Abstract
Objective
Currently, there is limited evidence regarding the use of direct oral anticoagulants (DOACs) for patients with liver cirrhosis (LC). We performed a meta-analysis to compare the efficacy and safety of DOACs versus warfarin in this population.
Methods
We searched PubMed, Cochrane Library, Web of Science, Embase and Scopus databases from inception to August 2024. Clinical studies comparing the use of DOACs with warfarin in cirrhotic patients were included. Hazard ratios (HRs) with 95% CIs were estimated by either fixed or random effects models. Primary efficacy outcomes were ischemic stroke/thromboembolism (IS/TE) and all-cause death, the primary safety outcomes were the bleeding risks.
Results
Sixteen studies were included (16 829 individuals). DOACs had similar benefits in preventing IS/TE (HR = 0.87, 95% CI: 0.69-1.10), but DOACs were significantly associated with reduced risk of all-cause death (HR = 0.87, 95% CI: 0.79-0.97). On the other hand, we observed significantly reduced risks of any bleeding (HR = 0.60; 95%CI: 0.37-0.95), major bleeding (HR = 0.72; 95%CI: 0.63-0.82), intracranial hemorrhage (ICH) (HR = 0.47; 95%CI: 0.30-0.73), and gastrointestinal bleeding (GIB) (HR = 0.72, 95% CI: 0.60-0.87) in patients receiving DOACs. Results were consistent in cirrhotic patients with AF. Furthermore, DOACs reduced the incidence of major bleeding (HR = 0.65, 95%CI: 0.55-0.78) and ICH (HR = 0.17, 95%CI: 0.04-0.76) in patients with moderate to severe cirrhosis.
Conclusion
Our study demonstrates that DOACs, compared with warfarin, exerted comparable efficacy and better safety and may represent a safer alternative to warfarin in cirrhotic patients.
Introduction
Patients with cirrhosis are at a higher risk of both bleeding and thrombosis due to an imbalance between pro- and anticoagulant homeostasis.1,2 Studies have confirmed that patients with cirrhosis are prone to experience thrombosis in the portal as well as systemic circulation venous, 3 which was linked to the altered hemostatic balance, in particular, an increase in thrombin generation as well as a decrease in coagulation factors except for factor VIII.4-6 Recent investigations indicated that patients with LC had a higher risk of stroke or venous thromboembolism (VTE) than those without LC.7-10 Additionally, atrial fibrillation (AF) is more common in patients with cirrhosis.11-14 A previous study reported higher risks of stroke and ICH in cirrhotic patients with AF than patients with AF alone. 7 Furthermore, available research shows that hypercoagulability may promote hepatic decompensation and the development of fibrosis, 15 whereas prophylactic anticoagulation appears to improve clinical outcomes in patients with cirrhosis by delaying the occurrence of hepatic decompensation. 16 Therefore, patients with cirrhosis may have AF in addition to other risk factors for thromboembolic events and treatment with anticoagulants should be considered.
Vitamin K antagonists (VKAs) are currently considered the standard of care for stroke prevention in AF, and they also serve as the standard treatment for VTE prevention in patients with cirrhosis. 6 VKAs, particularly warfarin, have been used for the management and prophylaxis of thrombosis in LC.7,17 Despite the proven efficacy, its narrow therapeutic index and dose adjustment requirements limited the use of warfarin. However, direct oral anticoagulants (DOACs) could have specific advantages in patients with LC. DOACs did not require frequent monitoring than VKAs and lower potential for food-drug and drug-drug interactions. 18 Research has shown that DOACs had similar efficacy to warfarin for the treatment of acute VTE or stroke in AF, often accompanied by lower bleeding rates.19-22 In addition, several studies suggest that DOACs showed similar efficacy and safety compared to warfarin in patients with liver disease.23-27 Accordingly, DOACs have gradually developed as an alternative to warfarin in clinical practice, including in patients with liver disease. 28 However, few data on the safety and efficacy of DOACs in cirrhotic patients because patients with cirrhosis have routinely been excluded from randomized clinical trials (RCTs) of DOACs. Although several studies have revealed that DOACs could remarkably decrease bleeding rates in cirrhotic patients compared to warfarin,29-31 which is limited by its small sample. As a result, concerns regarding the use of DOACs in cirrhotic patients were raised, and there remains a paucity of consensus regarding the choice of anticoagulants in patients with LC. Therefore, we conducted this meta-analysis to assess the safety and efficacy of DOACs versus warfarin in cirrhotic patients.
Methods
We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, 32 and preregistered in PROSPERO (registration number: CRD42018102664).
Search Strategy
We searched the potential studies through Web of Science, PubMed, Cochrane Library, Embase, and Scopus through August 31, 2024 for articles using the search keywords “novel oral anticoagulant,” “new oral anticoagulant,” “direct oral anticoagulant, “ “non-vitamin K antagonist,” “Oral Factor Xa Inhibitor,” “factor Xa inhibitors,” “edoxaban,” “rivaroxaban,” “dabigatran,” “apixaban,” “DOAC,” “NOAC,” “vitamin K antagonist,” “VKAs,” “warfarin,” “liver cirrhosis,” “cirrhotic,” “chronic liver disease” combined by the Boolean “AND/OR”.
Eligibility Criteria
The following inclusion criteria were used to select articles: (1) patients with cirrhosis were prescribed DOACs or warfarin; (2) included studies comparing the efficacy and safety outcomes of DOACs and warfarin; (3) primary efficacy outcomes included the incidence of IS/TE and all-cause death, whereas the primary safety outcomes included a safety composite of any bleeding, major bleeding, GIB and ICH; (4) studies were RCTs or observational design studies.
