Abstract
Background
Direct oral anticoagulants (DOACs) have increasingly become an alternative to warfarin in atrial fibrillation (AF) patients. Nonetheless, data on the effectiveness and safety of DOACs in periprocedural of catheter ablation (CA) in real-world practice was relatively rare.
Methods and Results
3385 AF patients underwent initial CA and never used oral anticoagulant before enrollment between April 2013 and December 2018 were involved from China Atrial Fibrillation (China-AF) Registry. Warfarin, rivaroxaban and dabigatran were used in 1896 (56.0%), 718 (21.2%), and 771 (22.8%) patients, respectively. Propensity score matching was used to balance covariates across study groups. No significant differences were observed in rivaroxaban-warfarin, dabigatran-warfarin and dabigatran-rivaroxaban cohort for thromboembolic (TE) and major bleeding (MB) incidence. Similar results were also revealed in low-dose rivaroxaban (RLD)-warfarin, low-dose dabigatran (DLD)-warfarin and DLD-RLD cohort. However, the risk of non-MB was higher not only on standard-dose of rivaroxaban but also on RLD when compared with warfarin and with DLD, respectively.
Conclusions
In this study, the incidence of TE and MB were both comparable in standard- or low-dose DOACs versus warfarin and between the two DOACs, whereas the risk of non-MB was higher in rivaroxaban than in warfarin and in RLD than in DLD.
Introduction
Catheter ablation (CA) is an established rhythm-control strategy for non-valvular atrial fibrillation (NVAF), particularly for symptomatic patients, 1 while carrying rare but disastrous complications of thromboembolic (TE) and major bleeding (MB) events.2, 3 Proper usage of periprocedural anticoagulation is critical to reduce the risk of these events. Direct oral anticoagulants (DOACs) have increasingly become an alternative to warfarin for stroke prevention in patients with NVAF.4, 5 Nonetheless, concerns about their safety remain given the lack of a reliable laboratory marker of DOAC anticoagulant activity.6, 7 Furthermore, East Asians are inclined to experience a higher risk of MB with oral anticoagulant (OAC) treatment than non-East Asians.8–10 To reduce the risk of MB, low-dose DOACs, generally rivaroxaban 15 mg or 10 mg once daily or dabigatran 110 mg twice daily, are preferred in Asian real-world clinical practice, even in those without a high risk of bleeding.11–13 However, the benefit and harm of low-dose DOACs among Asian patients with NVAF have not been fully evaluated up to now.
Previous studies mainly focused on the periprocedural outcomes of DOACs versus warfarin in NVAF,14, 15 and few data are available for Chinese patients. Further investigation is needed to determine which OAC is better than another, and whether low-dose DOACs can provide effective and safe protection during the periprocedural period of CA in Chinese NVAF patients. The current study aimed to evaluate the effectiveness and safety of rivaroxaban and dabigatran in standard-dose and low-dose patterns compared with warfarin in NVAF patients who were scheduled for first time CA, using data from the China Atrial Fibrillation (China-AF) Registry.
Methods
Study population
The China-AF Registry is a prospective, multicenter, hospital-based, ongoing registry study with consecutive AF patients, conducted in 31 tertiary and non-tertiary hospitals in Beijing, China, with 18 hospitals providing AF ablation. Details of the study design have been described previously. 16 The Human Research Ethics Committee at Beijing Anzhen Hospital approved this study and the ethics review boards at individual participating hospitals approved their participation. Data of 14 716 patients registered from April 2013 to December 2018 (since dabigatran and rivaroxaban were available in the Chinese market) were used for this retrospective analysis. All patients provided written consent to participate in the registry. In total, 8829 patients aged ≥18 years and admitted for CA because of AF were eligible (Figure 1). We excluded patients with valvular AF (moderate and severe mitral stenosis or mechanical heart valves), those with <3 months of follow-up, and those who had previous CA of AF. To focus on patients who were only prescribed OAC because of AF, patients with a history of deep venous thrombosis or pulmonary embolism were excluded. We also excluded those who had ever used any OAC previously to ensure a pure OAC-naïve cohort, and excluded those who had taken more than one OAC, or used OACs other than warfarin, rivaroxaban, or dabigatran during the perioperative period. The patients were followed up by trained staff for 3 months after the ablation in the outpatient department, or by telephone when necessary.
