Abstract
Cardiovascular disease is a prevalent complication in patients with end-stage renal disease (ESRD) on maintenance hemodialysis. In the ESRD patient population, cardiovascular mortality is 20 times higher compared to the general population. The strong relationship between both illnesses can be explained through cardiorenal syndrome (CRS). CRS encompasses a spectrum of disorders involving both the heart and kidneys in which acute or chronic dysfunction in one organ may induce a similar effect in the other organ. Current literature reveals that inflammation and thrombosis are integral to CRS development. Hence, this study aims to demonstrate whether thromboinflammatory biomarkers and laboratory parameters correlate with ESRD progression and the development of CRS. Ninety-five ESRD patients were recruited at Loyola University Medical Center hemodialysis unit. Epic chart analysis was used to determine patients with CRS. Biomarkers (C-reactive protein, tumor necrosis factor alpha, interleukin-6, Annexin V, L-fatty acid binding protein, monocyte chemoattractant protein 1, nitric oxide, von Willebrand factor, D-dimer, and plasminogen activator inhibitor-1) were profiled using the enzyme-linked immunosorbent assay method in patients with and without CRS in the ESRD cohort. All biomarkers were significantly elevated in ESRD patients compared to normal controls (P < .05) and laboratory parameters, ferritin (521.99 ± 289.33) and PTH (442.91 ± 1.50). Through EPIC chart analysis 47% of ESRD patients have CRS. D-dimer and TNF-α were significantly elevated in patients with CRS compared to patients without CRS. This study suggests that biomarkers, D-dimer, and TNF-α, can be good predictors of CRS in ESRD patients.
Keywords
Introduction
In 2018 the incidence of end-stage renal disease (ESRD) in the United States was 390.2 per million and prevalence was 242 per million. 1 Adults older than 75 years had a 4-fold greater incidence of ESRD than the average adult and more than double the incidence of adults in the 45- to 64-year-old age group. 2 It is a highly prevalent disease and is linked to several complications in various systems including cardiovascular, endocrine, gastrointestinal, hematologic, neurologic, and musculoskeletal. These complications contribute to higher morbidity, mortality, and poor quality of life. Of these complications, cardiovascular disease is the leading cause of mortality in ESRD patients. Cardiovascular mortality is 20 times higher in ESRD patients compared to the general population.3,4 Several cardiovascular problems can occur in ESRD patients, including atrial fibrillation (AF), peripheral artery disease (PAD), heart failure (HF), and coronary artery disease (CAD).5,6 The strong relationship between both illnesses in the ESRD patient population can be explained through cardiorenal syndrome (CRS). CRS encompasses a spectrum of disorders involving both heart and kidneys in which acute or chronic dysfunction in one organ may induce a similar effect in the other organ.7,8 The diagnosis, prognosis, and risk stratification of CRS are poorly understood; thus, it is imperative to understand the syndrome's pathophysiology.
Cardiovascular complications occur due to an accelerated atherosclerosis process in dialysis patients. 9 The accelerated atherosclerosis process in ESRD patients occurs through various pathophysiological mechanisms such as endothelial dysfunction, inflammation, oxidative stress, thrombosis, and tissue fibrosis. Endothelial dysfunction is characterized by stimulation of proinflammatory and prothrombotic states paralleled by a systemic alteration in vascular wall structure. 10 Endothelial stretch from peripheral venous congestion causes conversion of vascular endothelium from a quiescent to a proinflammatory phenotype leading to accelerated atherosclerosis of the vessels, thus cardiorenal syndrome. 11 Thus, it is appropriate to say that there is a growing role of thrombotic inflammatory processes in the pathogenesis of CRS.
Biomarkers are essential in diagnosing acute and chronic heart and renal disease as they can indicate early cardiac or renal injury and give better understanding of the prognosis and management. Established cardiac biomarkers include troponin, myeloperoxidase (MPO), vascular endothelial growth factor, and procalcitonin. Established renal biomarkers include creatinine, microalbuminuria, cystatin C, and kidney injury molecule (KIM-1). Previous studies have shown several cardiac and renal biomarkers elevated in CRS patients, but few have investigated thromboinflammatory biomarkers involved in the processes of endothelial dysfunction and atherosclerosis.12,13 Given previous literature and the process of endothelial dysfunction, thromboinflammatory biomarkers can provide a telling narrative of ESRD progression to CRS. In this study we sought to quantify the levels of biomarkers that have not previously been reported such as C-reactive protein (CRP), tumor necrosis factor alpha (TNF-α), interleukin-6 (IL-6), Annexin V, L-fatty acid binding protein (L-FABP), monocyte chemoattractant protein 1 (MCP-1), nitric oxide (NO), von Willebrand factor (vWF), D-dimer, and plasminogen activator inhibitor-1 (PAI-1) in a group of ESRD patients with CRS in comparison to without CRS to determine whether there is a relationship existing between them. Identification of this relationship may provide insight into the complex pathophysiology of CRS and may be useful for the development of potential prognostic, diagnostic, and therapeutic interventions.
