Abstract
En-stage renal disease (ESRD) is a growing public health problem. The atherosclerotic cardiovascular complications are the leading causes of mortality and morbidity in the ESRD. In this study, we sought to quantify the levels of thrombo-inflammatory biomarkers in an ESRD patients in comparison to healthy controls to determine their relevance in thrombo-inflammation and adverse outcomes. The levels of D-Dimer, C-reactive protein (CRP), plasminogen activator inhibitor 1 (PAI-1) antigen, functional PAI-1, thrombin activatable fibrinolysis inhibitor, tissue plasminogen activator, von Willebrand factor, and anti-PF4 IgG and microparticle (MP) activity were quantified by using commercially available ELISA immunoassays for each of the ESRD (n = 73) and control plasma samples (n = 10). The levels of endogenous glycosaminoglycans (GAGs) were quantified by utilizing a Heparin Red Probe (Redprobes UG, Germany). The collected data were analyzed to demonstrate the relationship between various parameters. All the tested biomarkers were increased in ESRD patients in comparison to healthy controls (p < 0.05). These biomarkers have shown significant correlations within each other except for anti-PF4 Ig G and MPs. The CRP levels were significantly higher in patients who had coronary artery disease (CAD) (p < 0.05), but there was no significant difference in other biomarkers according to the cardiovascular outcomes. In the multivariate analysis, the CRP (odds ratio: 1.19; 95% confidence interval: 1.01–1.41; p: 0.03) value was an independent predictor of CAD. In this study, we demonstrated increased levels of 10 different biomarkers in ESRD patients. The CRP levels can be a good predictor of CAD in ESRD patients.
Keywords
Introduction
Chronic kidney disease (CKD) and its terminal stage end-stage renal disease (ESRD) is a growing public health problem. The CKD affects more than 10% of the general population, affecting more than 850 million people globally. 1 It is estimated that 15% of the US adults or 37 million people to have CKD in the United States. 2 There are more than two million and 785.000 ESRD patients worldwide and in the United States, respectively.3,4 Moreover, the prevalence of CKD and ESRD appears to be rising due to the increase of the traditional risk factors of atherosclerosis among the general population and the widespread availability of renal replacement programs. 5 However, roughly 90% of the patients with CKD are not aware of their disease. 2 Therefore, a better understanding of the complex pathophysiology of these disease states may provide better insight for developing better diagnostic, prognostic, and therapeutic tools.
While CKD is defined by the presence of structural damage to the kidneys or decreased kidney function for more than 3 months, the degree of proteinuria and the glomerular filtration rate (GFR) are the two most important indicators of the CKD severity.6,7 The progressive course of CKD is generally associated with a gradual decline in the GFR to ESRD. ESRD is the most severe stage of CKD that is defined by a GFR of less than 15 mL/min.6,7 Once ESRD is diagnosed, the only treatment option is renal replacement therapy with peritoneal or hemodialysis. 8 Unfortunately, even with the modern techniques of hemodialysis, the death rates are high, varying from 20% to 50% over 2 years in this patient population. 9 Kidney transplantation is available for only 10% of ESRD patients due to long organ waiting times. 4
Patients with CKD and ESRD are exposed to a higher comorbidity, worse quality of life, higher healthcare expenses, and higher overall mortality mainly due to atherosclerotic cardiovascular diseases (CVDs). 10 Therefore, CKD is established as an independent risk factor for CVD and aggressive risk factor reduction is recommended as a part of standard therapy in many of the practice guidelines.11,12 Advanced age, male gender, smoking, hypertension, obesity, diabetes mellitus, dyslipidemia, and genetic susceptibility have previously described traditional atherosclerotic risk factors for the development of CKD, preceding ESRD. 13 However, these traditional risk factors cannot completely explain the high frequency of CVD in this patient population. While complex pathophysiological pathways contribute to the development and progression of CKD and ESRD, emerging evidence suggests that inflammation has a major role in its development.
Irrespective of the cause of a kidney disease, there is strong evidence that an inflammatory state exists in patients with CKD and ESRD.14,15 Regarding this, recent studies have shown that the levels of inflammatory biomarkers such as C-reactive protein (CRP), tumor necrosis factor alpha (TNF-α), interleukin-1 (IL-1), and IL-6 are elevated in CKD and ESRD patients.16,17 Furthermore, this elevation has also shown to be associated with the progression of CKD to ESRD with higher rates of cardiovascular complications and mortality in this patient group.17–20 Interestingly, major improvements were observed at the levels of inflammatory biomarkers after kidney transplantation; this has indicated that inflammation is not only a cause but also a consequence in the development of a kidney disease and its associated complications. 21 Regarding this, several dialysis-related factors have been associated with persistent and low-grade inflammation in ESRD. 22 However, no major differences were observed in serum inflammatory biomarkers between long-term dialysis patients and those who were not dialyzed. 22 Additionally, impaired renal clearance has been identified as a cause of accumulation of uremic toxins, advanced glycosylation products, and inflammatory mediators, which further lead to oxidative stress and endothelial dysfunction in CKD and ESRD patients. 23 Moreover, chronic infections (eg, Chlamydia pneumoniae, Helicobacter pylori, and periodontitis), over activation of the renin-angiotensin-aldosterone system, fluid overload and hypertension, and the increased levels of homocysteine and impaired clearance of leptin are some other causes that are likely to contribute to inflammation in CKD and ESRD. 23 Nevertheless, the exact mechanisms are not well established and the complex interplay of inflammation in the pathogenesis of CKD and ESRD remains unclear.
