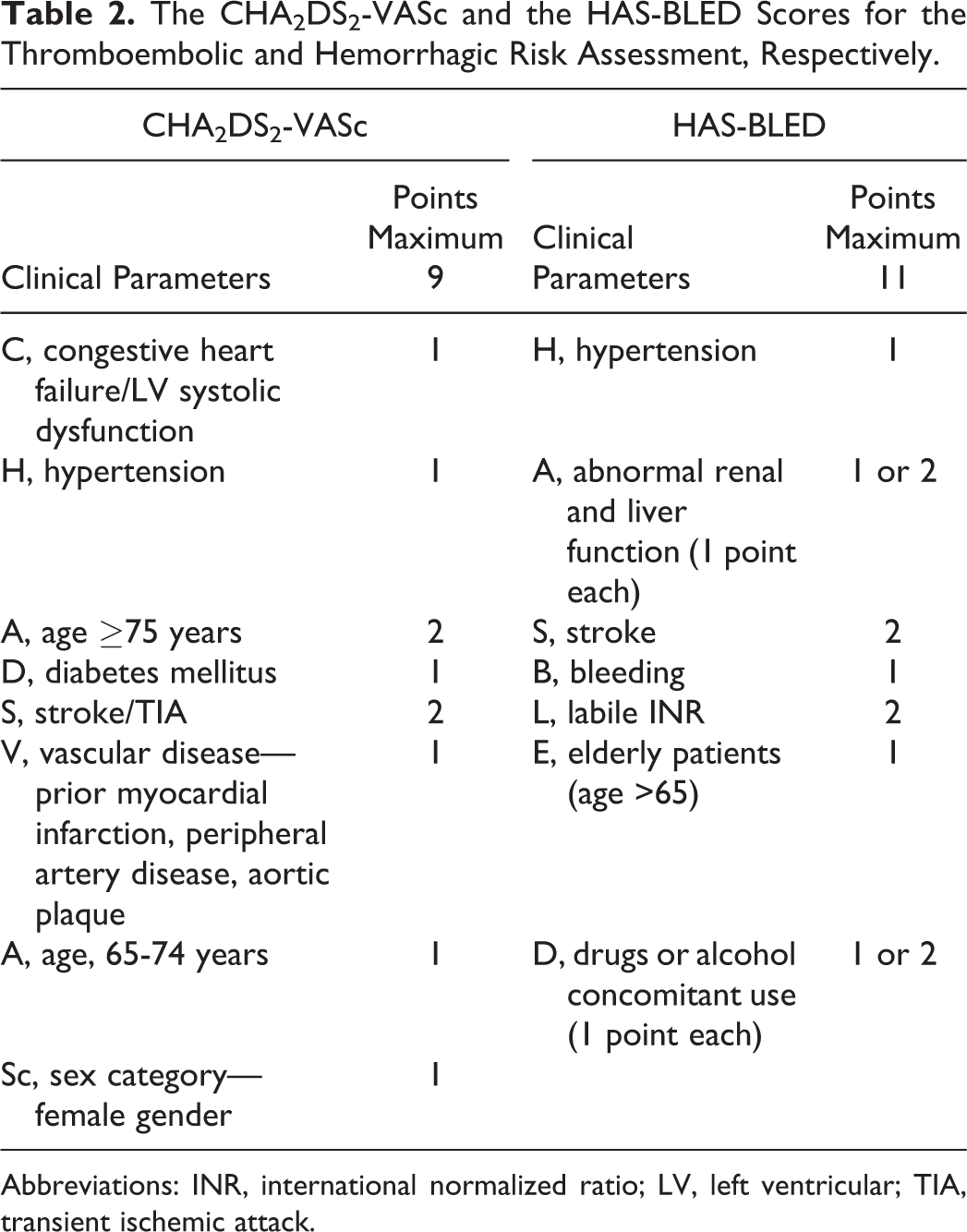
Abbreviations: INR, international normalized ratio; LV, left ventricular; TIA, transient ischemic attack.