Material and Methods
Blood samples were collected from 95 consented ESRD patients recruited with an approved Institutional Review Board (IRB) protocol at the Loyola University Medical Center hemodialysis unit. The only inclusion criteria were that the ESRD patients should have been on dialysis for greater than 3 months. The blood samples were collected in tubes containing 3.2% sodium citrate, and the tubes were then centrifuged at 3000g for 15 min. The plasma supernatant was separated, aliquoted, and frozen at −80 °C until the samples were analyzed. All the samples were given a study number. The samples were then blindly analyzed in the Hemostasis and Thrombosis Research Laboratories at the Loyola University Chicago. Controls were composed of 50 plasma samples from normal individual human volunteers (25 male; 25 female), obtained from a commercial vendor, George King Biomedical (Overland Park, Kansas).
Commercially available sandwich enzyme-linked immunosorbent assay (ELISA) kits were used to quantify the thromboinflammatory biomarkers including CRP, TNF-α, IL-6, Annexin V, L-FABP, MCP-1, NO, vWF, D-dimer, and PAI-1 in ESRD patients with and without CRS and control plasma samples. The sandwich ELISA kits were purchased from Aviva Systems Biology (San Diego, California), R&D Systems (Minneapolis, Minnesota), Neobiolabs (Cambridge, Massachusetts), and Antibodies Online. CRS, other illness co-morbidities and laboratory parameters were obtained from Epic chart analysis.
Statistical analyses were performed using Microsoft Excel and GraphPad Prism Software. For each biomarker, we calculated the mean ± SEM (SD) for the ESRD and normal individual human samples as well as for patients with and without CRS in the ESRD cohort. The Mann-Whitney U test was used to compare between the mean ESRD and mean of the 50 healthy individual volunteers for each biomarker as well as between patients with CRS and without CRS in the ESRD cohort. A P-value <.05 was considered statistically significant. A spearman correlation analysis was then performed to test the interrelationship between the biomarkers in patients with and without CRS.
Results
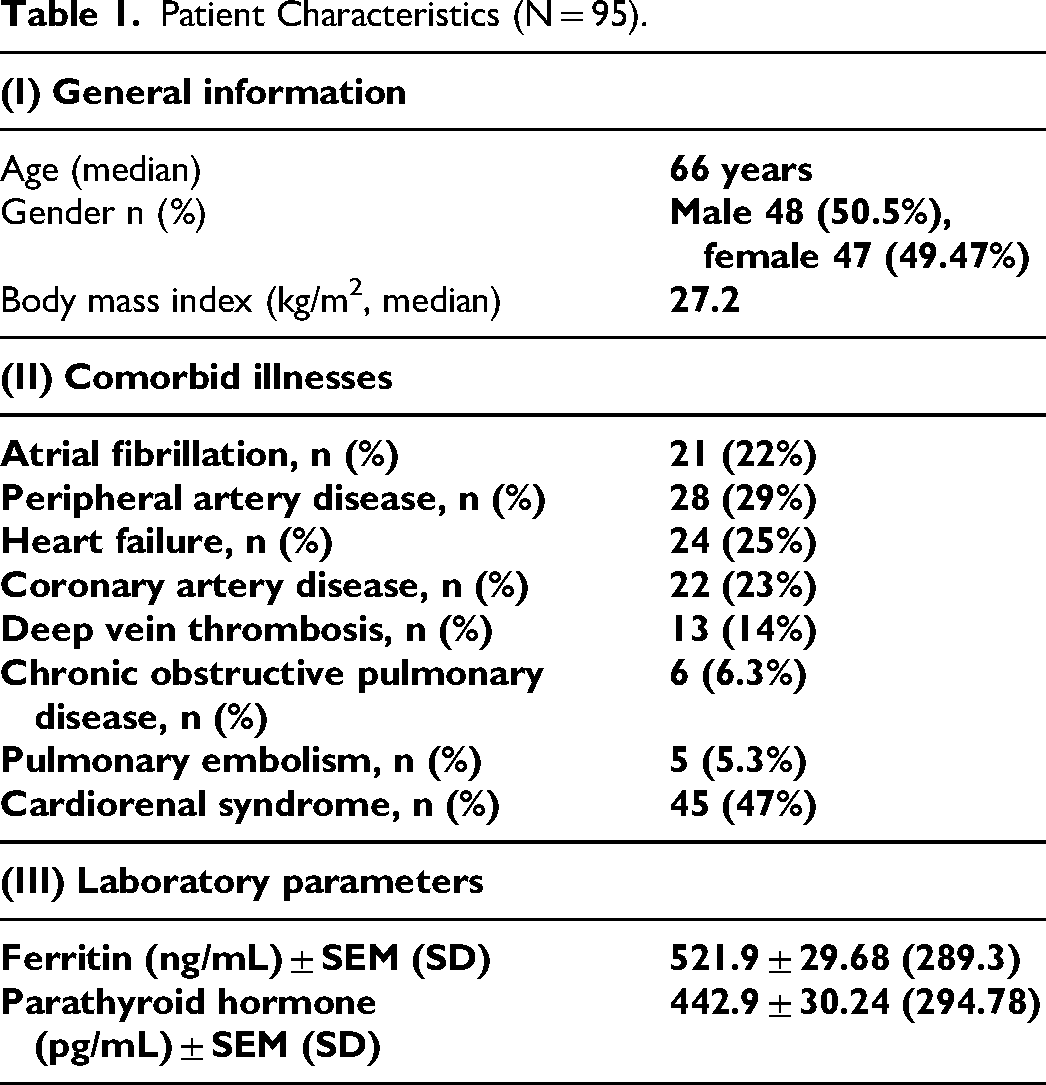
Of the 95 patients that were included in the final analysis, the median age of the patients was 66 with 48 male patients (50.5%) and 47 female patients (49.47%). The patient population had myriad of comorbid illness in which 21 patients (22%) had AF, 28 patients (29%) PAD, 24 patients (25%) HF, 22 patients (23%) CAD, 13 patients (14%) deep vein thrombosis (DVT), 6 patients 6.3% chronic obstructive pulmonary disease (COPD), 5 patients (5.3%) pulmonary embolism (PE), and 45 patients (47%) CRS. The details of patient information including demographics, laboratory parameters, and comorbid illnesses are shown in Table 1. Laboratory parameters, ferritin (521.9 ± 29.68 ng/mL) and parathyroid hormone (PTH) (442.9 ± 30.24 pg/mL) were elevated in ESRD patients. There was no significant difference observed in the laboratory parameters in ESRD patients with CRS and without CRS, shown in Figure 1.
