Abstract
Background
Portal venous thrombosis (PVT) is a serious complication in cirrhosis, with management debated due to differing anticoagulant guidelines. This network meta-analysis assessed various anticoagulants for PVT in cirrhotic patients.
Methods
PubMed, Cochrane Central, and ScienceDirect were searched till May 2025. A frequentist network meta-analysis was conducted in RStudio version 4.3.3. The ranking was based on P-scores.
Results
Nineteen studies were included in the analysis. Direct oral anticoagulants (DOACs) (RR = 2.38; 95%CI:[1.17, 4.85]; p = 0.02), fondaparinux (RR = 18.16; 95%CI:[2.09, 158.13]; p = 0.009), low-molecular-weight heparin (LMWH) (RR = 11.96; 95%CI:[1.58, 90.79]; p = 0.01), LMWH-Warfarin sequential therapy (RR = 2.32; 95%CI:[1.01, 5.31]; p = 0.04), and Vitamin K antagonists (VKA) (RR = 1.71; 95%CI:[1.03, 2.83]; p = 0.04) showed a significant increase in complete recanalization, with fondaparinux ranked highest (P-score = 0.94). DOACs (RR = 0.23;95%CI:[0.07, 0.71]; p = 0.01), LMWH (RR = 0.24;95%CI:[0.11, 0.52]; p = 0.0003), LMWH-Warfarin sequential therapy (RR = 0.40;95%CI:[0.21, 0.77]; p = 0.006) demonstrated a significant reduction in PVT progression, with Antithrombin-III ranked highest (P-score = 0.87). Fondaparinux was ranked highest (P-score = 0.90) for reducing esophageal variceal bleeding, while LMWH was ranked highest (P-score = 0.72) for decreasing overall bleeding. DOACs significantly decreased mortality (RR = 0.74; 95% CI: [0.67, 0.81]; p < 0.0001), with LMWH ranked highest (P-score = 0.80) for mortality.
Conclusion
DOACs, LMWH, and LMWH-Warfarin sequential therapy significantly improved recanalization and reduced PVT progression. LMWH was ranked highest in reducing bleeding and mortality, while fondaparinux ranked highest regarding esophageal bleeding and complete recanalization.
Keywords
Introduction
Portal vein thrombosis (PVT) is defined as venous thromboembolism of the intrahepatic portal vein, splenic vein, and mesenteric vein. 1 The pathogenesis of PVT is related to increased vascular resistance and decreased portal vein blood flow, creating an ideal environment for thrombus formation. 2 PVT is a common complication in patients with liver cirrhosis, with its incidence reported to be from 5% to 20%, with a higher incidence in patients with advanced-stage or decompensated liver cirrhosis.3,4 Liver cirrhosis can lead to portal hypertension, esophageal varices, and ascites, and PVT can further worsen these conditions. 5
Considering these life-threatening complications, PVT needs to be treated promptly. However, guidelines regarding management of PVT in cirrhotic patients remain conflicted. The American Association for the Study of Liver Diseases (AASLD) recommends that patients with PVT be treated with anticoagulant therapy for 3 months. In contrast, the European Association for the Study of the Liver (EASL) recommends at least 6 months of anticoagulation for acute PVT
Low-molecular-weight-heparin (LMWH) and warfarin are the most heavily studied anticoagulants, with some studies reporting a 60%–80% recanalization rate with no increased risk of bleeding.13,14 However, in recent times, there has been growing interest in the efficacy of Direct Oral Anticoagulants (DOACs), which include direct factor Xa inhibitors such as rivaroxaban and apixaban, along with direct factor IIa inhibitors like dabigatran. These have shown comparable, if not superior, outcomes when compared to traditional anticoagulants such as LMWH and vitamin K antagonists (VKA).15–18 DOACs do not require regular monitoring or subcutaneous injections as with LMWH and have fewer drug-drug interactions, even in patients with hereditary thrombophilia. 19
A recent study by Tadakoro showed that the overall response rate for thrombolysis was higher with edoxaban at 76.7% compared to warfarin at 29.4% and achieved long-term recanalization with intact hepatic reserves compared to warfarin. 16 However, a recent study by Joseph et al. showed significantly higher resolution rates of PVT in patients treated with warfarin compared to those treated with DOAC. 20 The current literature is either conflicting or shows comparable safety and efficacy of using conventional anticoagulation with DOAC. 17 The most recent network meta-analysis by Li et al demonstrated that combination therapies, such as LMWH-DOAC sequential therapy, are among the most effective regimens for achieving complete recanalization of PVT. 21 In this network meta-analysis, we aim to compare the efficacy of DOACs, fondaparinux, LMWH, LMWH-warfarin sequential therapy, VKA, and antithrombin III in halting the progression of PVT and recanalization of concurrent PVT, as well as their safety in reducing the risk of major bleeding events and mortality.
