Abstract
Background:
β-Blockers are first-line agents for reduction in symptoms, hospitalization, and mortality in patients with heart failure having reduced ejection fraction (HFrEF). However, the safety and efficacy of continuous β-blocker therapy (BBT) in patients who actively use cocaine remain controversial, and available literature is limited. We aimed to evaluate the effect of BBT on hospital readmission and mortality in patients having HFrEF with concurrent cocaine use.
Methods:
We conducted a retrospective study of patients with a diagnosis of HFrEF between 2011 and 2014 based on International Classification of Diseases 9-Clinical Modification codes. We included patients aged 18 and older who tested positive for cocaine on a urine toxicology test obtained at the time of index admission. Patients were followed for 1 year. Multivariate logistic regression was used to assess the effect of BBT on the 30-day, all-cause and heart failure–related readmissions.
Results:
The 30-day readmission rates for BBT versus no BBT groups were 20% versus 41% (odds ratio [OR]: 0.17, 95% confidence interval [CI] = 0.05-0.56, P = .004) for heart failure-related readmissions and 25% versus 46% (OR: 0.19, 95% CI = 0.06-0.64, P = .007) for all-cause readmissions.
Conclusion:
The BBT reduced 30-day, all-cause and heart failure–related readmission rate but not 1-year mortality in patients having HFrEF with concurrent cocaine use.
Introduction
Background
β-Blockers are 1 class of medication that has been shown to prevent maladaptive left ventricular (LV) remodeling in patients with low LV ejection fraction. 1,2 The mean all-cause mortality reduction in patients with heart failure having reduced ejection fraction (HFrEF) treated with β-blockers is 35%. 3 The current heart failure guidelines from American College of Cardiology (ACC)/American Heart Association (AHA)/Heart Failure Society of America (HFSA) recommend use of β-blocker therapy (BBT) in all patients with HFrEF unless contraindicated. 1,4,5
However, some studies caution against the use of β-blockers in patients who actively use cocaine. 6 –8 In general, many reviews recommend against BBT in patients with clinical evidence of cocaine toxicity 2,4,6 –9 presenting with acute decompensated heart failure. This was based on the theory that β-receptor blockade leaves the α-receptors unopposed in the setting of active cocaine use in humans. 4,6 –8,10,11 Unopposed α-adrenergic receptor stimulation may theoretically worsen cocaine-related cardiovascular complications such as acute LV dysfunction, cardiogenic shock, 12 acute coronary syndrome, hypertensive emergency, aortic dissection, stroke, and sudden death. 2,6,13 On the other hand, recommendation for BBT in patients with HFrEF concurrently using cocaine is unclear, at worst controversial, and available literature is limited. A systematic review of the pharmacologic treatment of cocaine cardiovascular toxicity revealed β-blockers to be safe and efficacious. 14 Furthermore, the authors of all published cases of unopposed α-adrenergic receptor stimulation felt this inconsistent and unpredictable phenomenon was most likely the result of cocaine alone and not a side effect of β-blocker use. 15
As unopposed α-receptor activation theoretically may occur in active cocaine users continuously exposed to nonselective BBT for heart failure management, β-1 selective β-blockers such as metoprolol, and combined α/β-blockers such as labetalol and carvedilol would be expected to obviate this safety concerns. There is no specific practice guideline that addresses continuous outpatient BBT in patients having HFrEF with concurrent cocaine use. Therefore, we undertook this study to describe the clinical experience and impact of BBT on hospital readmission and mortality in patients having HFrEF with concurrent cocaine use seen in our institution.
Materials and Methods
Study Design
We performed a single-center retrospective cohort study of patients with a diagnosis of heart failure based on International Classification of Diseases, Ninth Revision, Clinical Modification codes seen at Grady Memorial Hospital, Atlanta, Georgia, between January 2011 and December 2014. Grady Memorial Hospital is about a 1000-bed safety net hospital that provides medical care for residents of the city of Atlanta and its metropolis.