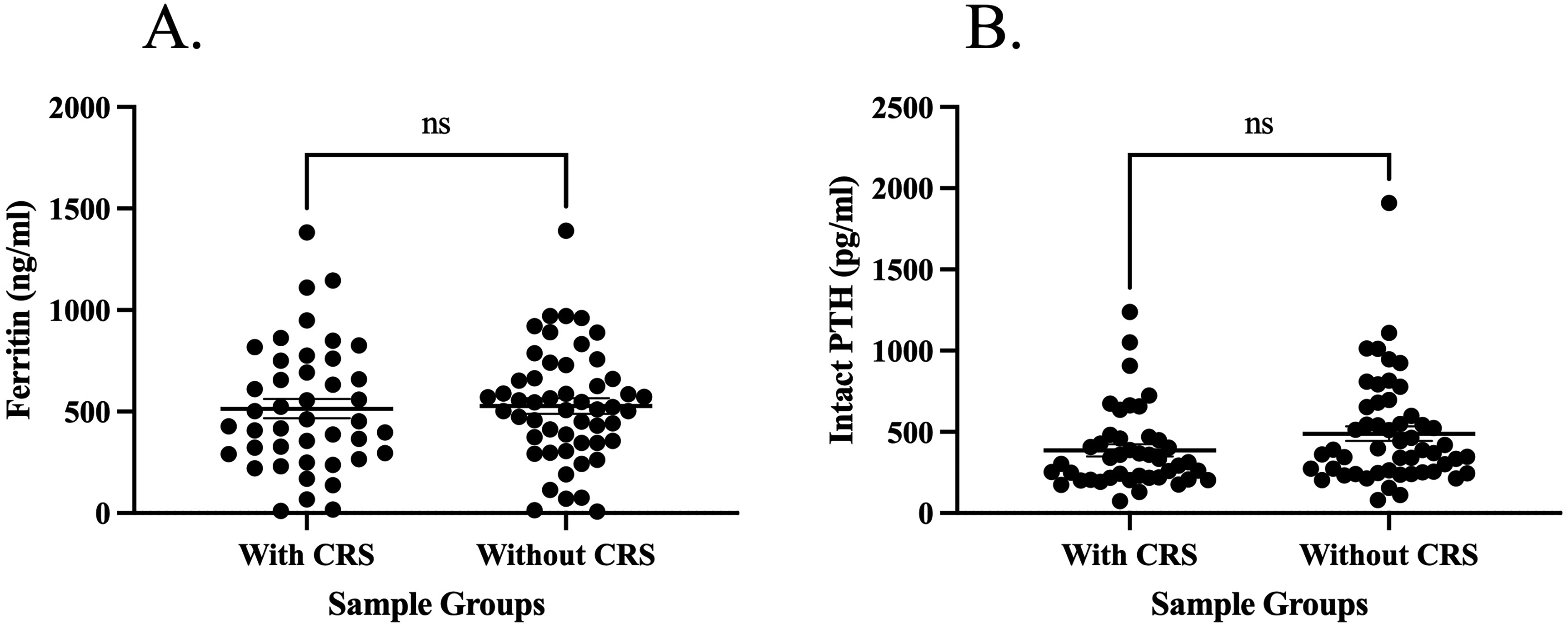
Concentrations of laboratory parameters in ESRD patients with CRS compared to patients without CRS. Results are reported as means, with the error bars representing the standard error of the mean. (A) The comparison of Ferritin; (B) The comparison of PTH. Abbreviations: CRS, cardiorenal syndrome; ESRD, end-stage renal disease.
Patient Characteristics (N = 95).