Methods
This systematic review and network meta-analysis was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines 22 and followed the Cochrane Handbook for Systematic Reviews of Interventions guidelines. 23 The protocol of this review was registered on PROSPER under the ID: CRD420251135942.
Literature Search
Electronic databases, including PubMed, ScienceDirect, and Cochrane Central, were searched from inception to May 2025. The bibliographies of the included studies were also searched for potential topics. The MeSH terms and keywords used were “Liver Cirrhosis,” “portal vein thrombosis,” “Anticoagulants,” “Direct Oral Anticoagulants,” “Low Molecular Weight Heparin,” “Warfarin,” “Fondaparinux,” and “Antithrombin-III.” The details of the search strategies used in different databases are shown in Supplementary Table 1.
Study Selection and Eligibility Criteria
The articles retrieved through initial database searching by two independent authors were imported into EndNote. After duplicate removal, the remaining articles were passed through primary title and abstract screening. Articles retrieved after primary screening were subjected to secondary screening through a full-text review. Any discrepancy was resolved by a third author. The entire study selection process is depicted in the PRISMA flowchart (Figure 1).

PRISMA Flowchart of the Study Selection Process.
The inclusion criteria encompassed patients over 18 years old with a confirmed diagnosis of PVT in cirrhosis. The interventions involved various pharmacological anticoagulants, including DOACs, LMWH, LMWH-Warfarin sequential therapy, VKAs, fondaparinux, and antithrombin-III. The comparison group consisted of patients who received no anticoagulation. The study included both randomized clinical trials (RCTs) and observational studies.
Patients under 18 years old, those with malignant PVT, pre-existing extrahepatic thrombosis, unspecified anticoagulation regimens, non-pharmacological anticoagulation strategies, or study designs such as reviews, abstracts, and editorials were excluded.
Data Extraction and Endpoint Definition
The variables of interest were extracted by two authors independently on an Excel sheet. Any discrepancies during the extraction process were resolved through discussion with a third author. The baseline variables extracted included study ID, location, study design, sample size, the anticoagulant used, mean age, gender distribution, Child-Pugh scores, model for end-stage liver disease (MELD) score, presence of esophageal varices, International Normalization Ratio (INR), platelet count, creatinine level, and follow-up period.
The outcomes extracted were complete recanalization, PVT progression, esophageal variceal bleeding, overall bleeding, and mortality. Complete recanalization refers to the total resolution of a fully occlusive PVT, with the vessel transitioning to a non-occlusive state. PVT progression is characterized by either additional vessel occlusion or the extension of the thrombus into the segments of the superior mesenteric or splenic veins.
Risk of Bias
The risk of bias of the included studies was assessed by two independent authors using the Cochrane Risk of Bias (RoB 2.0) tool 24 and the Newcastle-Ottawa Scale (NOS) 25 for RCTs and observational studies, respectively. The RoB 2.0 tool assesses bias across five domains: bias resulting from the randomization process, bias due to deviations from the intended intervention, bias caused by missing outcome data, bias in the measurement of outcomes, and bias in the selection of reported results. The risk of bias is then marked as low, uncertain, or high. The NOS evaluates bias across three domains: selection, comparability, and exposure. Studies with an overall score of 7 or higher are considered high quality.
Statistical Analysis
A frequentist network meta-analysis was conducted using the “meta” and “netmeta” packages in RStudio version 4.3.3. Risk ratios (RRs) with 95% confidence intervals (CIs) were pooled under a random-effects model, with no anticoagulation group serving as the reference. Heterogeneity was evaluated through the Cochrane Q test and Higgins I2 statistics. 26 A p-value of less than 0.05 was considered statistically significant. To examine discrepancies between direct and indirect estimates, node splitting analysis was performed. Network graphs were created using NMA Studio, where each node represents a different intervention, and the thickness of the connecting lines indicates the number of studies comparing those interventions. The illustrations, such as the forest plots and league tables, were designed to depict the network structure. Interventions were ranked using the P-score, with 1 indicating the most effective or safest, and 0 indicating the least. Publication bias was assessed both visually via funnel plots and statistically through Egger's regression test.
