Abstract
Atrial fibrillation (AF) is prevalent in nearly 27% of patients with stage 5 chronic kidney disease on hemodialysis (CKD5-HD), suggesting a strong association between these 2 pathologies. It is hypothesized that the relationship between these 2 diseases may be mediated by inflammation. Angiopoietin-2 (Ang-2), a pro-inflammatory biomarker of endothelial instability, inflammation, and vascular remodeling, is elevated in CKD5-HD and AF, yet has not been evaluated in patients with concomitant AF and CKD5-HD. The aim of this study is to analyze circulating levels of inflammatory and thrombotic biomarkers in patients with concomitant AF and CKD5-HD. Plasma levels of Ang-2 were measured via sandwich enzyme-linked immunosorbent assay method in CKD5-HD patients (n = 96), patients with AF (n = 38), and controls (n = 50). Angiopoietin-2 was markedly elevated in CKD5-HD with comorbid AF as compared to CKD5-HD alone, and AF alone, respectively (13.05 ± 1.56 vs 9.57 ± 0.71 ng/mL; P = .00169; vs 2.48 ± 0.57 ng/mL; P < .0001). The results of this study suggest an additive effect of Ang-2 with coexistence of AF and CKD5-HD, which may be useful in the detection of AF within this patient population.
Introduction
Atrial fibrillation (AF) is prevalent in nearly 27% of patients with stage 5 chronic kidney disease on hemodialysis (CKD5-HD), and accelerates the progression of CKD, suggesting a strong relationship between these 2 pathologies within the cardiorenal axis 1,2 Additionally, the presence of AF or CKD5-HD predisposes patients to the other condition. 1,3 Atrial fibrillation is independently associated with increased hospitalizations in hemodialysis patients, as well as a 1.72-fold increased mortality risk, and a 9.8-fold increase risk of ischemic stroke. 4,5 The burden comorbid AF places on CKD5-HD patients warrants improved diagnostic and prognostic measures.
Inflammation as the Link Between AF and CKD5-HD
The link between these 2 conditions may be explained by their shared pathogeneses. Fibrosis and subclinical cardiac remodeling, increased activation of the sympathetic nervous system, thromboembolic events, endothelial dysfunction and oxidative stress, and inflammation play a role in the development of both CKD and AF. 6 –18
Increased pro-inflammatory activity is strongly associated with renal impairment and decreased glomerular filtration rate, as well as development of AF and disruption of atrial conduction. 16,19 –22 The role of classic mediators of inflammation has been well reported in these conditions individually, but little research evaluating the pro-inflammatory state linking these conditions has been conducted.
This study sought to investigate circulating levels of 11 biomarkers of inflammation, hemostasis, and thrombosis within AF patients, CKD5-HD, and both comorbidities. Angiopoietin-1 (Ang-1) and Angiopoietin-2 (Ang-2) are both growth factor glycoproteins involved in regulation of endothelium stability, vascular permeability, and angiogenesis. 23 The CD40 ligand (CD40-L) plays an important role in platelet and endothelial activation through the expression of adhesion molecules, as well as the interplay between thrombosis and inflammation within atherothrombotic disease. 24 C-reactive protein (CRP) and plasminogen activator inhibitor-1 (PAI-1) are acute phase reactants associated with cardiovascular disease, mortality, and vascular events in AF patients. 25,26 D-dimer is a fibrin degradation product that serves as an indicator of thrombosis, especially in the setting of deep vein thrombosis and pulmonary embolism. 27,28 Intracellular adhesion molecule-1 (ICAM-1) is a surface glycoprotein that may serve as a molecular marker of atherosclerosis and coronary heart disease. 29 NATCH, LRR, and PYD domains containing protein 3 (NALP3), also known as cryopyrin, is a pyrin-containing protein associated with inflammasome formation and apoptosis. Procalcitonin (PCT) is a propeptide upregulated during inflammation and cellular injury, and it has been characterized as a novel indicator of sepsis. Tumor necrosis factor (TNF)-α and von Willebrand factor (vWF) are pro-inflammatory peptides upregulated in multiple cardiovascular diseases and inflammatory settings. 20,30 Biomarker profiling may provide a noninvasive measure of AF in patients with CKD5-HD and be useful in risk stratification of this population.
