Abstract
In patients with end-stage renal disease (ESRD), heart failure with reduced ejection fraction (HFrEF) is a common comorbidity. Thromboinflammatory processes in both conditions represent complex pathophysiology, demonstrated by dysregulation of thromboinflammatory biomarkers, and commonly resulting in the combined pathology of cardiorenal syndrome. We sought to investigate the effects of HFrEF on these biomarkers in patients with ESRD, and observe the relationship to mortality. Blood samples from 73 patients with ESRD (mean age 67 ± 13 years, 56% male) and 40 healthy controls were analyzed via enzyme-linked immunosorbent assay and other chromogenic methods for angiopoietin-2 (Ang2), endogenous glycosaminoglycans, fatty acid binding protein, interleukin-6, lipopolysaccharide, free fatty acids, NT-pro B-type natriuretic peptide, tumor necrosis factor α, vascular endothelial growth factor, and von Willebrand factor. Patients were stratified into those with or without HFrEF (EF < 50%). Patients had highly prevalent comorbidities including coronary artery disease 46%, diabetes 69%, hypertension 97%, and smoking 49%. Most biomarkers were upregulated in ESRD compared to controls. Patients with HFrEF and ESRD had greater interleukin-6 and NT-pro B-type natriuretic peptide and lesser lipopolysaccharide compared to ESRD only. Spearman correlations between most biomarkers were increased in HFrEF + ESRD over ESRD only. Ang-2 was associated with mortality in this cohort. The dysregulation of thromboinflammation in ESRD is somewhat amplified in comorbid HFrEF. Correlation among biomarkers in this cohort indicates the mechanisms of thromboinflammatory biomarker generation in ESRD and HFrEF share an integrative process. Ang2, interleukin-6, and lipopolysaccharide show promise as biomarkers for risk stratification among patients with both HFrEF and ESRD.
Keywords
Introduction
Heart failure is a highly prevalent condition, affecting approximately 6 million Americans. The pathophysiology of heart failure with reduced ejection fraction (HFrEF) is multifaceted and involves complex interplay between various molecular and systemic processes. Emerging evidence highlights the crucial involvement of inflammation in this pathophysiology. Chronic systemic inflammation triggered by widespread inflammatory cytokine release stimulates cardiac remodeling, and plays a critical role in major adverse cardiac events and mortality in addition to progressing other chronic inflammatory diseases such as end-stage renal disease (ESRD).1,2
Approximately half of heart failure patients have some degree of chronic kidney disease. 3 The pathophysiology of ESRD in these patients is similarly multifactorial. The release of pro-inflammatory cytokines and resultant activation of fibroblasts promotes renal fibrosis and progressive loss of functional nephrons.4,5 Aberrant immune responses further fuel inflammation, tubulointerstitial damage, and progressive renal dysfunction. 6
In the ESRD population, cardiovascular mortality is 20 times higher than in the general population. 7 Likewise, renal insufficiency has shown to be an independent prognostic factor in both diastolic and systolic dysfunction, and concomitant disease is associated with greater rates of hospitalization and all-cause mortality.3,8 The high interconnectivity of both illnesses can be explained through cardiorenal syndrome (CRS). CRS encompasses a spectrum of disorders involving both the heart and the kidneys in which acute or chronic dysfunction in one organ may induce acute or chronic dysfunction in the other organ. 9 The most recent professional consensus classifies CRS into 5 subtypes primary based upon the organ that initiated the insult as well as the acuity of the disease. 9 The more integrative definition of CRS states regardless of which organ fails first, the same neurohormonal systems are activated leading to the progression of damage and failure of both organs.
Although it is recognized that biomarkers play an important role in diagnosis of acute and chronic heart and renal disease independently, biomarkers have not yet been integrated into the standard diagnosis, treatment, or prognostication of CRS. 2 However, recent evidence supports key biomarkers of renal and cardiac injury have been shown to be appropriate markers of disease progression in these patients. 2 Several traditional inflammatory biomarkers such as interleukin-6 (IL-6), tumor necrosis factor α (TNFα), lipopolysaccharide (LPS), fatty acid binding protein (FABP), and free fatty acids (FFA) have been shown to be upregulated in, or associated with morbidity and mortality in either renal disease, heart disease, or both.6,10–15 The same holds true for classic biomarkers of endothelial damage such as endogenous glycosaminoglycans (eGAG), von Willebrand factor (vWF), Angiopoietin-2 (Ang2), vascular endothelial growth factor (VEGF), and NT-pro B-type natriuretic peptide (NT-proBNP).1,5,16–24
Determining simultaneous fluctuations of biomarkers involved in kidney function and cardiac function may be helpful in risk stratification of patients with ESRD. Thus, we investigated a combination of biomarkers and cardiac indices to diagnosis and establish a risk stratification system in patients with ESRD with superimposed heart failure. The goal of our study was to develop a greater understanding of CRS and help provide a diagnostic and prognostic tool to evaluate the risk of developing cardiac dysfunctions in patients with ESRD, and vice versa.
Methods
Whole blood samples were collected from 73 patients with ESRD undergoing routine hemodialysis at Loyola University Medical Center under an Institutional Review Board approved protocol. The whole blood samples were collected in 3.8% (0.109 mol/L) sodium citrate tubes, centrifuged for platelet poor plasma, and frozen at −70 °C prior to analysis. Control citrated plasma samples (n = 40) were collected from nonsmoking normal healthy patients (NHP), obtained from a commercial vendor George King Biomedical (Overland Park, Kansas). The NHP are not age or sex matched.
Biomarkers including Ang2, NT-proBNP, L-FABP, IL-6, LPS, TNFa, VEGF, and vWF were analyzed via commercial enzyme-linked immunosorbent assay methods. The levels of eGAGs were quantified by utilizing the Heparin Red Probe, a fluorescence assay which quantifies heparins and other endogenous polyanionic sulfated glycosaminoglycans (eGAG) which can reflect endothelial damage in plasma samples (Redprobes UG, Germany). 25 FFA were quantified via other specific chromogenic methods.