Results
Search Results
Following the initial search of electronic databases, including PubMed, ScienceDirect, and Cochrane Central, 1,249 articles were identified. After removing duplicates (n = 312), the remaining 937 articles underwent title and abstract screening. The articles passing this primary screening (n = 141) were then reviewed in full. In the end, 19 articles13,15,16,2027–41 were included in this network meta-analysis.
Study Characteristics
Nineteen studies13,15,16,2027–41 pooling a total of 9,512 patients and published between 2012 and 2024 were included. Five of the included studies were RCTs, while the rest were observational studies. The anticoagulants included were DOACs, LMWH, LMWH-Warfarin sequential therapy, VKAs, fondaparinux, and antithrombin-III. Five of the included studies were from the United States, seven were from China, three were from Japan, two were from Italy, and the rest were from Korea and Belgium. The age of the patients included ranged from 45 to 70 years. The baseline characteristics of the included studies are presented in Table 1. The imaging modalities used to verify the absence of thrombosis are shown in Supplementary Table 2.
Baseline Characteristics of the Included Studies.

Note: RC: Retrospective cohort; PC: Prospective cohort; DOAC: Direct oral anticoagulants, LMWH: Low molecular weight heparin; AT-III: Antithrombin-III; qd: once daily; MELD: Model for end-stage liver disease; VKA: Vitamin K antagonist; INR: International Normalized Ratio; all the data are presented as mean ± standard deviation, and median (range).
Risk of Bias
The risk of bias in the RCTs was evaluated using the RoB 2.0 tool. 24 All RCTs demonstrated a low risk of bias, except for Wang et al, 40 which raised some concerns due to bias in the measurement of outcome. The traffic light plot from the RoB 2.0 assessment is shown in Supplementary Figure 1. The bias risk for observational studies was assessed using NOS. Most included observational studies were of high quality, while three studies —Niu et al, 37 Senzolo et al, 39 and Joseph et al 20 —were rated as moderate quality. The NOS quality assessment is detailed in Supplementary Table 3.
Network Meta-Analysis
Complete Recanalization
Fourteen studies, including seven interventions, and 14 pairwise comparisons were included in the network meta-analysis of complete recanalization. Compared to no anticoagulation, DOACs (RR = 2.38; 95%CI:[1.17, 4.85]; p = 0.02), fondaparinux (RR = 18.16; 95%CI:[2.09, 158.13]; p = 0.009), LMWH (RR = 11.96; 95%CI:[1.58, 90.79]; p = 0.01), LMWH-Warfarin sequential therapy (RR = 2.32; 95%CI:[1.01, 5.31]; p = 0.04), and VKA (RR = 1.71; 95%CI:[1.03, 2.83]; p = 0.04) significantly increased the rate of complete recanalization except antithrombin-III (RR = 3.00; 95%CI:[0.57, 15.91]; p = 0.20) which didn't cause any significant increase (Figures 2A and 3A). The network plot of complete recanalization is shown in Figure 4A. Fondaparinux was ranked best (P-score = 0.94) regarding complete recanalization, whereas VKA was ranked worst (P-score = 0.28). The ranking of different anticoagulants based on the P-score is shown in Table 2 and Figure 5A. The heterogeneity detected was low (tau2 = 0.1133, I2 = 39%).