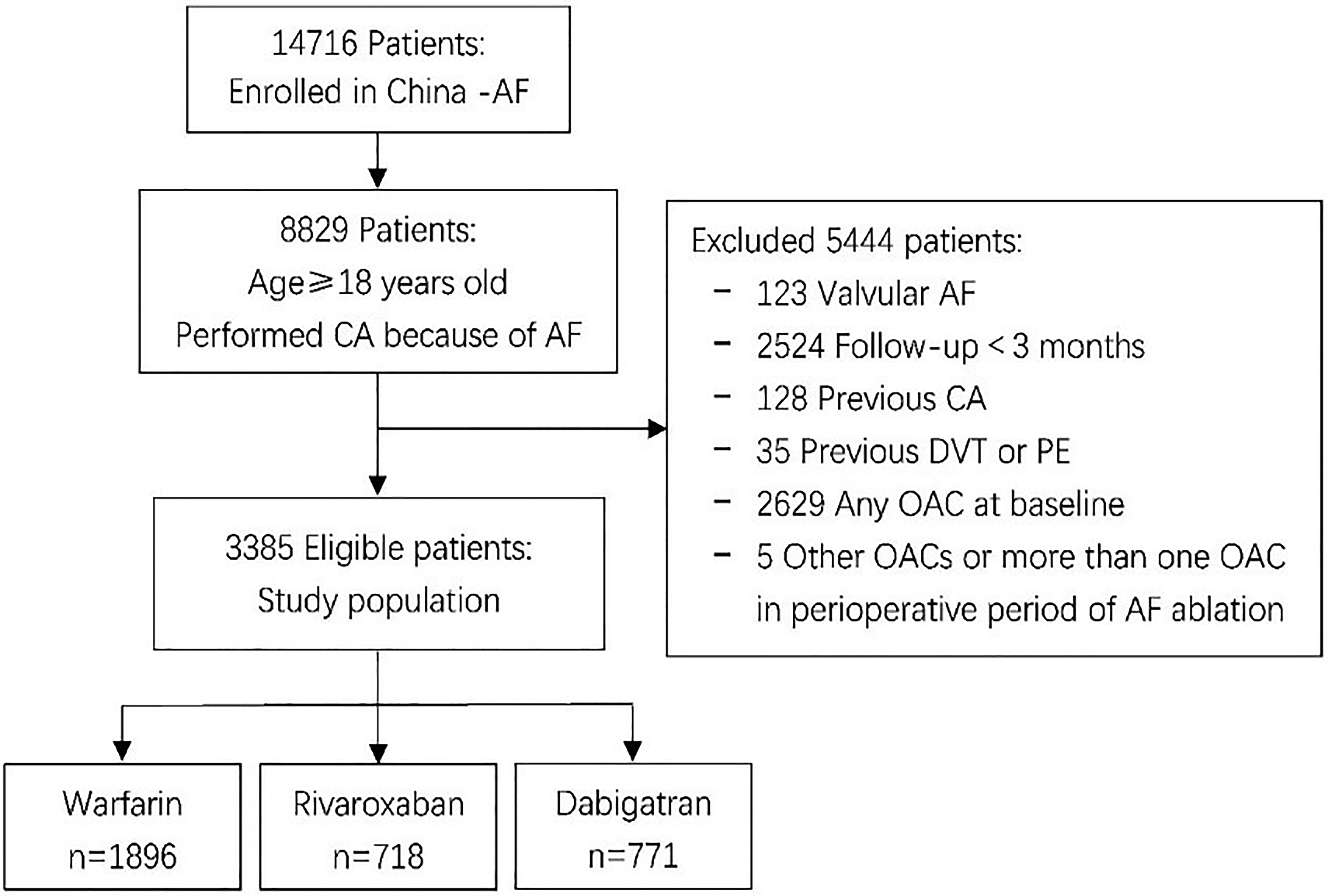
Flowchart of the study. Abbreviations: AF, atrial fibrillation; CA, catheter ablation; DVT, deep venous thrombosis; OAC, oral anticoagulant; PE, pulmonary embolism.
Ablation Protocol
Radiofrequency energy was typically used; however, cryoablation energy was also included in our study. CA can proceed only when no left atrial thrombi have been detected by pre-ablation transesophageal echocardiography (TEE) within 48 h. During the procedure, patients received intravenous unfractionated heparin interrupted to achieve a target activated clotting time (ACT) of 300 to 350 s. The detailed ablation strategy has been described previously. 17 Circumferential pulmonary vein isolation was achieved in all of the included patients. Left atrial linear ablation and/or complex fractionated atrial electrograms were also performed for patients with non-paroxysmal AF.