CKD and ESRD are complex medical conditions that affect multiple organ systems. Bleeding and thrombotic events are common which can result from the disruption of the hemostatic process with an associated inflammatory state and endothelial damage. 24 While impairment in platelet function appears to be the dominant abnormality in uremic bleeding, the elevated levels of d-dimer, fibrinogen, tissue factor, von Willebrand factor (vWF), plasminogen activator inhibitor 1 (PAI-1), thrombin activatable fibrinolysis inhibitor (TAFI), tissue plasminogen activator (tPA), and microparticles (MPs) have been shown in ESRD patients, indicating a prothrombotic disease state with impaired fibrinolysis in ESRD.24–26 Additionally, the formation of anti-platelet factor 4 (anti-PF4) antibodies have also been shown and associated with adverse outcomes in CKD and ESRD. 27 However, the relevance of these findings remains unclear, and not much is known on the interrelationship of these factors.
Atherosclerotic cardiovascular complications including myocardial infarction (MI), stroke, and peripheral arterial disease are the leading causes of mortality and morbidity in ESRD, and endothelial dysfunction seems to be another key factor for these adverse outcomes. 28 The endothelial biomarkers were evaluated in previous studies in CKD and ESRD patients. 29 However, current knowledge on their clinical use is limited. Heparin Red probe is a novel fluorescence assay which quantifies heparins (including unfractionated heparin, low molecular weight heparins, and non-anticoagulant heparins) and other endogenous polyanionic sulfated glycosaminoglycans (GAGs) with a sensitivity range of 0–6 µg/ml in plasma samples. The probe enables direct detection of heparins and other sulfated GAGs in the circulation by a simple mix-and-read microplate assay which is independent of any anticoagulant activity.30,31 This method offers a simple and rapid assay for measuring endogenous GAGs which can reflect endothelial damage in plasma samples.
In this study, we sought to quantify the levels of thrombo-inflammatory biomarkers including D-Dimer, CRP, PAI-1 antigen, functional PAI-1, TAFI, tPA, endogenous GAGs, vWF, anti-PF4 Ig G, and MPs in a group of ESRD patients in comparison to healthy controls to determine whether there is a relationship existing between them. Identification of this relationship may provide insight into the complex pathophysiology of CKD and ESRD and may be useful for the development of potential prognostic, diagnostic, and therapeutic interventions.
Materials and Methods
Whole blood samples were drawn from 73 adult ESRD patients who were treated at the Loyola University Medical Center Hemodialysis Unit under an Institutional Review Board (IRB)-approved protocol. All the patients were hemodialysis patients. Plasma samples were collected in 3.8% (0.109 mol/L) sodium citrate tubes, processed for platelet-poor plasma, and stored at −70 °C prior to analysis. Control plasma samples from healthy, non-smoking adults, aged 19 to 53, were purchased from a commercially available source (George King Biomedical, Overland Park, Kansas, USA). The levels of D-Dimer, CRP, PAI-1 antigen, functional PAI-1, TAFI, tPA, vWF, anti-PF4 IgG, and MPs were quantified by using commercially available ELISA immunoassays for each of the ESRD and control plasma samples. The levels of endogenous GAGs were quantified by utilizing the Heparin Red Probe (Redprobes UG, Germany). The clinical information including the patient demographics, comorbid illnesses, laboratory results, and treatment characteristics was collected through the review of patient electronic medical records.
The calculations were performed using SPSS Statistics (IBM) and Prism (GraphPad, La Jolla, California, USA) software. Statistical differences between the ESRD groups and normal controls were evaluated utilizing nonparametric Mann–Whitney U, Student’s t-tests, and Kruskal–Wallis ANOVA test. Correlation analysis was performed by utilizing Spearman correlation coefficients. Univariate and multivariate logistic regression analysis was performed to identify the possible predictors of cardiovascular outcomes of coronary artery diseases (CADs), cerebrovascular diseases, and peripheral artery diseases. Any variable having a p value ≤ 0.1 in univariate analysis is included in the multivariate analysis. A p < 0.05 was considered statistically significant.
Results
Of the 73 patients that were included in the final analysis, the median age of the patients was 69.0 years, with 31 female patients (42.5%) and 42 male patients (57.5%). The patient population had a high frequency of CVD events in which 30 patients (41.1%) had CAD, 11 patients (15.1%) had cerebrovascular disease, 31 patients (42.5%) had peripheral artery disease, 13 patients (17.8%) had heart failure (EF ≤ 50%), and 16 patients (21.9%) had atrial fibrillation confirmed diagnosis. The details of patient information including demographics, comorbid illnesses, laboratory results, and treatment characteristics are shown in Table 1.