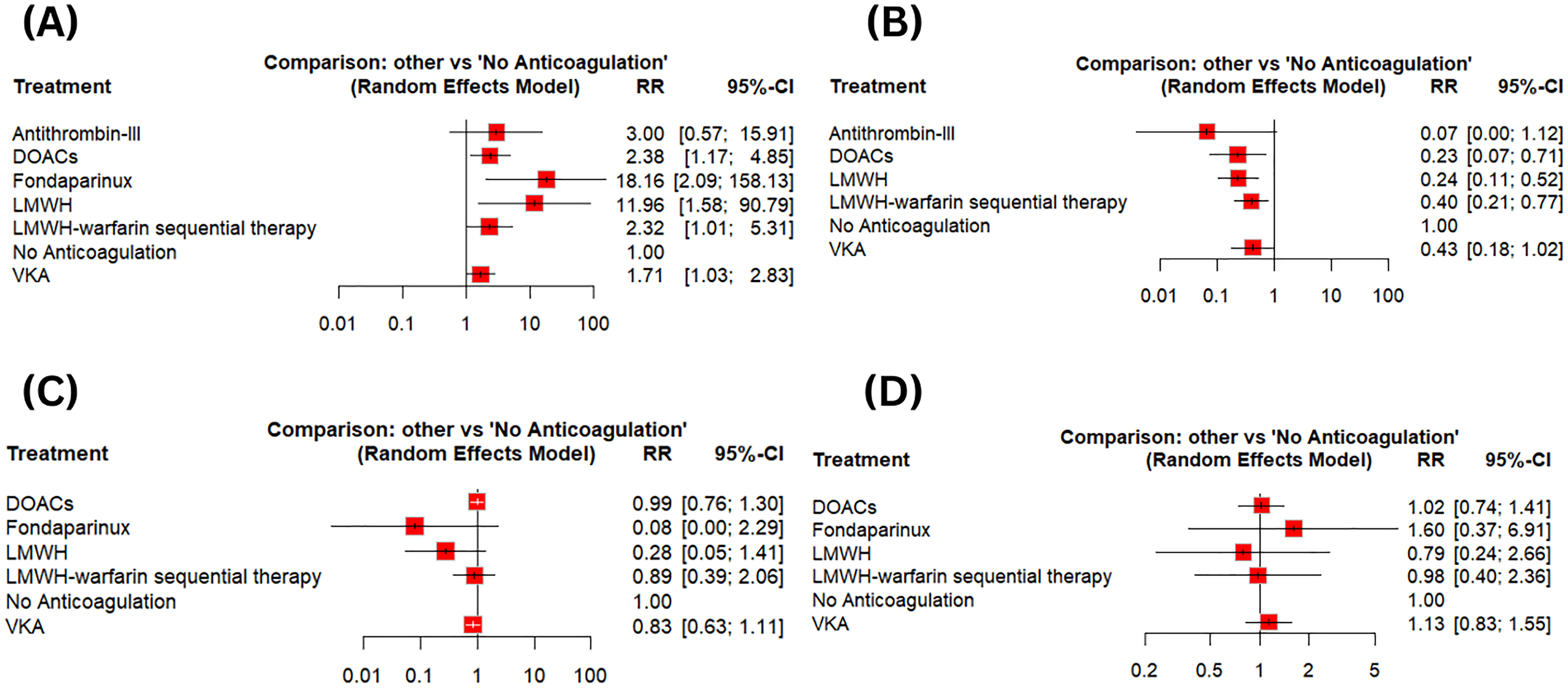
Forest Plots (A) Complete Recanalization (B) Portal Venous Thrombosis (PVT) Progression (C) Esophageal Variceal Bleeding (D) Overall Bleeding.

League Tables (A) Complete Recanalization (B) Portal Venous Thrombosis (PVT) Progression (C) Esophageal Variceal Bleeding (D) Overall Bleeding.

Network Plots (A) Complete Recanalization (B) Portal Venous Thrombosis (PVT) Progression (C) Esophageal Variceal Bleeding (D) Overall Bleeding.

P-Score Ranking Graphs (A) Complete Recanalization (B) Portal Venous Thrombosis (PVT) Progression (C) Esophageal Variceal Bleeding (D) Overall Bleeding.
Treatment Ranking Based on P-Scores.

Note: PVT: Portal venous thrombosis; DOAC: Direct oral anticoagulants, LMWH: Low molecular weight heparin; AT-III: Antithrombin-III; VKA: Vitamin K antagonist.
