Abstract
Background
Acute venous thromboembolism (VTE) in children presents unique challenges due to the limitations of standard anticoagulation therapies. Herein, we aimed to systematically review randomized controlled trials (RCTs) evaluating the efficacy and safety of direct oral anticoagulants (DOACs) in pediatric patients with acute VTE.
Methods
PubMed and Embase databases were searched for RCTs comparing DOACs to standard anticoagulation in pediatric VTE patients. Efficacy outcomes included VTE recurrence and all-cause mortality, while safety outcomes comprised major bleeding and other adverse events.
Results
Three RCTs with 790 participants were included. When compared with standard anticoagulation, DOACs demonstrated a reduced risk of VTE recurrence (risk difference[RD] = −3%, 95% confidence interval[CI]: −6% to 0%, P = 0.04) and an increased risk of any adverse event (RD = 8%, 95% CI: 1% to 14%, P = 0.02). No significant differences were found in all-cause mortality, major bleeding, clinically relevant non-major bleeding, or total bleeding between the DOAC and control groups.
Conclusion
DOACs, primarily dabigatran and rivaroxaban, are non-inferior to standard anticoagulants in reducing VTE recurrence in pediatric patients, with comparable safety profiles. Further research is essential to confirm these findings.
Keywords
Introduction
Venous thromboembolism (VTE), encompassing deep vein thrombosis and pulmonary embolism, is a significant cause of morbidity and mortality in children. 1 Currently, the standard of care for anticoagulation therapy in pediatric patients with acute VTE includes vitamin K antagonists (VKAs), unfractionated heparin (UFH), or low-molecular-weight heparins (LMWHs).2,3 However, VKAs like warfarin have certain drawbacks, such as the need for frequent monitoring of anticoagulant activity, complex dose adjustments, and the potential for drug-drug or drug-food interactions. LMWHs, while effective, often require injections and can lead to complications such as heparin-induced thrombocytopenia and decreased bone mineral density. Given these limitations, it is crucial to explore alternative drug options for pediatric patients with acute VTE.
Direct oral anticoagulants (DOACs), namely dabigatran, rivaroxaban, apixaban, and edoxaban,4–8 have been found to have greater net clinical benefits compared to VKAs in AF patients9,10 or in the treatment of acute VTE in adults. 11 However, the previous studies assessing the effect of DOACs have largely focused on adult patients; and thus their role in pediatric VTE treatment remains less defined.12,13 Currently, there have been several prospective randomized clinical trials (RCTs, eg, EINSTEIN-Jr, DIVERSITY, and Eghbali et al)14–16 that have examined the use of DOACs as potential therapeutic alternatives for acute VTE in pediatric patients. Therefore, a systematic review and meta-analysis of these RCTs were conducted to evaluate the efficacy and safety outcomes of DOACs when compared to to standard anticoagulants in this specific population.
Methods
Data Sources and Search Strategy
The PubMed and Embase databases were systematically searched until May 2024 to identify relevant published studies. During our search, we focused on RCTs that specifically investigated the efficacy and safety outcomes of DOACs compared to the standard of care for anticoagulation therapy in pediatric patients with acute VTE. The search strategy was included keywords related to DOACs, VTE, and pediatric populations. Supplemental Table 1 shows the search strategies of this meta-analysis. The reference lists of prior reviews were also checked for additional studies.
Eligibility Criteria and Study Selection
Two independent reviewers screened titles and abstracts of the identified studies, followed by full-text assessment of potentially eligible studies. Discrepancies were resolved through consensus or by involving a third reviewer. We included studies according to the following eligibility criteria:
Population: pediatric patients (under 18 years) diagnosed with acute VTE. There were no restrictions on the types of acute VTE included in this analysis. Intervention: treatment with any DOAC (dabigatran, rivaroxaban, apixaban, edoxaban). Comparison: standard anticoagulation therapy (VKAs, UFH, or LMWH). Outcomes: efficacy (VTE recurrence and all-cause mortality) and safety (major bleeding, clinically relevant non-major bleeding, total bleeding, and any adverse event) outcomes
17
(
Supplemental Table 2
). Study design: RCTs. Post hoc analyses of the included RCTs were not included.
Data Extraction
A standardized data extraction form was used to collect information on study characteristics, participant demographics, intervention details, and outcomes. Extracted data mainly included: study identifiers (authors, publication year), study type, registry number, data source, participant characteristics (age, sex, VTE type), intervention and comparison details (DOAC type and dosage, standard anticoagulation therapy), treatment time, and outcome measures.