Anticoagulation Strategy
After admission, the type of OAC that determined the treatment group and the timing of the initiation of the OAC was based on the discretion of the treating physician. OAC therapy could be started before, after, or on the procedure day (only in the warfarin group). If OAC therapy was started before the procedure, warfarin was uninterrupted throughout the periprocedural period, while therapy with DOACs was interrupted before the CA (at least 1-2 doses were put on hold before ablation). The dosage of warfarin was adjusted to achieve a therapeutic international normalized ratio (INR) value in the range of 2.0–3.0. Low-molecular-weight heparin (LMWH) was used as a bridge for patients treated with warfarin whose INR was <2.0 and for patients whose rivaroxaban or dabigatran was discontinued at least 24 h before ablation. LMWH was also used in patients with a CHA2DS2-VASc score ≥2 and who had not used an OAC before the procedure. Rivaroxaban was administered at a dosage of 10 mg, 15 mg, or 20 mg once daily, and dabigatran was at a dosage of 150 mg or 110 mg twice daily. After CA, OAC treatment was continued for 3 months continuously and the subsequent anticoagulation regimen was also determined by the treating physician.
Study Outcomes
The effectiveness outcomes were TE complications, including ischemic stroke, transient ischemic attack (TIA), and peripheral arterial thromboembolism (PT). Definitions of the effectiveness outcomes were consistent with the 2017 HRS/EHRA/ECAS/APHRS/SOLAECE expert consensus statement on catheter and surgical ablation of atrial fibrillation. 18 The safety outcome was a composite of clinically relevant MB and non-MB events. An event was considered to be a MB event if either one of the definitions of the International Society on Thrombosis and Haemostasis (ISTH) for a major bleed, or the Global Use of Strategies to Open Occluded Coronary Arteries (GUSTO) for a severe/life-threatening bleed was satisfied.16, 19, 20 Non-MB events were defined as clinical bleeding events that did not fulfill ISTH or GUSTO criteria for MB events.
Definitions
The estimated glomerular filtration rate (eGFR) was calculated with the modified equation for Chinese patients with chronic kidney disease. 21 The CHA2DS2-VASc score was calculated by giving 1 point to each patient for a history of congestive heart failure, hypertension, diabetes mellitus, vascular disease, female sex, and age 65 to 74 years, and 2 points for previous stroke or transient ischemic attack or thromboembolism and age ≥75. 22 The modified HAS-BLED score was calculated by giving 1 point each for hypertension, abnormal liver function, abnormal renal function, stroke, bleeding history, advanced age (>65 years), drugs, eg, antiplatelet or nonsteroidal anti-inflammatory drugs (NSAIDs) and alcohol use or abuse. 23
Statistical Analysis
Continuous variables were presented as mean ± standard deviation (SD) and were analyzed by paired t-test or Wilcoxon rank-sum test. Categorical variables were presented as numbers and percentages (%), and were analyzed by Pearson's chi-square test or Fisher's exact test as appropriate. The Shapiro-Wilk test was used to check the normality distribution of data.
The baseline characteristics between the different OAC groups were compared. To minimize the imbalance of baseline characteristics between study patients with different OACs, three comparable pairs (rivaroxaban vs warfarin, dabigatran vs warfarin, and rivaroxaban vs dabigatran) were matched at a ratio of 1:1 through propensity score matching (PSM).24, 25 Propensity scores were generated by multivariable logistic regression, incorporating variates related to the outcome but not to the treatment allocated, 25 including baseline demographic characteristics (age, sex, body mass index), comorbidities, CHA2DS2-VASc score, modified HAS-BLED score, and concomitant medications (Table 1). Because the INR stability was not available for every patient, we used a modified HAS-BLED score instead of the HAS-BLED score. The nearest neighbor matching method with a caliper of 0.01 without replacement was used to match the patients. The balance of covariates was checked based on standardized mean differences with a threshold of 10% (a standardized mean difference of less than 0.1 was considered well balanced). 24 The distribution of the propensity scores was reviewed for each matched population to ensure balance.