Patient Characteristics (N = 73).

Abbreviations: HDL, high-density lipoprotein; LDL, low-density lipoprotein.
All of the thrombo-inflammatory biomarkers and endogenous GAGs were significantly elevated in ESRD patients compared to healthy controls. The ESRD patients exhibited varying degrees of increase in the D-Dimer, CRP, PAI-1 antigen, functional PAI-1, TAFI, tPA, endogenous GAGs, vWF, anti-PF4 IgG and MPs, as shown in Figure 1 (p < 0.05). D-Dimer showed the most pronounced increase (2649.54%) followed by tPA (1423.68%), PAI-1 antigen (500.72%), anti-PF4 IgG (366.66%), endogenous GAGs (348.14%), MPs (251.13%), functional PAI-1 (250.35%), and CRP (229.96%). The composite data, including the average, standard deviation, and percent increase in the levels of each biomarker and endogenous GAGs in comparison to healthy controls are shown in Table 2.

Levels of thromboinflammatory biomarkers, anti-PF4 Ig G and endogenous GAGs in ESRD patients and healthy controls.
The Levels of Biomarkers and Endogenous GAGs in ESRD Patients and Healthy Controls.

In the correlation analysis, D-Dimer was correlated with PAI-1 antigen (r = −0.33) and CRP (r = 0.32). The CRP was correlated with D-Dimer (r = 0.32), tPA (r = 0.26), PAI-1 antigen (r = 0.25), and functional PAI-1 (r = −0.24). The PAI-1 antigen was correlated with tPA (r = 0.54), D-Dimer (r = −0.33), endogenous GAGs (r = 0.31), CRP (r = 0.25), and functional PAI-1 (r = −0.25). Functional PAI-1 was correlated with PAI-1 antigen (r = −0.25) and CRP (r = −0.24). The TAFI was correlated with tPA (r = 0.20). Endogenous GAGs were correlated with PAI-1 antigen (r = 0.31) and vWF (r = 0.24). The vWF was correlated with tPA (r = 0.28) and endogenous GAGs (r = 0.24). Anti-PF4 IgG and MPs were not correlated with any of the tested biomarkers. The results of correlation analysis are depicted in Figure 2.
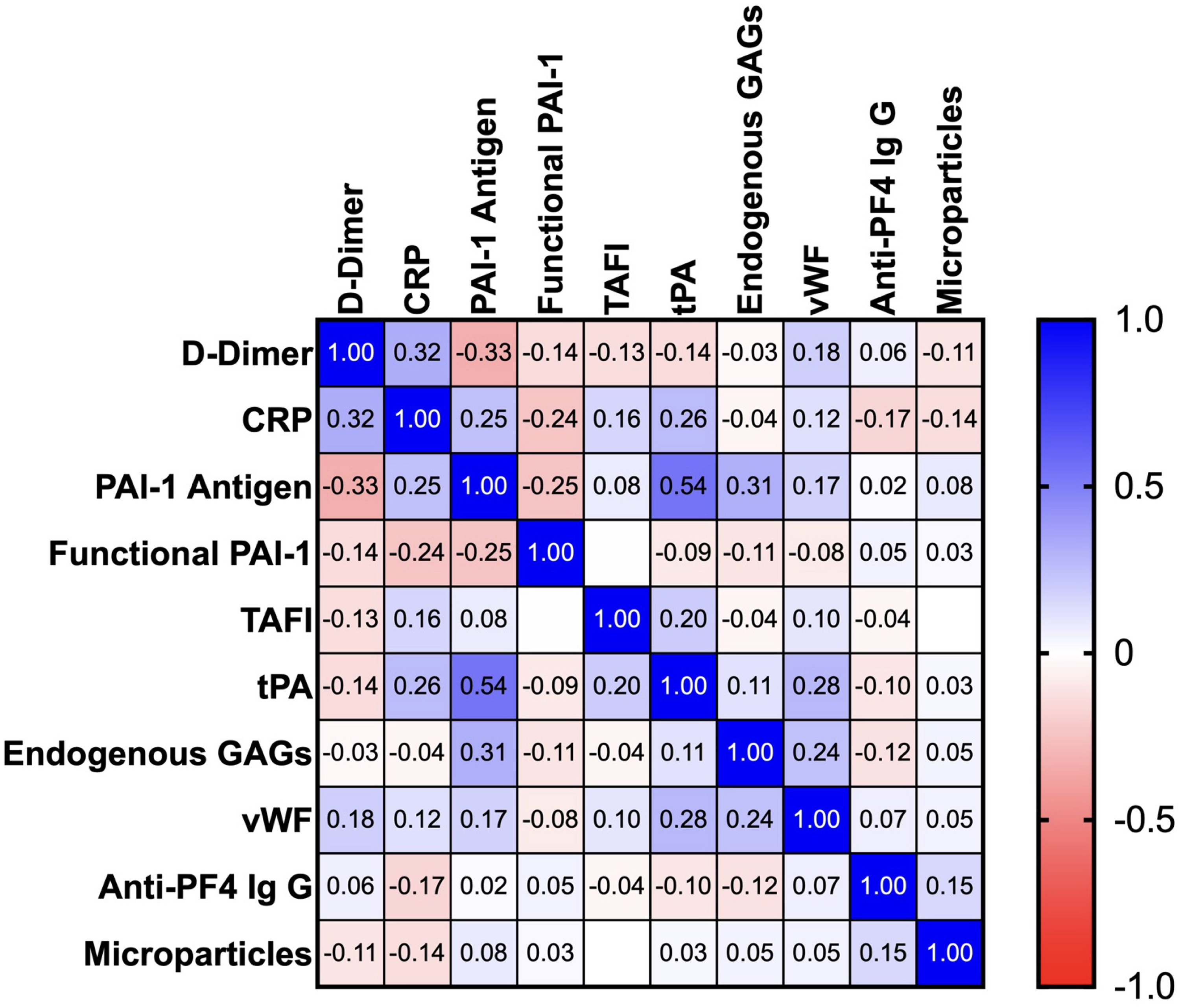
Correlation analysis of thromboinflammatory biomarkers, anti-PF4 Ig G and endogenous GAGs in ESRD patients.