Selection of Participants
We included all adult patients aged 18 years and older who had HFrEF and who tested positive for cocaine on a urine toxicology test obtained at the time of index admission. Using our institution’s heart failure registry, we identified 6025 consecutive patients with a diagnosis of heart failure between January 2011 and December 2014. Three hundred and twenty-five (5% of the total population) patients tested positive for cocaine using a urine toxicology screening test obtained during index admission. Fifty-seven patients had an ejection fraction greater than 40% and were excluded. The final sample size used for analyses consisted of 268 patients. Electronic medical records were reviewed by trained investigators to ensure accuracy and completeness of data points. An institutional review board waiver of consent was obtained. Ethical approval was also obtained from our institution’s research oversight committee. Medication refill for patients who obtained their medication from our hospital pharmacy was confirmed by retrospective chart review. Also, we verified that each patient had outpatient β-blocker prescription for at least 1 year. Continuous outpatient BBT was defined as outpatient prescription and refills for carvedilol or metoprolol succinate for the entire follow-up period.
Data Collection and Processing
We abstracted data from our heart failure registry using the electronic medical record and included patient medical history, relevant laboratory result, number of hospital readmissions, and medications prescribed inpatient and outpatient. We conducted a manual review of each patient’s electronic medical record to verify accuracy of data. We specifically ascertained that a β-blocker was prescribed during the study period and also documented the number of readmissions and reason for readmission within the study period.
Measures
Outcomes
The primary outcome was 30-day, all-cause and heart failure–related readmissions. The secondary outcome was 1-year, all-cause mortality rate.
Covariates
Covariates included in the analysis were age, gender, smoking status, alcohol abuse status, patient comorbidities including hypertension, diabetes, hyperlipidemia, coronary artery disease, asthma, chronic obstructive pulmonary disease, chronic kidney disease, stroke, and peripheral vascular disease.
Primary Data Analysis
The study population characteristics and outcomes were described using mean and standard deviation (SD) for continuous variables and number with percentage for categorical variables. A χ2 test was used in a bivariate analysis to assess the effect of BBT on number of readmissions. A multivariate logistic regression model including all significant covariates was used to assess the effect of BBT on number of readmissions in an adjusted model. We accounted for potential confounders in the adjusted model. In all analyses, a significant P value level was set to .05. We reported the 30-day readmissions rate for all-cause and heart failure–related readmissions. The 1-year mortality rate was also reported. Analyses were performed using SAS, version 9.4 (SAS Institute, Cary, North Carolina).
Results
Our study included 268 patients with a diagnosis of HFrEF who tested positive for cocaine on a urine toxicology obtained at the time of index admission. The mean age of our study population was 54 (SD 7) years. Baseline characteristics including comorbidities are described in Tables 1 and 2. Result of multivariate analysis is presented in Table 3.
Baseline Characteristics Based on β-Blocker Therapy on Discharge.
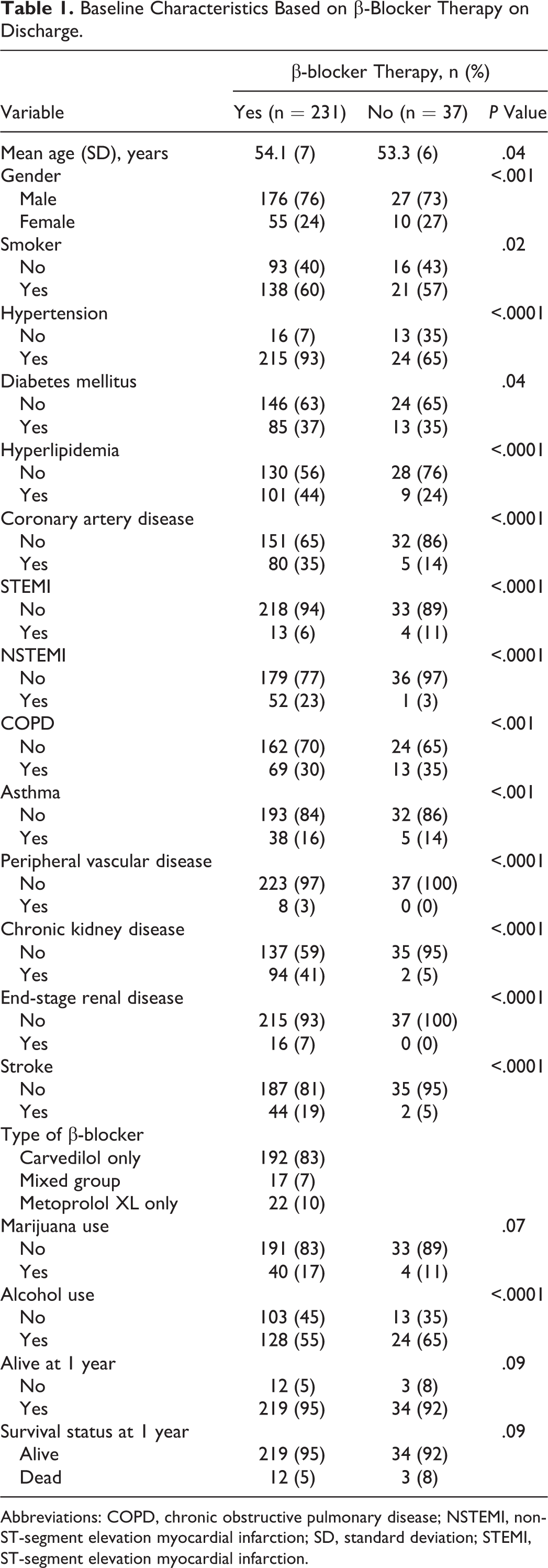
Abbreviations: COPD, chronic obstructive pulmonary disease; NSTEMI, non-ST-segment elevation myocardial infarction; SD, standard deviation; STEMI, ST-segment elevation myocardial infarction.