Clinical information was collected from patient electronic medical records (EMR) in a chart review for demographic data and medical history including echocardiography records. Patient groups were stratified into 2 groups, those with or without heart failure with reduced ejection fraction defined by those having a confirmed by echocardiogram ejection fraction (EF) <50% at any point in the 6 months before or after the plasma collection date, these parameters intending to exclude those with recovered EF or those with normal EF at time of sample collection but later worsening of systolic function. Statistical differences between the ESRD groups and NHP were evaluated utilizing nonparametric Mann-Whitney U (Wilcoxon rank-sum) tests. Correlation analysis was performed by utilizing Spearman correlation coefficients. At 2 years following sample collection, the EMR was reviewed for patient mortality with time of death or survival recorded. The relationships of biomarkers to survival were analyzed using Cox proportional hazards model. The optimal cut-off value for baseline serum Ang-2 to predict all-cause mortality was 1715 pg/mL (AUC 0.706) based on the regression analysis. Survival curves were compared using Log-rank (Mantel-Cox) test. The calculations were performed using R (Posit Software, Boston, Massachusetts) and Prism (GraphPad, La Jolla, California) software.
Results
The mean age of the patients with ESRD was 69 ± 13.14 years, 58% were male. Sixty-one out of 73 (84%) patients had ESRD only; 12 had both ESRD and HFrEF per the inclusion criteria above. Multiple comorbidities were present in this patient population. Coronary artery disease was present in 41%, diabetes in 55%, hypertension in 93%, obesity in 41%, and smoking in 49%. The details of the patient population are shown in Table 1.
Patient Characteristics.

Abbreviations: EF, ejection fraction; ESRD, end-stage renal disease.
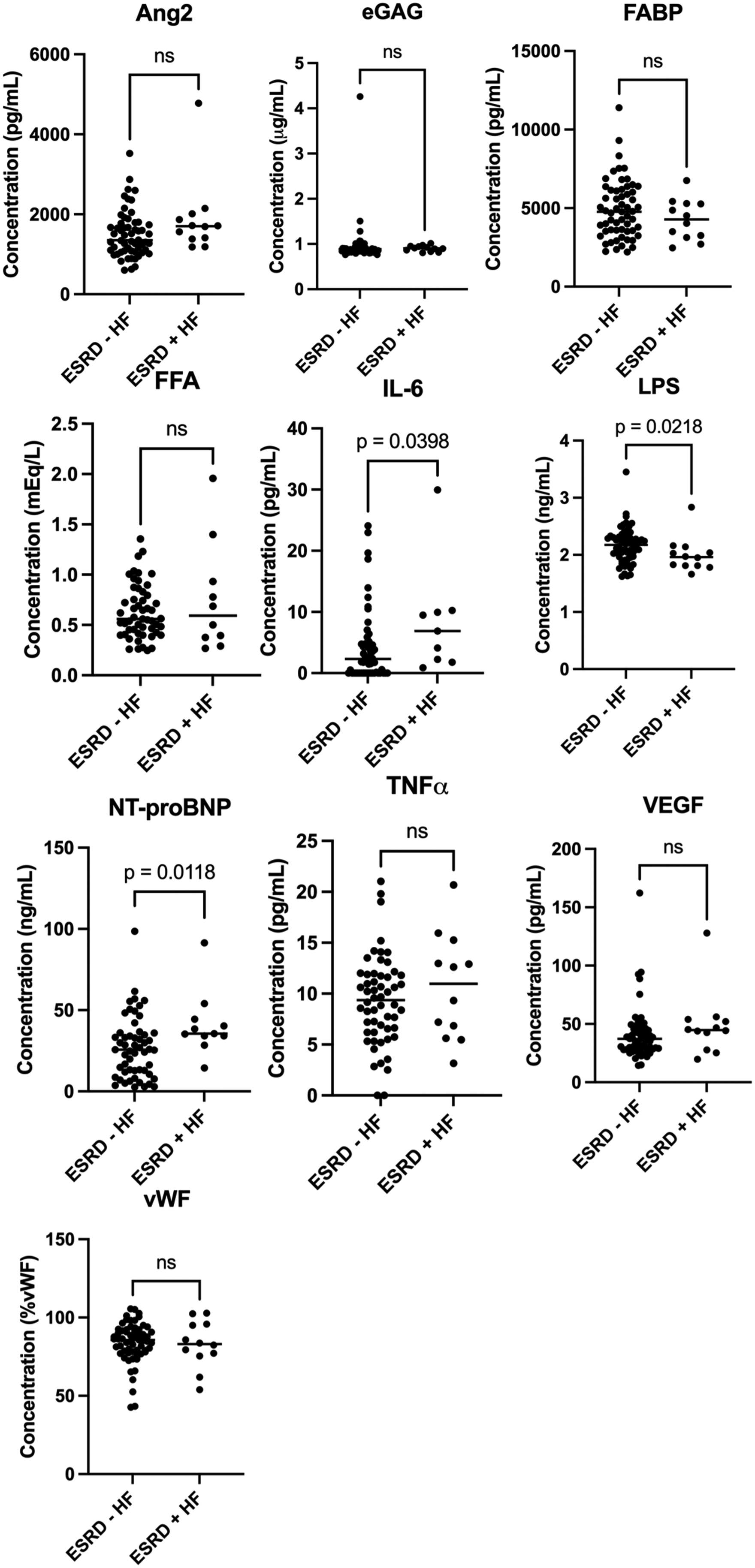
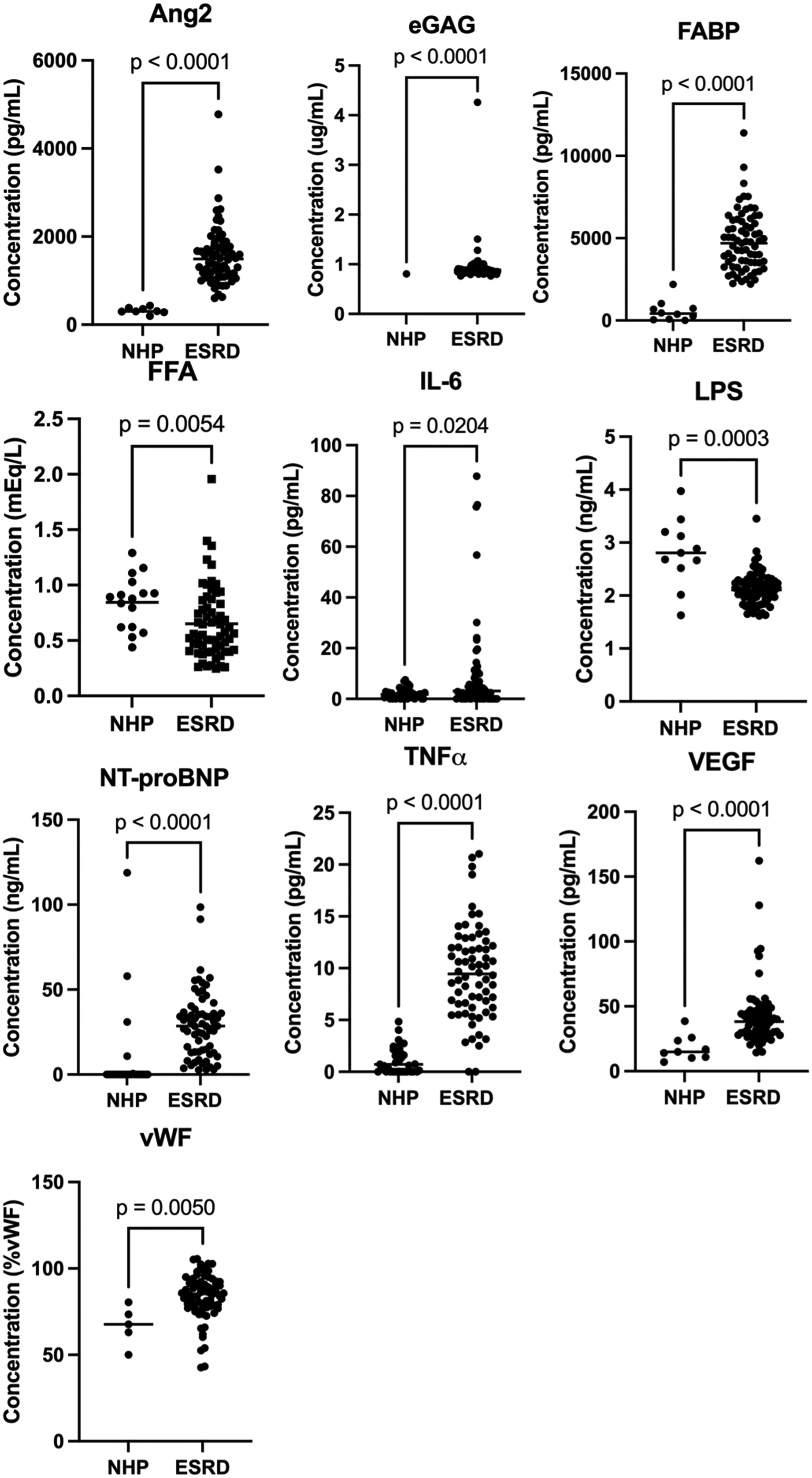
There was a significant (P < .05) change in all biomarker levels in those patients with ESRD compared to NHP (Figure 1). The greatest fold increase was noted in eGAG (13.5 fold) followed by TNFα (9.0 fold), FABP (8.2 fold), Ang2 (4.9 fold), NT-proBNP (4.4 fold), VEGF (3.4 fold), vWF (1.3 fold), and IL-6 (1.1 fold) (Table 2). LPS (0.8 fold) and FFA (0.6 fold) decreased in patients with ESRD versus NHP (Table 2). Compared to those patients with ESRD alone, a significant increase (P < .05) was noted in IL-6 (2.0 fold) and NT-proBNP (1.5 fold) in those with both ESRD and HFrEF. A significant decrease (0.9 fold) was noted in LPS (Figure 2 and Table 2).
Biomarkers in ESRD compared to NHP. Abbreviations: ESRD, end-stage renal disease; NHP, normal healthy patients.
Biomarkers in ESRD + HFrEF patients compared to ESRD alone patients. Abbreviations: ESRD, end-stage renal disease; HFrEF, heart failure with reduced ejection fraction.
Measured Quantity, All Biomarkers.
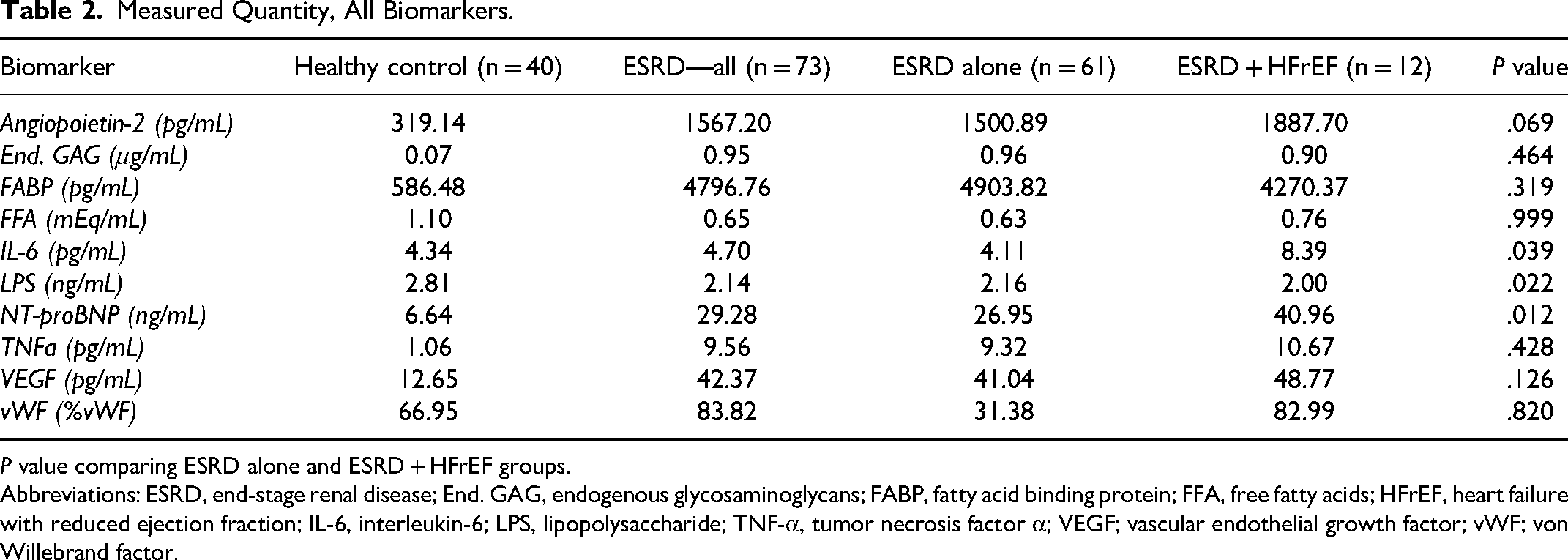
P value comparing ESRD alone and ESRD + HFrEF groups.