The CRP levels of ESRD patients were significantly higher in patients with CAD, in comparison to the patients without CAD (p: 0.01). The biomarker and endogenous GAG levels did not show any significant difference according to cerebrovascular and peripheral artery diseases (p > 0.05). The composite data regarding these comparisons are represented in Tables 3, 4 and 5.
The Levels of Biomarkers and Endogenous GAGs According to CAD in ESRD Patients.

The Levels of Biomarkers and Endogenous GAGs According to Cerebrovascular Disease in ESRD Patients.

The Levels of Biomarkers and Endogenous GAGs According to Peripheral Artery Disease in ESRD Patients.

In univariate analysis, to have a cerebrovascular disease and peripheral artery disease diagnosis, to be under statin, antiplatelet and beta blocker therapy, and the levels of high-density lipoprotein and CRP were significantly associated with CAD. The triglyceride levels and having a CAD diagnosis were significantly associated with cerebrovascular disease. The ESRD causes other than hypertension or diabetes mellitus, having a CAD diagnosis and being under antiplatelet therapy were significantly associated with peripheral artery disease. The results of univariate analysis are presented in Table 6.
Univariate Analysis of Predictors for Cardiovascular Outcomes.

Abbreviations: CAD, coronary artery disease; CD, cerebrovascular disease; PAD, peripheral artery disease; RR, relative risk; HDL, high-density lipoprotein; LDL, low-density lipoprotein.
In the multivariate analysis, having a peripheral artery disease diagnosis (odds ratio [OR]: 7.03; 95% confidence interval [CI]: 1.75–28.19; p < 0.01), being under beta blocker therapy (OR: 4.87; 95% CI: 1.06–22.46; p: 0.04) and the levels of CRP (OR: 1.19; 95% CI: 1.01–1.41; p: 0.03) were significant predictors for CAD. The levels of triglycerides (OR: 1.02; 95% CI: 1.004–1.045; p: 0.01) and being under calcium channel blocker therapy (OR: 13.19; 95% CI: 1.48–117.18; p: 0.02) were significant predictors for the cerebrovascular disease. Only having a CAD diagnosis (OR: 4.98; 95% CI: 1.64–15.11; p < 0.01) was a significant predictor of the peripheral artery disease. The results of multivariate analysis are presented in Table 7.
Multivariate Analysis of Predictors for Cardiovascular Outcomes.

Discussion
In this study, we were able to determine the levels of 10 different biomarkers in a relatively large group of ESRD patients. Of these biomarkers, D-dimer and CRP are highly related to hemostatic activation and inflammation; PAI-1 antigen, functional PAI-1, TAFI, and tPA are more representative for fibrinolytic dysregulation; vWF and endogenous GAGs are markers of endothelial dysfunction; and anti-PF4 Ig G antibodies with MPs are more representative for the activation of platelets and immune dysregulation in ESRD patients. The major findings of this study can be summarized as: (1) The levels of all the tested biomarkers were elevated in ESRD patients in comparison to healthy controls. (2) These biomarkers have shown significant correlations to each other except for anti-PF4 Ig G and MPs. (3) The CRP levels were significantly higher in patients who had CAD, but there was no significant difference in any of the biomarkers according to the other vascular complications such as cerebrovascular or peripheral artery diseases. (4) The levels of CRP were significantly associated with CAD in both univariate and multivariate analyses; none of the remaining tested biomarkers have shown a significant association with cerebrovascular or peripheral artery disease.