Baseline Characteristics of Study Population (After PSM).

Data are shown as mean ± SD or n/N (%). Abbreviations: AF, atrial fibrillation; ACEI, angiotensin-converting enzyme inhibitor; ARBs, angiotensin-receptor blockers; BMI, body mass index; CAD, coronary artery disease; CHF, chronic heart failure; DCM, dilated cardiomyopathy; DM, diabetes mellitus; DVT, deep venous thrombosis; eGFR, estimated glomerular filtration rate; HCM, hypertrophic cardiomyopathy; MI, myocardial infarction; PAD, peripheral artery disease; PSM, propensity score matching; PT, peripheral arterial thromboembolism; SMD, standardized mean differences; TIA, transient ischemic attack; a other CAD includes history of percutaneous coronary intervention, history of coronary artery bypass graft and ≥50% coronary stenosis was confirmed by coronary angiography; b Anti-arrhythmic drug refers to amiodarone, propafenone and sotalol; c CCBs refers to non-dihydropyridine calcium channel blockers.
Because all variables were well balanced after PSM except for oral anticoagulation, we used univariable logistic regression analysis for each PSM matched cohort to estimate the odds ratio (OR) of each effectiveness and safety outcome with 95% confidence intervals (CIs). The χ2 test (≥5 events) and Fisher's exact test (<5 events) were performed for intergroup comparison of the incidence.
In the current study, most patients on DOACs were using a low dose (rivaroxaban 10 mg or 15 mg once daily, dabigatran 110 mg twice daily). Because dose-based interaction effects may be observed with MB and TE events, subgroup analyses were performed in patients taking a low dose of DOACs. Patients were reassigned to three subgroup cohorts [low-dose rivaroxaban (RLD) versus warfarin, low-dose dabigatran (DLD) versus warfarin, and RLD versus DLD], and the analytic steps previously described were repeated.
Statistical analyses were performed using SAS version 9.4 (SAS Institute, Cary, NC, USA). A 2-tailed p-value of <.05 was considered statistically significant.
Results
Patient Characteristics
In total, 3385 eligible patients were enrolled in our study, including 1896 (56.0%) patients on warfarin, 718 (21.2%) patients on rivaroxaban, and 771 (22.8%) patients on dabigatran (Figure 1). Pre-matched data (Supplementary Table 1) showed that the mean age of the total patient cohort was 59.5 years, about two-thirds (67%) were under 65 years, women accounted for 33.7%, and paroxysmal AF made up of 71.7% of the overall population. The average CHA2DS2-VASc scores in the warfarin, rivaroxaban and dabigatran groups were 1.81, 1.75, and 1.64, respectively, and the average modified HAS-BLED scores were 1.39, 1.16, and 1.21, respectively.
After PSM, there were 628 matched pairs in the rivaroxaban-warfarin cohort, 757 matched pairs in the dabigatran-warfarin cohort, and 537 matched pairs in the rivaroxaban-dabigatran cohort. All covariates were well balanced in each cohort (Table 1).
Clinical Outcomes
Effectiveness Outcomes
Before PSM, 26 TE events (0.77%) occurred in the current study: 11 (0.58%) in the warfarin group, 6 (0.84%) in the rivaroxaban group, and 9 (1.17%) in the dabigatran group (Supplementary Table 2).
The incidence rates of TE for the three matched cohorts after PSM are shown in Figure 2(A)-(C). No significant differences were observed in the TE incidence rate in the rivaroxaban-warfarin cohort (OR: 2.01, 95% CI: 0.50-8.07), the dabigatran-warfarin cohort (OR: 4.54, 95% CI: 0.98-21.09), or the rivaroxaban-dabigatran cohort (OR: 2.35, 95% CI: 0.60-9.14).

Odds ratios (95% ci) of TE, MB and non-MB events in rivaroxaban versus warfarin PSM cohorts (A), dabigatran versus warfarin PSM cohorts (B) and rivaroxaban versus dabigatran PSM cohorts (C). Abbreviations: CI, confidence interval; MB, major bleeding; Non-MB, non-major bleeding; OR, odds ratio; PSM, propensity score matching; TE, thromboembolism.