Abbreviations: ESRD, end-stage renal disease; End. GAG, endogenous glycosaminoglycans; FABP, fatty acid binding protein; FFA, free fatty acids; HFrEF, heart failure with reduced ejection fraction; IL-6, interleukin-6; LPS, lipopolysaccharide; TNF-α, tumor necrosis factor α; VEGF; vascular endothelial growth factor; vWF; von Willebrand factor.
In Spearman's rank correlations for both groups, correlations were noted in those with both ESRD alone and with HFrEF (Figures 1 and 2). In those with ESRD alone, Ang2 was positively correlated with NT-proBNP. eGAG were positively correlated with FFA, VEGF, and vWF while negatively correlated with IL-6. FABP was positively correlated with IL-6 and negatively correlated with vWF. IL-6 was positively correlated with FABP, NT-proBNP and negatively correlated with eGAG, VEGF. LPS was positively correlated with VEGF. FFA was positively correlated with eGAG, VEGF, and vWF. NT-proBNP was positively correlated with Ang2, IL-6, TNFα, and negatively correlated with vWF. TNFα was positively correlated with NT-proBNP. VEGF was positively correlated with eGAG, LPS, and FFA, and negatively correlated with IL-6. vWF was positively correlated with eGAG and FFA, and negatively correlated with FABP, IL-6, and NT-proBNP (Figure 3).
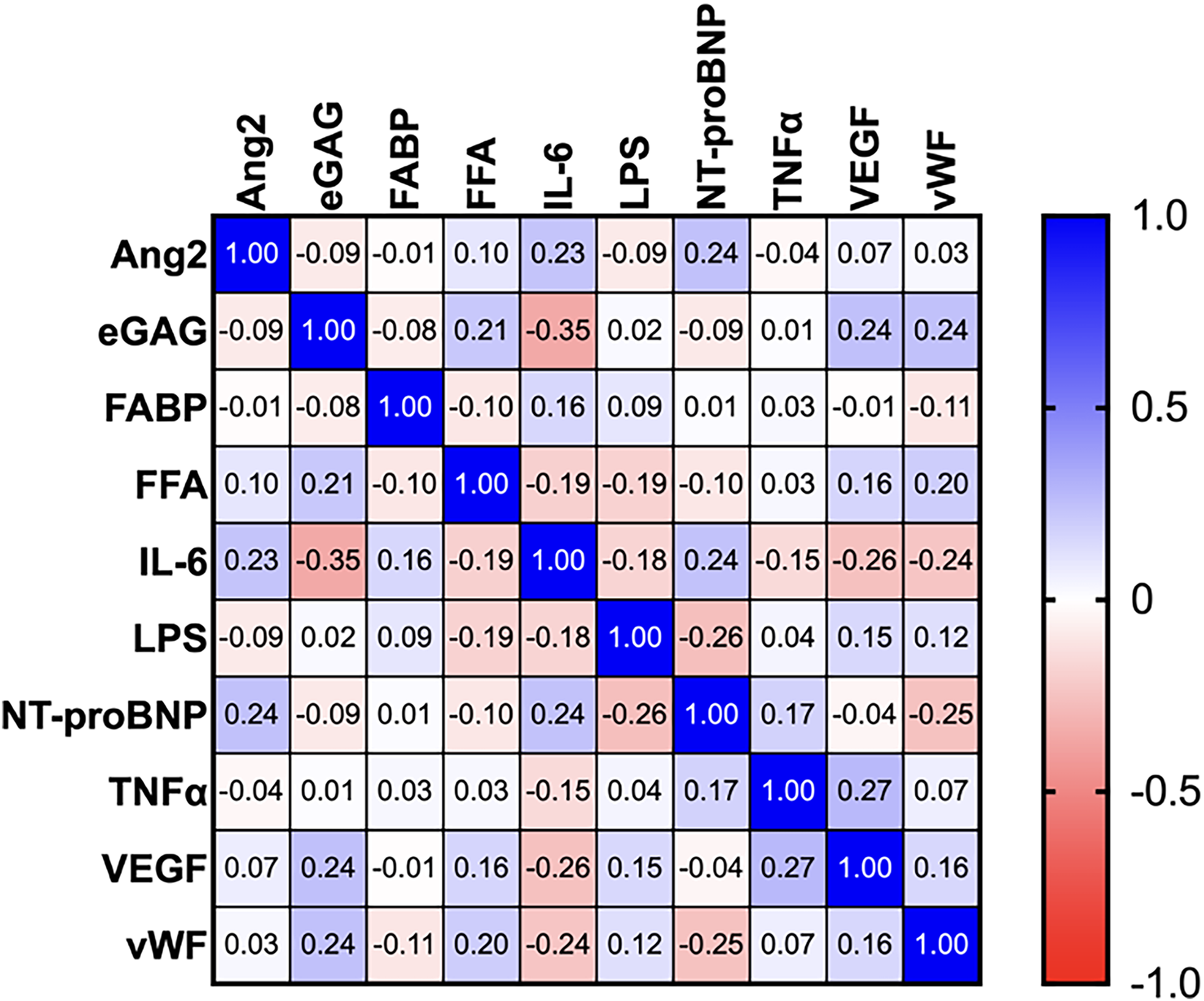
Spearman correlations among all biomarkers in all patients with ESRD, with listed correlation coefficient values (r). Abbreviation: ESRD, end-stage renal disease.
In patients with both ESRD and HFrEF, correlations were markedly greater overall, as seen represented in Figure 2. Ang2 was positively correlated with eGAG, IL-6, and FFA while negatively correlated with FABP and VEGF. eGAG was positively correlated with Ang2, LPS, VEGF, and vWF while negatively correlated with FABP. FABP was positively correlated with vWF while negatively correlated with Ang2. IL-6 was positively correlated with Ang2 and LPS while negatively correlated with VEGF. LPS was positively correlated with eGAG, IL-6, TNFα, and vWF while negatively correlated with FFA. FFA was positively correlated with Ang2 while negatively correlated with FABP, IL-6, and LPS. NT-proBNP was negatively correlated with LPS and vWF. TNFα was positively correlated with LPS, VEGF, and vWF while negatively correlated with Ang2. VEGF was positively correlated with eGAG, TNFα, and vWF while negatively correlated with Ang2 and IL-6. vWF was positively correlated with eGAG, LPS, TNFα, and VEGF while negatively correlated with NT-proBNP (Figure 4).