The HAS-BLED score (see Table 2 for details and acronym expansion) is considered simple, with better predictability than other scores for bleeding risk assessment. A high HAS-BLED score itself is rarely a reason to stop oral anticoagulation. However, patients with potentially reversible risk factors for bleeding included in the score as uncontrolled blood pressure, labile INRs, and concomitant aspirin should be monitored. 13 At a CHA2DS2-VASc score of 1, apixaban and both doses of dabigatran (110 mg twice daily and 150 mg twice daily) have been reported to have a positive net clinical benefit, while in patients with CHA2DS2-VASc score ≥2, DOACs have been reported to be superior to warfarin, with a positive net clinical benefit, irrespective of bleeding risk. 13
Besides clinical scores, recently the prognostic role of the small vessel disease in patients eligible for anticoagulation has been highlighted. In this setting, brain magnetic resonance imaging (MRI) has been proposed as an additional tool to stratify hemorrhagic risk. Cerebral microbleeds are currently thought to be biomarkers of bleeding-prone small vessel pathology and thus could predict future risk of developing symptomatic intracerebral hemorrhage. The presence of cortical microbleeds or at least 5 subcortical microbleeds would discourage VKA anticoagulation, while DOACs would be acceptable due to the lower risk of intracranial bleeding. Repeating brain MRI after starting DOACs would be reasonable to assess the potential microbleeds progression and treatment safety. 14 However, to date no conclusive evidence supports the role of MRI in bleeding risk stratification.
Which Drug Interactions Are Associated With DOACs?
Although DOACs have fewer reported drug–drug interactions than warfarin, they are known to carry some concomitant drug restrictions. All DOACs are substrates of P-glycoprotein (P-gp) and are metabolized by cytochrome P450 3A4 (CYP3A4) to varying degrees, with dabigatran and edoxaban having the lowest metabolic dependence on CYP3A4. 15 Concomitant use of P-gp or CYP3A4 modulators may alter DOAC exposure: P-gp or CYP3A4 inducers decrease DOAC exposure and thereby increase the risk of venous thromboembolism, while P-gp or CYP3A4 inhibitors generally increase DOAC exposure and may lead to increased bleeding risk. The effect of drug–drug interactions on DOAC exposure varies across the DOACs. 16
From neurologists’ perspective, patients with stroke often have other disease, requiring concomitant medications, for example: Antiepileptic drugs (AEDs) are known to affect P-gp and CYP3A4 systems in different ways. Carbamazepine, levetiracetam, phenobarbital, phenytoin, and valproic acid have been found to induce P-gp activity and, thus, might decrease the effect of DOACs. Carbamazepine, oxcarbazepine, phenytoin, phenobarbital, and topiramate are CYP3A4 inducers and might decrease the effect of DOACs. These pharmacokinetic properties of carbamazepine (for P-gp and CYP3A4), oxcarbazepine, and phenytoin (for CYP3A4) are derived from human studies, while the other abovementioned AEDs have been studied only in animal or in vitro models. For other AEDs data are not available. Furthermore, to date, knowledge about the clinically relevant interactions between AEDs and DOACs is not conclusive, since only sporadic case reports or series for dabigatran (with phenobarbital and phenytoin) and rivaroxaban (with carbamazepine, oxcarbazepine, and valproic acid) are currently available.
17
Further studies and results from subgroup analyses of DOACs pivotal studies are advocated. Statin therapy may affect several steps of the blood coagulation cascade, including downregulation of tissue factor expression, increased protein C activity and factor Va inactivation, and enhanced thrombomodulin expression. The debate regarding a possible contribution of statins in the risk of hemorrhage is ongoing: high-dose atorvastatin has been associated with increased rates of hemorrhagic stroke in a large randomized controlled trial of patients with previous stroke or TIA.
18
A meta-analysis assessing the efficacy and safety of statins in patients with acute ischemic stroke or TIA could not confirm contribution.
19
However, statins are considered being protective against mortality in patients with AF, due to an observed high cardiovascular burden associated with AF.