The exclusion criteria were: (1) studies with insufficient data or study details; (2) certain publications, including reviews, meeting abstracts, and case reports.
Data Extraction and Quality Assessment
Two authors extracted available information independently (C.CW and H.J.). Extraction from literature: study title, year, first author's name and publication time, geographical location, duration of follow-up, population number, demographic and characteristic data of cirrhotic patients, and Child-Pugh classification. In addition, the risk of bias was assessed using the Newcastle–Ottawa scale (NOS) for the observational studies. We regarded the NOS score of ≥7 as a high-quality. 33 The detailed quality assessment of each study is provided in Table S1.
Statistical Analysis
RevMan5.4 and stata15 software were used to analyze data. In this study, heterogeneity was examined by the I2 statistic. When I2 values <50% and P>.1, a fixed-effects model was chosen to pool effect sizes; otherwise, the random effect model was utilized. Hazard ratios (HRs) with 95% CIs were estimated by either fixed or random effects models. Sensitivity analysis was further conducted by sequentially deleting each study and reanalyzing all outcomes. Publication bias was evaluated by funnel plots and Egger's test.
Results
In the preliminary retrieval, we identified 1494 eligible articles by searching electronic databases. After removing duplicates and no relevant articles, we screened the titles, abstracts, and full-text screening. Finally, sixteen eligible studies12,13,23,34-46 were included in our meta-analysis (Figure 1).
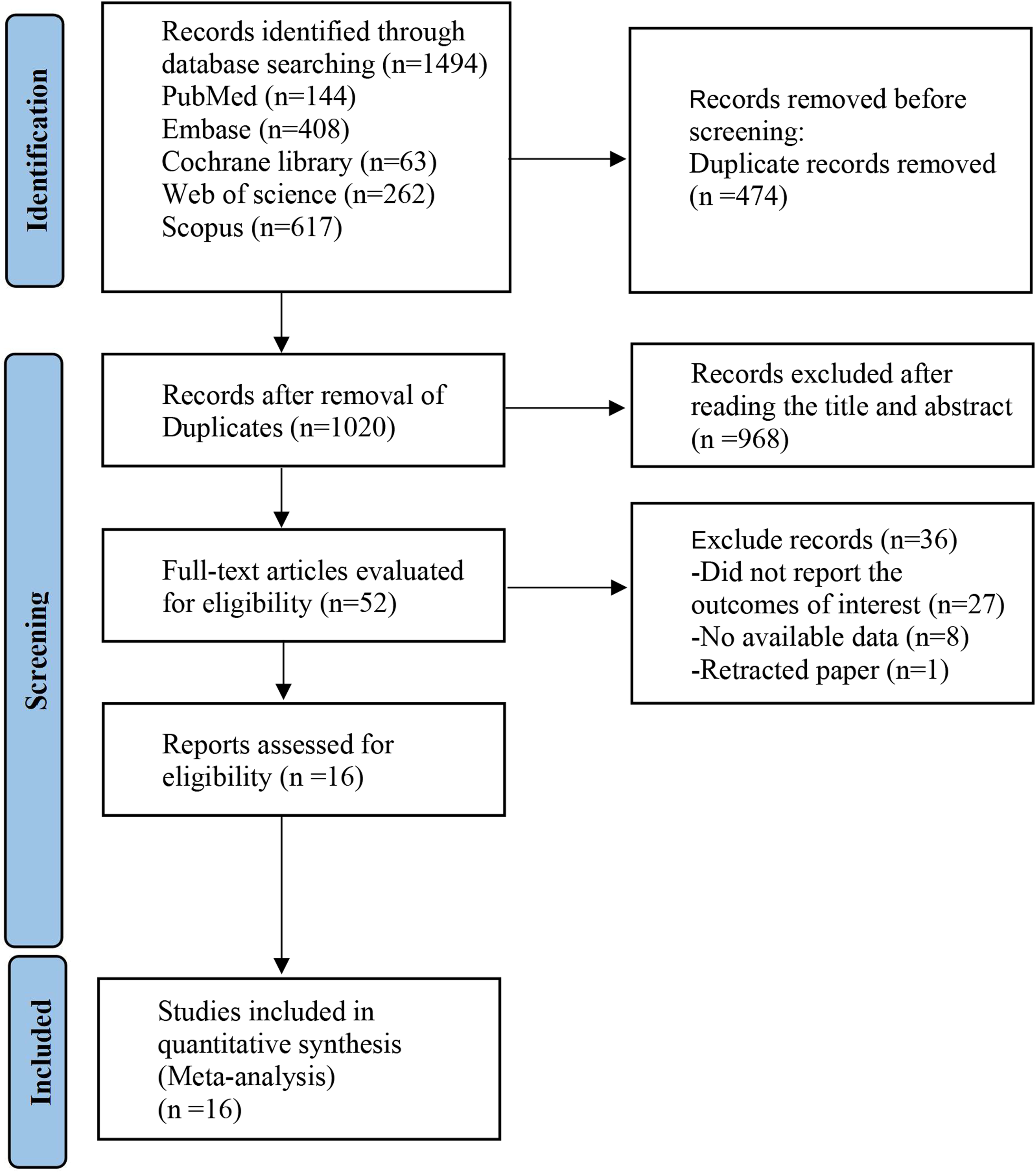
The flow chart of the literature search and screening process.