Angiopoietin-2 and its Role in Inflammation, CKD5-HD, and AF
Angiopoietin-2 is a 70 kDa glycoprotein growth factor that is overexpressed in endothelial dysfunction, inflammation, and vascular remodeling. 31,32 The angiopoietin family of glycoproteins serve as ligands of the Tie2 receptor tyrosine kinase, ranging from full agonists, such as Ang-1, to partial agonists or antagonists, as in the case of Ang-2. 31,33 The Tie2 receptor tyrosine kinase is almost exclusively expressed in endothelial and hematopoietic cells, and tight regulation of Tie2 signaling is essential for vascular homeostasis and endothelial cell survival. Constitutive expression of the full agonist Ang-1 maintains endothelial quiescence in mature, healthy vasculature by binding and stabilizing Tie2, while Ang-2 prevents Ang-1-mediated vascular homeostasis by competing for its binding site. 31,34,35 Under homeostatic conditions, ligand binding of the Tie2 receptor by Ang-1 results in upregulation of PI3K/Atk cell survival signals, prevention of capillary leak through cell–cell junction stabilization, and tonic inhibition of NF-κB-dependent inflammatory gene expression, such as those involved in the regulation of leukocyte adhesion molecules. 36 Under inflammatory conditions, Ang-2 is released from Weibel-Palade bodies in a paracrine manner to compete for Tie2 bining. 37 Displacement of Ang-1 results in a pro-inflammatory state mediated through NF-κB gene expression, and increased capillary leak as a result of Rho-kinase activation and cell–cell junction breakdown. 36 After binding to endothelial cells, Ang-1 and Ang-2 are released into circulation where they can accumulate and become capable of binding to other healthy endothelial cells within the vasculature. 38
Elevated Ang-2 levels have been demonstrated in inflammatory conditions, such as acute coronary syndrome, congestive heart failure, and sepsis, as well as the conditions focused in this study, CKD5-HD and AF. 39 –44 Angiopoietin-2 levels have not been evaluated when considering both comorbid conditions together. As both AF and CKD5-HD are mediated by inflammatory processes and endothelial instability, it was hypothesized that plasma concentrations of inflammatory and thrombotic biomarkers, with a focus on Ang-2, may be elevated in AF, elevated in CKD5-HD, and further elevated when both comorbid conditions are present.
Materials and Methods
Plasma samples from patients with CKD5-HD (n = 97) and AF patients (n = 23-40) treated at the Loyola University Medical Center in Maywood, IL were analyzed in this study. Ages of the CKD5 patients ranged from 19 to 87 with a mean age of 60 years, while ages of AF patients ranged from 18 to 81 with a mean of 59 years. Medications of the CKD5-HD population included aspirin (n = 53), anticoagulants (n = 17), antihypertensives (n = 34), and calcium-channel blockers (n = 46). Sex distribution of the CKD5-HD cohort was 50.52% female and 49.48% male. Whole blood samples from CKD5-HD patients prior to dialysis, and AF patients were collected in 3.2% (0.109 mol/L) sodium citrate tubes and centrifuged at 1100 × g for 15 minutes within 2 hours from draw time. Platelet-poor plasma supernatant was separated, aliquoted, and stored at −8°C until time of analysis.
Normal Controls
Healthy, nonsmoking, drug-free normal volunteer human plasma (age range from 19 to 54 years, mean of 33 years) purchased through a commercial source (George King Biomedical, Overland Park, Kansas) served as controls.
Eligibility Criteria
Participation in this study required patients to be 18 years or older receiving hemodialysis treatment for stage 5 CKD at the outpatient hemodialysis clinic at Loyola University Medical Center, or treatment for AF at Loyola University Medical Center. Patients in critically ill condition, those requiring mechanical ventilation, and pregnant or nursing females were excluded from this study.
Electronic Medical Record Chart Review
Epic electronic medical record charts of the CKD5-HD patients were reviewed for age, sex, medications, white blood cell count, platelet count, and cardiac comorbidities. Comorbidities of interest included prior diagnosis of AF (paroxysmal, persistent, long-standing persistent, permanent, and nonvalvular subtypes), coronary artery disease, heart failure, left ventricular hypertrophy, and myocardial infarction as indicated by International Classification of Diseases diagnostic codes, catheterization laboratory results, or other physician-signed documentation prior to whole blood draw date. Patients with prior ablation therapy for AF were excluded from AF analysis. Levels of each biomarker compared CKD5-HD patients diagnosed with a comorbid condition stated above to CKD5-HD patients without the condition.
Sample Analysis
Circulating levels of biomarkers in sample plasma were measured using commercial sandwich enzyme-linked immunosorbent assay (ELISA) method. Biomarkers of interest included Ang-1, Ang-2, CRP, CD40-L, D-dimer, ICAM-1, NALP3, PAI-1, PCT, TNF-α, and vWF. Commercial ELISA kits were purchased through Abcam, Cambridge, United Kingdom (PCT), Hyphen Biomed, Neuville-sur-Oise, France (CRP, D-dimer, PAI-1, vWF), Lifespan BioSciences, Inc, Seattle, Washington (NALP3), and R&D Systems, Minneapolis, Minnesota (Ang-1, Ang-2, CD40-L, ICAM-1, TNF-α).
Statistical Analysis
Data analysis was completed using Microsoft Excel and GraphPad Prism version 7 software. Comparisons between groups utilized nonparametric Mann-Whitney t test and Kruskal-Wallis analysis of variance statistical analyses for nonuniformly distributed data. All results were expressed as mean ± standard error of the mean. P values less than .05 were considered statistically significant.
Results
Baseline Clinical Characteristics of the CKD5-HD Cohort
Table 1 demonstrates the prevalence of each cardiac comorbidity found in the CKD5-HD cohort, compared to documented prevalence. Atrial fibrillation was prevalent in 25.0% of CKD5-HD patients (n = 24). The CKD5-HD cohort included 97 patients, subdivided into CKD5-HD alone (n = 72), and CKD5-HD with comorbid AF (n = 24). One patient was excluded from CKD5-HD analysis of AF due to prior ablative therapy.