20
In conclusion, since available data are either limited or not in agreement, other known risk factors for hemorrhagic risk should be monitored during statin therapy. Serotonin-modulating antidepressants include selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs). SSRIs and, to a lesser extent, SNRIs have been reported to increase intracerebral bleeding risks,
21
especially when coadministered with anticoagulants,
22,23
but specific data about their association with DOACs are lacking.
Elderly Patients
Patients with AF and aged > 75 years have an individual yearly stroke risk of >4% and also an increased hemorrhagic risk. In fact, age has been included in both thromboembolic and hemorrhagic risk assessment scores. 8 Anticoagulation is more effective than antiplatelet therapy in reducing the risk of stroke in elderly patients, however, with possible thromboembolic complications. 24 Compared to warfarin, DOACs have a more favorable risk–benefit profile in the elderly population due to significant reductions in stroke, ICH, and mortality. In addition, DOACs have a wide therapeutic window and only a few highly predictable interactions with other drugs. The latter aspect is especially relevant in elderly patients who are often in polypharmacy. 25
Which Factors Should Be Evaluated?
Some cautions should be exercised when treating elderly patients with elevated risk of bleeding. It is important to define and monitor all known risk factors which may increase the risk of hemorrhages in the elderly patients, such as high blood pressure, concomitant use of nonsteroidal anti-inflammatory drugs, and alcohol abuse. 24
Furthermore, thrombocytopenia and anemia are frequently encountered in elderly patients, and anticoagulation is generally contraindicated when platelet count is lower than 50 000/dL, and when it ranges between 50 000 and 100 000/dL. Anemia predicts thromboembolic events, bleeding complications, and mortality in patients with AF but is not per se a contraindication for starting anticoagulation; strict control and follow-up are advisable. 24
Renal impairment is also common in the elderly patients, especially in those with permanent AF, as it may worsen both thromboembolic and hemorrhagic risk outcomes. However, kidney dysfunction does not generally contraindicate anticoagulation. 26,27 Concerning DOACs, renal function should be assessed prior to prescription. Dabigatran is contraindicated in patients with creatinine clearance <30 mL/min, while rivaroxaban and edoxaban are not recommended for patients with creatinine clearance <15 mL/min. Only apixaban has been recently approved for use in patients with end-stage renal dysfunction and a need for hemodialysis (2.5 mg twice daily if body weight ≤60 kg or older than 80 years). 28 According to the currently available data, the other DOACs are not suitable for patients undergoing hemodialysis. 9,24,29 Direct oral anticoagulants can safely be used for patients with moderate renal dysfunction with dose adjustment, while in patients with mild impairment or normal renal function, no dose adjustment is needed.
Body weight is another crucial factor to consider because of its relation to drug distribution volumes. The knowledge of using DOACs in patients with low (<50 kg) or extremely high (>150 kg) body weight is currently limited; however, according to the published trials patients with extremely high body weight seemed to have similar outcome measures. 30 A recent meta-analysis of NVAF and venous thromboembolism prevention reported that low body weight was associated with an increased risk of thromboembolic events, while high body weight did not significantly differ in terms of outcome measures. This finding is apparently paradoxical, since low body weight would suggest an increased concentration of anticoagulants but could be potentially explained by underlying prothrombotic conditions such as chronic illnesses and malignancies. In addition, bleeding outcomes were similar among weight subgroups. Thus, current evidence does not support further dose adjustment of DOACs in patients with extremely high body weight. 31
Although frequent falls have been associated with an increased risk of ICH and mortality in anticoagulated patients, the overall risk is low and particularly lower than the risk of stroke in elderly patients with AF. For instance, in patients with a high risk of frequent falls and a CHADS2 score ≥3, anticoagulation benefits have been reported to be higher than the risk of ICH. However, in patients with posttraumatic ICH, anticoagulants should be stopped. In this subgroup, DOACs should be preferred to warfarin because of their lower risk of ICH. 5,6 To further reduce ICH during treatment, other risk factors need to be evaluated, including concomitant therapy, orthostatic hypotension, and visual impairment. Only when these factors are corrected, can anticoagulation be prescribed. 24
Dementia
Neurologists often have to deal with patients having dementia. The association of AF and dementia usually leads to a highly complex setting, due to the many clinically relevant items to consider, as summarized in Figure 2. In fact, dementia can result from several diseases, which are associated with distinct ischemic and hemorrhagic risks, different clinical expressions, comorbidities, concomitant medications, and potential adverse events.