The increased levels of D-Dimer in ESRD have been shown in recent studies; however, its relationship with inflammation and inflammatory biomarkers is less well understood. For instance, Huang et al have shown that the levels of D-dimer, vWF, fibrinogen, Factor VII (FVII), and FVIII were elevated in patients with CKD in comparison to the healthy controls, and this elevation was in parallel with the progression of CKD to ESRD. 32 Gubensek et al have shown an increased prevalence of elevated D-dimer levels in chronic hemodialysis patients even without additional comorbidities. 33 Vaziri et al have shown increased levels of D-dimer, vWF, FVII, and FXIII antigens in ESRD patients. 34 However, although the levels of D-dimer were evaluated, the relationship of D-dimer with inflammatory biomarkers has not been demonstrated in these studies. Regarding this, two recent publications of our group have evaluated the levels of D-dimer along with inflammatory biomarkers in ESRD patients. The study reported by Sharain et al has evaluated D-dimer, CRP, neuron-specific-enolase, neutrophil gelatinase-associated lipocalin, tumor necrosis factor receptor 1, and thrombomodulin (TM) in CKD and ESRD patients in comparison to healthy controls. 18 This study demonstrated a significant upregulation of all the tested biomarkers in these patients. Another study of our group by Nelson et al has evaluated the biomarkers including D-dimer, thrombin-antithrombin III (TAT) complex, prothrombin fragment (F1.2), fibrinopeptide A (FPA), CD40 ligand, myeloperoxidase, TNF-α, monocyte chemotactic protein-1, and nitric oxide (NO), which has shown increased levels of all the tested biomarkers in ESRD patients in comparison to healthy controls. 35 In our current study, while the D-dimer levels have shown the most prominent increase among all the tested biomarkers, both D-dimer and CRP levels were elevated in ESRD patients. Furthermore, both D-dimer and CRP levels were correlated with each other, and they also showed significant correlations with PAI-1 antigen and functional PAI-1. This may be the reflection of a possible relationship between hemostatic activation, inflammation, and fibrinolytic dysregulation in ESRD patients.
The fibrinolytic dysregulation is another important finding of our study which was also investigated in previous studies with conflicting results. In an early study by Irish et al, the levels of PAI-1 antigen were significantly lower in ESRD patients compared to healthy controls. 36 Mezzano et al could not show any significant difference in the levels of PAI-1 antigen between ESRD patients and healthy controls. 37 Alwakeel et al investigated coagulation inhibitors and fibrinolytic parameters in ESRD patients, reporting no significant difference in tPA and PAI levels in ESRD patients. 38 Lottermoser et al determined the levels of tPA and its main antagonist PAI-1 in CKD and ESRD patients in comparison to healthy controls. 39 In this study, the plasma concentrations of PAI-1 and tPA antigen as well as the PAI-1:tPA molar ratio were unchanged in CKD patients in comparison to healthy controls. However, tPA concentrations were markedly decreased while PAI-1 antigen concentrations remained in the control range causing a significant increase in the PAI-1:tPA molar ratio in the ESRD group. The authors of this study concluded that fibrinolysis is markedly disturbed in ESRD patients due to a decreased availability of tPA. Segarra et al have investigated the levels of PAI-1, tPA, and TM as endothelial cell glycoproteins in ESRD patients. 40 In this study, when compared with healthy controls, dialysis patients showed increased levels of CRP, TM, tPA, and PAI-1, and PAI-1 was an independent predictor of CAD in the multivariate analysis. In our current study, we were able to examine the PAI-1 levels with two different methods in terms of the antigen level and functionality. We have observed that both the antigen levels and activity of PAI-1 were elevated in ESRD patients although the elevation of functional PAI-1 was less than the elevation of PAI-1 antigen. Interestingly, the levels of tPA were also increased in our patient cohort. In line with this, Sabovic et al have determined the levels of D-dimer, fibrinogen, PAI-1 antigen and activity, tPA, TAT, and F1 + 2, before and immediately after the regular hemodialysis sessions in ESRD patients to evaluate the influence of hemodialysis-related factors on their levels. 41 In this study, the markers of coagulation and fibrinolysis activation remained unchanged during the process of hemodialysis. However, in post-hemodialysis samples, tPA activity was significantly increased which has provided an association between dialysis-related factors with the levels of tPA in hemodialysis patients. In our current study, there was a positive correlation (r = 0.54) between tPA and PAI-1 antigen, but functional PAI-1 and tPA were not correlated. Considering the fact that both PAI-1 antigen and tPA are released from endothelium, it may be possible that the increases of PAI-1 antigen and tPA are due to the ongoing endothelial damage or hemodialysis-related factors in our patient cohort in the current study. Supporting this, the levels of PAI-1 antigen were correlated with endogenous GAGs and the levels of tPA were correlated with vWF, which are released in response to glycocalyx degradation leading to endothelial dysfunction. It is important to note that we measured the activity of vWF in this study. Although the activity of vWF is usually comparable with the antigen levels, other factors such as uremia, inflammation, platelet activation, or individual polymorphisms may also have an influence on vWF activity.