CKD5-HD Cohort Cardiac Comorbidity Prevalence Versus Documented Cardiac Comorbidity Prevalence.
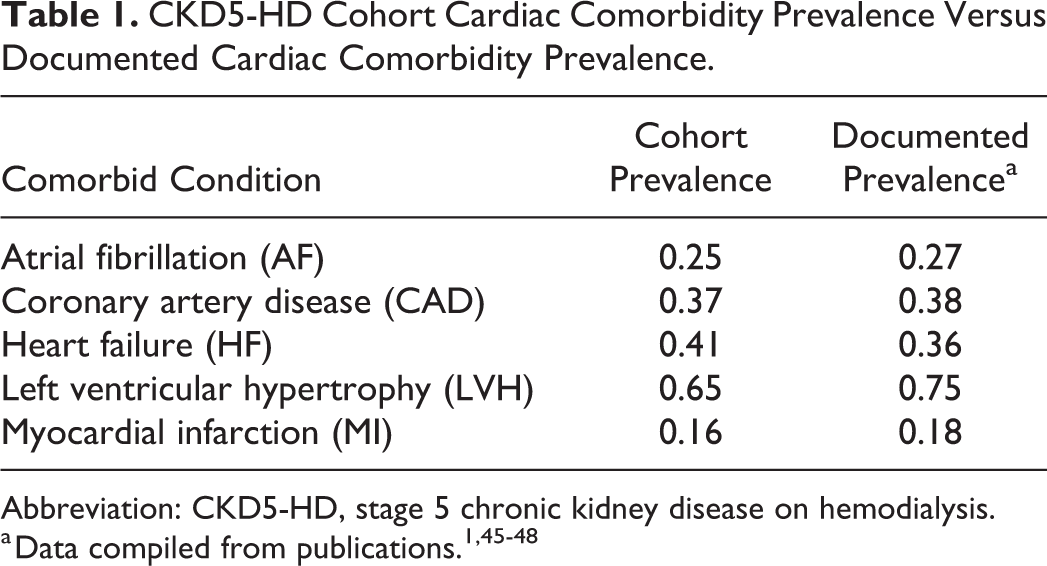
Abbreviation: CKD5-HD, stage 5 chronic kidney disease on hemodialysis.
Comparison of Thrombotic and Inflammatory Biomarkers in the CKD5-HD and AF Cohorts Versus Controls
The CKD5-HD group demonstrated significantly elevated levels of each biomarker when compared to controls, with exception of PAI-1 (Table 2). The Ang-1/Ang-2 ratio was significantly higher in the control population than in CKD5-HD patients (0.11 ± 0.08 vs 0.08 ± 0.01; P < .0001). Statistically significant differences between CKD5-HD and control levels all resulted in a positive percent change, except for that of the Ang-1/Ang-2 ratio which resulted in a −26.01% ± 10% decrease.
Comparison of Biomarker Levels Between Controls and CKD5-HD Patients.a
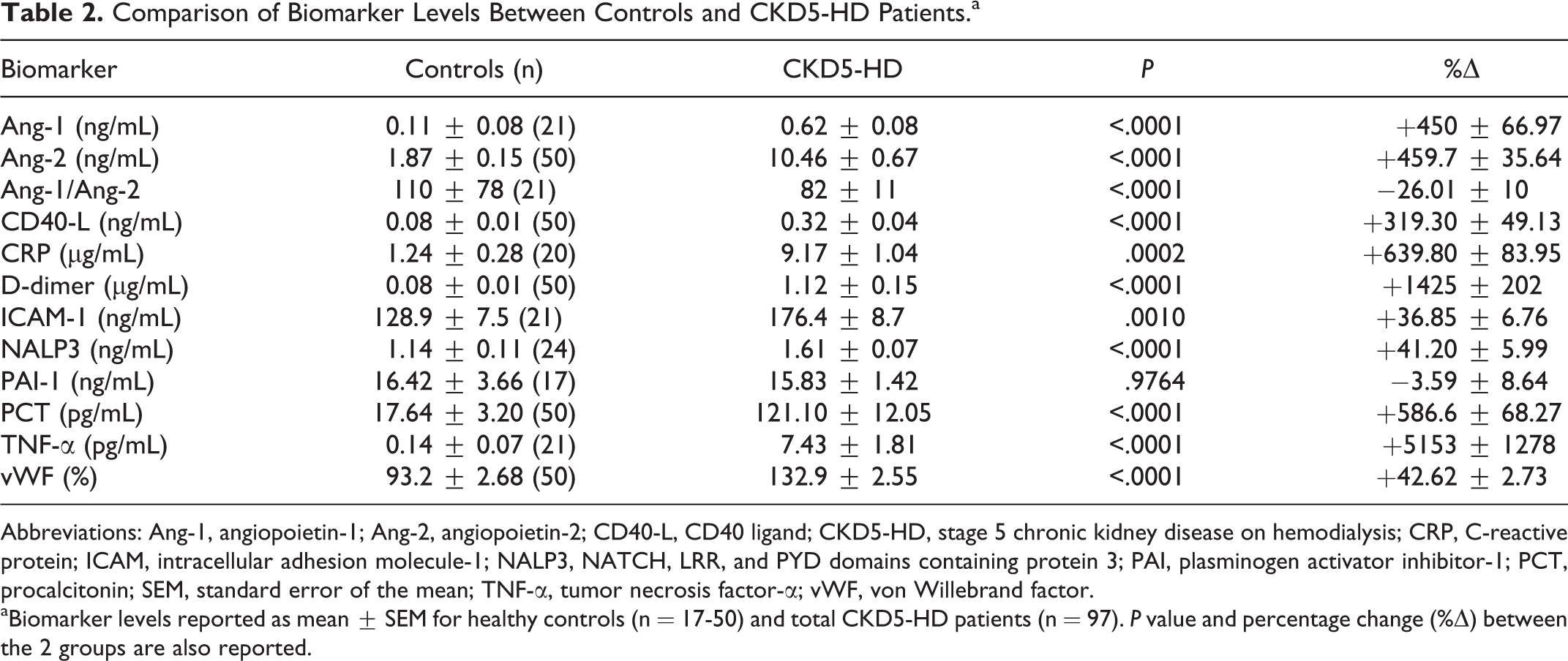
Abbreviations: Ang-1, angiopoietin-1; Ang-2, angiopoietin-2; CD40-L, CD40 ligand; CKD5-HD, stage 5 chronic kidney disease on hemodialysis; CRP, C-reactive protein; ICAM, intracellular adhesion molecule-1; NALP3, NATCH, LRR, and PYD domains containing protein 3; PAI, plasminogen activator inhibitor-1; PCT, procalcitonin; SEM, standard error of the mean; TNF-α, tumor necrosis factor-α; vWF, von Willebrand factor.