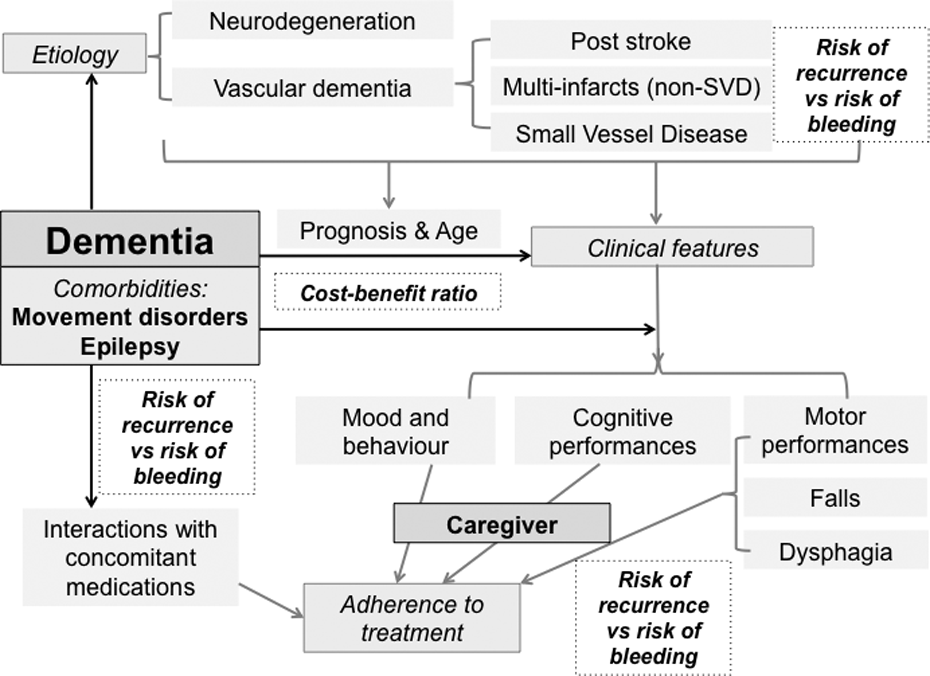
Factors to be considered in the prescription of direct oral anticoagulants (DOACs) for patients with dementia.
Generally, in patients with dementia, DOACs should be preferred over VKA due to the higher manageability and the lower risk of major bleeding, which is also the case in patients with vascular dementia due to small vessel disease. 24 However, adherence to DOACs is of major importance due to their short half-lives. To avoid missing or doubling doses, patients with dementia should be supervised by caregivers while taking medications.
To ensure the best adherence to treatment, bearing in mind cost–benefit balance, and before prescribing anticoagulants, neurologists have to carefully evaluate patients with dementia and focus on their cognitive and mental status, gait performances, and global disability and prognosis, to assess the kind of supervision may be required for the patients, and discuss the features of the treatment with caregivers.
During DOACs Therapy
What Should Be Done in the Case of Stroke?
When a recurrent stroke occurs during anticoagulant therapy, it is fundamental to determine its origin so to choose the most appropriate intervention. If revascularization is required in patients with extracranial large artery atherosclerotic infarction, endarterectomy is generally preferred to stenting to avoid dual antiplatelet therapy.
32
Embolism from a cardiac source may require estimating the time of therapeutic range and identifying causal factors for stroke recurrence. Therapeutic shift from warfarin to DOACs may be advisable in these cases; especially with either apixaban or rivaroxaban in patients with prior stroke.
32
Lacunar infarcts are expression of small vessel disease, thus require assessing the presence of hypertension, dyslipidemia, and diabetes mellitus to ameliorate their pharmacological treatment.
32
For other determined causes, such as dissection, hypercoagulable states, or sickle cell disease, current guidelines recommend specific management strategies.