Baseline Characteristics
The baseline clinical characteristics of the individual studies are illustrated in Table 1. Sixteen observational cohort studies were included in the analysis, including fifteen retrospective cohort studies and one prospective cohort study. 35 Among these, nine studies were from the USA, two were from Korea, two were from Canada, with the others were from Italy, Taiwan and New Zealand, respectively. Six studies only involved cirrhosis, and ten studies included cirrhotic patients with AF. This study involved 16 829 cirrhosis patients, with 8861 (52.7%) receiving DOACs and 7968 (47.3%) receiving warfarin. Most patients were male, average age ranged from 46 to 79. Regarding the Child-Pugh score, most cirrhotic patients in this study had mild or moderate status. In five studies,37,40,41,45,46 patients were classified further into the moderate cirrhosis and severe cirrhosis groups, and data on the use of DOACs in comparison to warfarin were analyzed in this population.
Basic Characteristics of the Included Studies.
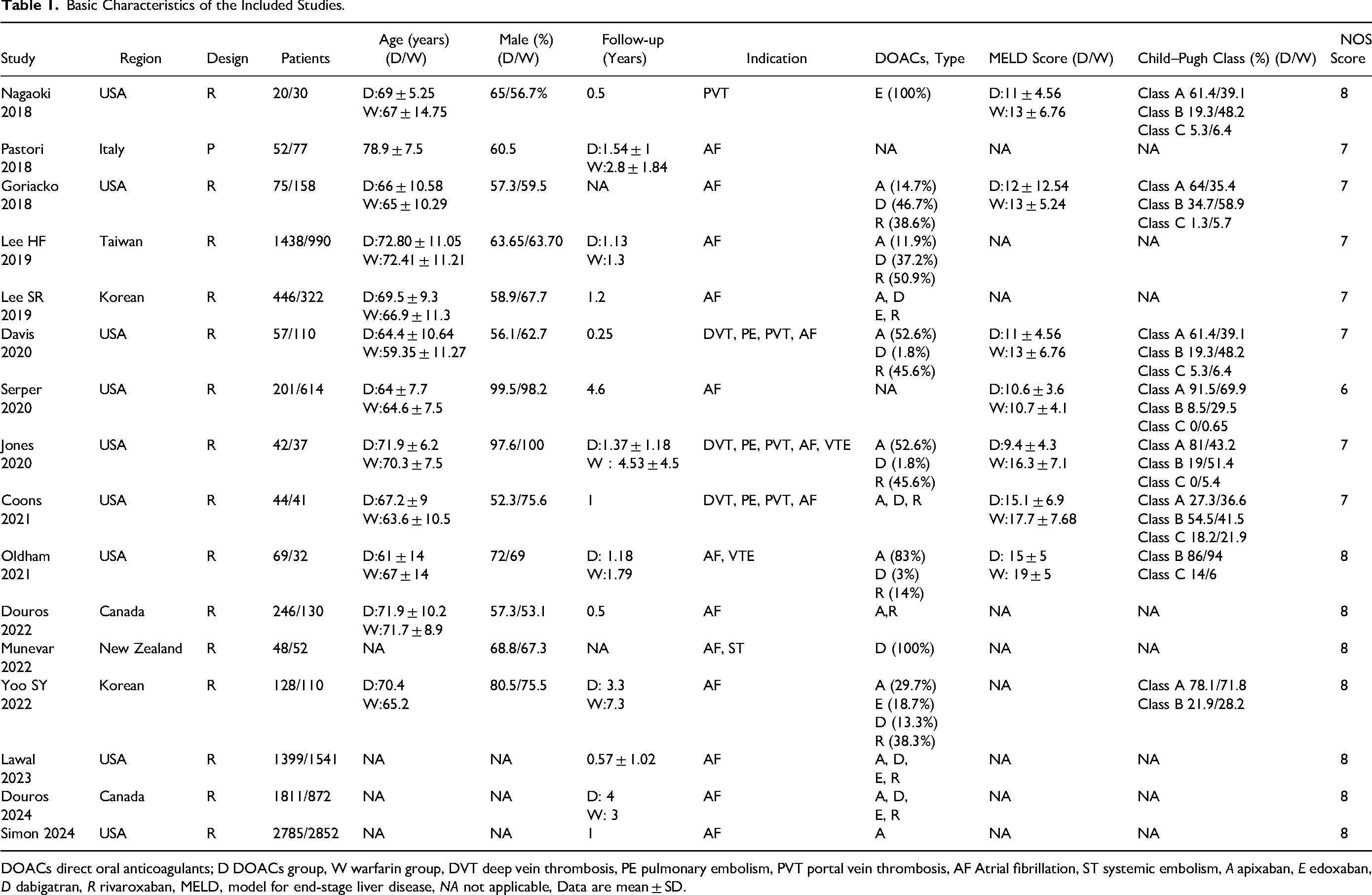
DOACs direct oral anticoagulants; D DOACs group, W warfarin group, DVT deep vein thrombosis, PE pulmonary embolism, PVT portal vein thrombosis, AF Atrial fibrillation, ST systemic embolism, A apixaban, E edoxaban, D dabigatran, R rivaroxaban, MELD, model for end-stage liver disease, NA not applicable, Data are mean ± SD.
Efficacy Outcomes
IS/TE
Eleven studies reported IS/TE. As shown in Figure 2A, our results indicated that there was no statistical difference in IS/TE between DOACs and warfarin groups (HR = 0.87, 95% CI: 0.69-1.10).