aBiomarker levels reported as mean ± SEM for healthy controls (n = 17-50) and total CKD5-HD patients (n = 97). P value and percentage change (%Δ) between the 2 groups are also reported.
As demonstrated in Table 3, AF patients had significantly elevated levels of Ang-1 (P < .0001), Ang-2 (P = .0257), CD40-L (P < .0001), ICAM-1 (P = .0206), PAI-1 (P = .0493), and TNF-α (P < .0001) when compared to controls, but not CRP (P = .8012) or D-dimer (P = .0650). All statistically significant differences between normal plasma and AF patient plasma resulted in a positive percent change. The Ang-1/Ang-2 ratio, as well as levels of NALP3, PCT, and vWF were not determined (ND) in the AF population.
Comparison of Biomarker Levels Between Controls and AF Patients.a
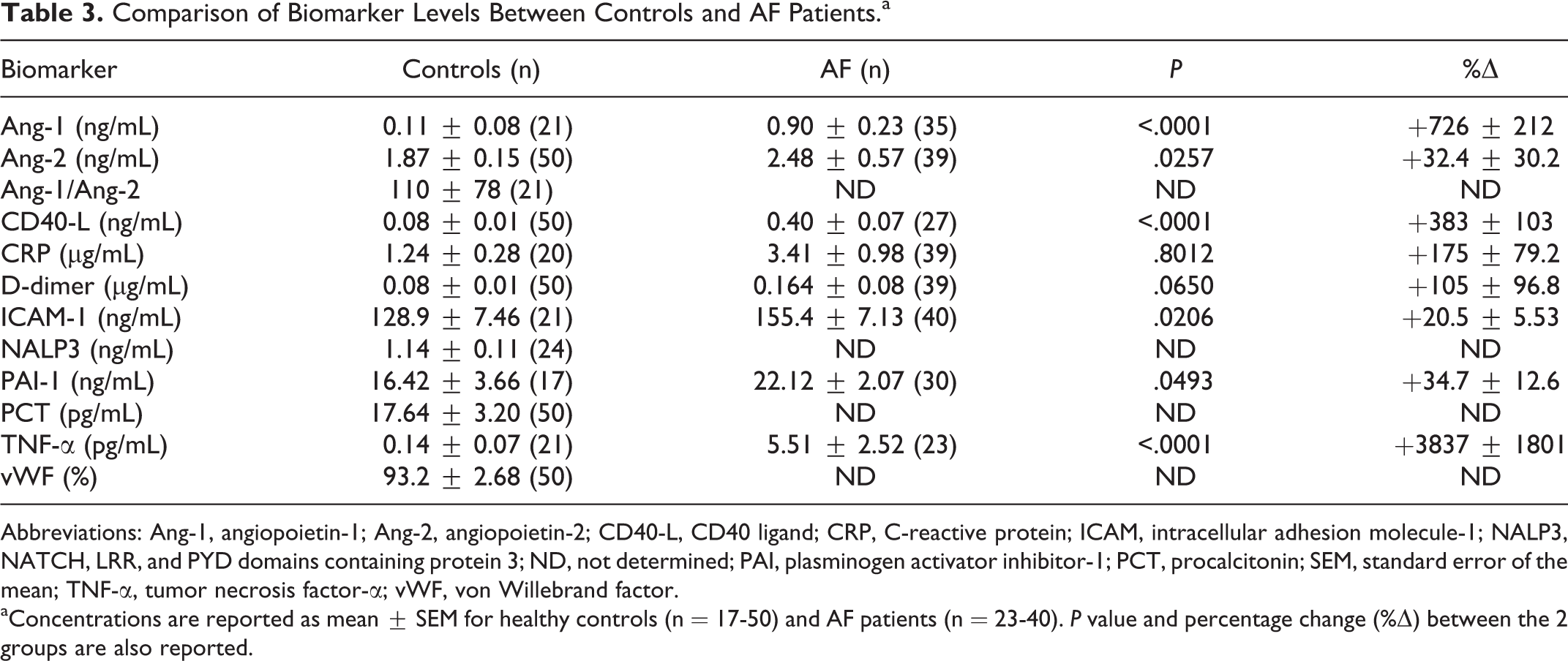
Abbreviations: Ang-1, angiopoietin-1; Ang-2, angiopoietin-2; CD40-L, CD40 ligand; CRP, C-reactive protein; ICAM, intracellular adhesion molecule-1; NALP3, NATCH, LRR, and PYD domains containing protein 3; ND, not determined; PAI, plasminogen activator inhibitor-1; PCT, procalcitonin; SEM, standard error of the mean; TNF-α, tumor necrosis factor-α; vWF, von Willebrand factor.