All the inflammatory biomarkers were elevated in the ESRD cohort compared to the normal controls. The composite data, including the average, standard deviation, and percent increase in the levels of each biomarker in comparison to healthy controls are shown in Table 2. A comparison of the inflammatory biomarkers in ESRD patients with CRS and without CRS is shown in Figure 2. This graph shows that TNF-α was the only inflammatory biomarker that was significantly elevated in ESRD patients with CRS compared to those without. For TNF-α the range in the CRS group was (2.68 ± .47 pg/mL) and the range in without CRS groups was (2.23 ± .52 pg/mL). The analysis of the biomarkers in this ESRD cohort showed the following results. There was no significant difference in CRP with CRS (13.62 ± 2.18 μg/mL) compared to without CRS (15.43 ± 2.47 μg/mL). There was no significant difference in IL-6 with CRS (6.22 ± 1.09 pg/mL) compared to without CRS (3.42 ± .81 pg/mL). There was no significant difference in MCP-1 (145.58 ± 25.73 pg/mL) compared to without CRS (153.41 ± 36.16 pg/mL). There was no significant difference in L-FABP with CRS (84099.10 ± 12824.99 pg/mL) compared to without CRS (123524.89 ± 16967.45 pg/mL). There was no significant difference in Annexin V (7.39 ± 1.13 ng/mL) with CRS compared to without CRS (7.72 ± 1.07 ng/mL).
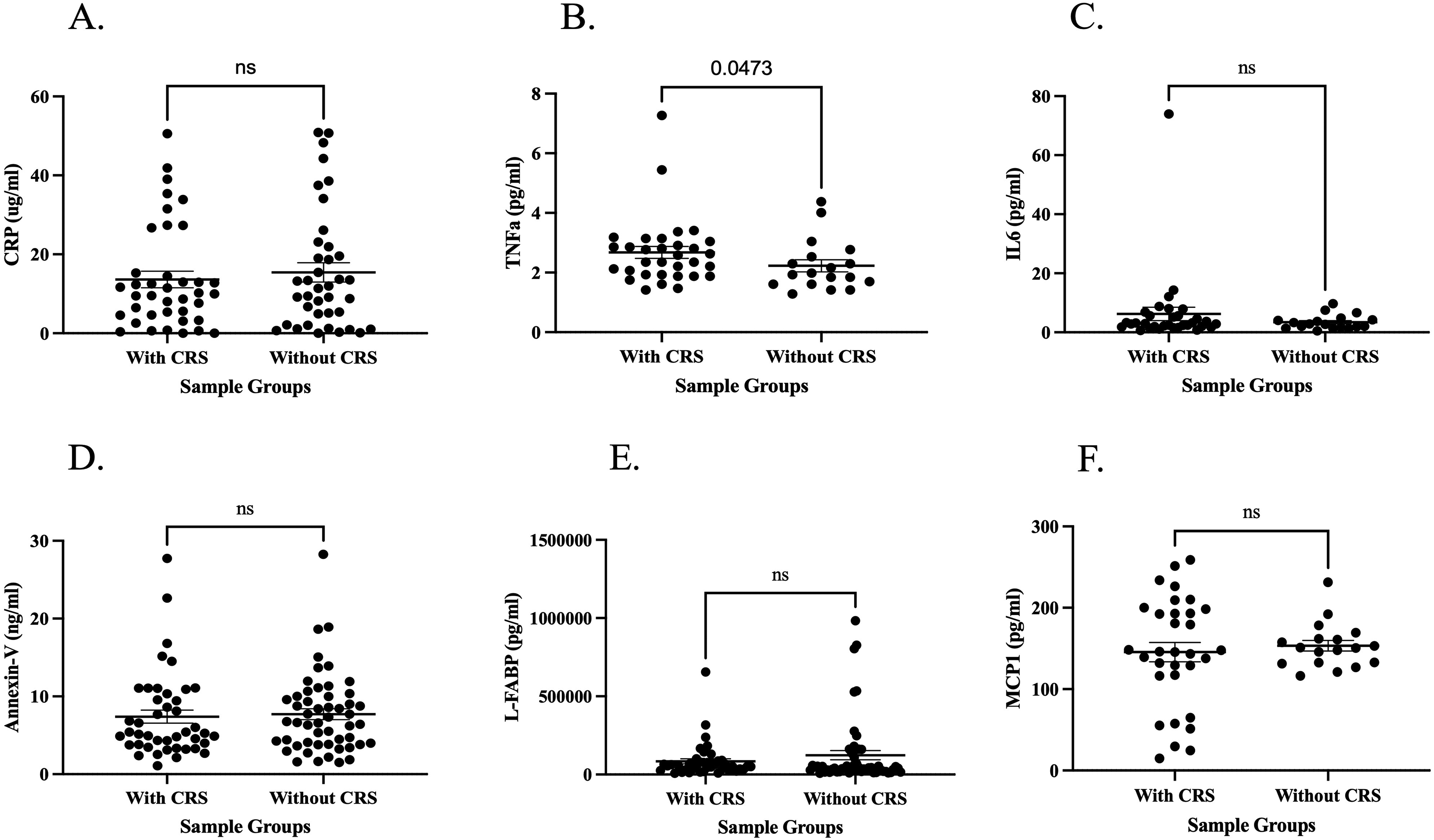
Concentrations of inflammatory biomarkers in ESRD patients with CRS compared to patients without CRS. Results are reported as means, with the error bars representing the standard error of the mean. (A) The comparison of CRP; (B) the comparison of TNF-α; (C) the comparison of IL-6; (D) the comparison of Annexin V; (E) the comparison of L-FABP; and (F) the comparison of MCP-1. Abbreviations: CRP, C-reactive protein; CRS, cardiorenal syndrome; ESRD, end-stage renal disease; IL-6, interleukin-6; L-FABP, L-fatty acid binding protein; MCP-1, monocyte chemoattractant protein 1; TNF-α, tumor necrosis factor alpha.