Proportion of Patients With 30-Day Readmission by β-Blocker Therapy.a

Abbreviation: OR, odds ratio.
a Sample size = 268; β-blocker therapy = 231; and no β-blocker therapy = 37.
Multivariate Analysis With Adjusted Odds Ratios for Outcomes (30-Day Readmission and 1-Year Mortality).
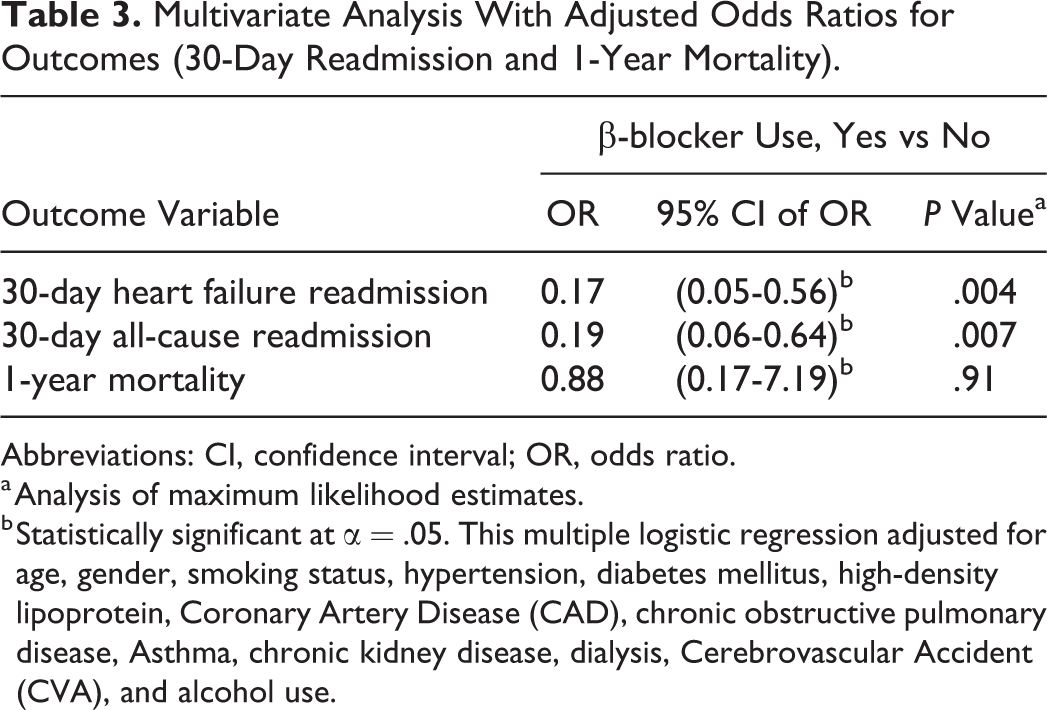
Abbreviations: CI, confidence interval; OR, odds ratio.
a Analysis of maximum likelihood estimates.
b Statistically significant at α = .05. This multiple logistic regression adjusted for age, gender, smoking status, hypertension, diabetes mellitus, high-density lipoprotein, Coronary Artery Disease (CAD), chronic obstructive pulmonary disease, Asthma, chronic kidney disease, dialysis, Cerebrovascular Accident (CVA), and alcohol use.