Portal Venous Thrombosis Progression
The network meta-analysis included 12 studies, including six interventions and 12 pairwise comparisons. Compared to no anticoagulation, DOACs (RR = 0.23;95%CI:[0.07, 0.71]; p = 0.01), LMWH (RR = 0.24;95%CI:[0.11, 0.52]; p = 0.0003), LMWH-Warfarin sequential therapy (RR = 0.40;95%CI:[0.21, 0.77]; p = 0.006) significantly reduced the PVT progression whereas antithrombin-III (RR = 0.07;95%CI:[0.004, 1.12]; p = 0.06), and VKA (RR = 0.43;95%CI:[0.18, 1.02]; p = 0.054) showed results comparable to no anticoagulation (Figures 2B and 3B). The network plot of the PVT progression is shown in Figure 4B. Antithrombin-III was ranked best (P-score = 0.87) regarding PVT progression, whereas VKA was ranked worst (P-score = 0.37). The P-score-based ranking details regarding different anticoagulants are given in Table 2 and Figure 5B. A low heterogeneity was observed (tau2 < 0.0001, I2 = 15.3%).
Esophageal Bleeding
Twelve studies, comprising six interventions and nineteen pairwise comparisons, were included in the network meta-analysis of esophageal bleeding. Compared to no anticoagulation, none of the interventions, including the DOACs (RR = 0.99; 95%CI:[0.76, 1.30]; p = 0.94), fondaparinux (RR = 0.08; 95%CI:[0.003, 2.29]; p = 0.14), LMWH (RR = 0.28; 95%CI:[0.05, 1.41]; p = 0.12), LMWH-Warfarin sequential therapy (RR = 0.89; 95%CI:[0.39, 2.06]; p = 0.79), and VKA (RR = 0.83; 95%CI:[0.63, 1.11]; p = 0.21), showed a significant decrease in the rate of esophageal bleeding (Figures 2C and 3C). The network plot of esophageal bleeding is shown in Figure 4C. Fondaparinux was ranked best regarding esophageal bleeding (P-score = 0.90), whereas DOACs were ranked worst (P-score = 0.24). The ranking of different anticoagulation regimens based on the P-scores is shown in Table 2 and Figure 5C. No heterogeneity was detected (tau2 < 0.0001, I2 = 0%).
Overall Bleeding
The network meta-analysis encompassed 17 studies, covering six interventions and 24 pairwise comparisons. Compared to no anticoagulation, none of the interventions, including the DOACs (RR = 1.02; 95%CI:[0.74, 1.41]; p = 0.89), fondaparinux (RR = 1.60; 95%CI:[0.37, 6.91]; p = 0.53), LMWH (RR = 0.79; 95%CI:[0.24, 2.66]; p = 0.71), LMWH-Warfarin sequential therapy (RR = 0.98; 95%CI:[0.40, 2.36]; p = 0.96), and VKA (RR = 1.13; 95%CI:[0.83, 1.55]; p = 0.43), showed a significant decrease in the rate of overall bleeding (Figures 2D and 3D). The network plot of the overall bleeding is shown in Figure 4D. LMWH was ranked best (P-score = 0.72) in terms of overall bleeding, whereas Fondaparinux was ranked worst (P-score = 0.24). The details regarding P-score ranking are given in Table 2 and Figure 5D. The overall heterogeneity observed was low (tau2 = 0.03; I2 = 11.2%).
Mortality
Seven studies, including four interventions and nine pairwise comparisons, were included in the network meta-analysis. Compared to no anticoagulation, DOACs (RR = 0.74; 95%CI: [0.67, 0.81]; p < 0.0001) significantly reduced the mortality whereas the VKA (RR = 0.92; 95%CI:[0.85, 1.00]; p = 0.05) and LMWH (RR = 0.42; 95%CI:[0.08, 2.33]; p = 0.32) showed no significant decrease (Supplementary Figures 2 and 3). The network plot of mortality is shown in Supplementary Figure 4. LMWH was ranked best (P-score = 0.80) regarding mortality, whereas VKA was ranked worst (P-score = 0.39). The P-score ranking details are given in Table 2 and Supplementary Figure 5. No heterogeneity was detected (tau2 < 0.0001, I2 = 0%).
Inconsistency and Publication Bias
The nodal split analysis was performed to assess the inconsistency between the direct and indirect evidence generated. The node split analysis details for each endpoint are presented in Supplementary Tables 4-8 and Figure 6A-D, as well as Supplementary Figure 6. The publication bias was evaluated visually with funnel plots and statistically using Egger's regression test. No publication bias was observed from the funnel plot symmetry, and this was further confirmed by the Egger's regression test results, as shown in Supplementary Figures 7–11.
