Abstract
Angiopoeitin-2 (Ang2) is a vascular growth factor involved in regulating angiogenesis and endothelial remodeling. Higher Ang2 levels have been associated with mortality in the general population and among male hemodialysis patients, but its effects on concomitant heart failure with reduced ejection fraction (HFrEF) and end-stage renal disease (ESRD) are unknown. Plasma samples from 73 ESRD patients and 40 healthy patients were analyzed for Ang2 concentrations using ELISA. Patient groups were stratified into those with or without HFrEF (EF < 50%). At two years following sample collection, the medical record was reviewed for mortality. The optimal cut-off value for Ang2 to predict all-cause mortality was 1671 pg/mL (AUC 0.73, sensitivity 0.714, specificity 0.750) based on the regression analysis. Statistical analyses included Mann–Whitney U tests, Cox proportional hazards model, and Log-rank test. Multiple comorbidities were present; coronary artery disease 46%, diabetes 69%, hypertension 97%, and smoking 49%. Patients with one- and two-year mortality had higher Ang2. Ang2 levels above the optimal cut-off are associated with mortality within the entire ESRD sample and within the group with both ESRD and HFrEF. In the Cox proportional hazards analysis, Ang2 levels were associated with mortality within the larger ESRD sample but not in the group with ESRD and HFrEF. Ang2 has potential as a non-specific biomarker for prognostication in patients with cardiorenal syndrome given its association with mortality, despite modest sex-based differences. Future research should be conducted with larger samples to evaluate if it has prognostic value in individuals with HFrEF and ESRD of varying severity and temporality.
Introduction
Angiogenesis, the process by which the body maintains and creates new blood vessels is highly regulated in part by angiopoietins. Angiopoietins 1 and 2 are vascular growth factors which are stimulated by inflammatory cytokines and involved in regulating both angiogenesis and endothelial remodeling through their interactions with tyrosine receptor kinase Tie1. 1 Primarily expressed in blood and lymphatic endothelial tissues, their exact effects are context-dependent in the contrasting settings of physiologic and inflamed epithelium and are capable of being both agonistic and antagonistic to in vivo angiogenesis. 1 Angiopoietins have recently been the subject of much investigation due to their potential as biomarkers. Angiopoietin-2 (Ang2) in particular has been shown to be mediate small vessel remodeling in diseases characterized by inflammation vascular permeability, and is associated with mortality in a wide variety of disease conditions, including sepsis, heart disease, acute respiratory distress syndrome, and more.1–4 It is thought that Ang2 contributes to the progression of disease, as opposed to reflecting changes after the fact. Its effects mediated by Tie protein kinases are destabilizing, increasing vascular remodeling, permeability and directly contributing to worsening function. Temporality, a pinnacle of causality, has been confirmed by work identifying Ang2 elevations prior to the development of heart failure in prior work. 3
However, the effort to apply Ang2 as a risk-stratifying biomarker has been impeded in part by observed sex-based differences in the impact of Ang2 on clinical outcomes. In end-stage renal disease (ESRD) patients on hemodialysis, for example, the association between Ang2 and mortality has only been shown to present in male patients. 5 Further investigation into the etiology of this difference is an objective of our group and others which must be resolved before Ang2 may be utilized clinically. An additional limitation is the role of Ang2 in multiple different diseases. Despite increasing numbers of patients with concomitant kidney disease and heart failure with reduced ejection fraction (HFrEF), there is not currently data showing the relation of Ang2 levels to prognosis in those with cardiorenal syndrome, the intersection of both diseases. Fan et al and Mohebi et al identify angiopoietins as showing promise as biomarkers for risk stratification in cardiorenal syndrome, however there is currently a paucity of data identifying primary biomarkers in this condition which has only somewhat recently been strictly defined.6,7 This effort is worth the costs of investigation for the potential value of both risk stratification and future therapeutics targeting Ang2, as preliminary data shows they may have greater promise than therapeutics targeting vascular endothelial growth factor. 8
While known that Ang2 levels impact survival in ESRD and HFrEF independently, this study aims to answer if the impact of Ang2 levels on survival and if previous findings of sex-based differences in ESRD are conserved in this comorbid population.