Another important finding of our study was the results of TAFI activity observed in our patients. Related to this, a recent study by Malyszko et al assessed the TAFI antigen and activity, TAT, F1 + 2, and plasmin–antiplasmin complexes in CKD and ESRD patients. 25 In this study, the levels of TAFI antigen and activity were elevated in only diabetic CKD and ESRD patients, while they did not show any significant difference in non-diabetic patients. Gad et al evaluated the levels of CRP, TAFI, malondialdehyde (MDA), and soluble intracellular adhesion molecule-1 (sICAM-1) in CKD and ESRD patients compared to healthy controls. 42 The CRP, TAFI, MDA, and sICAM-1 were significantly elevated in renal impairment patients. However, only TAFI and MDA were significantly higher in hemodialysis patients with CVD in this patient cohort. In a previous publication of our group, Tobu et al evaluated the levels of CRP, NO, TAFI antigen, and TAFI activity in ESRD patients. 43 In this study, the measured levels of all the tested biomarkers were increased in ESRD patients when compared to healthy controls. In the current study, the levels of TAFI activity were increased in our patient samples and the TAFI activity was correlated with the levels of tPA. It is important to note that approximately 55% of our patient cohort had a diabetes mellitus diagnosis which could also impact our reported findings. Nevertheless, the reported results provide a rationale for the mechanisms of fibrinolytic dysregulation in ESRD population.
The elevated levels of vWF as a marker of endothelial dysfunction have also been reported in ESRD patients. An early study by Gris et al has shown increased levels of vWF, tPA, uPA, and TM in hemodialysis patients. The study of Kim et al has shown higher levels of vWF and D-dimer in continuous ambulatory peritoneal dialysis patients. 44 Borawski et al have evaluated the levels of plasma soluble vWF in 110 hemodialysis patients. 45 In this study, the levels of vWF were increased and directly associated with the presence of CVD, patient age, fibrinogen levels, and the use of enoxaparin during hemodialysis. Multivariate analysis has shown that low pre-dialysis blood pressure, hypoalbuminemia, and hyperfibrinogenemia were independent predictors of high vWF levels. In the study of Bolton et al, vWF and adhesion molecules VCAM-1 and ICAM-1 and E-selectin were increased in hemodialysis patients. 46 Additionally, the inflammatory biomarkers of IL-6 and TNF-α levels were also positively correlated with vWF levels in this study. The study reported by Kirmizis et al has shown significantly elevated levels of plasma fibrinogen, D-dimer and vWF levels in ESRD patients with CVD. 47 Péquériaux et al have evaluated vWF and platelet activation markers in 671 hemodialysis patients in which the levels of vWF have shown a positive correlation with total mortality. 48 The results of the study published by van der Vorm et al were similar showing significantly elevated levels of active vWF in CKD, hemodialysis, and peritoneal dialysis patients. 49 Ocak et al prospectively evaluated 956 hemodialysis patients. 50 In this study, high vWF levels and low ADAMTS13 activity were associated with increased mortality risk in hemodialysis patients. Our findings were similar to the recent reports, that the levels of vWF were elevated in ESRD patients in comparison to healthy controls. The underlying mechanism of this finding is likely related to the ongoing endothelial dysfunction that is generally observed in ESRD patients. Moreover, we utilized a novel method to measure the levels of endogenous GAGs which has also shown increased levels in our patient group. Although this method needs to be further evaluated in more well-designed, controlled studies in different pathological disease states, it is important to note that we were able show elevated levels of endogenous GAGs in various pathological plasma samples including ESRD in one of our recent studies. 51 Interestingly, while the levels of vWF were correlated with the levels of tPA, endogenous GAGs were correlated with the levels of PAI-1 antigen. This finding might be due to the fact that while the levels of vWF are significantly associated with endothelial cell damage, endogenous GAGs are significantly associated with glycocalyx degradation. A recent report of our group has demonstrated the relevance of absolute measurements of GAGs to investigate absolute kinetics of heparin. 52 The current technology using the Heparin Red Probe measures collective levels of endogenous GAGs; however, newer methods are developed to measure different species of GAGs, and it will be helpful to identify the relevance of specific GAGs in various disease states. Additional studies to demonstrate this relevance are warranted at this time.