aConcentrations are reported as mean ± SEM for healthy controls (n = 17-50) and AF patients (n = 23-40). P value and percentage change (%Δ) between the 2 groups are also reported.
Comparison of Biomarker Levels Between CKD5-HD (+)AF and CKD5-HD (−)AF Patients, and CKD5-HD Versus AF.a
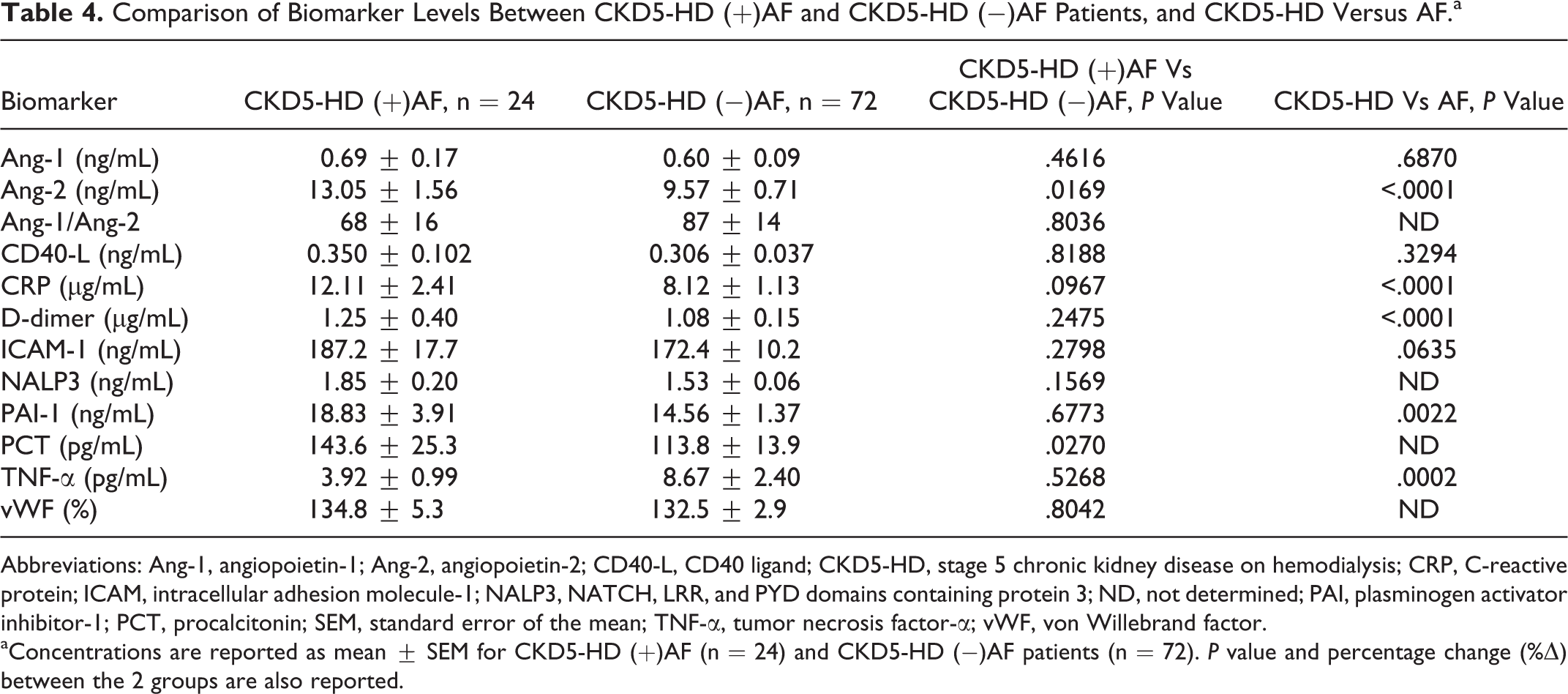
Abbreviations: Ang-1, angiopoietin-1; Ang-2, angiopoietin-2; CD40-L, CD40 ligand; CKD5-HD, stage 5 chronic kidney disease on hemodialysis; CRP, C-reactive protein; ICAM, intracellular adhesion molecule-1; NALP3, NATCH, LRR, and PYD domains containing protein 3; ND, not determined; PAI, plasminogen activator inhibitor-1; PCT, procalcitonin; SEM, standard error of the mean; TNF-α, tumor necrosis factor-α; vWF, von Willebrand factor.