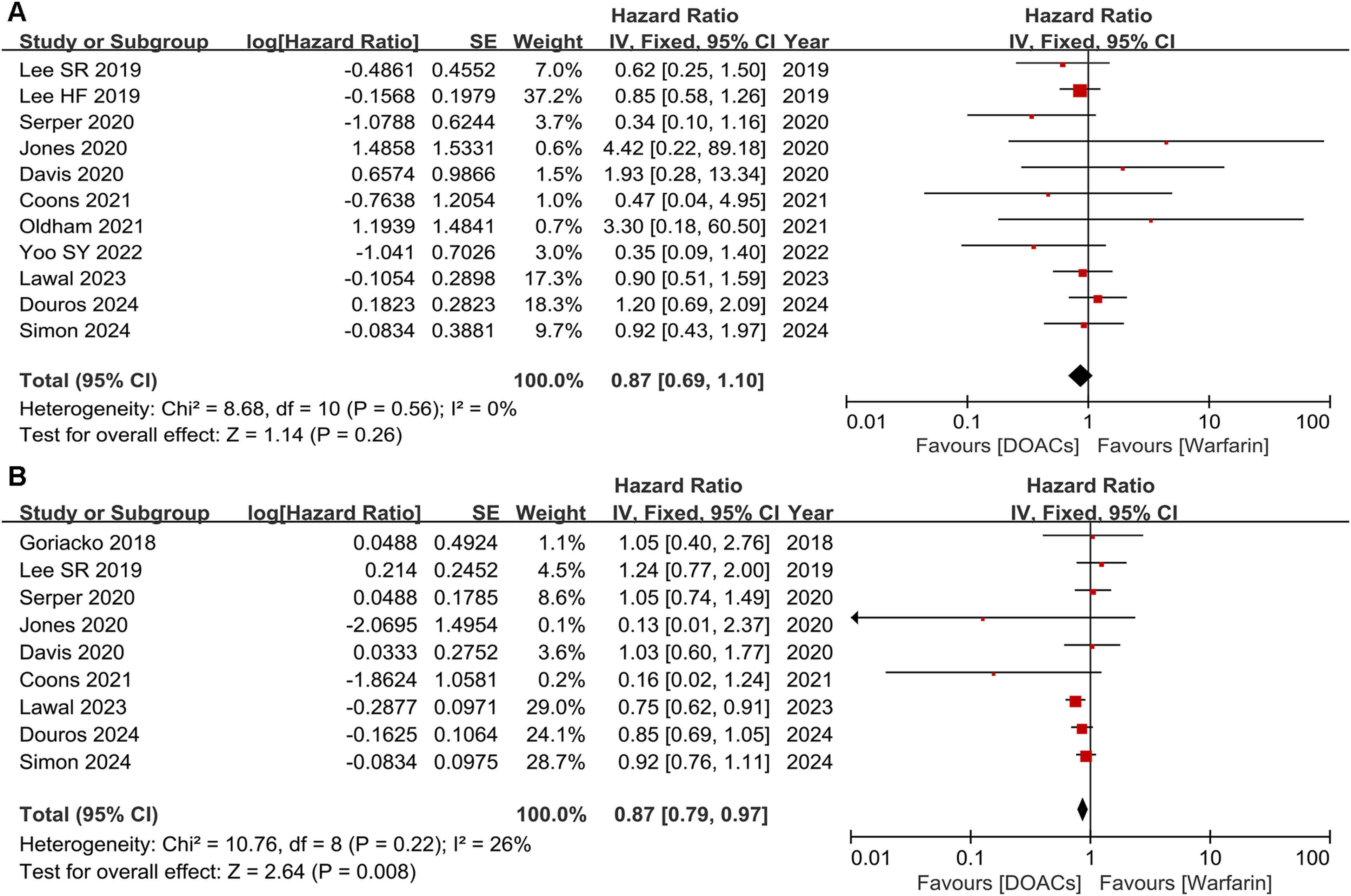
The efficacy outcomes of DOACs and warfarin in cirrhotic patients. (A) IS/TE; (B) all-cause death.
All-Cause Death
Nine studies reported all-cause death. As illustrated in Figure 2B, the results showed a slightly significant difference in all-cause death between patients taking DOACs and warfarin (HR = 0.87, 95% CI: 0.79-0.97).
Safety Outcomes
Any Bleeding
Eight studies reported any bleeding data. As presented in Figure 3A, DOAC users had a 40% lower risk of any bleeding than warfarin users (HR = 0.60, 95% CI: 0.37-0.95).