Comparison of Thromboinflammatory Biomarkers.
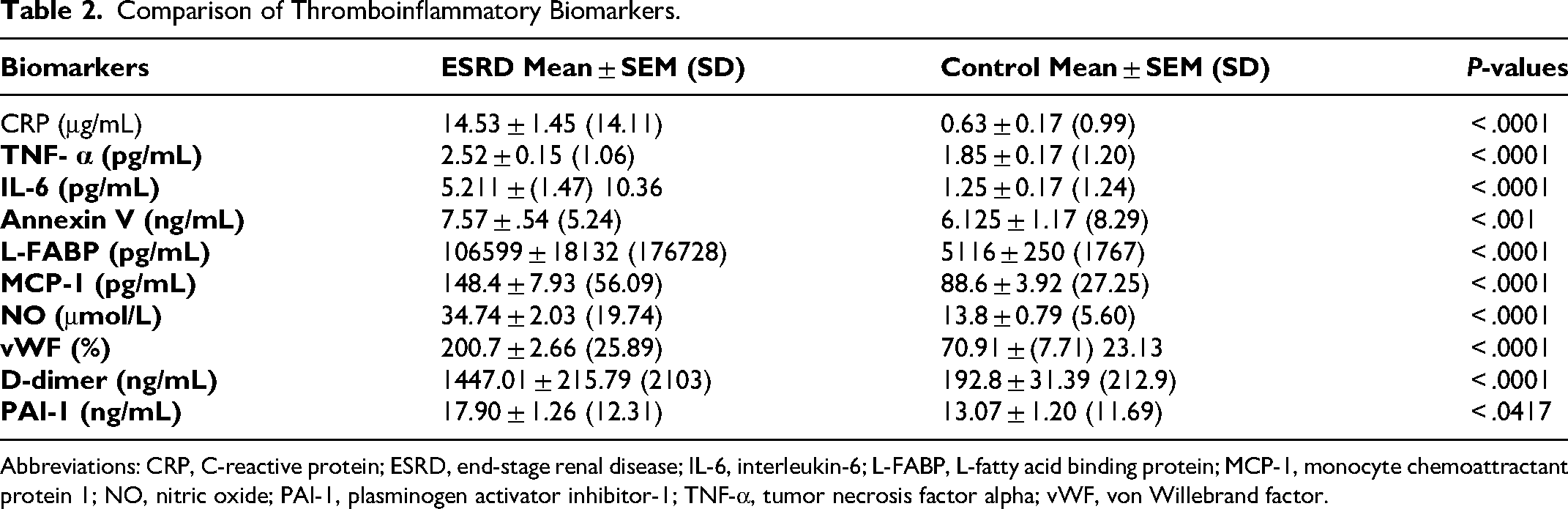
Abbreviations: CRP, C-reactive protein; ESRD, end-stage renal disease; IL-6, interleukin-6; L-FABP, L-fatty acid binding protein; MCP-1, monocyte chemoattractant protein 1; NO, nitric oxide; PAI-1, plasminogen activator inhibitor-1; TNF-α, tumor necrosis factor alpha; vWF, von Willebrand factor.
All the thrombotic biomarkers were elevated in the ESRD cohort compared to the normal controls. Additionally, antithrombotic biomarker NO was also elevated in the ESRD cohort compared to the normal controls. The composite data, including the average, standard deviation, and percent increase in the levels of each biomarker in comparison to healthy controls are shown in Table 2. A comparison of the thrombotic biomarkers and antithrombotic biomarker NO in ESRD patients with CRS and without CRS is shown in Figure 3. Figure 3 shows that D-dimer was significantly elevated in ESRD patients with CRS compared to those without. For D-dimer the range in the CRS group was (1938.95 ± 310.48 ng/mL) and the range in without CRS groups was (955.07 ± 152.93 ng/mL). There was no significant difference in vWF with CRS (205.95 ± 32.97%) compared to without CRS (195.40 ± 31.28%). There was no significant difference in NO with CRS (31.28 ± 5.07 μmol/L) compared to without CRS (38.39 ± 6.39 μmol/L). There was no significant difference in PAI-1 with CRS (16.62 ± 2.81 ng/mL) compared to without CRS (18.96 ± 2.92 ng/mL).