Safety Outcomes
Before PSM, there were 20 MB events (0.59%), consisting of 14 (0.74%) in the warfarin group and 3 each in the rivaroxaban (0.42%) and dabigatran groups (0.39%). Some non-MB complications were observed (182 or 5.38%), including 94 (4.96%), 52 (7.24%), and 36 (4.67%) in the warfarin, rivaroxaban, and dabigatran groups, respectively (Supplementary Table 2).
Figure 2(A)-(C) display the incidence rates of MB and non-MB events in the three PSM cohorts. No statistically significant differences in the MB incidence rate were revealed when the three OACs were compared with each other in the rivaroxaban-warfarin cohort (OR: 0.75, 95% CI: 0.17-3.36), the dabigatran-warfarin cohort (OR: 1.50, 95% CI: 0.25-9.01) or the dabigatran-rivaroxaban cohort (OR: 1.00, 95% CI: 0.20-4.98). When compared with warfarin, rivaroxaban was associated with a significantly higher risk of non-MB events (OR: 1.92, 95% CI: 1.17-3.13)
Subgroup Analysis
A total of 77.9% (n = 559) rivaroxaban patients and 95.2% (n = 734) dabigatran patients received the low dose of rivaroxaban (15 mg or 10 mg once daily) or dabigatran (110 mg twice daily). In the subgroup analysis of patients on low-dose DOACs and warfarin, baseline characteristics were similar in the RLD-warfarin cohort, the DLD-warfarin cohort, and the DLD-RLD cohort (Supplementary Table 3).
The PSM incidence rates of TE, MB, and non-MB events are listed in Figure 2(A)-(C). No statistical significance was observed in the TE incidence rates in the matched RLD versus warfarin cohort (OR: 1.34, 95% CI: 0.30-6.00), the DLD versus warfarin cohort (OR: 2.35, 95% CI: 0.60-9.11), or the DLD versus RLD cohort (OR: 2.35, 95% CI: 0.60-9.16); this was consistent with the main analysis. MB complications were also without statistical significance in the RLD versus warfarin cohort (OR: 0.50, 95% CI: 0.09-2.73), the DLD versus warfarin cohort (OR:0.50, 95% CI: 0.09-2.73), and the DLD versus RLD cohort (OR: 1.00, 95% CI: 0.14-7.13). There were no significant differences in the incidence rates of non-MB complications in the RLD versus warfarin cohort (OR: 1.47, 95% CI: 0.89-2.42) or the DLD versus warfarin cohort (OR: 0.94, 95% CI: 0.58-1.53), but a significantly lower incidence of non-MB events was found in DLD than in RLD (OR: 0.44, 95% CI: 0.58-1.53).
Discussion
The present study is based on the prospective China-AF Registry study, and evaluated the effectiveness and safety of rivaroxaban and dabigatran in patients with NVAF who were scheduled for first time CA and were newly receiving OACs during the periprocedural period. Our results showed the following: (1) rivaroxaban and dabigatran were associated with a comparable risk of TE and MB versus warfarin; (2) compared with warfarin, non-MB events were more frequent with rivaroxaban; (3) the incidence of TE, MB, and non-MB events with dabigatran was comparable to that of rivaroxaban; (4) subgroup analysis showed that RLD and DLD had a comparable risk of TE, MB, and non-MB events when compared with warfarin; and there were no significant differences in TE and MB between DLD with RLD, except the non-MB risk of RLD was significantly higher than that of DLD.