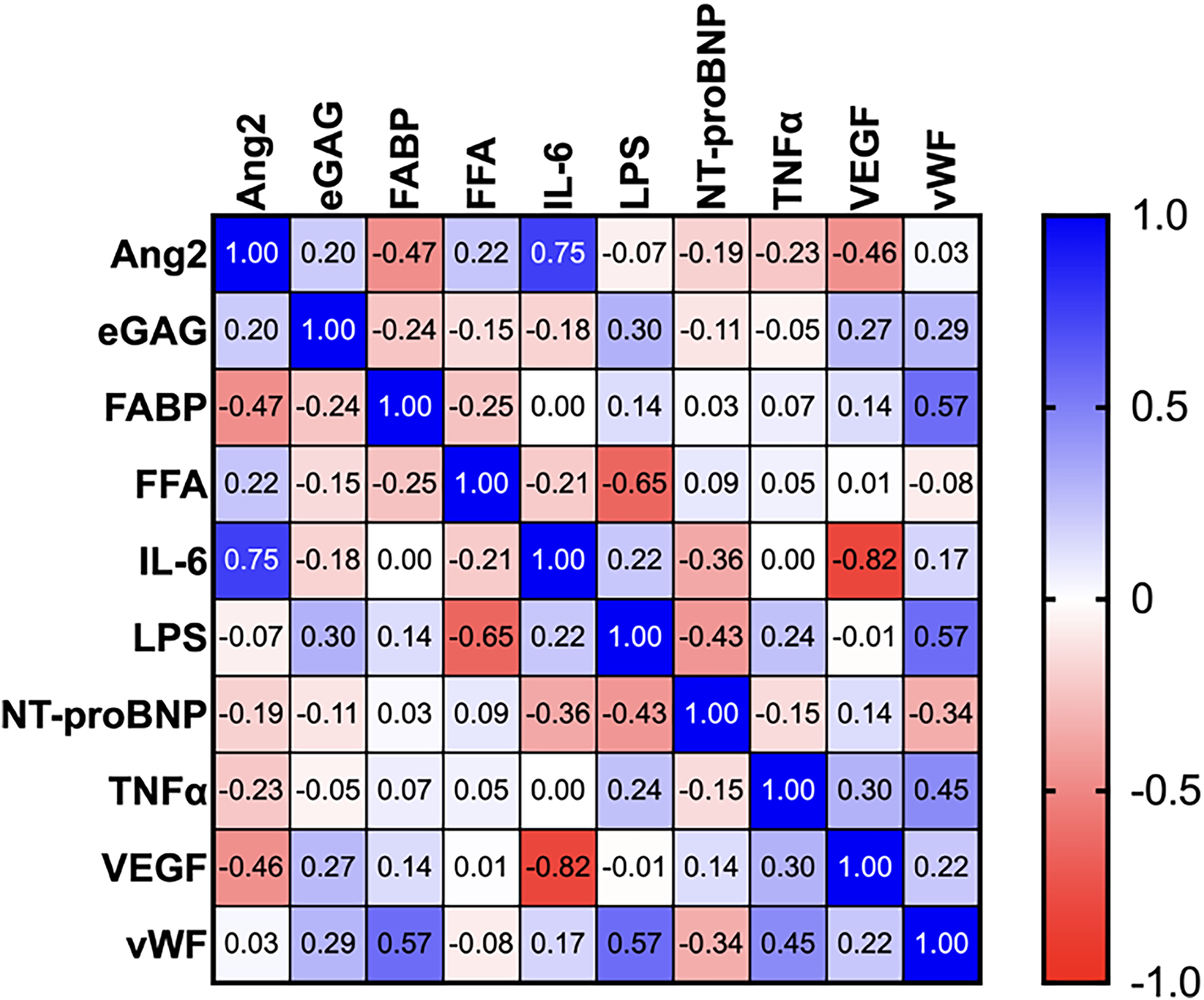
Spearman correlations among all biomarkers in all patients with ESRD + HFrEF, with listed correlation coefficient values (r). Abbreviations: ESRD, end-stage renal disease; HFrEF, heart failure with reduced ejection fraction.
Mortality in this cohort at 1 year was 12.7%. Most biomarkers did not predict survival or mortality. Patients with ESRD who died within 1 year of assessment had significantly higher Ang2 than those who survived (P = .040; Figure 5). Those who died also had lower LPS (P = .075), lower FFA (P = .072), and greater NT-proBNP (P = .135) though not statistically significant. No other biomarkers were significantly changed in this group. In Cox proportional hazard regression, Ang2 was predictive of mortality in this sample (P < .001), with concordance = 0.706, likelihood ratio test = 11.96 (P = .003) and score (log rank) test = 13.71 (P = .001) on 2 degrees of freedom. The survival curves distinguishing the optimal cut-off value of 1715 pg/mL of Ang2 were significantly different (P = .0338) (Figure 6).