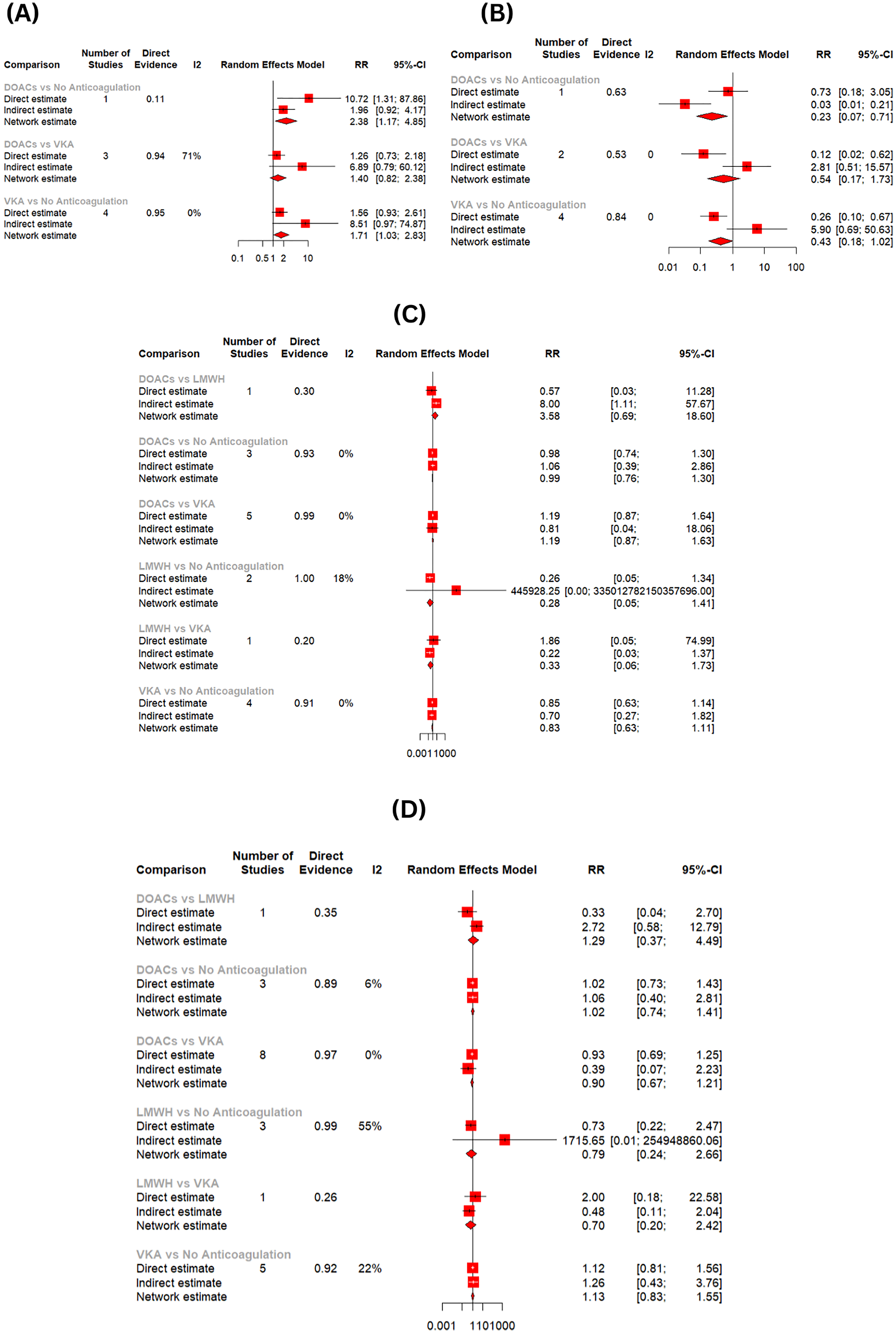
Node Split Analysis (A) Complete Recanalization (B) Portal Venous Thrombosis (PVT) Progression (C) Esophageal Variceal Bleeding (D) Overall Bleeding.