aConcentrations are reported as mean ± SEM for CKD5-HD (+)AF (n = 24) and CKD5-HD (−)AF patients (n = 72). P value and percentage change (%Δ) between the 2 groups are also reported.
Comparison of Thrombotic and Inflammatory Biomarkers in the CKD5-HD Cohort Versus the AF Cohort
Plasma from the CKD5-HD cohort contained significantly higher levels of Ang-2 (P < .0001), CRP (P < .0001), D-dimer (P < .0001), PAI-1 (P = .0022), and TNF-α (P = .0002) in comparison to the AF cohort (Table 4). Levels of Ang-1, CD40-L, and ICAM-1 had no significant difference between groups. Comparisons between NALP3, PCT, and vWF were ND.
Thrombotic and Inflammatory Biomarkers in the CKD5-HD Cohort Comparing (+)AF With (−)AF
The only comorbid cardiac condition with considerable differences between biomarker levels was concomitant AF (Table 4). Angiopoietin-2 was significantly increased in CKD5-HD patients with comorbid AF (13.05 ± 1.56 ng/mL) versus patients with CKD5-HD alone (9.57 ± 0.71 ng/mL; P = .0169). Procalcitonin levels were also significantly elevated in CKD5-HD (+)AF patients (143.6 ± 25.33 pg/mL) compared to CKD5-HD (−)AF patients (113.8 ± 13.86 pg/mL; P = .0270).
Analysis of Ang-2 Within Controls, AF, CKD5-HD (−)AF, and CKD5-HD (+)AF
Normal control plasma measured the lowest level of Ang-2 (1.87 ± 0.15 ng/mL), while levels were elevated in AF (2.48 ± 0.57 ng/mL), further elevated in patients with CKD5-HD without AF (9.571 ± 0.7088 ng/mL), and highest levels were seen in patients when both CKD5-HD and AF were present (13.05 ± 1.559 ng/mL). Normal control plasma levels of Ang-2 were significantly lower than levels in AF (P = .0257), CKD5-HD (−)AF (P < .0001) or CKD5-HD (+)AF (P < .0001). Comparing Ang-2 levels in AF to CKD5-HD (+)AF or (−)AF, both were significantly elevated in CKD5-HD patients (P < .0001). Angiopoietin-2 was significantly increased in CKD5-HD patients with comorbid AF compared to CKD5-HD patients without AF (P = .0169).
Analysis of variance evaluation of Ang-2 levels between controls, AF patients, CKD5-HD (+)AF and CKD5-HD (−)AF revealed a significant difference between all groups (P < .0001). Multiple comparisons demonstrated significant differences (P < .0001) between controls and CKD5-HD patients regardless of the presence of comorbid AF (Figure 1). Neither sex, age, nor medications were found to have an impact on the results for Ang-2.