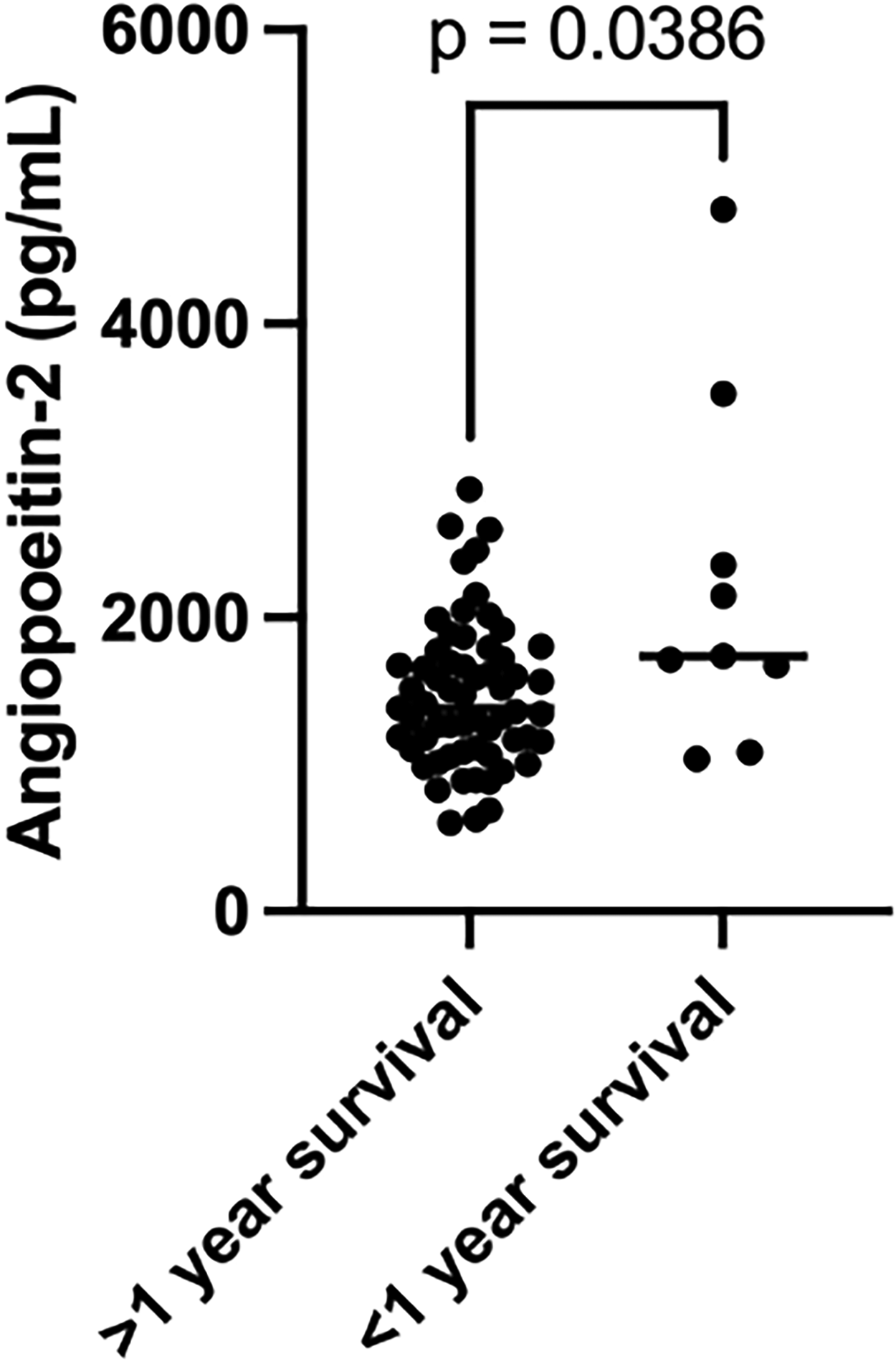
Angiopoeitin-2 levels in 1-year mortality.
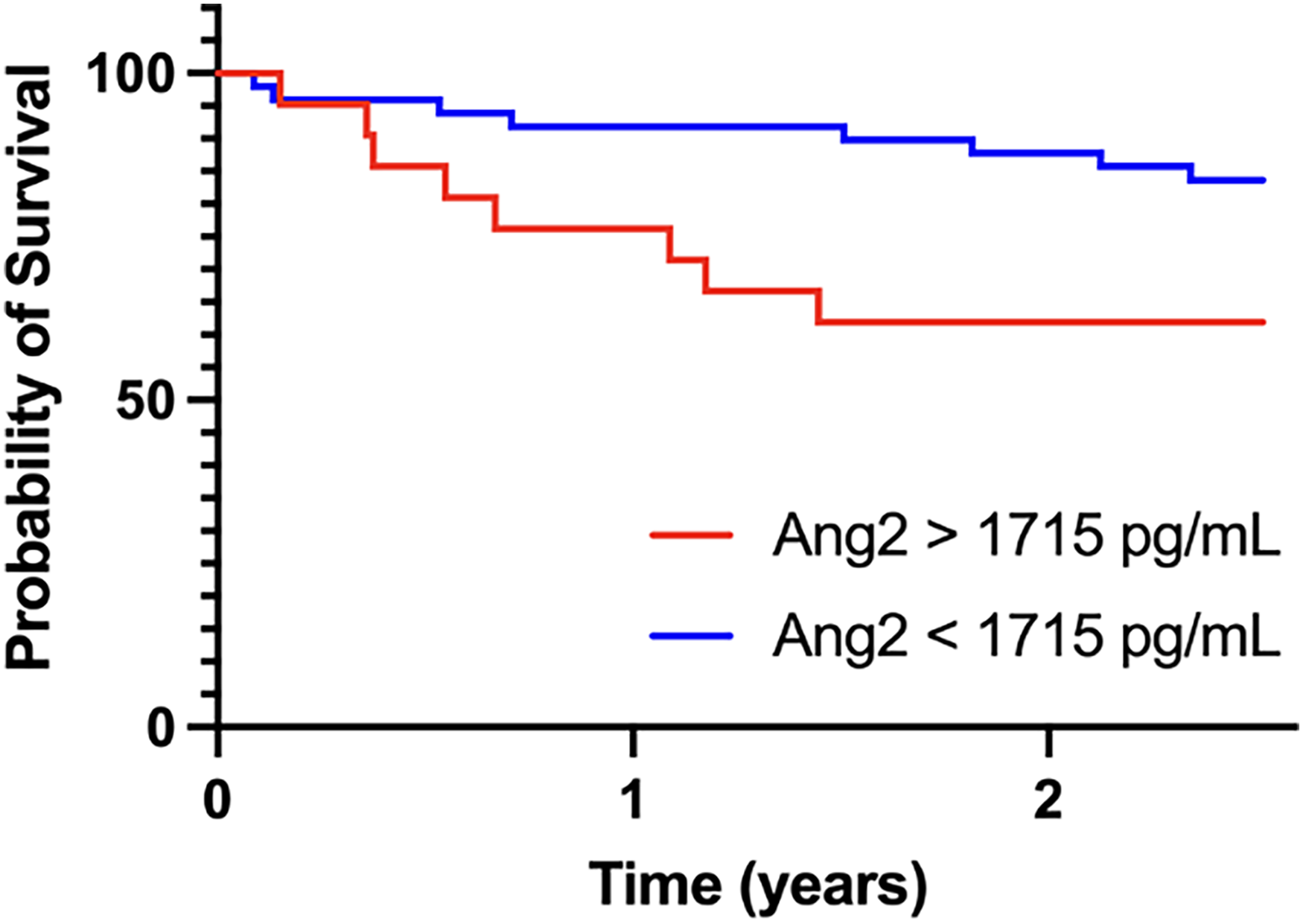
Kaplan-Meier survival curves for all-cause mortality. Patients were divided according to optimal cut-off values of Ang-2 concentrations.