Methods
To assess mortality, we carried out a prospective cohort study of 73 patients with ESRD currently undergoing routine hemodialysis at Loyola University Medical Center. Whole blood samples were collected under an Institutional Review Board approved protocol, centrifuged for platelet poor plasma, and frozen at −70 °C prior to analysis. The plasma samples were analyzed for Ang2 concentrations using a commercially available ELISA kit. With this sample size, the power to detect large differences (effect size d = 0.8) was 0.705. The power to detect medium differences (d = 0.5) was 0.346.
Patients were followed for two years following sample collection, at which time the EMR was reviewed for patient mortality with time of death or survival recorded. Patient demographics and other clinical information were collected from patient electronic medical records (EMR). Patient groups were stratified into two groups, those with or without HFrEF defined by those having a confirmed by echocardiogram ejection fraction (EF) less than 50% recorded at any point in the six months before or after the plasma collection date, these parameters intending to exclude those with recovered EF or those with normal EF at time of sample collection but later worsening of systolic function.
The statistical difference between those patients surviving more or less than one year was evaluated utilizing the nonparametric Mann–Whitney U (Wilcoxon rank-sum) test. The relationships of biomarkers to survival were analyzed using Cox proportional hazards model. The optimal cut-off value for baseline serum Ang-2 to predict all-cause mortality was 1715 pg/mL (AUC 0.706) based on the regression analysis. Survival curves were compared using Log-rank (Mantel-Cox) and Hazard Ratio (Mantel-Haenszel) tests. The calculations were performed using R (Posit Software, Boston, Massachusetts) and Prism (GraphPad, La Jolla, California) software.
Results
The mean age of the ESRD patients was 69 ± 13.14 years, 58% were male. Sixty-one out of seventy-three (84%) patients had ESRD only; twelve had both ESRD and HFrEF per the inclusion criteria above. Multiple comorbidities were present in this patient population. Coronary artery disease was present in 41%, diabetes in 55%, hypertension in 93%, obesity in 41%, and smoking in 49%. The details of the patient population are shown in Table 1.
Patient Characteristics.
Mortality in this cohort was 12.7% at one year and 19.7% at two years. Those patients who survived less than one year had 1.5 fold greater Ang2 than those who survived for more than one year (p = 0.0386). Those surviving for greater than two years had 1.4 fold greater Ang2 (p = 0.0074) (Figure 1). There was no significant difference in survival between the groups with and without HFrEF (Figure 2). There was no significant difference in Ang2 levels between those with HFrEF and those without (data available upon request).