32
Thrombolytic therapy with recombinant tissue plasminogen activator (rt-PA), should be started within a 4.5-hour time window from onset of stroke symptoms, and is not recommended for patients on anticoagulants. As the plasma half-life of DOACs ranges between 8 and 17 hours, thrombolytic therapy should be discouraged when DOACs are administrated within the last 24 to 48 hours, 33 unless rapid tests are available for checking anticoagulation status. Thrombolysis therapy should not be administered to patients on DOACs when laboratory assessed parameters including prolonged activated partial thromboplastin time (for dabigatran) and PT (for factor Xa inhibitors) indicate that the patient is in an anticoagulation state. 9 If DOACs have been administered within the last 48 hours and coagulation tests are not available or their results are abnormal, mechanical recanalization of occluded vessels may be considered as an alternative treatment. 9
Recently, thresholds of DOACs plasma levels have been proposed to supplement routine coagulation tests for any decision regarding the administration of thrombolysis when the last intake of DOACs is unknown. In a small study, the threshold levels were defined as follows: 62 ng/mL for dabigatran, 91 ng/mL for rivaroxaban, and 40 ng/mL for apixaban. Further studies are needed to confirm these results. 35
As discussed, to date dabigatran is the only DOAC having a specific antidote: idarucizumab, a monoclonal antibody fragment that binds with high-affinity dabigatran. This molecule has been recently approved by the US Food and Drug Administration (FDA) for reversing the anticoagulant effects of dabigatran in situations of emergency surgery/urgent procedures or life-threatening/uncontrolled bleeding. 16 Recently, an expert opinion published by Diener et al suggested and provided guidance on the use of this reversal agent followed by rt-PA and/or thrombectomy in patients with acute ischemic stroke who were pretreated with dabigatran. 34 Factor Xa inhibitors’ antagonists are currently under investigation (ciraparantag and andexanet alfa) and/or development. If these drugs are effectively approved as antidotes also patients experiencing ischemic stroke while on treatment with rivaroxaban, apixaban, or edoxaban will potentially be treated with reversal agents prior to intravenous thrombolysis (Figure 3).
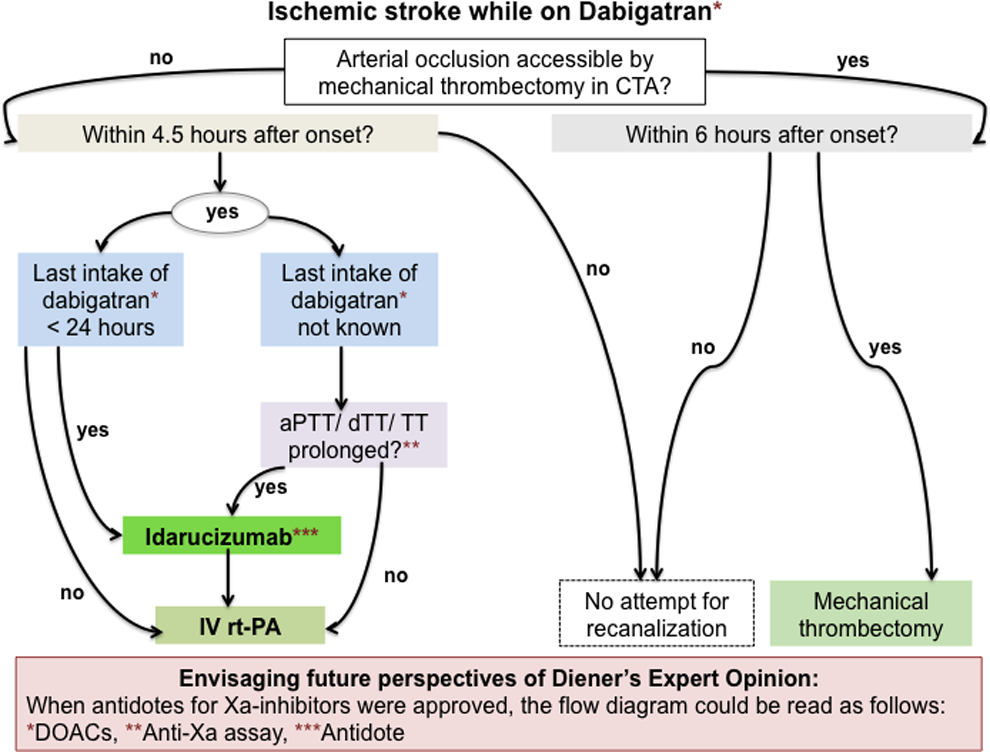
Extending Diener’s expert opinion for emergency treatment of patients with acute ischemic stroke while taking dabigatran according to future perspectives. Modified from Diener et al. 34
What Should Be Done in the Case of Acute Coronary Syndrome?