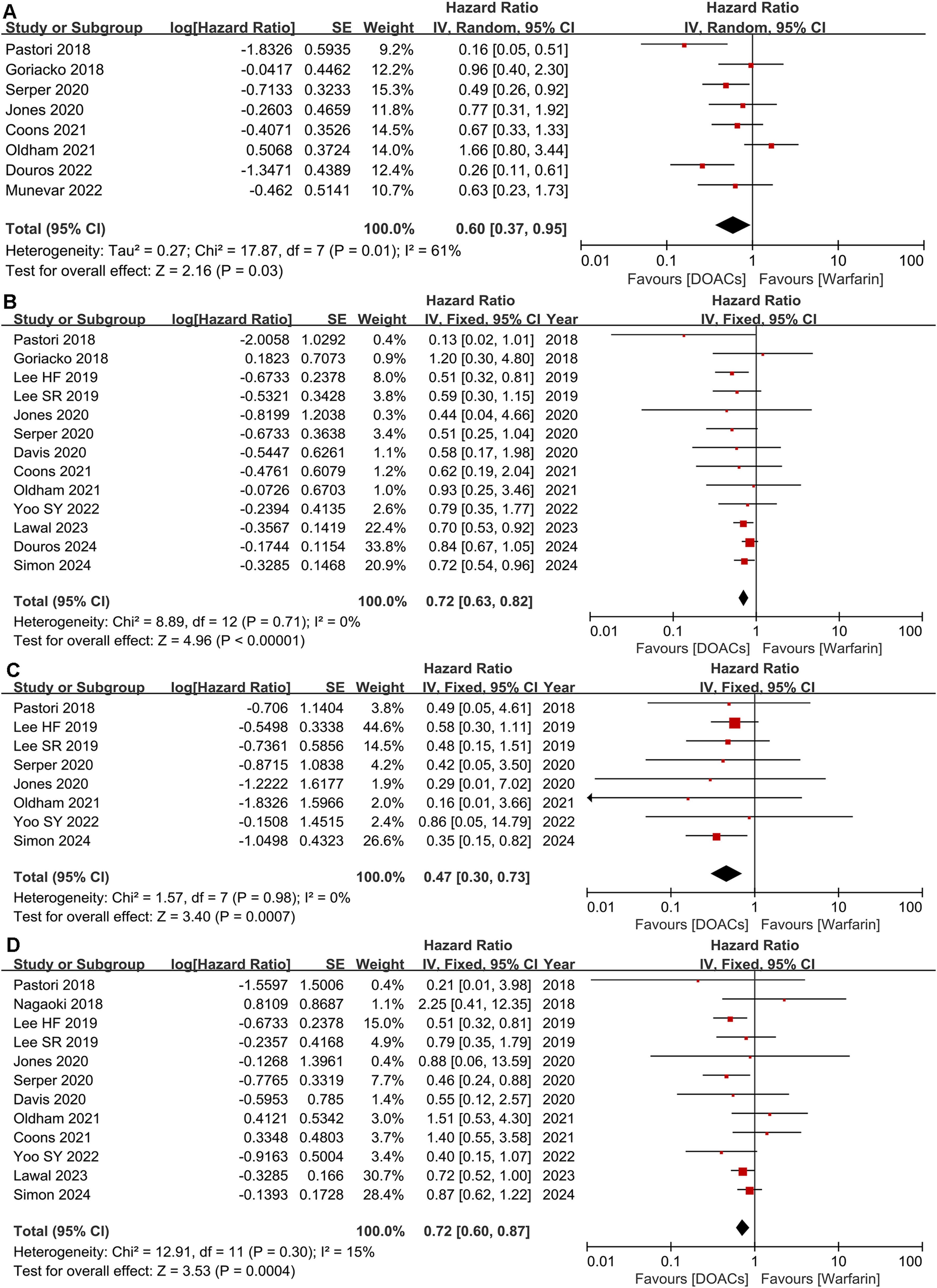
The safety outcomes of DOACs and warfarin in cirrhotic patients. (A) any bleeding; (B) major bleeding; (C) ICH; (D) GIB.
Major Bleeding
Thirteen studies reported major bleeding. As shown in Figure 3B, DOAC users experienced a lower risk of major bleeding compared with warfarin users (HR = 0.72, 95% CI: 0.63-0.82).
ICH
Eight studies reported the incidence of ICH. As depicted in Figure 3C, the use of DOACs resulted in a lower risk of ICH than warfarin users (HR = 0.47, 95% CI: 0.30-0.73).
GIB
Twelve studies reported gastrointestinal bleeding. The risk of GIB was lower in patients receiving DOACs compared with patients receiving warfarin (HR = 0.72, 95% CI: 0.60-0.87) (Figure 3D).
Subgroup Analyses
Subgroup Analyses of Patients with LC and AF
Subgroup analyses were conducted to assess whether the comparative efficacy and safety of DOACs versus warfarin were consistent in cirrhotic patients with AF. As shown in Figure 4A, our results showed that patients receiving DOACs did not experience a significant reduction in IS/TE (HR = 0.88, 95% CI: 0.67-1.15). On the other hand, DOACs experienced a significant reduction in all-cause death (HR = 0.87, 95% CI: 0.79-0.97) (Figure 4B). Additionally, in terms of safety, our results demonstrated a significant reduction in the risks of any bleeding (HR = 0.40, 95% CI: 0.20-0.79), major bleeding (HR = 0.72, 95% CI: 0.63-0.82), ICH (HR = 0.48, 95% CI: 0.31-0.75), and GIB (HR = 0.68, 95% CI: 0.56-0.82) in DOACs group compared with warfarin (Figure 4C–F).