Several studies have shown that the interplay of inflammation, endothelial damage, and the generation of anti-PF4 antibodies may contribute to the adverse outcomes of arterial and venous thrombotic events. Regarding this, Pena de la Vega et al prospectively evaluated the adverse outcomes of hemodialysis if they could be explained by subclinical heparin-induced thrombocytopenia (HIT). 27 In this study, the all-cause mortality rate was significantly higher for patients with higher levels of anti-PF4 antibodies compared to the patients that have lower levels. Interestingly, the prevalence of HIT in patients was not greater than the expected from the other patient populations in this study. Palomo et al investigated anti-PF4 antibodies in 207 hemodialysis patients, and this study could not show any statistically significant difference between anti-PF4 antibodies and thrombocytopenia or thrombosis. 53 Similarly, the study of Asmis et al could not also show any relationship between the anti-PF4 antibodies and the development of thrombocytopenia, arterial cardiovascular events, venous thromboembolism, vascular access occlusion, and mortality. 54 Carrier et al evaluated the association between PF4 antibodies and mortality in a prospective cohort of 419 asymptomatic hemodialysis patients. 55 The relative risk of death was significantly increased for patients with IgG-specific anti-PF4 antibodies after controlling the potential confounders. The authors suggested that IgG-specific anti-PF4 antibodies were associated with increased mortality in hemodialysis patients. In the cross-sectional study published by Tan et al, multivariate analysis has shown that the prevalence of anti-PF4 Ig G antibodies was an independent risk factor for the occurrence of arterial and venous thrombosis. 56 Tsai et al evaluated the role of anti-PF4 antibodies on access fistula thrombosis, PAD, and CAD in 111 hemodialysis patients. 57 While there was a significant difference in the generation of anti-PF4 antibodies for only access fistula thrombosis, it was not observed for PAD and CAD in this study. In the study of Zhao et al, the anti-PF4 antibodies were not related to the occurrence of clinical bleeding, thromboembolic events and risk of death within two years of follow-up in maintenance hemodialysis patients. 58 In summary, the reported findings of anti-PF4 antibodies in the past literature are diverse with different results for different outcomes. This may also be related to the utilization of different methods to detect anti-PF4 antibodies in hemodialysis patients, who exhibited a higher frequency of exposure to different types of heparins than other patient populations. In our current study, the levels of anti-PF4 antibodies were increased in our patient group who were treated under a regular hemodialysis protocol. Interestingly, we were not able to show any significant association between anti-PF4 Ig G antibodies with any of the thrombo-inflammatory biomarkers. We also did not observe any significant difference in the levels of anti-PF4 Ig G antibodies in patients with coronary, cerebrovascular, or peripheral arterial disease. Since various factors such as the severity of tissue damage, the amount of circulating PF4 protein, exposure characteristics of heparin therapy, regulatory pathways of immune response, and genetic predisposition may influence the generation of anti-PF4 antibodies, these results should be further investigated in future studies.
MPs are shed by endothelial and blood cells upon activation or apoptosis, under the form of membrane vesicles. They mainly originate from the platelets and endothelial cells; however, they can also be released from other blood cells. Few studies have investigated the role of MPs in ESRD. Amabile et al investigated the relationship between circulating MPs and arterial dysfunction in patients with ESRD. 59 In this study, endothelial MP levels have shown high correlation with arterial dysfunction whereas platelet-derived, erythrocyte-derived, and Annexin V positive MP levels did not. The results of the study by Faure et al were similar and revealed that the endothelial MP levels were significantly higher in CKD and hemodialysis patients compared to healthy controls. 26 The MPs from leukocytes, platelets, and Annexin V positive apoptotic cells were also increased in this patient group. Another study published by Amabile et al has shown that the elevated levels of endothelial MPs were significantly associated with all cause and cardiovascular mortality in ESRD patients. 60 The study of Ryu et al has shown a possible relationship between the increased levels of endothelial and platelet MPs and vascular access failure in ESRD patients. 61 In our current study, we observed elevated levels of MP activity in the ESRD cohort. We did not observe any relevance between the MP activity and other quantified biomarkers. It is important to note that the method we used to measure MP activity in this study had shown the procoagulant activity of MPs by generating thrombin in the plasma, not where they were originated from. Additionally, in our patient cohort, approximately 63% of the patients were on antiplatelet therapy and 23.3% of the patients were on anticoagulant therapy which may have further affected our results. In any case, these results show upregulation of MP activity in ESRD patients. Additional studies are needed to understand the complex pathophysiology on this subject.
Another important finding in our study was that we compared biomarker levels in patients with CAD, cerebrovascular disease, and peripheral artery disease, which are the main vascular complications that cause mortality and morbidity in patients with ESRD. Although previous studies have shown that various biomarkers or diagnostic tests may have a predictive potential on these cardiovascular complications in the general population, knowledge in specific disease groups such as ESRD is not clear. There are a few studies related to these vascular complications with focus on CKD patients, particularly in those with ESRD. For instance, the importance of lipid-lowering therapy, the use of antiplatelet drugs or anticoagulant therapies and the decision of timing and the type of revascularization therapy, are still the subject of a discussion for CAD in ESRD patients. 62 Similarly, the diagnosis and treatment of ESRD patients with cerebrovascular disease vary, and it is not certain how these patients will be treated with antiplatelet and anticoagulant treatments, lipid-lowering treatments, treatment of concomitant carotid disease and atrial fibrillation, and thrombolytic therapy and interventional methods. 63 The situation is similar in terms of the peripheral arterial disease, and the diagnosis, medical treatments and invasive treatments such as revascularization and amputation of these patients are not certain in ESRD patients. 64 This is partly due to the patients with kidney disease being underrepresented in randomized clinical trials of CVD interventions. 65 Furthermore, although the established methods have certain success rates in the general population, they are less successful in ESRD patients and newer approaches are essential.62–64 In this study, our results have shown that only CRP levels were significantly higher in ESRD patients diagnosed with CAD. There was not any significant difference found in the levels of other biomarkers in terms of CAD. In addition, our results did not show any significant difference in the biomarker levels in patients diagnosed with cerebrovascular disease and peripheral arterial disease. It is important to note that vascular comorbid illnesses showed wide simultaneities in the study group, and the majority of patients with cerebrovascular and peripheral artery disease had also concomitant CAD. This may have affected our biomarkers results regarding the cerebrovascular and peripheral arterial disease, so that these results should be further investigated.
