Abstract
There is a high incidence of mortality due to cardiovascular events seen in patients with end-stage renal disease (ESRD) undergoing hemodialysis. Through utilization of biochip microarrays and enzyme-linked immunosorbent assay methods, we assessed various cardiac, inflammatory, thrombotic, and cerebral biomarkers to determine their roles in the hemostatic activation or inflammatory processes in ESRD. In this study, 72 patients on maintenance hemodialysis were recruited and 53 normal healthy male and female adults volunteers. Blood samples from patients with ESRD were drawn prior to maintenance hemodialysis. Of the 15 statistically significant biomarkers, 10 were elevated in the ESRD group, most notably soluble tumor necrosis factor receptor 1, heart-type fatty acid binding protein, and von Willebrand factor activity. These results are consistent with previously reported studies proposing ESRD represents a poly-pathologic process with the activation of hemostatic and inflammatory processes, which contribute to the significant sequelae of cardiovascular disease seen in these patients.
Introduction
End-stage renal disease (ESRD) is the fifth and final stage of chronic kidney disease (CKD) necessitating dialysis or renal transplantation for survival. The global increase in patients with CKD progressing to ESRD threatens to reach epidemic proportions in the near future, especially with the steady rise in life expectancy of the world’s population. 1,2 Currently, there are more than 300,000 patients in the United States having ESRD and receiving hemodialysis as a life-sustaining treatment. 3 The life expectancy of patients undergoing dialysis is about 4 times lower than that of the general population with an annual mortality rate of 15%. 4
More than 60% of the morality seen in patients with chronic kidney failure, the disease preceding the development of ESRD, can be accounted for by atherosclerosis. Furthermore, cardiovascular disease accounts for approximately half of the deaths seen in patients with ESRD. 5 –7 Chronic inflammation, which has widespread predominance in ESRD, is a known factor contributing to atherosclerosis and also plays a crucial role in the pathogenesis of cardiovascular disease. Patients with ESRD experience cardiovascular events at a particularly high and disproportionate incidence based on their Framingham risk score compared to the general population for similar aged individuals. 4 A key reason for this increased number of cardiovascular events and consequently high cardiovascular mortality in ESRD is due to the prevalence of cardiovascular disease in these patients prior to the initiation of renal replacement therapy. 8 Additional features contributing to the high percentage of cardiovascular deaths seen in this patient population are leukocytosis and increased levels of other inflammatory markers, which serve as risk factors for both initial and recurrent cardiovascular events. Therefore, systemic vascular changes are thought to be a contributing factor for both the pathogenesis and the thrombotic comorbidities evident in ESRD. 9,10
The higher incidence of cardiovascular and other thrombotic events in patients having ESRD raises the question as to whether there are factors or indicators that could allow for risk stratification within this patient population. In this study, we attempt to address this question by profiling various cardiac, inflammation, and thrombotic markers in this specific patient group. The cardiac markers of interest are myoglobin (Myo), creatine kinase MB (CK-MB), heart-type fatty acid binding protein (H-FABP), cardiac troponin I (cTnI), glycogen phosphorylase BB (GPBB), and carbonic anhydrase III (CA3). The thrombotic markers included are tissue plasminogen activator–plasminogen activator inhibitor type 1 complex (tPA-PAI-1 complex), plasminogen activator inhibitor type 1 (PAI-1), von Willebrand factor (vWF), antithrombin (AT), antiannexin V immunoglobulin M (IgM), and the group of inflammatory markers studied were interleukin 1β (IL-1β), myeloperoxidase (MPO), sL-selectin, and intercellular adhesion molecule 1 (ICAM-1). Considering the high occurrence of cerebrovascular events seen in ESRD, the cerebral markers neuron-specific enolase (NSE), neutrophil gelatinase-associated lipocalin (NGAL), soluble tumor necrosis factor receptor 1 (sTNFR1),
Material and Method
Seventy-two patients with ESRD on maintenance hemodialysis treatment at the Loyola University outpatient dialysis unit were recruited for this study under a study protocol (LU107346) approved by the institutional review board of Loyola University Medical Center. Study criteria included patients older than the age of 18 years who had received hemodialysis treatment for at least 3 months prior to the initiation of the study. Patients with active infection, hospitalization at the time of blood draw, life expectancy less than 6 months, on immunosuppressive treatment, and those unable to give informed consent were excluded from this study. The control group consisted of 53 healthy male and female adult volunteers.