Risk of Bias Assessment
The risk of bias in the included studies was assessed using the Cochrane Risk of Bias Tool 2.0 (RoB 2), 18 which evaluates bias in randomization, deviations from intended interventions, missing outcome data, measurement of the outcome, and selection of reported results.
Statistical Analysis
In order to conduct our statistical analyses, we followed the guidelines outlined in the Cochrane Handbook for Systematic Reviews of Interventions version 6.2, 19 and used Review Manager version 5.4 software (the Cochrane Collaboration 2014, Nordic Cochrane Centre Copenhagen, Denmark).
We collected information on the number of events and sample size from each included study. To combine the results across studies, we used a random- or fixed-effects model, depending on the degree of heterogeneity (I²) in the data (random effects if I² > 50%, fixed effects if I² ≤ 50%). The effect measures for our analysis were expressed as absolute risk differences (RDs) with corresponding 95% confidence intervals (CIs). Subgroup analyses and meta-regression were planned but not performed due to the limited number of studies.
Results
Study Selection
The PRISMA flow diagram (
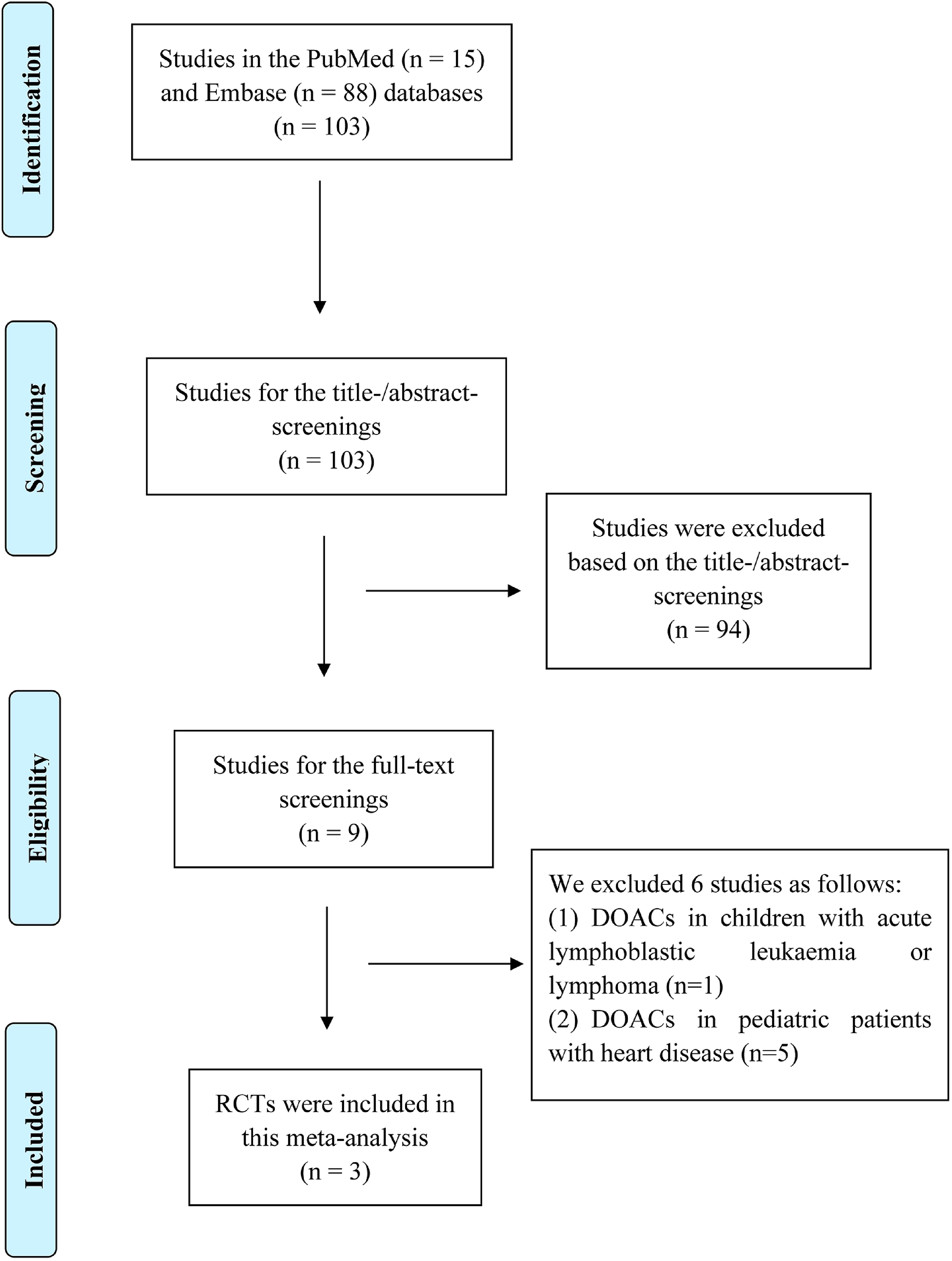
The PRISMA flow diagram for the study selection process.