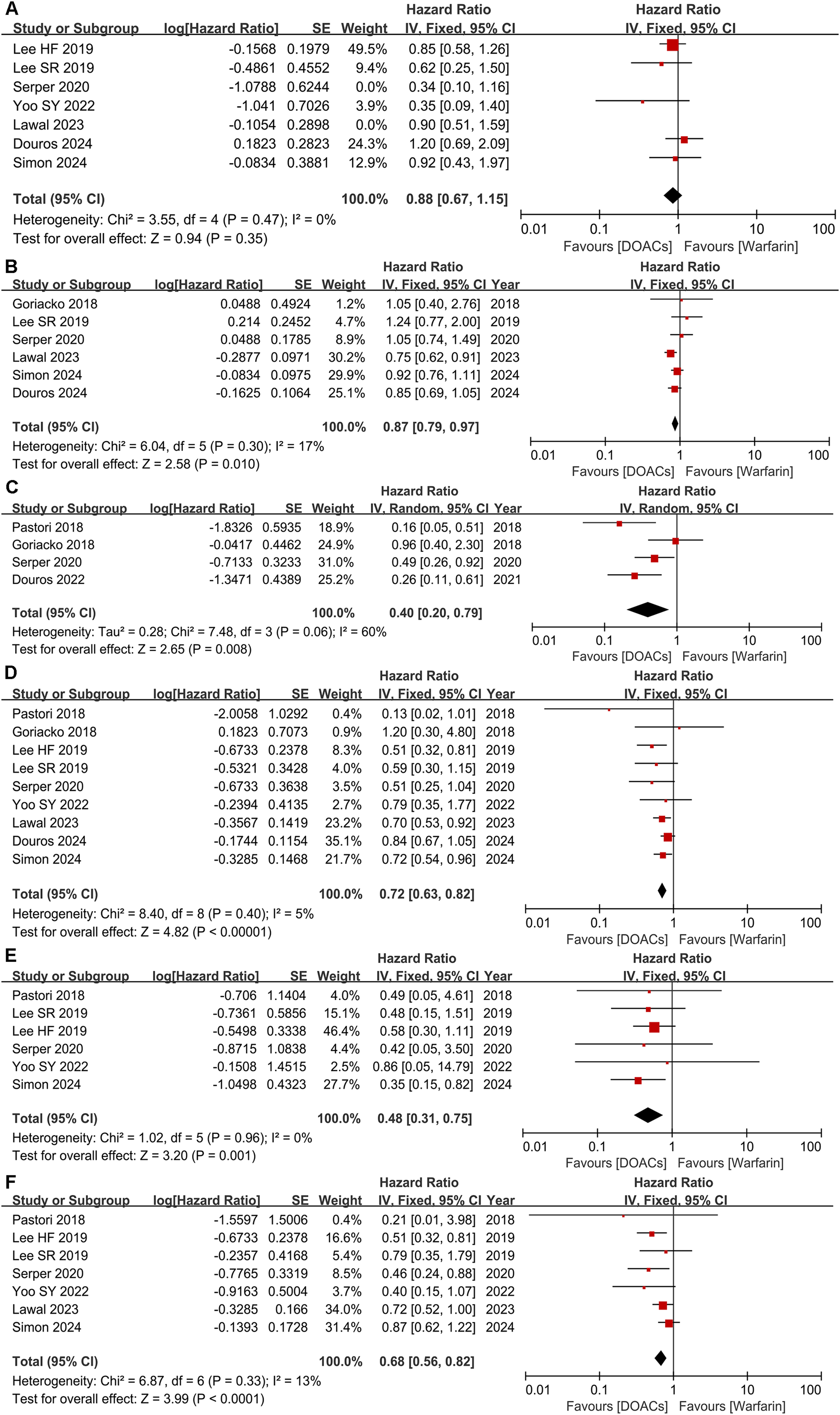
Comparing the efficacy and safety of DOACs with warfarin in patients with cirrhosis and AF. (A) IS/TE; (B) all-cause death; (C) any bleeding; (D) major bleeding; (E) ICH; (F) GIB.
Subgroup Analysis of Cirrhotic Patients with Moderate to Severe Status
A more detailed analysis was also performed in patients with moderate to severe cirrhosis (Child-Pugh B and C). Our results revealed no significant variation in the risk of IS/TE (HR = 1.43, 95% CI: 0.78-2.60) and GIB (HR = 0.75, 95% CI: 0.49-1.15) between these two groups (Figure 5A and D). Furthermore, DOAC users had decreased risks of major bleeding (HR = 0.65, 95% CI: 0.55-0.78) and ICH (HR = 0.17, 95% CI: 0.04-0.76) than warfarin users (Figure 5B and C).
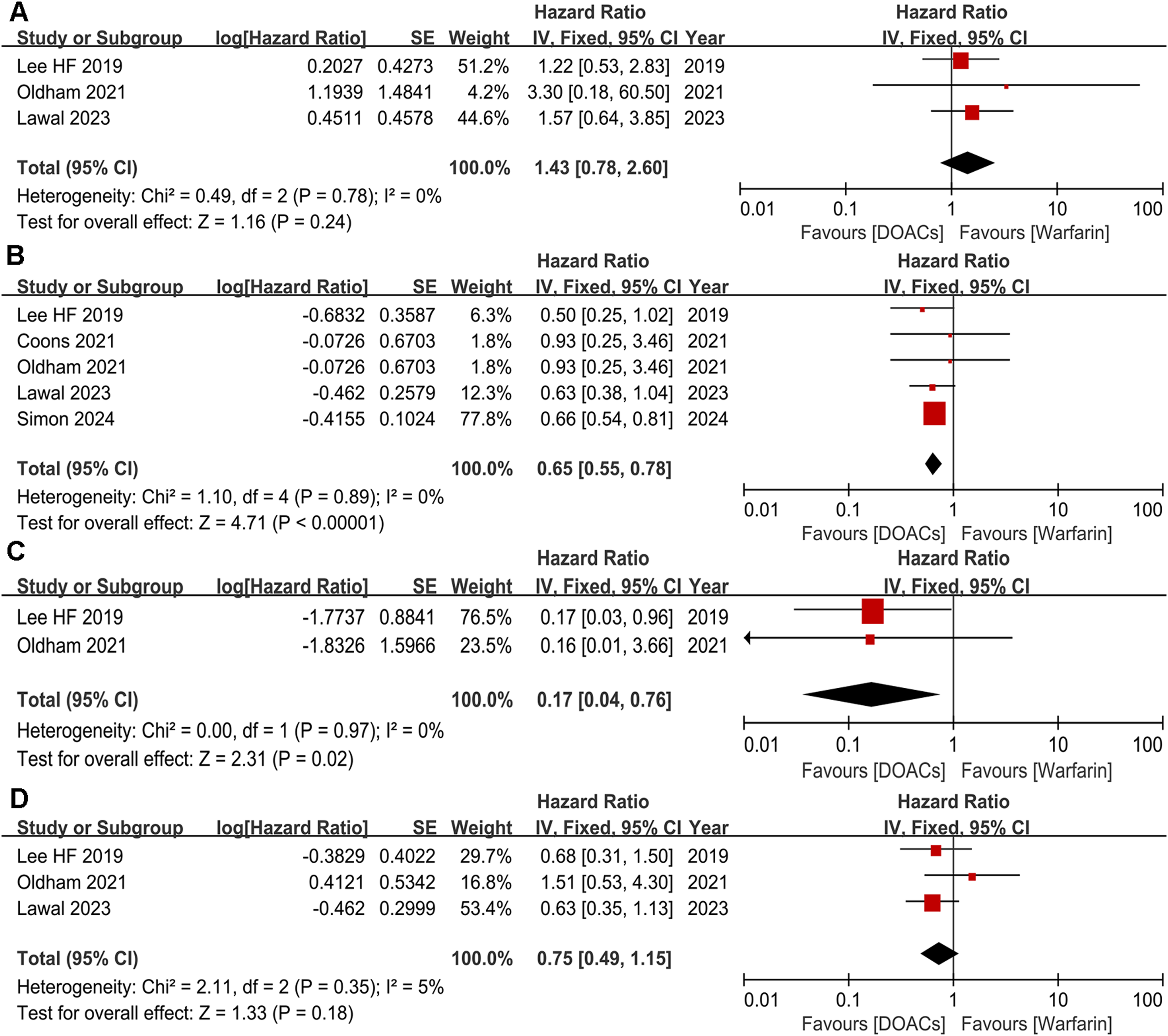
Comparing the efficacy and safety of DOACs with warfarin in moderate-severe cirrhosis patients. (A) IS/TE; (B) major bleeding; (C) ICH; (D) GIB.
Sensitivity Analysis and Publication Bias
The sensitivity analysis revealed that none of the studies significantly altered the results except all-cause death and any bleeding (sensitivity analysis in Supplementary Figure 1 corresponding to Figure 2A and B and Figure 3A–D), suggesting that the meta-analysis results of major safety outcomes were robust. Egger's test and funnel plots indicated no significant publication bias (Funnel plot in supplementary Figure 2 corresponding to Figure 2A and B and Figure 3A–D).
Discussion
In our current study, our results revealed that in comparison with warfarin, DOACs had significantly lower rates of all-cause death, any bleeding, major bleeding, ICH and GIB in cirrhotic patients, but there was no significant difference in the risks of IS/TE. Similar findings were also observed in cirrhotic patients with AF. Moreover, we found that DOACs were associated with lower risks of major bleeding and ICH than warfarin among moderate-severe cirrhosis patients. In summary, our study provides evidence that DOACs may be a safer alternative to warfarin for cirrhotic patients.
Cirrhosis patients are at a higher risk of bleeding and thrombosis.31,47 Besides, individuals with esophageal and gastric varices and thrombocytopenia are prone to gastrointestinal bleeding. 48 Therefore, anticoagulant therapy in cirrhotic patients is challenging. Warfarin is commonly used in cirrhotic patients for prophylaxis and treatment of thrombosis.49,50 However, warfarin is considered burdensome for many patients due to the frequent monitoring and dietary restrictions, which influence its efficacy. 