The effect of DOACs in patient with acute coronary syndrome (ACS) and AF has not been evaluated yet, since patients with AF who require oral anticoagulation have been systematically excluded from recent ACS trials, and patients with recent ACS have been excluded from phase III stroke prevention trials. Two randomized clinical trials are currently ongoing to address the safety of DOACs in terms of bleeding in those patients undergoing percutaneous coronary intervention (PCI) in association with single or dual antiplatelet therapy (NCT01830543 and RE_DUAL PCI). 36
In general, patients with NVAF having ACS or undergoing PCI, stroke and bleeding risk assessments must be carried out using the validated scores (CHA2DS2-VASc score and HAS-BLED score). 36
In an ACS setting, triple therapy composed of dual antiplatelet therapy and DOACs is associated with at least a double risk of major bleeding, as is the case with combining any OAC with antiplatelet therapy. Data regarding this elevated risk of bleeding are currently limited, but it seems reasonable to continue an existing OAC (ie, whether VKA or DOACs). 36 Guidelines recommend that the period of triple therapy composed of OACs and dual antiplatelet therapy, should be as short as possible, followed by an OAC plus a single antiplatelet therapy (preferably clopidogrel 75 mg/d or aspirin 75-100 mg/d). The duration of triple therapy should be dependent on acute versus elective procedures, bleeding risk, and type of stent. When a DOAC is used in combination with clopidogrel and/or low-dose aspirin, the lower tested dose may be considered for stroke prevention in AF (ie, dabigatran 110 mg twice daily, rivaroxaban 15 mg once a day, or apixaban 2.5 mg twice daily). 33 Novel P2Y12 receptor inhibitors (prasugrel or ticagrelor) are contraindicated in this setting for their associated bleeding risk. 36
In patients undergoing PCI, general measures can be applied to reduce the risk of bleeding, such as using low doses of aspirin (75-100 mg once a day), preferring clopidogrel to ticagrelor or prasugrel, choosing to use bare-metal stents or new generation stents to reduce the duration of triple therapy, and using the radial approach to minimize the risk of access site bleeding. 36
In patients with stable coronary artery disease and AF undergoing PCI triple therapy (OAC, aspirin 75-100 mg daily, and clopidogrel 75 mg daily) should be given a minimum of 4 weeks (and no longer than 6 months) after PCI; after which dual therapy with OAC (whether a DOAC or a VKA) and clopidogrel 75 mg/d (or alternatively, aspirin 75-100 mg/d) should be continued for up to 12 months. In the case of high bleeding risk (HAS-BLED > 3), a dual therapy consisting of OAC (a DOAC or a VKA) and clopidogrel 75 mg/d should be considered over triple therapy in the 4 weeks after PCI. 36
What Should Be Done in the Case of ICH?