Discussion
Our findings support that the use of biomarkers in risk stratification in CRS is both possible and potentially impactful, given the significant changes which are seen in patients with ESRD both with and without HFrEF. Of special note were the changes in inflammatory biomarkers LPS and IL-6 seen in those with CRS, and the changes in endothelial biomarkers Ang2 and eGAG seen in the 1-year mortality group.
The most common biomarker used clinically for the prognosis of heart failure is NT-proBNP, which is a prohormone strongly associated with myocardial stretching in heart failure. In addition, in those with acute heart failure, the NT-proBNP ratio to mature BNP precedes worsening renal failure and has been proposed as a biomarker for risk stratification in CRS. 16 Here, we find that NT-proBNP is significantly upregulated both in those with ESRD, and those who have concomitant HFrEF. In those with ESRD, NT-proBNP was positively correlated with Ang2, IL-6, and TNFα. In CRS, NT-proBNP was negatively correlated with LPS. In both ESRD alone and with HFrEF, NT-proBNP was negatively correlated with vWF (r = −0.21, −0.34). This may reflect differences in the effect of CRS on the endothelium of the heart versus that of the peripheral vessels. Those patients experiencing mortality within 1 year of measurement had a 35% higher NT-proBNP than those who survived (P = .076), in line with the current understanding that progressive cardiac damage contributes to the demise of those with CRS.
Inflammation is an early-stage phenomenon in the development of both heart failure and ESRD. 2 Inflammatory biomarkers such as IL-6 and TNFα have been implicated in the pathophysiology of CRS, as both have previously been found to be predictive of outcome in chronic renal failure and heart failure independently.6,15 While a proinflammatory milieu has been identified in CRS, its effect on morbidity, mortality, and prognosis is not fully characterized. 15 In this study, measurements of IL-6 were far more sensitive to the presence of HFrEF than that of TNFα. IL-6 was upregulated in those with concomitant HFrEF while TNFα was not, and in fact they were not correlated with each other in either Spearman analysis. IL-6 was correlated with VEGF in both analyses (r = −0.23, −0.82) and to a markedly greater degree in the population with ESRD and HFrEF, suggesting a strong inverse relationship between inflammatory and angiogenic signaling in CRS. Contrarily, TNFα was positively correlated with VEGF, suggesting that it is not helpful to consider TNFα and IL-6 as a monolith in the pathogenesis of inflammation in this condition. Similarly, IL-6 was positively correlated with Ang2 while TNFα was negatively correlated with Ang2 in the ESRD and HFrEF groups. Altogether, these data suggest that while both markers are upregulated in ESRD, the pro-inflammatory milieu in CRS is distinct from that in ESRD alone, and is a promising area for future investigation as well.
A known effect of inflammation in the intestinal system is endotoxemia, the translocation of bacterial endotoxins into systemic circulation, also known as the “leaky gut hypothesis.” One of these endotoxins, LPS, is commonly utilized as the biomarker for endotoxemia, and is elevated in heart failure especially when clinically severe. 13 LPS signaling through toll-like receptors then evokes damage to the myocardium and worsens heart failure, as seen through the increased adverse cardiac events seen in those with worse endotoxemia. 13 This finding has also been seen in patients with CRS type I, acute kidney injury as a result of acute decompensated heart failure. 11 However, this is in contrast to the Adda-Rezig et al finding of decreased LPS levels seen in those with chronic kidney disease (CKD) despite the association of CKD with increased gut bacterial translocation. 12 Thus the exact role of LPS in chronic states of CRS is still under active investigation. Similarly to Adda-Rezig et al, we found significantly decreased LPS in those with ESRD compared to NHP (−24%). Expanding upon that result, we also found significantly decreased LPS in those with concomitant HFrEF compared with ESRD alone (−7.4%). These results are especially notable given the decreased levels of LPS (−9%) in those deceased within 1 year (P = .058). The reasons for the lower LPS in CRS contrary to the leaky gut/endotoxemia hypothesis is currently unknown and worthy of future investigation, especially in the context of its strong positive correlations with reliable inflammatory biomarkers IL-6 and TNFα, and endothelial damage biomarkers eGAG and vWF. Future directions in the study of LPS fluctuations in CRS may involve further dividing LPS based upon different carbon structures, which have been shown to instigate inflammatory changes according to saccharide composition as per Adda-Rezig et al.
Endothelial changes are also a hallmark of heart and renal disease as vascularization is the logical consequence of decreased perfusion in the setting of heart failure, and commonly occurs at sites of inflammation such as diseased renal tissue in ESRD. 17 Properly functioning endothelium depends on normal repair and turnover via pro- and anti-angiogenic factors. VEGF is a key regulator and pro-angiogenic factor expressed both in cardiac myocytes and glomeruli. In CKD, overexpression of VEGF causes renal fibrosis, whereas VEGF in heart failure and CRS has been investigated not only as a biomarker but also as a potential treatment modality, as diseased myocardium sometimes fails conventional treatment due to insufficient blood flow.18,19 We identified a significant 3.4 fold increase in VEGF in patients with ESRD over NHP, and a lesser 1.2 fold increase in those with concomitant HFrEF (P = .126), suggesting similarly to the other biomarkers that the magnitude of changes seen in CRS are not purely additive to those in ESRD alone. VEGF did not appear to impact mortality in our study, and in fact was strongly negatively correlated to Ang2, the only biomarker that was significantly higher in those who experienced mortality within 1 year. Likewise, VEGF was strongly negatively correlated with pro-inflammatory IL-6 in this study, which lends support to the finding in Ricciardi et al that VEGF may serve as a protective factor in CRS. 18
Ang2, released from endothelial cells, antagonizes pro-angiogenic factors and supports vessel regression in the absence of VEGF, but in the presence of VEGF assists in endothelial proliferation. 1 This proliferation may be aberrant in states of pathologically elevated Ang2 such as in ESRD and congestive heart disease, contributing to the development of endothelial abnormalities. 1 This is likely true in the patients discussed here, as Ang2 was markedly elevated both in ESRD over NHP and in ESRD with HFrEF over ESRD alone. Furthermore, Ang2 was predictive of mortality in the Cox regression analysis. This is the second observation of increased Ang2 predicting mortality in renal disease after Chu et al 26 but the first to be reported in CRS. Ang2 was also positively correlated with several biomarkers in the CRS population with eGAG, IL-6, and FFA, suggestive of endothelial damage and pro-inflammatory signaling. These results suggest that Ang2 may make for a highly beneficial biomarker for the purpose of risk stratification in patients with CRS and is a highly valuable subject for future research.