There is a growing body of evidence to suggest the similar effectiveness and safety of DOACs compared with warfarin in AF patients undergoing CA.14, 15, 26–30 Limitations of these studies were the different demographic characteristics of the patients, the low number of TE and MB events, and the non-randomized or retrospective study designs. In the present study, we found that neither rivaroxaban nor dabigatran were associated with a higher risk of TE and MB compared with warfarin. Previous studies31, 32 have confirmed that despite ≥4 weeks of continuous DOAC therapy, left atrial thrombi were still being detected by TEE. Harada et al 32 reported that low-dose treatment / inappropriate dose reduction of DOACs significantly increased the incidence of left atrial thrombus. Providência et al conducted a systematic review 33 involving 155 patients treated with DLD and 1237 patients with standard-dose dabigatran; no difference was found between the two dabigatran dosages concerning TE and MB. An outcome analysis from a prospective multicenter registry study 34 observed that RLD exhibited comparable efficacy and safety with uninterrupted warfarin. Limitations of these studies were the low number of observed events 33 and differences in the baseline characteristics. 34 In the present study, the rates of TE and MB events did not significantly differ among the RLD versus warfarin cohort, the DLD versus warfarin cohort, and the DLD versus RLD cohort. These findings reinforce the current expert consensus recommendations on the use of DOACs at the time of AF ablation. 18
The major concern when using DOACs during CA of AF is the risk of bleeding, especially MB such as cardiac tamponade. 18 The markedly higher incidence of cardiac tamponade during AF ablation compared with routine cardiac electrophysiology procedures can be attributed to a number of important procedural differences, including extensive intracardiac catheter manipulation and ablation, the common need for two or more transseptal punctures, and systemic anticoagulant strategies. 18 Two worldwide surveys of AF ablation reported incidences of cardiac tamponade of 1.2% and 1.3%.35, 36 In our study, cardiac tamponade occurred in 10 cases (0.3%), of which nine occurred during the ablation procedure, and one occurred insidiously several hours after ablation. Furthermore, only one case occurred 6 days after initiation of warfarin, the other nine had not yet initiated OACs or had initiated warfarin less than 3 days previously. These results provide evidence of the inherent risk of the ablation procedure. The lower rate of pericardial tamponade in our study may be related to the non-robust effect of oral anticoagulants before ablation, which leads us to speculate that initiation of OACs post-ablation may be safer but no less effective than initiation before ablation for patients who are naïve to OAC treatment.
Even though non-MB events are not the main markers for evaluating safety in our study, the occurrence of MB events is troublesome. Previous studies14, 15, 27 have revealed that DOACs have a risk of minor bleeding similar to or less than that of warfarin. Our study showed that rivaroxaban had a higher risk of non-MB than warfarin, and when the low-dose DOACs were compared, RLD had a higher incidence of non-MB than did DLD. The underlying reason for this phenomenon may be that more attention was paid to bleeding risk in the early stages of DOAC application in China.
Limitations
First, the AF patients in our study were recruited from Beijing, and might not represent the whole AF population of China. Second, variables were well matched by using PSM, but undetected potential confounders still existed. Third, detailed data about how long OACs remained in each patient were not included in the analysis. Fourth, the sample sizes were insufficient and there was a low incidence of effectiveness and safety outcomes; thus, we were only able to generate hypotheses. Finally, few patients were treated with apixaban and edoxaban until December 2018, so data about apixaban and edoxaban were missing in the present data set. Despite these limitations, we believe that our study provides real-world evidence for the selection of OACs during the periprocedural period of NVAF ablation.
Conclusions
In this real-world practice study among Chinese NVAF patients who were new users of OACs and admitted for the first time for CA of AF, both standard and low-dose DOACs were associated with a comparable risk of TE and MB when compared with warfarin. The risk of TE and MB were similar when standard and low-dose rivaroxaban and dabigatran were compared. Rivaroxaban was associated with a higher risk of non-MB events versus warfarin, and the risk of non-MB events was higher in low-dose rivaroxaban than in low-dose dabigatran.
Supplemental Material
sj-docx-1-cat-10.1177_10760296221123306 - Supplemental material for Effectiveness and Safety of DOACs in Atrial Fibrillation Patients Undergoing Catheter Ablation: Results from the China Atrial Fibrillation (China-AF) Registry
Supplemental material, sj-docx-1-cat-10.1177_10760296221123306 for Effectiveness and Safety of DOACs in Atrial Fibrillation Patients Undergoing Catheter Ablation: Results from the China Atrial Fibrillation (China-AF) Registry by Zhimin Dong, Xiaoxia Hou, Xin Du, Liu He, Jianzeng Dong and Changsheng Ma in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Declaration of Conflicting Interests
Changsheng Ma has received honoraria from Bristol-Myers Squibb, Pfizer, Johnson & Johnson, Boehringer-Ingelheim and Bayer for giving lectures. Jianzeng Dong has received honoraria from Johnson & Johnson for giving lectures. The other authors have no disclosures.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the the National Key Research and Development Program of China, (grant number D 2016YFC1301002, D 2020YFC2004803, M 2016YFC0900901)
Ethics Approval
Ethical approval to report this case series was obtained from the Human Research Ethics Committee at Beijing Anzhen Hospital (approval no. D11110700300000) on June 10, 2011. Ethics review boards in each participating hospital approved their participation. Verbal informed consent was obtained from the patients for their anonymized information to be published in this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