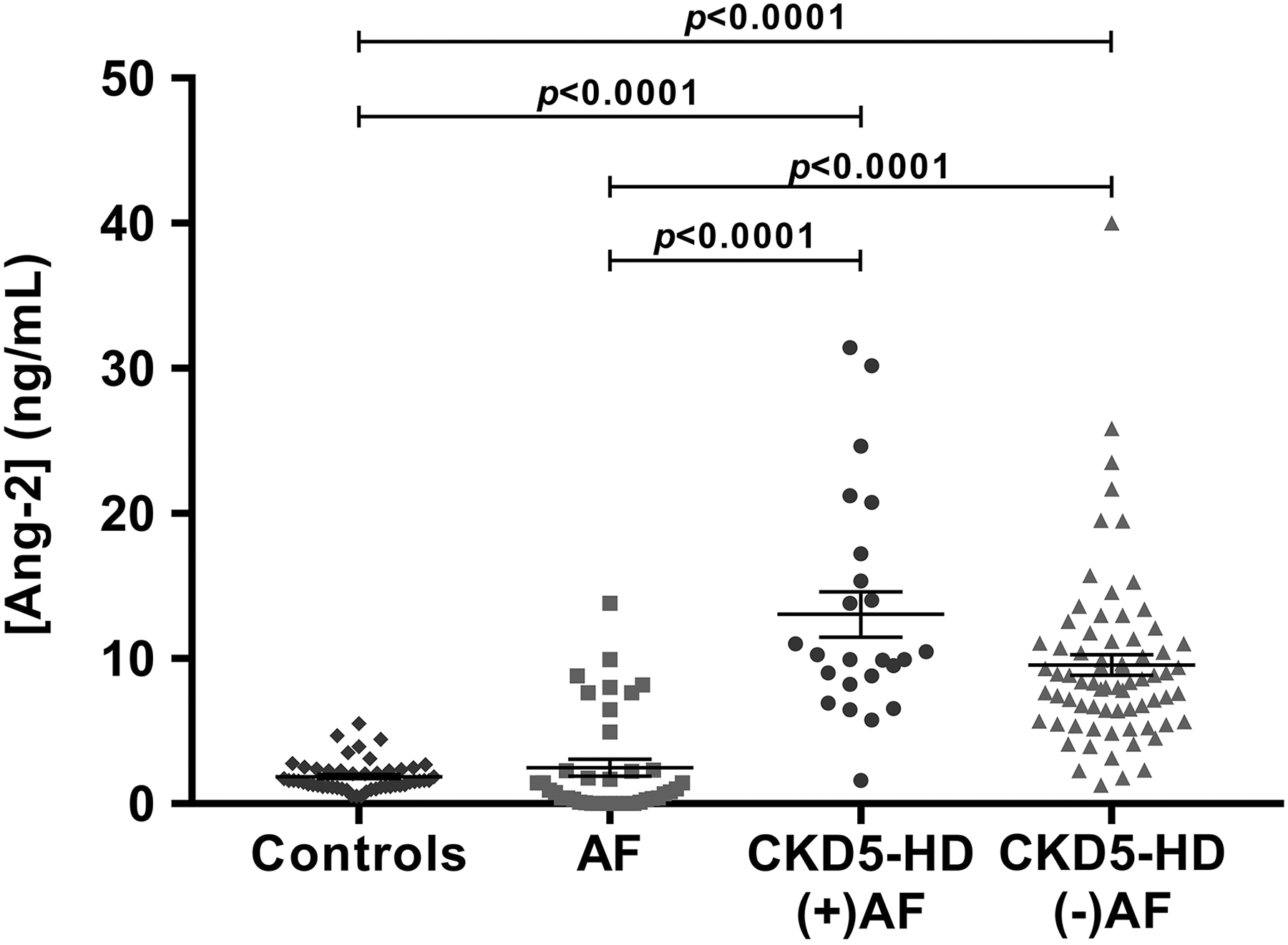
Comparison of Ang-2 levels between controls, AF, CKD5-HD (+)AF, and CKD5-HD (−)AF. Mean levels were 1.87 ± 0.15 ng/mL for controls, 2.48 ± 0.57 ng/mL for AF, 13.05 ± 1.56 ng/mL for CKD5-HD (+)AF, and 9.57 ± 0.71 ng/mL for CKD5-HD (−)AF. AF indicates atrial fibrillation; Ang-2, angiopoietin-2; CKD5-HD, stage 5 chronic kidney disease on hemodialysis.
Discussion
It is generally thought that endothelial dysfunction, hemostatic dysregulation, and the multiple foci of inflammation, all present within the CKD5-HD vascular milieu, may contribute to the exacerbation of AF within this patient population. 3,12 The results of this study are consistent with the well-documented presence of enhanced inflammation and hemostatic dysfunction in CKD5-HD and AF. 16,19 –22 We found that almost all circulating inflammatory markers were elevated in the CKD5-HD patient cohort, with exception of PAI-1. Multiple studies have demonstrated PAI-1 depletion in hemodialysis patients due to interactions with unfractionated heparin and resultant removal. 49,50 Most markers were also elevated in the AF cohort. In a comparison between CKD5-HD and AF patients, there were significant increases in levels of most biomarkers in the former, demonstrating an increased burden of inflammation associated with CKD and subsequent hemodialysis treatment. This may be explained by reduced continuous physiological fluid and toxin removal, in addition to activation of inflammatory responses through blood-surface contact during the hemodialysis procedure. 51 This hemodialysis-influenced pro-inflammatory state may contribute adversely to vascular health.
We also demonstrated that there is a further stepwise increase in Ang-2 in CKD5-HD (+)AF group compared to CKD5-HD alone, and AF alone, respectively. Our study is the first to demonstrate that there may be an additive increase in inflammation in CKD5-HD with comorbid AF, as suggested by the observed upregulation of Ang-2 levels. Further analysis of this biomarker through receiver–operator curves and positive predicative values can only be carried out in a randomized study including an appropriate number of participants. The only other biomarker to discern the presence of AF within the CKD5-HD cohort was PCT, yet as this biomarker has not been investigated in the AF cohort; further analysis of PCT is required.