CRP is a marker for inflammation that appears to predict cardiovascular events in healthy populations. For example, in the prospective Physicians’ Health Study (PHS) by Ridker et al, a high plasma concentration of CRP was associated with a 2-fold increase in risk of stroke, a 3-fold increase in risk of MI, and a 4-fold increase in risk of developing peripheral vascular disease in apparently healthy men. 66 In the Women Health Study by Ridker et al, apparently healthy postmenopausal women who developed cardiovascular events had higher baseline CRP levels had as well as a 5-fold increase in risk of any vascular event and a 7-fold increase in risk of MI or stroke. 67 In the JUPITER (Justification for the Use of Statins in Primary Prevention: An Intervention Trial Evaluating Rosuvastatin) trial, Ridker et al tested the hypothesis that people with elevated hs-CRP levels, but without hyperlipidemia, would benefit from statin treatment. 68 This is because, the statin therapy has been shown to reduce CRP levels independently of its effect on cholesterol. This trial was terminated early due to a 44% reduction in the primary endpoint of all vascular events. There was also a 54% reduction in MI, a 48% reduction in stroke, a 46% reduction in the need for arterial revascularization, and a 20% reduction in all-cause mortality. The results of this study were of major importance, due to the fact that an intervention interacting with the inflammatory pathways was reducing the incidence of adverse outcomes. The higher incidence of coronary and vascular adverse outcomes in the ESRD population shown in our results provides a better insight for biomarkers of inflammation and related pathways. In our current study, the level of CRP was also an independent predictor of CAD in ESRD. Additional studies are needed to understand this complex pathophysiology.
Study Limitations and Strengths
Our study also has several limitations which should be noted. This is a retrospective study. The retrospective nature of this study also meant that it was difficult to exclude other drivers of thrombo-inflammation from our patient cohort which is difficult to assess from the medical record review. This study is based on a single sample analysis and due to logistic reasons, follow-up samples were not collected and analyzed. Moreover, the ESRD cohort is not compared with age-matched controls and the populations were not balanced. Future studies should consider collecting sequential samples in follow-up analysis and age-matched controls. We were also not able to exclude those patients taking medications which may impact the plasma levels of some biomarkers assessed. Additionally, we were not able to consider the anticoagulant history and the type of anticoagulant used during hemodialysis. This study included 73 ESRD patients and 10 healthy controls providing us two cohorts to demonstrate the difference of biomarker levels to determine the role of ESRD on cardiovascular outcomes. Since this was a pilot study, our observation clearly suggests the need for a larger study with an increased number of both groups to validate our data. The relatively small number of normal samples was included as a reference point and additional trials including a larger number of normal individuals may be needed; however, the fluctuations in thromboinflammatory biomarkers in the normal cohorts are not as obvious as in the patients with ESRD. Despite these limitations and the apparent lower number of representative patients in the ESRD group and normal individuals, this study provides some observational differences in the levels of biomarkers in the two groups which require additional expanded studies. We also were not able to add other inflammatory biomarkers in addition to CRP. Despite these limitations, the study provides considerable insight in the upregulation of thrombo-inflammatory biomarkers and their associations with each other and adverse outcomes.
This study also has several strengths. Firstly, all of the patients assessed presented to and were evaluated at the same university hospital. Workup for each patient followed the Loyola University Medical Center, Nephrology and Hemodialysis team protocols, and each plasma sample collected was stored and measured in the same fashion. We were able to stratify the patients according to cardiovascular outcomes such as coronary artery, cerebrovascular and peripheral artery disease. Moreover, our IRB-approved program is still ongoing and recruiting additional patients and provides a platform to expand the current profiling of the biomarkers to better determine their diagnostic and prognostic values.
Conclusions
In this study, we investigated the levels of 10 different biomarkers of thrombo-inflammation in a relatively large group of ESRD patients. Our results, including the relationship between the tested parameters and their relevance for the major vascular adverse outcomes, are of major importance. The increased levels of CRP can be a reliable predictor of CAD in ESRD patients.
Footnotes
Acknowledgments
The authors are thankful to the staff of the Hemostasis Research Laboratories for processing and analysis of the samples. We are also thankful to the staff of the outpatient hemodialysis unit, in particular, Ms. Pamela Abulencia for their expert assistance in facilitating blood collection. A special thanks to Professor Kraemer of the University of Heidelberg for providing the generous gift of Heparin Red reagents used in this study. We are also thankful to Mr Jonas Kingo and Ms. Catherine Sandon for providing some of the kits used in this study. The authors also acknowledge the helpful advice and guidance from Professor Robert Linhardt, Rensselaer Polytechnic Institute (Troy, NY) for conducting the study. We are thankful to Dr Seth Robia and Dr Alain Heroux for their support. The skillful assistance of Ms. Erin Healy-Erickson in the preparation of this manuscript is gratefully acknowledged.
Declaration of Conflicting Interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