Study Characteristics
The baseline characteristics of the included trials are presented in
Baseline Characteristics of the Included Studies of this meta-Analysis.
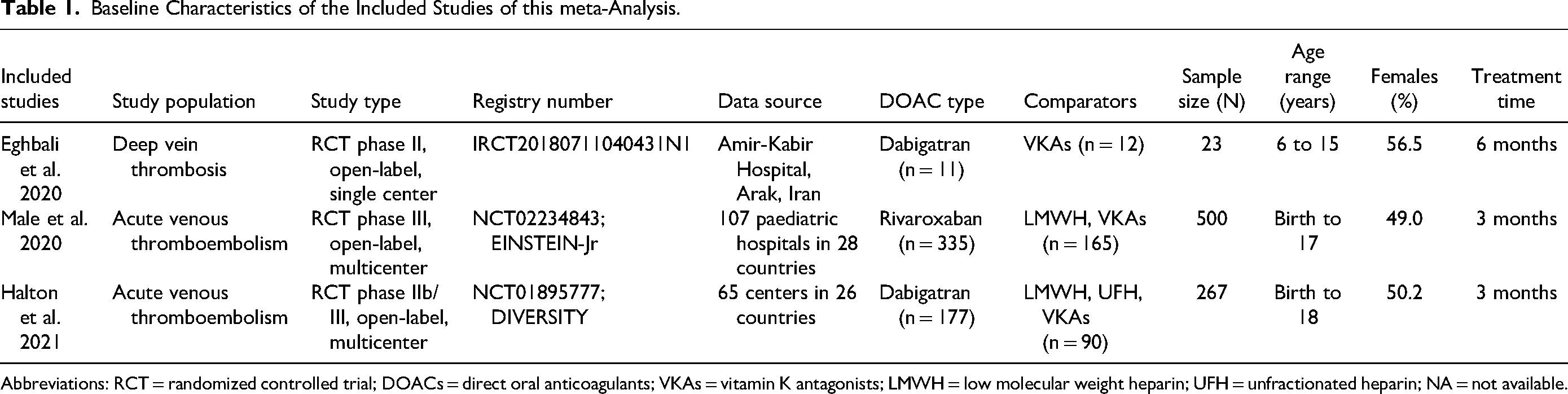
Abbreviations: RCT = randomized controlled trial; DOACs = direct oral anticoagulants; VKAs = vitamin K antagonists; LMWH = low molecular weight heparin; UFH = unfractionated heparin; NA = not available.
The risk of bias in the included studies was assessed using the RoB 2 tool. 18 Halton et al and Male et al were considered to have a low risk of bias, while the study by Eghbali et al was deemed as having a potential high risk of bias.
Efficacy and Safety Outcomes
All three studies (Eghbali et al, Halton et al, and Male et al)14–16 reported outcomes related to VTE recurrence, all-cause death, total bleeding, and major bleeding. Additionally, Halton et al 14 and Male et al 15 provided data on clinically relevant non-major bleeding and any adverse event.