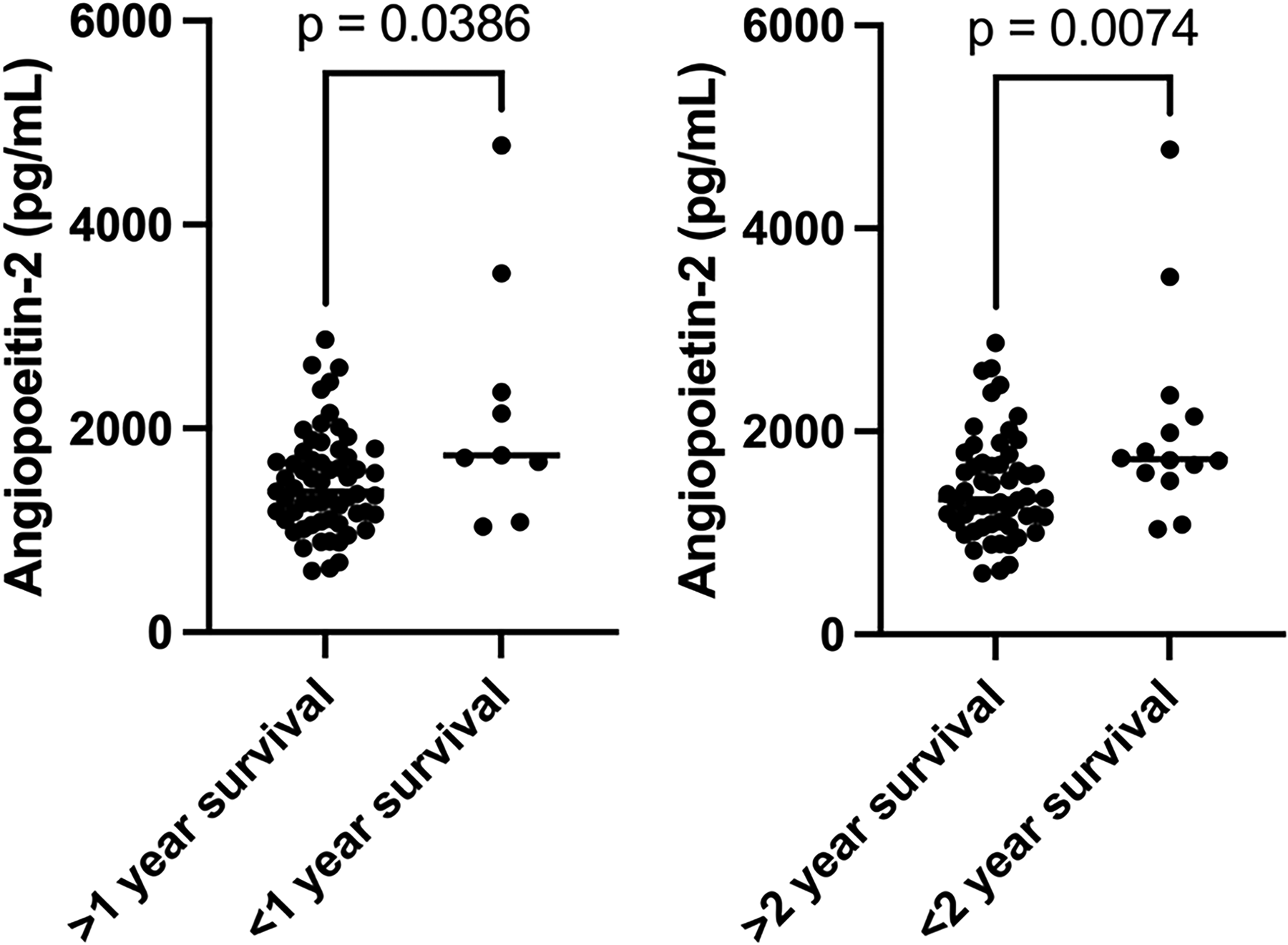
Ang2 levels in 1- and 2-year survival (p = 0.0386, 0.0074).