Discussion
PVT is a frequent and serious complication in cirrhosis, contributing to hepatic decompensation and increased mortality. 42 Anticoagulation promotes thrombus resolution but raises concerns about bleeding in patients with cirrhosis. Despite a rebalanced hemostatic state, 43 the optimal anticoagulant remains uncertain. While DOACs, LMWH, VKAs, fondaparinux and antithrombin-III show promise, direct comparisons are limited. This network meta-analysis evaluates six anticoagulants versus no treatment for efficacy and safety in cirrhotic PVT patients, aiming to guide evidence-based decisions through treatment ranking.
Regarding complete recanalization, all five anticoagulation regimens, including DOACs, fondaparinux, LMWH, LMWH-Warfarin sequential therapy, and VKA, showed a significant increase, except for antithrombin III, which showed nonsignificant results compared to no anticoagulation. DOACs, fondaparinux, LMWH, LMWH-Warfarin sequential therapy, and VKA all showed insignificant changes regarding overall bleeding and esophageal variceal bleeding compared to no anticoagulation. DOACs showed a statistically significant decrease in mortality compared to no anticoagulation, whereas LMWH and VKA showed comparable results regarding mortality compared to no anticoagulation. DOACs, LMWH, and LMWH-warfarin sequential therapy showed a statistically significant decrease in the PVT progression compared to no anticoagulation. Antithrombin III and VKA showed comparable results regarding the PVT progression compared to no anticoagulation.
Our findings regarding complete recanalization are consistent with prior meta-analyses that highlighted the efficacy of anticoagulants in promoting thrombus resolution in cirrhotic PVT patients.34,44,45 Interestingly, fondaparinux was ranked as the best treatment in achieving recanalization, despite limited statistical power in clinical practice due to smaller cohort sizes. Previous observational studies have demonstrated fondaparinux's favorable pharmacokinetic profile in liver-impaired patients, supporting this ranking in patients with liver impairment. 38 Conversely, antithrombin III showed comparable results to no anticoagulation, aligning with the literature, which suggests its role may be more preventative than therapeutic. VKAs, while statistically significant, were ranked the lowest, possibly due to variable INR control and hepatic metabolism challenges in cirrhotic patients. 43
Fondaparinux was ranked the best at reducing esophageal variceal bleeding. While not statistically significant, this finding suggests a clinically meaningful trend, likely attributable to its selective anti-factor Xa activity and renal clearance, which minimize hepatic side effects. On the other end of the spectrum, DOACs were ranked the worst, although evidence from observational studies remains inconclusive. For example, Chun et al 46 found that DOACs did not significantly increase the risk of variceal bleeding in cirrhotics; yet, concerns about the lack of reversal agents in acute settings persist.
Mortality reduction emerged most significantly with DOACs. VKAs showed non-significant results. These findings underscore recent shifts toward DOAC usage in patients with hepatic disease, supported by studies such as Chun et al. 46 which demonstrated superior safety and survival outcomes with rivaroxaban compared to traditional anticoagulants. LMWH, while not statistically superior, was ranked best in reducing mortality. This may reflect its dual benefit in thrombus resolution and anti-inflammatory effects, particularly relevant in decompensated cirrhosis, where systemic inflammation worsens outcomes. 42 These findings emphasize that treatment rankings should not be disregarded due to a lack of statistical significance, as they may signal emerging efficacy trends likely to achieve significance in future studies or larger populations.
All regimens, including DOACs and VKAs, showed no statistically significant difference in overall bleeding risk. However, LMWH ranked best, while fondaparinux ranked worst. While this seems paradoxical to its favorable variceal bleeding profile, the divergence may stem from the cumulative measure of overall bleeding (eg, soft tissue, gastrointestinal, procedural), wherein fondaparinux's once-daily dosing and long half-life may increase risk during acute events.
DOACs, although not the best-ranked, have been shown in emerging studies to confer lower bleeding risks than warfarin, particularly with careful dosing.47–49 Importantly, none of the anticoagulants caused a statistically significant increase in bleeding, 17 50–52 reinforcing the notion that with proper screening and prophylaxis (eg, variceal ligation), anticoagulation can be safely administered in cirrhotics.38,53
Three therapies, including DOACs, LMWH, and LMWH-warfarin sequential therapy, showed a statistically significant reduction in PVT progression, while VKA and antithrombin III showed an insignificant reduction. Interestingly, antithrombin III was ranked the best, although the difference was statistically insignificant. VKAs ranked worst, possibly due to INR variability and the slow onset of action, which are problematic in managing rapidly progressing PVT in cirrhotic patients.21,54
Strengths and Limitations
A key strength of this analysis is its comparative scope across multiple anticoagulants, including less-studied agents such as fondaparinux and antithrombin III. The use of network meta-analysis methodology allowed indirect comparisons and generated treatment hierarchies, offering clinically actionable insights even in the absence of strong statistical significance.