Blood samples were drawn immediately prior to each patient’s hemodialysis session via venous blood draw. Each sample was centrifuged within 2 hours of collection at 3,000 rpm for 15 minutes. Plasma from the samples were then aliquoted and frozen at −80°C. Similar methods were used for the blood draw from healthy volunteers. The frozen citrated plasma samples from both the ESRD and the healthy volunteer groups were then thawed and retrospectively analyzed using various techniques.
Enzyme-linked immunosorbent assay (ELISA) methods for multiple markers were performed using commercially available kits and guidelines provided by the manufacturer. These kits were PAI-1, TM, antiannexin V IgM, vWF activity, ATIII activity (American Diagnostic, Inc, Stamford, Connecticut), IL-1β, ICAM-1, sL-selectin (R&D Systems, Inc, Minneapolis, Minnesota), tPA-PAI-1 complex (HYPHEN BioMed, France), and MPO (Assay Designs, Inc, Ann Arbor, Michigan).
The Randox Evidence Investigator (Antrim, United Kingdom), a device measuring multiple biomarkers simultaneously on a microarray panel using chemiluminescence technology, was also used. Samples were spotted on Randox biochip microarrays, and the concentration of various biomarkers was accessed, specifically the Randox Cardiac Biochip (CK-MB, H-FABP, Myo, and cTnI), Randox Cardiac Plus Array Biochip microarray (CA3, CK-MB, GP-BB, H-FABP, Myo, and cTnI), and the Cerebral Array II Biochip microarray (CRP,
Results
In this study, the results of biomarkers evaluated by ELISA methods consist of the comparative data presented in Tables 1 and 2. These tables divide the parameters on the basis of whether biomarker levels were increased or decreased in the ESRD group when compared to values obtained from the group of healthy volunteers. All results are shown as the mean ± the standard error of the mean (SEM) for the 53 healthy individuals (designated as normal in the table) and 72 patients with ESRD. The variation in each parameter was determined by calculating the percentage of change in the normal values compared to the ESRD value ([mean ESRD value − mean Normal value]/mean normal value). P values are also given for each parameter in the tables.
The biomarkers compiled in Table 1 were all decreased in patients with ESRD compared to the control group. Plasminogen activator inhibitor type 1 was markedly decreased in patients with ESRD showing a 46% change when compared to the results of the normal. Antiannexin V IgM and tPA-PAI-1 complex were also decreased in patients with ESRD by 43% and 29%, respectively. Less dramatic reductions were notable in the AT with a change of 23% and sL-selectin with a percentage of change of 18%. In this study, ICAM-1 levels were not significantly different between the 2 groups.