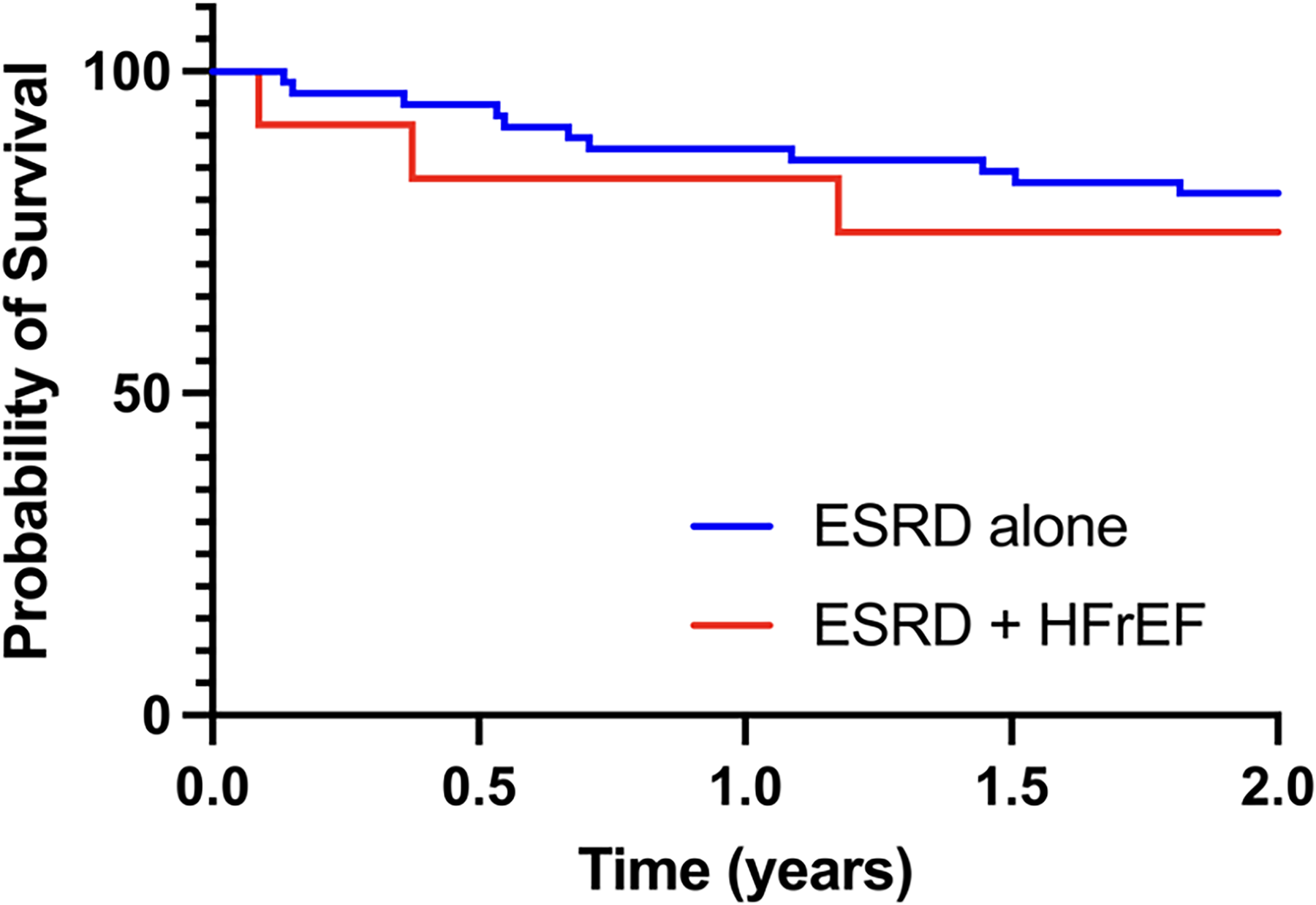
Survival in ESRD, ESRD + HFrEF patients (ns).
Receiver operator curve analysis determined the optimal cut-off to predict mortality as 1671 pg/mL (AUC 0.73, sensitivity 0.714, specificity 0.750). Ang2 levels above the optimal cut-off were highly associated with two-year mortality in Kaplan-Meier survival analysis (log-rank p = 0.0006). No patients with HFrEF and Ang2 below the optimal cut-off died within two years, in contrast with half of the patients with Ang2 levels above the optimal cutoff dying within two years. However, this finding did not reach the threshold for statistical significance (log-rank p = 0.0554) (Figure 3).