Endothelium is protected physiologically from shear stress and aberrant coagulation and inflammatory signals by the glycocalyx, a dynamic structure rich with eGAG that functions as a thin vascular barrier.20,22 The glycocalyx is responsive both to changes in forces exerted on the endothelium as well as neurohormonal factors such as aldosterone, which can be damaging to the tissue. 23 Damage to the endothelial glycocalyx can be detected in the blood as eGAG are released, serving as a biomarker for endothelial damage. The release of syndecan-1 and heparan sulfate, prominent eGAG in heart failure patients, is associated with greater risk of acute kidney injury and mortality likely due to impaired endothelium-dependent vasodilation and myocardial perfusion, increased afterload, and increased pathological cardiac remodeling.21,22 Markers of endothelial dysfunction are of increasing interest going forward due to their prominent role in the pathophysiology of CRS. 21 In this study, eGAG were significantly elevated in ESRD over NHP, but not in the group with HFrEF, suggesting that it may reach an upper limit of activation or play less of a role in HFrEF. However, among the patient who died within 1 year of assessment, those with HFrEF had 16% higher eGAG than those with ESRD alone (P = .036). This may represent an increase in circulating eGAG only in the most severe cases of CRS similarly to Ang2, and is another area deserving further research in the future.
Similarly to eGAG, vWF is another endothelial factor that has been under investigation as a biomarker in renal failure, and thus may serve a role in the risk stratification of CRS. It has been associated with tubular fibrosis and poor allograft outcomes in kidney transplant recipients independently of heart failure, and its expression is upregulated in the kidneys of mice with induced heart failure.5,24 As expected, it is positively correlated with eGAG in ESRD alone and with HFrEF. However, it is not upregulated significantly in the group with HFrEF over ESRD alone and was not associated with increased mortality in our analysis, and may serve as a better biomarker for predicting renal disease outcomes than those in CRS.
Also of interest is FABP which has been shown to be increased in both renal dysfunction and heart failure, respectively, and may be predictive of worsening renal failure in the setting of heart failure.6,10,14,15 We found that FABP was significantly upregulated in ESRD but not in concomitant HFrEF, and was noncontributory to mortality in 1 year. Despite this, on Spearman analysis FABP demonstrated alternating negative correlation to vWF in ESRD (r = −0.26), but positive correlation in ESRD with HFrEF(r = 0.57). This suggests a possible shared upregulation in both markers in CRS reflecting advanced stages of endothelial damage found in HFrEF but not necessarily in renal disease alone. These changes may also be related to aberrant angiogenic signaling when considered alongside the negative correlation with Ang2.
Study Strengths and Limitations
This study has several strengths. While molecular biomarkers are routinely used both academically and clinically for the diagnosis and prognostication of ESRD and HFrEF separately, these conditions frequently co-occur, and it is currently unknown whether these findings may be similarly applied to the convergence of both diseases, cardiorenal syndrome. Several marked correlations among biomarkers in this cohort suggest shared processes in the mechanisms of thromboinflammatory biomarker generation in ESRD and HFrEF. In particular, we find that inflammatory mediators LPS and IL-6 are strongly associated with this condition, with a novel finding that LPS levels decrease with HFrEF in patients with ESRD which has important implications for future study. This study also demonstrates Ang2 as a predictor of mortality in ESRD, a finding with important consequences to those caring for patients on hemodialysis. Methodologically, all of the patients in the study receive their dialysis at the same clinic and live in the same region, limiting environmental contributions as a cause of interpatient differences. Sample collection and processing were conducted according to rigid protocols and were stored and measured identically.
This is a retrospective study. Accordingly, confounding variables also impacting the inflammatory process in these patients cannot be excluded. These include other disease conditions, medications and therapies, age, sex, other demographic factors, and environmental impacts. These samples were also collected at one instance in time with no follow-up samples. Future studies should consider collecting sequential samples in follow-up analysis. In this sample, the proportion of patients with ESRD with HFrEF was below the national average and limited to only 12 patients. This severely limited the power of the study to detect significant results, and eliminated the possibility of pursuing age-matching and other means of population stratification. The mortality data is binary and does not take into account individual factors that predispose patients to death, particularly during the COVID-19 pandemic. Despite these limitations, this study provides some important and novel observational differences which require additional expanded studies.
Conclusions
In this study, a wide variety of biomarkers involved in thromboinflammatory processes were investigated in a group of patients with ESRD with a subset of those experiencing HF. Our findings, demonstrating mixed upregulation of inflammation in cardiorenal syndrome, and the recent finding that levels of Ang2 are an independent predictor of mortality in those with ESRD, are highly important to the field's understanding of this rising condition.
Footnotes
Acknowledgments
The authors are grateful for the support and guidance from the faculty and staff of the Hemostasis and Thrombosis Laboratories, Cardiovascular Research Institute, and the Department of Pathology at Loyola University Medical Center. The authors are thankful to Dr Lowell Steen, Chief of Cardiology, for his support to complete this study. The authors are also thankful to Dr Eva Wojcik, Chairperson of the Department of Pathology, for her support, and Director of the STAR program, Dr Colleen Fitzgerald, for providing the opportunity for this project through the Student Training in Approaches to Research (STAR) and Research Honors programs. The authors are thankful to Mr Jonas Kingo for providing some of the kits used for this study. A special thanks to Mr Srdjan Nikolovski and Ms. Erin Healy-Erickson for reviewing this manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