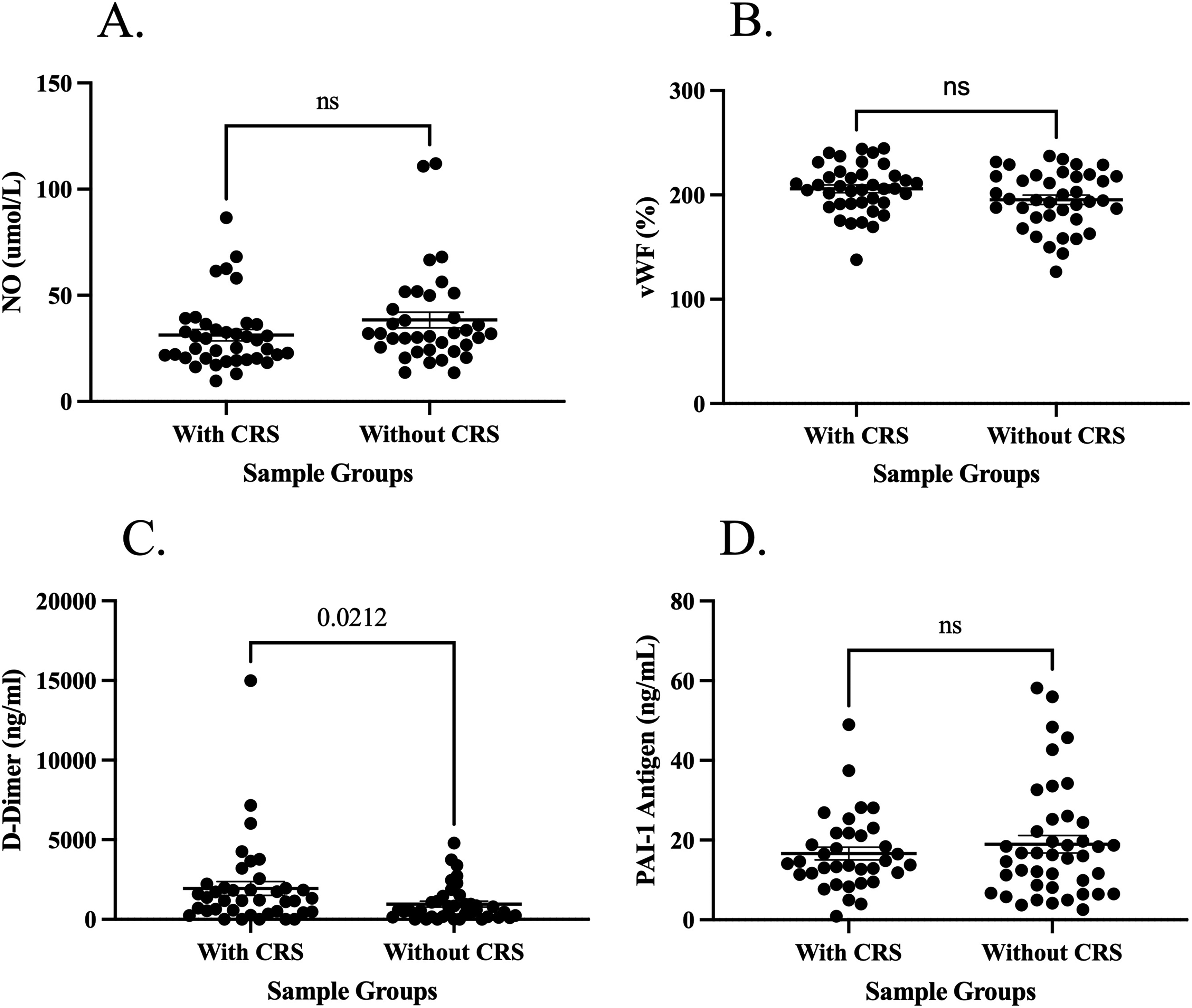
Concentrations of antithrombotic biomarker and thrombotic biomarkers in ESRD patients with CRS compared to patients without CRS. Results are reported as means, with the error bars representing the standard error of the mean. (A) The comparison of NO; (B) the comparison of vWF; (C) the comparison of D-dimer; and (D) the comparison of PAI-1. Abbreviations: CRS, cardiorenal syndrome; ESRD, end-stage renal disease; NO, nitric oxide; PAI-1, plasminogen activator inhibitor-1; vWF, von Willebrand factor.
In the correlation analysis for ESRD patients with CRS the following correlations were statistically significant. IL-6 correlated with D-dimer (r = 0.37) and PTH (r = −0.42). L-FABP correlated with MCP-1 (r = −0.45). MCP-1 correlated with PTH (r = −0.42). NO correlated with PAI-1 (r = −0.42). In the correlation analysis for ESRD patients without CRS the following correlations were statistically significant. CRP correlated with Annexin V (r = 0.49) and PAI-1 (r = 0.41). Annexin V correlated with L-FABP (r = 0.34). MCP-1 correlated with D-dimer (r = 0.47). VWF correlated with D-dimer (r = 0.36). The results of the correlations are depicted in Figure 4.