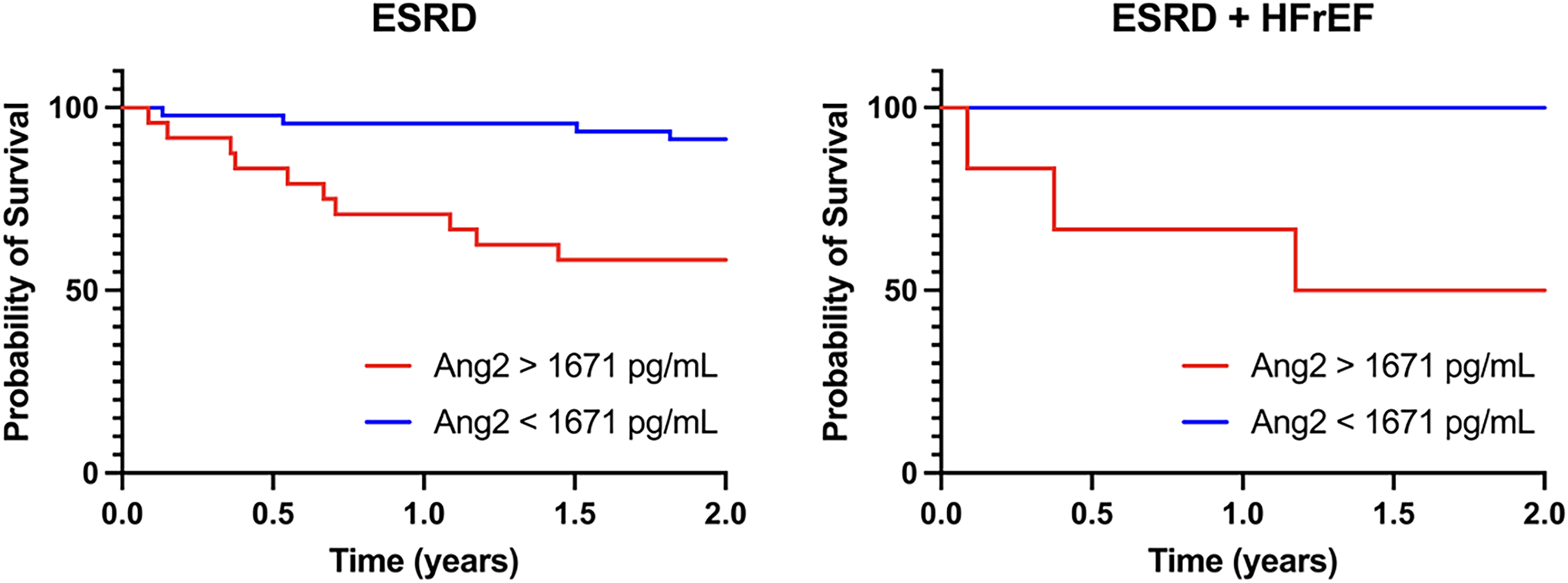
Ang2 levels above and below optimal cut-off in ESRD (p = 0.0006), ESRD + HFrEF (p = 0.0554) patients.
In the univariate Cox proportional hazard regression, Ang2 was predictive of mortality in our sample (HR = 1.001, p < 0.001), with concordance = 0.706, likelihood ratio test = 11.96 (p = 0.003) and score (log rank) test = 13.71 (p = 0.001) on 2 degrees of freedom. However, Ang2 was not significantly predictive in the subgroup with HFrEF.
An additional analysis was done to discern Ang2 prediction of mortality by sex. Mortality was markedly increased with increasing Ang2 levels in both males (p = 0.0049) and females (p = 0.0561), however in Cox analysis it was not identified as a significant factor in the variation of survival (Figure 4).