51 In contrast, DOACs do not require routine monitoring or dose adjustment, making them a favorable alternative. However, data on the use of DOAC in cirrhosis patients remains scanty as they are generally excluded from RCT studies. Additionally, most DOACs require hepatic metabolism, hence liver dysfunction may increase drug levels and bleeding risks. Hence, DOACs represent a challenge in cirrhotic patients. Nonetheless, with the clinical use of DOACs, accumulating evidence shows that DOACs appear to be a promising alternative in cirrhotic patients in terms of efficacy and safety.
Previous meta-analyses revealed that DOACs had similar efficacy to warfarin but a lower bleeding risk in patients with LC,29,52-54 while these analyses are constrained by a small sample size or a limited number of studies. A current meta-analysis of nine studies demonstrated that, in terms of safety, DOACs could reduce any bleeding, GIB, ICH, and all-cause death but not reduce the risk of major bleeding in cirrhotic patients compared to warfarin. 55 Compared with the aforesaid study, we conducted a more recent and comprehensive analysis with updated data to evaluate the use of DOACs in cirrhotic patients using anticoagulants for various reasons and provide new insights into the advantages of DOACs over warfarin. Our results revealed that DOACs achieved lower risks of bleeding events versus warfarin. DOACs decreased the risk of any bleeding by 40%, major bleeding by 28%, ICH by 53%, and GIB by 28%. One possible explanation of these differences in safety outcomes is that the slower onset and offset of anticoagulant activity of warfarin, in addition to its longer half-life, may result in an elevated risk of bleeding events relative to DOACs, which exert anticoagulant effects rapidly by acting on coagulation factors.56,57 Additionally, although significant differences existed in the risks of all-cause death and any bleeding, sensitivity analysis showed that the results had significant change after excluding several studies. Such discrepancy may be attributed to several factors, such as study design, regions, cirrhosis severity, type and dosages of DOACs. Notably, the majority of the meta-analysis results were robust in sensitivity analysis, including IS/TE, major bleeding, ICH and GIB. In addition, Egger's test and funnel plots indicated that these results had no publication bias. Taken together, DOACs have comparable or even better clinical benefits in efficacy outcomes and superior safety in cirrhotic patients compared with warfarin.