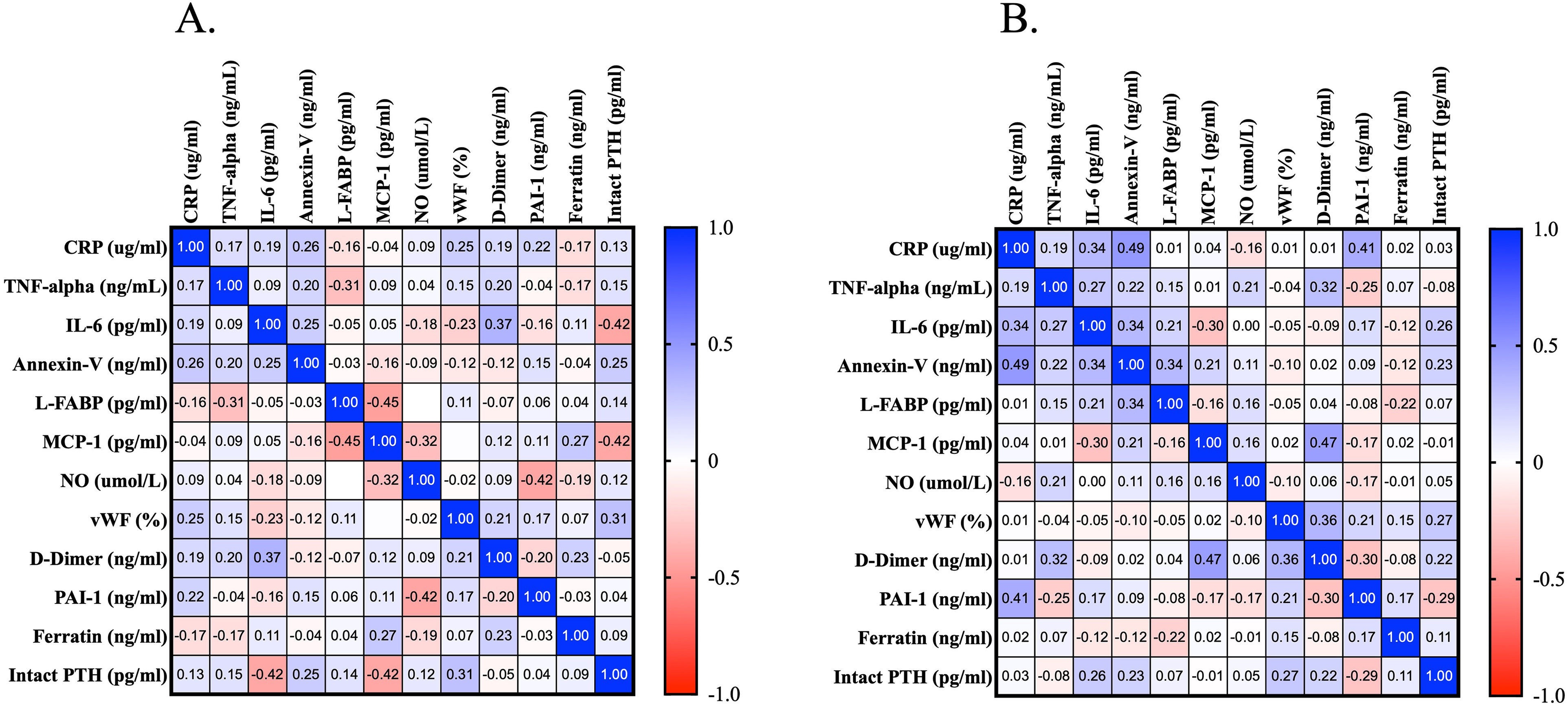
Correlation analysis of thromboinflammatory biomarkers in ESRD patients; (A) with CRS and (B) without CRS. Abbreviations: CRS, cardiorenal syndrome; ESRD, end-stage renal disease.
Discussion
CRS was grouped into 5 subtypes based on disease acuity and sequential organ involvement to facilitate reliable characterization of the clinical presentation of cardiorenal dysregulation for diagnostic and therapeutic purposes. The classification of CRS into subtypes overcame some initial ambiguity in defining CRS but not completely. In clinical practice identifying the initial insult and subsequent events that result in decompensated acute on chronic CRS/renocardiac pathways can be challenging. Thus, currently practitioners diagnose patients with cardiorenal syndrome without specifications of the type. Through EPIC chart analysis of 95 patients, 45 patients (47%) at a point in their disease progression were diagnosed by their clinicians with CRS without specifications of the type.
In patients affected by chronic kidney disease (CKD), CVD, and HF are highly prevalent. Its presence as well as its worsening is associated with a worse prognosis. Therefore, an accurate renal function evaluation plays a key role in stratifying patients’ prognosis. Glomerular filtration rate (GFR) estimation based on serum creatinine levels is the easiest way to assess overall kidney function in clinical practice and it is routinely used. However, several limitations in serum creatinine use exist in chronic and acute settings. To overcome the caveats related to creatinine, biomarkers are being studied to evaluate patients with ESRD prone to worsening cardiac function, essentially CRS. 14 The goal of this study was to evaluate if thromboinflammatory biomarkers could be useful in assessing the severity of endothelial dysfunction and thrombosis, both of which are implicated in the pathogenesis of CRS.10,11 Targeting these mechanisms and directly these markers may present therapeutic opportunity in addition to the conventional treatment of dialysis to attenuate CRS progression.