The elevation of Ang-2 within the CKD5-HD and AF may be explained by the role of this biomarker in the induction of inflammation and sensitization of the endothelium to downstream pro-inflammatory cytokines. 52 Angiopoietin-2 antagonizes the Tie2 receptor, outcompeting Ang-1 for upholding endothelial quiescence, thus causing endothelial disfunction and activation of the inflammatory response. Angiopoietin-2 expression has also been demonstrated to sensitize the endothelium to other mediators of inflammation, such as TNF-α. It is reasonable that this biomarker would be elevated in both conditions, as the role of inflammation in mediating both AF and CKD5-HD has been well established, yet the interesting finding in this study is the additive phenomenon of Ang-2 in the CKD5-HD patient population when analyzing comorbid AF. Angiopoietin-2 may be playing a crucial role in the regulation of inflammation in both these conditions, and therefore upstream of the other biomarkers in this study. Although Ang-1 and Ang-2 compete for Tie2 binding, the circulating levels of Ang-1 were not decreased, but significantly increased in both CKD5-HD and AF. However, analysis of the Ang-1/Ang-2 ratios was significantly deceased in the CKD5-HD cohort when compared to controls, demonstrating the antagonistic interaction between these 2 proteins, possibly due to upregulation of Ang-1 as a protective measure in an attempt to outcompete Ang-2. The Ang-1/Ang-2 ratio was not calculated in the AF cohort due to many of the patients exhibiting zero values of Ang-2, yet may be inferred to be decreased when comparing the +726% ± 212% positive change in Ang-1 levels compared to a +32.4% ± 30.2% positive change in Ang-2 levels. Lower Ang-1/Ang-2 ratios have demonstrated to be of use in multiple conditions, including predicting advancing disease and lower survival in multiple myeloma, and preeclampsia prediction. 53,54
As inflammation plays a large role in the mediation of both AF and CKD5-HD pathologies, it was expected that the levels of the different biomarkers of this study would be projected in a similar manner to that of Ang-2. None of the classical markers of inflammation or thrombosis were significantly elevated in patients with both comorbidities when compared to CKD5-HD alone. We speculate 3 reasons for this finding. Firstly, the chronic inflammation at play in these conditions may not follow a classical activation pathway, but may be part of an alternative inflammatory sequence, with Ang-2 as a large contributor. Secondly, as Ang-2 may mediate the expression and sensitization of the other inflammatory biomarkers investigated in this study, other homeostatic mechanisms may alter the levels of these more downstream mediators of inflammation. Thirdly, perhaps the overwhelming amount of inflammation in CKD5-HD compared to AF masks the presence of AF for some, but not other biomarkers. For example, levels of D-dimer in CKD5-HD patient plasma were nearly 7-fold higher than AF patient plasma. Thus, any slight elevation of D-dimer levels due to comorbid AF, in addition to the preexisting high levels in CKD5-HD, may be negligible.
One strength of this study is that the prevalence of each cardiac comorbidity within this CKD5-HD cohort closely reflects documented values, allowing this population to be more generalizable and comparable to other CKD5-HD populations (Table 1).
Limitations include a lack of age-matched controls. The control cohort used in this study was younger than the CKD5-HD and AF cohorts (means of 33 years, 60 years, and 59 years, respectively), although there was large overlap between age ranges. Another limitation of this study includes the single location of the study. All patients in both the CKD5-HD cohort and AF cohort were being treated at the Loyola University Medical Center; therefore, the demographics of study participants may not reflect those at other medical institutions. The sample size for each biomarker in the AF group ranged from 23 to 40 patients, which was primarily due to the limited amount of plasma available for the multiple parameters included in this study. Furthermore, it is difficult to conduct an age-based analysis due to the small cohort sizes.
Conclusions
This study demonstrated that Ang-2 is significantly elevated in AF and CKD5-HD patients, and further elevated in patients with both conditions. Our results suggest there is an additive effect of comorbid AF and CKD5-HD in relation to Ang-2. Further studies are needed to investigate the role of Ang-2 in the pathogenesis of these comorbidities, as well as its impact on health outcomes.
The elevation of Ang-2 plasma concentrations in CKD5-HD (+)AF patients compared to CKD5-HD (−)AF patients, as well as AF patients and normal controls, suggest that Ang-2 plays a significant role in the pathogenesis of AF within CKD5-HD, and may contribute to the overall increase in inflammation in CKD5-HD. Previous studies have demonstrated elevated levels of inflammatory and thrombotic biomarkers in CKD5-HD and AF, yet have not demonstrated comorbid AF to further amplify discernable levels in CKD5-HD patients. Additional investigation of the predictive value of Ang-2 in these patients should be considered to elucidate the relevance of increased Ang-2 level to adverse outcomes.
Footnotes
Authors’ Note
Ethical approval to report this case was obtained from the institutional review board of Loyola University Chicago. Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Acknowledgments
The authors are grateful for the support and guidance from the faculty and staff of the Thrombosis and Hemostasis Laboratories, Cardiovascular Research Institute, and the Departments of Pathology and Medicine at the Loyola University Chicago Medical Center. The authors are also thankful to Dr Eva Wojcik, Chairperson of the Department of Pathology, for her support, and Dr Gail Hecht for providing the opportunity of this project through the Student Training in Approaches to Research (STAR) and Research Honors programs. The authors are thankful to Mr Jonas Kingo for providing some of the kits used for this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the National Heart, Lung and Blood Institute of the National Institutes of Health under award number T35HL120835. The data in this article were presented at the American Heart Association Scientific Sessions 2017.