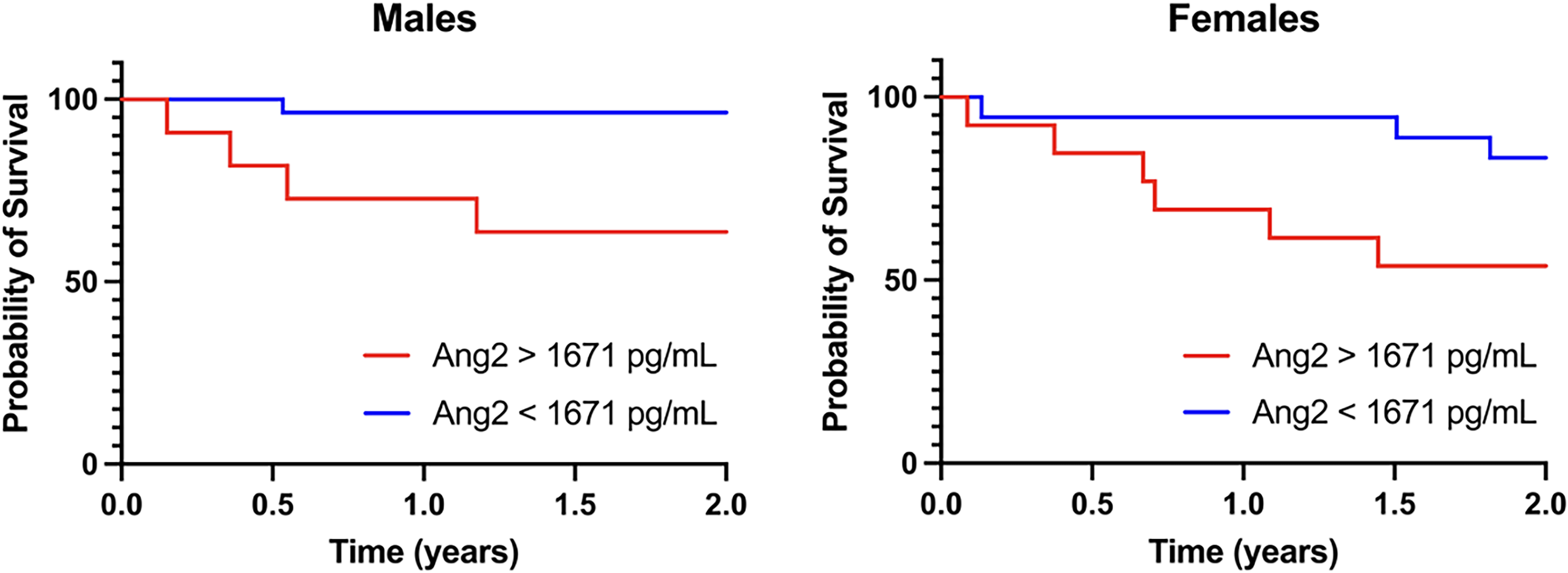
Ang2 levels above and below optimal cut-off in males (p = 0.0049) and females (p = 0.0561).
Discussion
As chronic disease becomes increasingly prevalent in the US and around the world, more patients suffer from vascular endothelial injury, characterized by reduced vasodilation and a pro-thromboinflammatory state. 9 While endothelial dysfunction may be caused by several different etiologies, from smoking and physical inactivity to metabolic syndrome, the impact of endothelial injury is perhaps the most relevant in patients with multiple chronic thromboinflammatory diseases as it appears to be causally involved in loss of function in multiple organs in addition to the endothelium. 10
Ang2 has been identified as a prognostic biomarker for several diseases, including its association with negative outcomes in ESRD and HFrEF.3,5 These results suggest it also has potential as a biomarker for mortality in patients with cardiorenal syndrome given its similar upregulation in ESRD patients with and without HFrEF. Additionally, we replicated modest sex-based differences in the effects of Ang2 on mortality between males and females.
We hypothesized that the levels of Ang2 may have been increased in HFrEF patients though the levels of Ang2 are not significantly different due to the limited power of this study given the small sample size. If in fact there is a true difference in expression, we hypothesize this is because the expression of Ang2 is partially dependent upon inflammatory signaling which is similar in magnitude between the two diseases but shares a common pathway of upregulating expression. These results imply that Ang2 may have potential in prognostication of cardiorenal syndrome similarly to other chronic diseases affecting the vasculature, though not in an additive fashion and notably not specific to this condition.