Anticoagulation therapy may increase the risk of anticoagulant-related ICH. In the RE-LY trial, the annual rates of ICH were 0.74% for warfarin, 0.30% for dabigatran 150 mg, and 0.23% for dabigatran 110 mg. Whereas the RE-COVER trial, which compared dabigatran and warfarin in acute venous thromboembolism, reported no incidence of ICH during 6 months of therapy, ROCKET-AF reported that the ICH incidence was 0.49% for rivaroxaban compared to 0.74% for warfarin. 37 A recent prospective observational study reported that DOAC-ICH was associated with smaller ICH volume and better clinical outcome, when compared to warfarin-ICH. 38 However, these findings have not been confirmed by further observational studies. The clinical course of ICH while on DOACs appears to be similar to VKA, with frequent hematoma expansion (independently of treatment with reversal agents), poor clinical outcome, and high mortality. 39 –41
Approved reversal agents are not currently available for reversing the anticoagulant effects of rivaroxaban, apixaban, or edoxaban. 16 As already mentioned, dabigatran can be specifically reversed by its antidote idarucizumab. In a clinical trial including 90 patients on dabigatran who had serious bleeding or required an urgent invasive procedure, idarucizumab was seen to completely reverse the anticoagulant effect of dabigatran within minutes. 42 Idarucizumab is approved by FDA for reversing dabigatran effects in situations of emergency surgery/urgent procedures or life-threatening/uncontrolled bleeding. 16
In the case of emergency or serious bleeding, for the other DOACs, nonspecific reversal approaches are available. Activated charcoal may reduce the absorptions of rivaroxaban and apixaban if administered 1 to 2 hours after ingestion. Prothrombin complex concentrate (PCC) with 4 factors has demonstrated to be effective in reversing the anticoagulant effect of rivaroxaban, apixaban, and edoxaban, as determined by measurements of prothrombin time (PT) or thrombin generation ETP, 16 and current expert recommendations suggest its use although no consensus on the matter has been established. 41,43
Ciraparantag binds directly to factor Xa agent (in particular edoxaban) via hydrogen bonds from or to various parts of the molecule. This mechanism of action blocks the anticoagulant effect of edoxaban and restores the PT in vitro. Similarly, andexanet alfa is a decoy of factor Xa, which binds to factor Xa inhibitors without triggering the prothrombotic activity. Thus, andexanet can quickly reverses the anticoagulant activity of apixaban and rivaroxaban in healthy patients without any toxic effects. 44
Neurosurgical evaluation is always required in patients with anticoagulant-related intracranial bleeding. In conditions that can be surgically amended, anticoagulation should be resumed as soon as possible, preferring DOACs over VKAs.
In anticoagulated patients experiencing ICH, the risks of hemorrhagic and thromboembolic complications may occur with similar frequencies, although their temporal risks tend to differ. Hemorrhagic complications have been predominantly observed within 72 hours of presentation, whereas thromboembolic complications have been predominantly recorded 3 to 5 days after medical presentation. 45 Early anticoagulant restarting may be optimal; however, individualized evaluations need to be carried out for all patients prior to selecting anticoagulant resumption time and intensity. 46 In addition, hypertension must be monitored as well as diabetes and other risk factors that are known to increase hemorrhagic risk. Hematologist’s consultation is advisable to identify malignancies or other potential causes of hemorrhagic diathesis. Furthermore, anticoagulation treatment is not recommended whenever hemorrhages are located at the lobar site or when amyloid angiopathy is diagnosed. In borderline situations, imaging techniques (MRI) may be helpful for investigating the presence of burden of leukoaraiosis, microvessel disease, microbleeds, and cortical siderosis. 14 In fact, these conditions are known to increase the hemorrhagic risk, and therefore anticoagulant therapy is generally contraindicated in this setting. When anticoagulation is contraindicated, surgical ablation or percutaneous left atrial appendage closure should be considered as they can lead to a reduction in thromboembolic events (Figure 4).