A Comparison of Various ELISA-Measured Parameters Decreased in ESRD.a
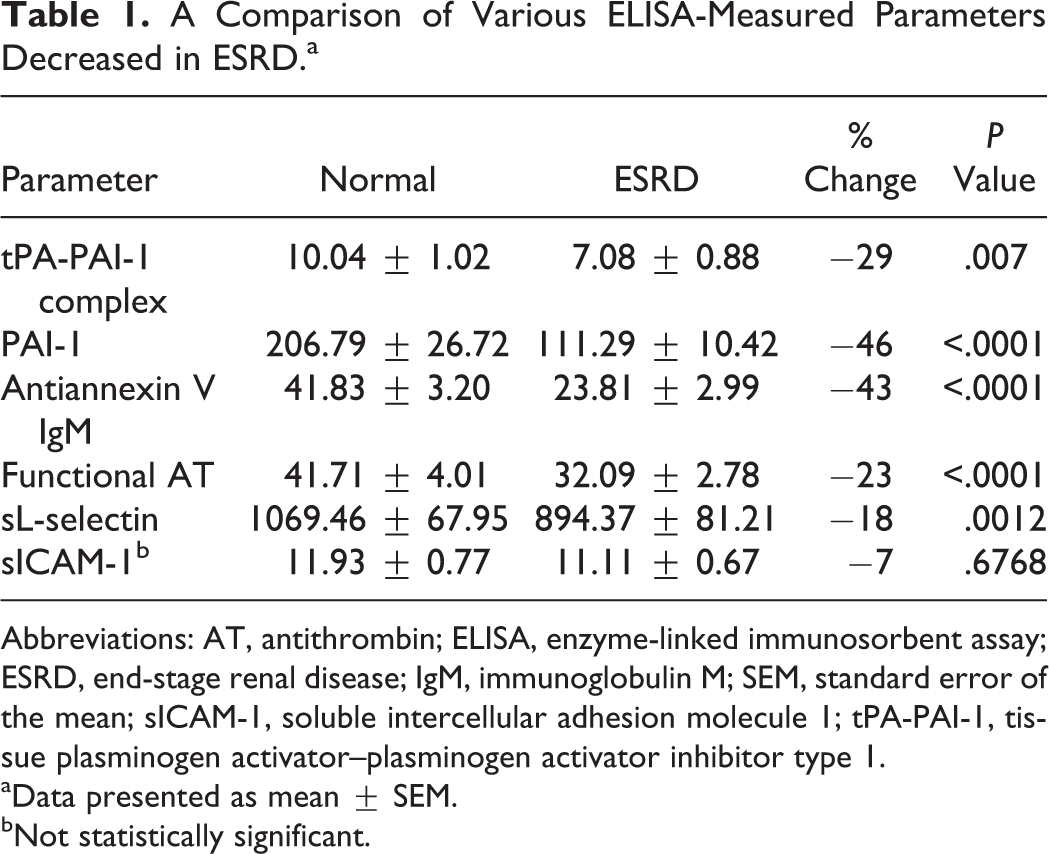
Abbreviations: AT, antithrombin; ELISA, enzyme-linked immunosorbent assay; ESRD, end-stage renal disease; IgM, immunoglobulin M; SEM, standard error of the mean; sICAM-1, soluble intercellular adhesion molecule 1; tPA-PAI-1, tissue plasminogen activator–plasminogen activator inhibitor type 1.
aData presented as mean ± SEM.
bNot statistically significant.
Biomarkers depicted in Table 2 all demonstrated an increase in the ESRD group when compared to the control group. The activity of vWF was the most significant showing a percentage of change of 1168%. Thrombomodulin was also markedly elevated in patients with ESRD having a change of 963%. Data for MPO and IL-1β also suggested higher levels of these biomarkers in the ESRD group, but these variations were not statistically significant. The composite circulating levels of tPA-PAI-1 complex, PAI-1, anti-annexin V IgM, functional AT, sL-selectin, and vWF activity in the ESRD group in comparison to the normal is given in Figure 1. As can be seen most of the parameters shown in this figure are decreased with the exception of vWF activity where a slight increase was noted.