However, limitations must be acknowledged. Heterogeneity in study design, sample sizes, and liver disease severity across included studies may introduce bias. Data on Child-Pugh scores, variceal prophylaxis, and renal function were variably reported, limiting subgroup analyses. Additionally, the low event rates and limited RCTs in cirrhotic populations necessitate cautious interpretation of treatment effects and rankings. Another limitation is that we were unable to investigate the unfractionated heparin (UFH) in PVT caused by cirrhosis since the included studies did not evaluate it therefore future studies should assess the role of UFH in PVT. Additionally, because this parameter was not reported in the included studies, we were unable to evaluate the anti-Xa activity of various anticoagulants. As a result, we recommend that future research evaluate the anticoagulants’ anti-Xa activity to gain a better understanding of their effectiveness in PVT caused by cirrhosis. Most of the included studies grouped multiple DOACs together without reporting data for each separately limiting our ability to analyze each of them separately. The results of this network meta-analysis should be interpreted cautiously due to the heterogeneity in follow-up periods and the absence of consistent data on study duration.
From a clinical standpoint, this analysis supports the growing use of DOACs and LMWH as effective and safe options in cirrhotic PVT. Fondaparinux emerged as a promising agent in both recanalization and bleeding outcomes, warranting larger randomized trials to confirm its profile. LMWH's consistent ranking across multiple outcomes further supports its continued use, especially in hospitalized or decompensated patients. Importantly, clinicians must appreciate that network meta-analysis rankings are probabilistic and not constrained by conventional p-value thresholds. A regimen ranked highest may show non-significant results now but reflects a consistent and reproducible trend toward benefit, thereby guiding therapeutic decisions until further evidence matures.
Conclusion
This network meta-analysis provides a comprehensive comparative overview of six anticoagulation strategies in patients with PVT and cirrhosis. DOACs, fondaparinux, LMWH, LMWH-Warfarin sequential therapy, and VKA significantly improved complete recanalization, with fondaparinux ranked as the most effective regimen. DOACs, LMWH, and LMWH-Warfarin sequential therapy showed a significant reduction in PVT progression, with antithrombin-III ranked as the most effective in reducing PVT progression. Fondaparinux was ranked highest for reducing esophageal variceal bleeding, while LMWH was ranked highest for reducing overall bleeding. Compared to no anticoagulation, DOACs significantly decreased mortality, and the LMWH was ranked highest for mortality reduction. These findings underscore the potential of emerging anticoagulants, such as fondaparinux and DOACs, in reshaping therapeutic approaches for cirrhotic PVT. Nonetheless, given the inherent limitations of current evidence and variability in study populations, large-scale RCTs are needed to confirm these trends and optimize individualized anticoagulation strategies in this complex clinical setting.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251405419 - Supplemental material for Comparative Effectiveness and Safety of Various Anticoagulation Regimens for Portal Venous Thrombosis in Cirrhosis: A Systematic Review and Network Meta-Analysis
Supplemental material, sj-docx-1-cat-10.1177_10760296251405419 for Comparative Effectiveness and Safety of Various Anticoagulation Regimens for Portal Venous Thrombosis in Cirrhosis: A Systematic Review and Network Meta-Analysis by Zain ul Abideen, Muhammad Hassan Waseem, Noor Ul Huda Ramzan, Hamna Raheel, Fariha Hasan, Pawan Kumar Thada and Prasun K. Jalal in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Ethical Approval
Not applicable.
Patient Consent
Not applicable.
Authors’ Contributions CRediT Roles
Study concept and design: ZUA and MHW; acquisition of data: ZUA and NUHR; analysis and interpretation of data: HS and ZUA; drafting of the manuscript: NUHR, FH, MHW, and PKT; critical revision of the manuscript: PKJ.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
Data will be made available upon reasonable request to the authors.
Status
This manuscript has not been published previously and is not under consideration for publication elsewhere.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