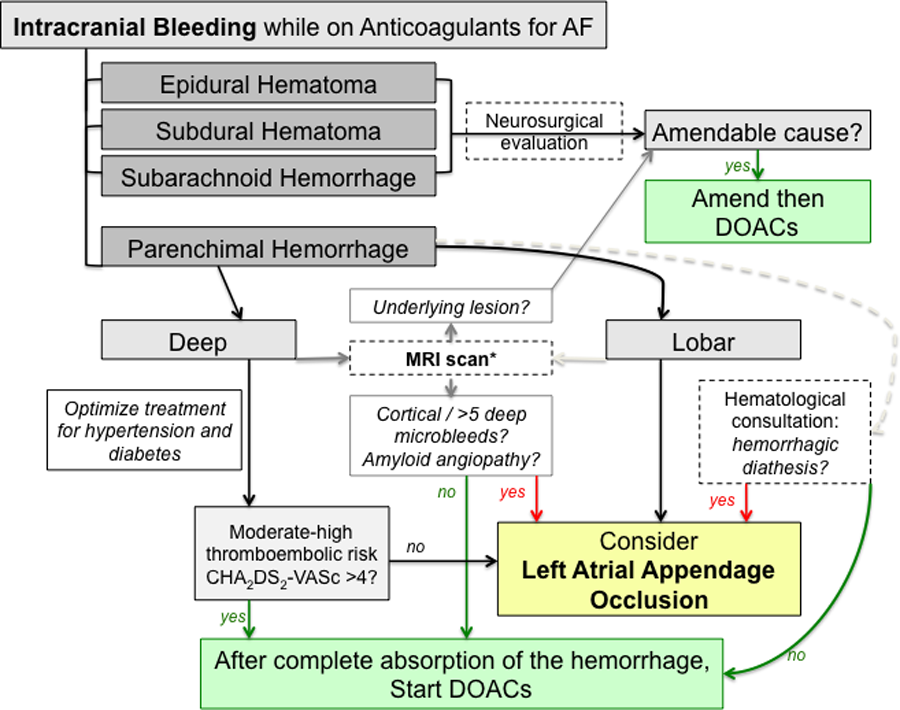
Decision-making process in anticoagulants resumption after anticoagulant-related intracranial bleeding. Modified from Becattini et al. 46
What Should Be Done if a Lumbar Puncture Is Needed?
Among neurological settings potentially affected by treatment with DOACs, the urgent need for invasive diagnostic testing has to be mentioned, the lumbar puncture above all. Similar to invasive procedures such as spinal and epidural anesthesia, a lumbar puncture requires complete hemostatic function and has to be considered under the “high risk of bleeding” category of interventions.
Due to the short half-lives of DOACs, especially in patients with normal renal function, restoration of hemostasis can be expected within 12 to 24 hours after the last dose intake. If an invasive procedure is required, it should be postponed if possible at least for 12 hours, but ideally 24 hours after the last DOAC dose intake to reduce the risk of bleeding. However, in case of procedures that carry a high risk of bleeding, including lumbar puncture, current guidelines recommend taking the last DOAC prior to 48 hours. 47
The availability of the antidote idarucizumab enables a quick reversal of the anticoagulant effect of dabigatran in patients requiring emergency diagnostic lumbar puncture, due, for example, to a suspicion of central nervous system infection. Supportive evidence from real-life experience is provided by some recent reports about patients treated with dabigatran, who underwent safe and successful emergency lumbar puncture after idarucizumab administration. 48,49 In the VKAs era, 50 the administration of PCC had been proposed as a valid reversal strategy in patients requiring urgent lumbar puncture; however, the same strategy to date has never been reported in patients treated with DOACs.
For procedures with immediate and complete hemostasis, including atraumatic spinal/epidural anesthesia or clean lumbar puncture (ie, nonbloody tap), treatment with DOACs can be resumed 6 to 8 hours after the intervention. 47
Conclusion
Direct oral anticoagulants are a valid and reliable option for the prevention of stroke and management of thromboembolic disease in patients with NVAF. Given their limited and well-established interactions with food and drugs and their wide therapeutic windows, DOACs do not require routine monitoring which is often a significant barrier to many patients who require anticoagulant therapy. Furthermore, DOACs are associated with reductions in ICH, therein favoring their use in neurodegenerative diseases and in elderly patients.
Footnotes
Authors’ Note
The authors thank the unconditional grant from Bristol-Myers Squibb and Pfizer Alliance.
Acknowledgments
The authors thank Elisa Sala, PhD, independent medical writer, for her medical editorial assistance with our report.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by unconditional grant from Bristol-Myers Squibb and Pfizer Alliance.