For the pooled analysis, a fixed-effects model was applied due to the acceptable heterogeneity observed across the included studies, with I² values ranging from 0% to 46%. When compared with standard anticoagulation, the use of DOACs in pediatric patients with acute VTE was associated with a reduced risk of VTE recurrence (RD = −3%, 95% CI: −6% to 0%, P = 0.04; I² = 0%) and an increased risk of any adverse event (RD = 8%, 95% CI: 1% to 14%, P = 0.02; I² = 0%) (
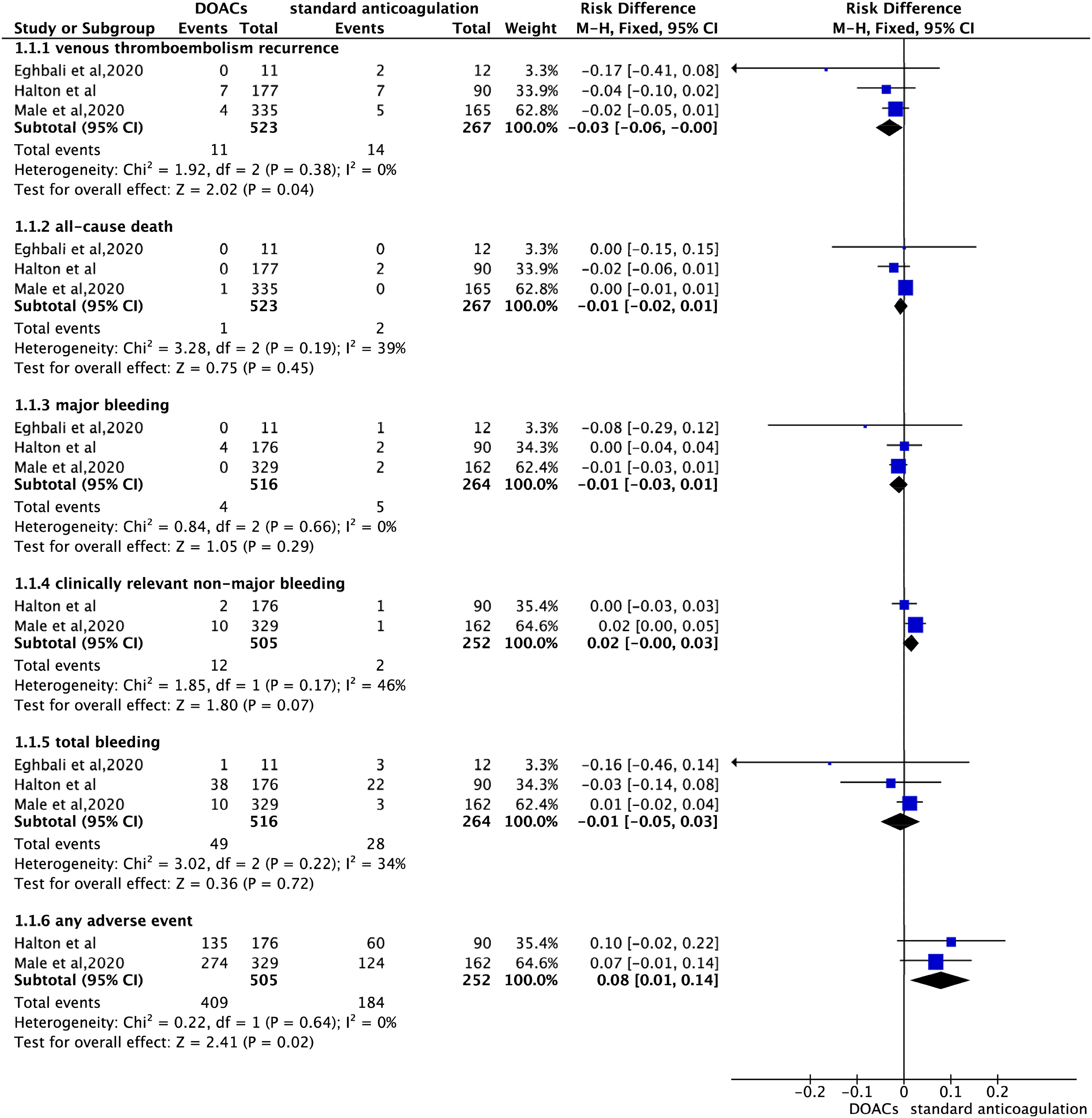
The efficacy and safety outcomes of direct oral anticoagulants in pediatric patients with acute venous thromboembolism.
However, there were no statistically significant differences in the rates of all-cause death (RD = −1%, 95% CI: −2% to 1%, P = 0.45; I² = 39%), major bleeding (RD = −1%, 95% CI: −3% to 1%, P = 0.29; I² = 0%), clinically relevant non-major bleeding (RD = 2%, 95% CI: −0% to 3%, P = 0.07; I² = 46%), or total bleeding (RD = −1%, 95% CI: −5% to 3%, P = 0.72; I² = 34%) between the DOAC and control groups (
Discussion
Based on the pooling of data from three initial trials, our current study found that the use of DOACs in pediatric patients with acute VTE was associated with a reduced risk of VTE recurrence when compared to traditional anticoagulants, while there were no differences in all-cause death, major bleeding, clinically relevant non-major bleeding, and total bleeding between the two studied groups. Therefore, DOACs (mainly dabigatran and rivaroxaban) were at least non-inferior to the standard of care for the treatment of VTE in pediatric patients.