This study is important in adding to the discussion began by Chu et al regarding sex-based differences in Ang2-associated mortality. While the result for females’ mortality split by optimal cut-off was just short of the threshold for significance (p = 0.0561), males and females have remarkably similar patterns of survival when viewed together (Figure 4). Sex-based differences are not unusual in the study of endothelial biomarkers; similar findings have also been seen in studies of vascular endothelial growth factor and hepatocyte growth factor in addition to Ang2.5,11 This may be related to the impact of both sex hormone signaling and genetic differences on endothelial physiology, which has been well described. 12 Tsuzuki et al, for example, have found that estradiol in particular alters the expression of angiopoietins through variable mRNA transcription, though this phenomenon is still incompletely understood. 13 Further research may attempt to replicate these methods with a larger sample size for potential etiological differences between males and females accounting for an unequal concordance with mortality and Ang2.
These results add to the growing body of evidence that Ang2 may be useful not just academically but clinically and could be ready for clinical utilization in patients with ESRD and a range of comorbidities.
Study Strengths and Limitations
This is the first paper specifically investigating Ang2 in cardiorenal syndrome, a disease of increasing prevalence which deserves more specific attention. The inclusion of reliable predictors of mortality in this condition would likely have important consequences to those caring for patients with more severe and highly complex disease. It is also possible that novel angiopoietin-acting therapeutics may one day be developed following greater knowledge of the effects of fluctuations in this pathway. The replication, albeit to a markedly lesser degree, of sex-based differences from Chu et al is similarly critical in guiding scientific understanding of the role of sex based differences in both endothelial damage and chronic disease progression. Methodologically, our protocol limited environmental contribution to inter-patient differences by ensuring that all of the patients in the study receive their dialysis at the same clinic and live in the same region. Sample collection and processing were conducted according to rigid protocols and were stored and measured identically with ELISA kits procured from a single manufacturer.
As these samples were collected in 2021 and the patients were followed through 2021–23, COVID-19 is a notable confounder. Ang2 levels are increased in those patients with COVID-19, particularly in those with more severe cases, and its detrimental effects on the vasculature are well documented.14,15 It is feasible that patients impacted by COVID-19 or other acute illnesses demonstrated endothelial damage and subsequent elevation in the corresponding biomarkers to an extent that overshadows the impact of chronic diseases such as ESRD and HFrEF. Further, this is a retrospective study. For this reason, other confounding variables also impacting the inflammatory process in these patients cannot be excluded. Lastly, this study was underpowered (0.705) to detect even large differences in effect due to the sample size. Despite these limitations, this study provides some important and novel observational differences which require additional expanded studies.
Conclusions
In this study, the impact of Ang2 on mortality was investigated in a group of patients with ESRD with a subset of those experiencing HF. Our findings, demonstrating increased mortality associated with Ang2 levels among both the ESRD population and HFrEF sub-group, with more pronounced impact in males, are highly important to the field's understanding of cardiorenal syndrome.
Footnotes
Acknowledgements
The authors are grateful for the support and guidance from the faculty and staff of the Hemostasis and Thrombosis Laboratories, Cardiovascular Research Institute, and the Department of Pathology at Loyola University Medical Center. The authors are thankful to Dr Lowell Steen, Chief of Cardiology, for his support to complete this study. The authors are also thankful to Dr Eva Wojcik, Chairperson of the Department of Pathology, for her support, and Director of the STAR program, Dr Colleen Fitzgerald, for providing the opportunity for this project through the Student Training in Approaches to Research (STAR) and Research Honors programs. A special thanks to Ms. Erin Healy-Erickson for reviewing this manuscript and Mr Isaiah Johnston for statistics support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