Cirrhotic patients with AF are prone to thrombotic complications and necessitate anticoagulant therapy. However, the efficacy and safety outcomes of anticoagulant therapy in cirrhotic patients with AF remain controversial. Kuo and colleagues found that warfarin could decrease the incidence of stroke in LC and AF patients and obtain better clinical net benefits compared with the non-anticoagulant therapy group. 7 Yet, another study reported that warfarin failed to produce a significant reduction in the incidence of ischemic stroke in individuals with cirrhosis and AF; instead, it increased the incidence of hemorrhagic complications. 58 A recent study using MarketScan claims data shows that DOAC use has increased substantially and surpassed warfarin in patients with cirrhosis and AF 59 ; nevertheless, more than half of patients remain untreated, underscoring the need for optimal treatment. Therefore, the optimal management of such patients remains uncertain. Previous meta-analyses comparing DOACs with warfarin found that DOACs have similar efficacy with lower bleeding risk.29,60,61 However, these studies may be limited by small sample sizes. In contrast, we performed an updated meta-analysis and more comprehensively compared different clinical outcomes between DOACs and warfarin in patients with cirrhosis and AF compared to previous studies. Our results revealed that DOACs significantly lowered the risks of all-cause death and bleeding events, including any bleeding, major bleeding, and ICH, as well as GIB, but no significant differences in rates of IS/TE. Our meta-analysis expanded the results of prior research to a comprehensive comparison of the effects and safety of DOACs with warfarin. Taken together, our results further reveal that DOACs may be slightly more effective and safer in individuals with cirrhosis and AF.
In many past studies, clinical data on the effect of DOACs has primarily concentrated on mild-moderate liver cirrhosis, but the role of DOACs in advanced liver cirrhosis patients has not been well-studied. Currently, DOACs are safe in Child-Pugh class A and B liver cirrhosis (except rivaroxaban), and in the Child-Pugh C class are contraindicated.62,63 Additionally, anticoagulated cirrhotic patients who have esophageal varices are at an increased risk of bleeding. 48 Despite several studies assessing the effect of DOACs in cirrhotic patients based on Child-Pugh classifications or liver cirrhosis severity, included patients were those with mild-moderate cirrhosis, designated as Child-Pugh A or B.39,53,64 However, a recent study concluded that the safe use of DOACs in advanced cirrhosis patients may be feasible. 65 Another study including 101 patients with cirrhosis 41 suggested that DOACs were recommended for Child-Pugh B or C patients based on safety, efficacy, and convenience. However, due to the limited data and the greater proportion of compensated cirrhosis patients in these studies, the effect of DOACs could not be extrapolated to patients with advanced cirrhosis. One prior meta-analysis revealed that DOACs only improved major bleeding in advanced cirrhosis when compared to warfarin. 60 However, this study assessed the efficacy and safety of DOACs versus warfarin using only two studies. In contrast, our results of subgroup analysis including five studies revealed that DOACs had lower rates of major bleeding and ICH than warfarin. In line with our study, a registry-based cohort analysis shows that DOACs had a statistically significant reduction in ICH and GIB compared to warfarin in patients with Child-Pugh Class C cirrhosis. 66 In general, our study demonstrated that DOACs may represent a relatively safer therapy for moderate to severe cirrhosis patients. However, this result required cautious interpretation due to the inclusion of only five studies.
In this study, our results of subgroup analysis indicated that DOACs have equal efficacy to warfarin and superior safety in cirrhotic patients with AF or moderate-severe status. Collectively, DOACs are generally favored over warfarin in terms of safety outcomes in cirrhotic patients.
Limitations
Our study has several limitations. First, the studies included in our research were retrospective studies. Further, we did not investigate the type or dose of DOACs for cirrhosis patients because of the scarcity of studies. Prospective studies or RCTs are required to ascertain the potential benefits of DOACs. In addition, although we have demonstrated that DOAC users achieved a reduced incidence of major bleeding and ICH versus warfarin and showed similar efficacy outcomes in moderate to severe cirrhosis patients, further studies are required to underpin our results since only five retrospective studies were included. Above all, this study reinforced the current limited available evidence, demonstrating that DOACs exerted comparable efficacy and superior safety profile relative to warfarin in this patient population, including moderate-severe cirrhosis.
Conclusion
Taken together, our study demonstrated that DOACs exerted comparable efficacy and superior safety than warfarin in the treatment of cirrhosis or combined with AF, even in advanced cirrhosis. However, this study still has the abovementioned limitations. In the future, larger RCTs and prospective studies are necessary to confirm its efficacy and safety.
Supplemental Material
sj-docx-1-cat-10.1177_10760296241301402 - Supplemental material for Comparison of the Efficacy and Safety of Direct Oral Anticoagulants and Warfarin in Cirrhotic Patients: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-cat-10.1177_10760296241301402 for Comparison of the Efficacy and Safety of Direct Oral Anticoagulants and Warfarin in Cirrhotic Patients: A Systematic Review and Meta-Analysis by Chunwei Cheng, Juan Hua and Liang Xiong in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the The Doctoral Research Initiation Fund of Jiangxi University of Technology (2024),
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