Increased oxidative stress is an important determinant of endothelial dysfunction and cardiovascular and renal damage. Oxidative stress induces inflammatory cytokine production, impairs endothelial function, and induces mitogen-activated protein kinases which are all involved in cardiac hypertrophy and remodeling, as well as myocardial dysfunction.15,16 The heart and kidney have high mitochondrial content which presents one of the sources of reactive oxygen species (ROS) production under pathological conditions, leading to cardiorenal damage.17,18 ROS contributes to tissue inflammation with increased levels of inflammatory mediators, including IL-6, CRP, and TNF-α which are detected in both acute and chronic kidney disease and acute and chronic HF. 19 Irrespective of the cause of kidney disease, there is strong evidence that an inflammatory state exists in patients with CKD and ESRD. 20 Regarding this, recent studies have shown that the levels of inflammatory biomarkers such as CRP, TNF-α, IL-1, and IL-6 are elevated in CKD and ESRD patients.21,22 Furthermore, this elevation has also been shown to be associated with the progression of CKD to ESRD with higher rates of cardiovascular complications and mortality in this patient group. 23 In vivo experiments showed a significant increase of serum levels of proinflammatory cytokines in patients with type 1 CRS compared to those with acute HF without renal insufficiency which included IL-6, IL-18, MPO, and neutrophil gelatinase-associated lipocalin (NGAL). 19 The interaction of proinflammatory cytokine IL-6 with the renin angiotensin system may represent an important pathogenic mechanism in the atherosclerotic process which is accelerated in ESRD patients.24,25 In this study, similarly, CRP, TNF-α, and IL-6 had a statistically significant elevation in ESRD patients compared to normal controls. Additionally, TNF-α had a statistically significant elevation in ESRD patients with CRS compared to those without. Also, L-FABP is expressed in tubular epithelial cells and is excreted into urine with cytotoxic lipids. It has been shown that urinary L-FABP is associated with ischemic tubular injury and with the risk for acute kidney failure in type 1 CRS. 26 In this study, L-FABP had a statistically significant elevation in ESRD patients compared to normal controls, but no statistically significant difference was seen between ESRD patients with CRS to those without. In addition to a few inflammatory biomarkers that have already been studied, this study reported on inflammatory biomarkers that have not such as MCP-1 and Annexin V. Both, MCP-1 and Annexin V had a statistically significant elevation in ESRD patient compared to normal controls, but no statistically significant difference was seen between ESRD patients with CRS to those without.
Most inflammatory biomarkers have been studied, but thrombotic biomarkers have not. Given the pathophysiology of CRS, we hypothesized that thrombotic markers would be elevated in ESRD patients compared to normal controls and to a higher degree in ESRD patients with CRS compared to those without. This reported on biomarkers NO, vWF, D-dimer, and PAI-1. A statistically significant elevation was seen in all thrombotic biomarkers in ESRD patients compared to normal controls. Even antithrombotic biomarker NO was elevated in ESRD patients compared to normal controls. This result could be attributed to the timing samples were collected. In the early stages of ESRD, protective effects for vascular remodeling include vasodilation through NO. 7 Additionally, D-dimer was significantly elevated in ESRD patients with CRS compared to ESRD patients without CRS. The results of this study indicate biomarkers, TNF-α and D-dimer can be useful predictors of CRS in ESRD patients. Also, some of the thromboinflammatory biomarkers have shown significant correlations between each other. IL-6 was significantly correlated with D-dimer in patients with CRS (r = 0.37). This may reflect the relationship between inflammation and hemostatic fibrinolytic dysregulation in CRS patients.
This study has several limitations which should be noted. This is a retrospective study. The retrospective nature of this study also meant that it was difficult to exclude other drivers of thromboinflammation from our patient cohort which is difficult to assess from the medical record review. Also, this study had a small cohort size of ESRD patients, lack of random sampling and nonavailability of age matched controls. It is important to acknowledge that studies on biomarkers have just begun. Future studies are essential to observe current biomarkers and find novel biomarkers in CRS to improve CRS's prognosis, diagnosis, and treatment. In conclusion, endothelial dysfunction and oxidative stress play a role in the interplay between renal disease and cardiac disease by promoting atherosclerosis through vascular remodeling. Thromboinflammatory biomarkers can be used to better assess the severity of renal dysfunction and to detect patients at higher risk of renal function worsening to more accurately identify patients that are prone to CRS. Specifically, TNF-α and D-dimer can be useful predictors of CRS in ESRD patients.
Footnotes
Acknowledgments
The authors are grateful for the support and guidance from the faculty and staff of the Hemostasis and Thrombosis Laboratories, Cardiovascular Research Institute, and the Department of Pathology at Loyola University Medical Center. The authors are also thankful to Dr. Eva Wojcik, Chairperson of the Department of Pathology, for her support, and Director of the STAR program, Dr. Colleen Fitzgerald, for providing the opportunity for this project through the Student Training in Approaches to Research (STAR) and Research Honors programs. The authors are thankful to Mr. Jonas Kingo for providing some of the kits used for this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the National Heart, Lung and Blood Institute of the National Institutes of Health under award number T35HL120835.