Approximately 86% of patients with HFrEF who tested positive for cocaine on index admission received a prescription for a β-blocker at the time of discharge from our institution. The 30-day readmission rates for BBT versus no BBT groups were 20% versus 41% (adjusted odds ratio [OR]: 0.17, 95% confidence interval [CI] = 0.05-0.56, P = .004) for heart failure–related readmissions and 25% versus 46% (adjusted OR: 0.19, 95% CI = 0.06-0.64, P = .007) for all-cause readmissions. After adjusting for all covariates, the rate of heart failure–related readmissions within 30 days of discharge from index admission was 5.9 times lower in BBT patients compared to those who did not receive BBT. The incidence of all-cause readmissions within 30 days of discharge from index admission was 5.3 times lower in patients that received outpatient BBT compared to those who did not.
The 30-day readmission rates for the treatment group in our study population were higher compared to the overall rates reported in general for patients with heart failure in our institution. The historical 30-day, all-cause readmission rate for all patients with heart failure in our institution during this period was 13.7%. The difference in the 30-day heart failure–related readmission rate between our institutional historical average and BBT group in this study is 6.3% (13.7% vs 20%). This margin of difference was greater compared to the difference in the margin of all-cause readmission rate between our institutional historical average and the BBT group in this study 3% (22% vs 25%). However, this comparison should be viewed with caution, as the institutional historical rates reflects all phenotypes of heart failure (HFrEF and heart failure with preserved ejection fraction) and includes both cocaine and noncocaine users.
The 1-year, all-cause mortality rates for BBT versus no BBT groups were 5% versus 8% (OR: 0.88, 95% CI = 0.17-7.19, P = .91). Although the odds of death in 1 year was lower in patients that received BBT, there was no significant difference in 1-year mortality rate between the BBT and the no BBT groups.
Discussion
Principal Findings
Our study indicates that majority of patients having HFrEF with concurrent cocaine use were prescribed β-blockers. It also showed that BBT in these patients significantly decreased both heart failure–related and all-cause readmissions but had no effect on 1-year mortality.
Although studies have included ejection fraction as one of the clinical measures most consistently improved by β-blockade in patients with HFrEF, 16 we encountered considerable limitation in evaluating the effect of BBT on ejection fraction in this retrospective cohort. This was due to significant variation in interval of follow-up echocardiograms and measurements of ejection fraction for the different patients included in the study.
In order to systematically evaluate this relationship, baseline and follow-up assessment of LV ejection function at prespecified time will be required in study participants. Another challenge is the fact that a vast majority of patients were receiving BBT due to its proven clinical benefit, leaving a small sample size for comparison. In our study, 86% of our study population was on BBT. Moreover, the retrospective design of this study is compounded by lack of a meaningful way of estimating the frequency and magnitude of exposure to cocaine. This latter concern also creates a challenge even with a prospective study design. According to the current heart failure guidelines from ACC/AHA/HFSA, the use of a β-blocker in patients with HFrEF is a class 1a indication. 1 Therefore, it would be unethical to propose a study that would withhold BBT.
Cocaine has been shown to have a variety of acute effects on the cardiovascular system, including suppression of myocardial contractility resulting in acute congestive heart failure in toxic levels, 13 reduction in coronary artery caliber, arrhythmias, increase in heart rate and blood pressure, and sudden death. 6 The chronic effects of cocaine use on the heart include myocarditis, suppression of LV systolic function, 17 dilated cardiomyopathy, 6,13 and increased susceptibility to a superimposed insult. 18 Dattilo et al 13 suggested that the protective effect of BBT on cardiac myocytes may be more important in determining clinical outcomes than its effect on coronary arteries. Moreover, reversal of cocaine-induced cardiomyopathy has been achieved with BBT in patients who use cocaine. 19 However, this beneficial effect of BBT was reported after abstinence from cocaine use. Furthermore, Littmann et al 2 reported that patients with HFrEF can occasionally experience complete echocardiographic and clinical recovery when treated with maximum dose carvedilol, despite concurrent cocaine use. However, metoprolol succinate and bisoprolol were not tested in their study. The improvement in ejection fraction and clinical recovery appears to be related to the ability of β-blockers to reverse LV remodeling and block the cardiovascular effect of the neurohormonal activation that occur in patients with HFrEF.
We did not evaluate for the impact of BBT on LV ejection fraction in patients with concurrent cocaine use in this study because of the previously mentioned limitations. The reduction in mortality seen in patients with HFrEF treated with β-blocker 20,21 may be mediated through the remodeling of LV systolic function.