The use of DOACs in the pediatric population has found significant advancements in recent years, with studies such as the DIVERSITY and EINSTEIN-Jr trials providing valuable insights into their safety and efficacy. The single-armed phase 3 trial by Brandão et al 25 has demonstrated dabigatran etexilate's favorable safety in preventing recurrent VTE in children aged 3 months to 18 years, with low rates of VTE recurrence and major bleeding events, and no treatment-related deaths. The findings from our current meta-analysis of three RCTs (EINSTEIN-Jr, DIVERSITY, and Eghbali et al)14–16 further indicated that DOACs, specifically dabigatran and rivaroxaban, might be considered as viable alternatives to standard anticoagulation in pediatric patients with acute VTE. The marked decrease in VTE recurrence with DOACs is a notable benefit, with no significant increase in major bleeding or other safety outcomes, offering potential advantages over traditional VKAs and LMWHs. In addition, the DIVERSITY and EINSTEIN-Jr trials reported that the pharmacokinetic and pharmacodynamic relationships in children were similar to those observed in adults, suggesting that age- and weight-adjusted dosing in children can achieve comparable drug exposures in adults.
However, while the 3-month study duration was sufficient to evaluate thrombus resolution and recurrence,14,15 the long-term effects of DOACs on preventing the development of post-thrombotic syndrome and other late complications are yet to be determined. In addition, the small number of RCTs and the limited data on apixaban and edoxaban restrict the generalizability of the findings. Clinical trials often exclude very sick children, those with certain comorbidities, and younger age groups (particularly infants) also limiting the generalizability of the findings. Overall, while the initial findings on the use of DOACs in children are promising, there is a need for continued research to address current gaps and challenges, and to expand the evidence base for the use of DOACs in the pediatric population.
It was noted that our current study had several limitations. First, the number of included studies and the sample size were relatively small, which might have limited the reliability of our results. Second, the current RCTs did not provide data on the use of apixaban and edoxaban in pediatric patients with acute VTE; and thus, our findings only apply to dabigatran and rivaroxaban. Third, although our meta-analysis included the variability in patient characteristics, DOAC types and doses, and follow-up duration, we were unable to conduct subgroup analyses due to limited data availability. Fourth, future trials should include a more diverse patient population, including those with less common underlying conditions, to better understand the full spectrum of DOAC use in children. Finally, observational studies12,26,27 in larger pediatric patient populations will help confirm the benefits of DOACs from RCTs and provide data on the long-term use of DOACs in children.
Conclusion
Our present study suggested that DOACs, specifically dabigatran and rivaroxaban, were viable treatment options for pediatric patients with acute VTE, which were at least non-inferior to the standard of care. However, given the limitations of our study, further research is needed to fully establish the role of DOACs in pediatric VTE management.
Supplemental Material
sj-pdf-1-cat-10.1177_10760296241271386 - Supplemental material for Direct Oral Anticoagulants for Treating Acute Venous Thromboembolism in Children: Meta-Analysis of Randomized Controlled Trials
Supplemental material, sj-pdf-1-cat-10.1177_10760296241271386 for Direct Oral Anticoagulants for Treating Acute Venous Thromboembolism in Children: Meta-Analysis of Randomized Controlled Trials by Xin Yu, Wengen Zhu, Chen Liu and Renrong Lu in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Author Contributions
Initials (Xin Yu and Wengen Zhu), conceptualization; Initials (Yun Wan and Chen Liu), methodology; Initials (Xin Yu and Wengen Zhu), investigation; Initials (Xin Yu and Wengen Zhu) wrote the manuscript; Initials (Renrong Lu and Chen Liu), validation, revision. All authors read and approved the final manuscript.
Availability of Data and Materials
All relevant data and materials are presented in the paper.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not required.
Funding
This study was funded by Key R&D Projects of Guangzhou Science and Technology Program (2023B03J1243), Guangdong Natural Science Foundation (2024A1515013289), Sanming City Science and Technology Joint Funding Project (2023-S-110), and the Science Foundation of the Fujian Province (2022J11454).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