Similarly, Tsuchihashi-Makaya et al demonstrated that BBT at the time of discharge was associated with a long-term survival benefit in a diverse cohort of patients with heart failure. 22 However, no information was provided on status of cocaine use in their study. In our study, there was no significant difference in 1-year, all-cause mortality for patients with HFrEF on BBT with concurrent cocaine use compared to those who did not receive BBT. Readmission rate as high as 50% has been reported in patients with congestive heart failure within the first few months of discharge from an index admission. 23 It is well established that the frequency and duration of heart failure–related hospitalizations are the major contributors to the huge annual economic burden of heart failure. 24 –27 Although current evidence shows that BBT decreases heart failure–related readmissions in patients with HFrEF, 24,28 we are not aware of any report in the literature with similar finding among patients with HFrEF concurrently using cocaine. In their study, Nguyen et al reported no significant differences in heart failure readmissions, death, major adverse cardiovascular events (MACE), or all composite end points in patients having heart failure with cocaine use versus patients having heart failure without cocaine use when treated with a β-blocker. 29 Our study did not compare MACE or other clinical end points of BBT in patients with HFrEF based on their cocaine use status. However, we demonstrated that patients having HFrEF with a history of cocaine use have significantly reduced 30-day, all-cause and heart failure–related readmissions when discharged on a β-blocker. Nguyen et al also reported no significant difference in mortality rates between patients having heart failure with cocaine use treated with a cardioselective β-blocker versus a non-cardioselective β-blocker. 29 Our study did not evaluate the pharmacologic profiles of different β-blockers. Nevertheless, we demonstrated that there was no 1-year mortality difference in patients having HFrEF with concurrent cocaine use discharged on a β-blocker versus patients not discharged on a β-blocker. Clinicians appear to be correctly disregarding the cocaine and β-blocker contraindication dogma and treating cocaine-associated heart failure with β-blockers with impunity.
Clinical Implications
In the United States, cocaine use has risen to epidemic proportions in the recent years, and cardiovascular complications associated with cocaine have become progressively more frequent. 19 With the growing number of cocaine users and the large numbers of cocaine-related cardiovascular diseases such as HFrEF, increasing numbers of patients are on continuous BBT. There is need for further studies to determine the clinical outcomes of BBT in this cohort of HFrEF. The impact on MACE, mortality, and readmissions will all be important clinical end points.
Limitations
We defined continuous BBT as outpatient prescription and refills for the period of follow-up. We were unable to confirm medication adherence for patients who obtained their medication from an external pharmacy. It is possible that some of these patients received an outpatient prescription but did not actually refill them. Moreover, medication refill does not necessarily indicate adherence with medication. Also, data on readmissions were generated solely from our center. We did not account for readmissions to other hospitals during the study period. The retrospective nature of this study cohort limits our ability to make any inferences regarding frequency and total duration of cocaine use. We recognize the limitation of the small sample size on the interpretation of our result.
Conclusion
Clinicians continue to prescribe β-blocker for most patients with HFrEF regardless of concurrent cocaine use. Our data showed that BBT significantly reduced readmission rate in patients with HFrEF who are concurrent cocaine users. While acute use of β-blockers during acute cocaine intoxication is still controversial, the use of β-blockers at discharge should be encouraged, particularly in patients with reduced systolic function. Large observational studies are needed to further elucidate the efficacy and safety of BBT as well as the pharmacologic profiles of different β-adrenergic antagonists in this population. At this time, there is insufficient clinical evidence to recommend withholding BBT in this population. However, close clinical follow-up of the patients while on BBT is recommended.
Footnotes
Authors’ Note
Some of the results described in this manuscript were presented at the Heart Failure Society of America scientific meeting in September 2017. All authors read and approved the final manuscript.
Acknowledgments
The authors thank Grady Memorial Hospital information technology department for their assistance with this study.
Authors’ Contribution
Egbuche, O contributed to conception and design; acquisition, analysis, and interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Ekechukwi, I and Maduabum, N contributed to acquisition, analysis, and interpretation; critically revised the manuscript; and gave final approval. Effoe, V. contributed to acquisition, critically revised the manuscript, and gave final approval. Millard, H. contributed to design and analysis, critically revised the manuscript, and gave final approval. Maihemuti, A. contributed to conception and design and acquisition, critically revised the manuscript, and gave final approval. Cross, J. contributed to conception, acquisition, and gave final approval. Adedinsewo, D. contributed to conception and design, acquisition and analysis, critically revised the manuscript, and gave final approval. Onwuanyi, A. contributed to conception and design, contributed to interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