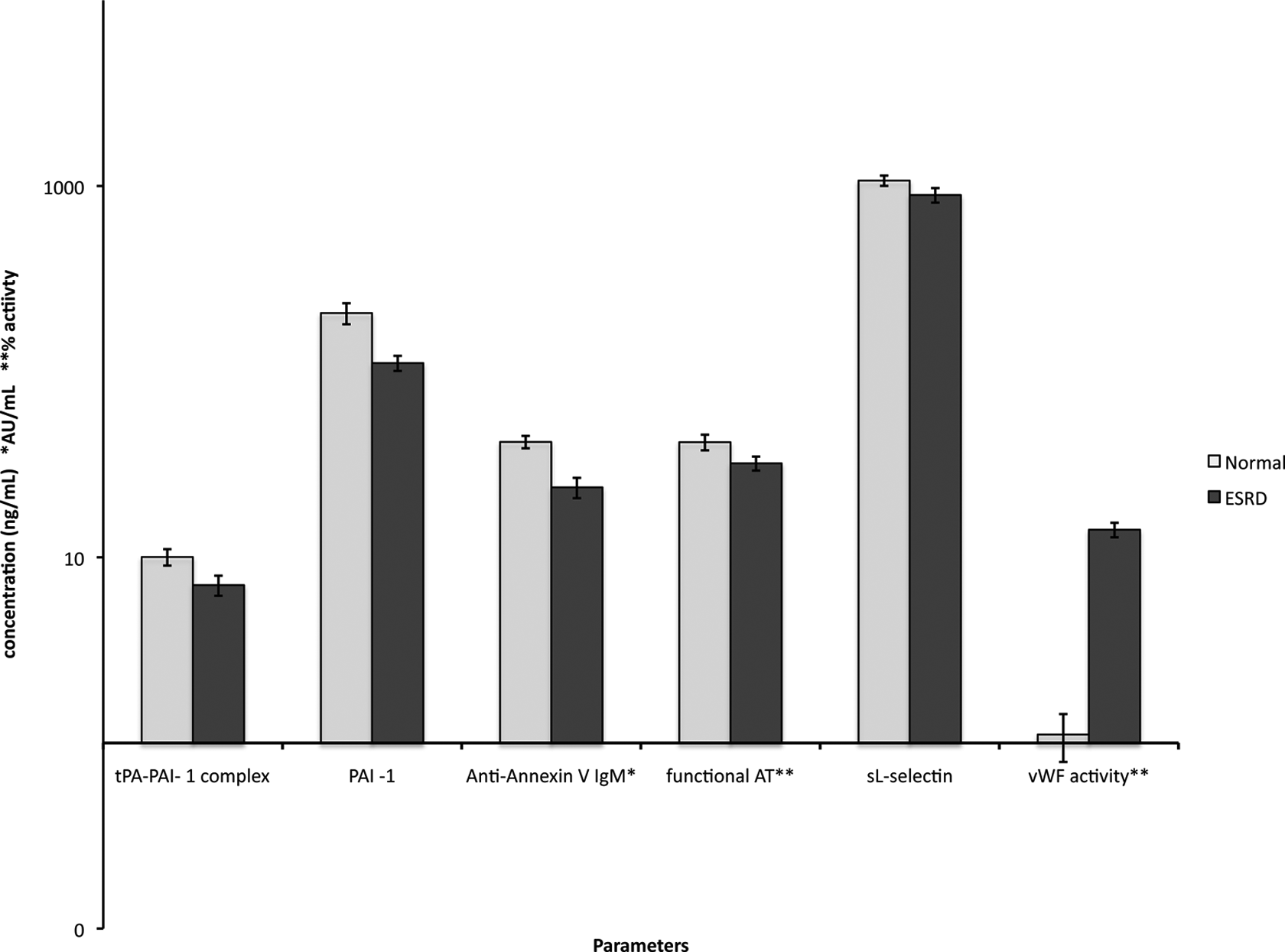
Circulating concentrations of tissue plasminogen activator–plasminogen activator inhibitor type 1 (tPA-PAI-1) complex, PAI-1, antiannexin V IgM, functional antithrombin (AT), sL-selectin, and von Willebrand factor (vWF) activity in the end-stage renal disease (ESRD) group in comparison to the normals. All results represented here show the mean ± standard error of the mean (SEM).
A Comparison of Various ELISA-Measured Parameters Increased in ESRD.a
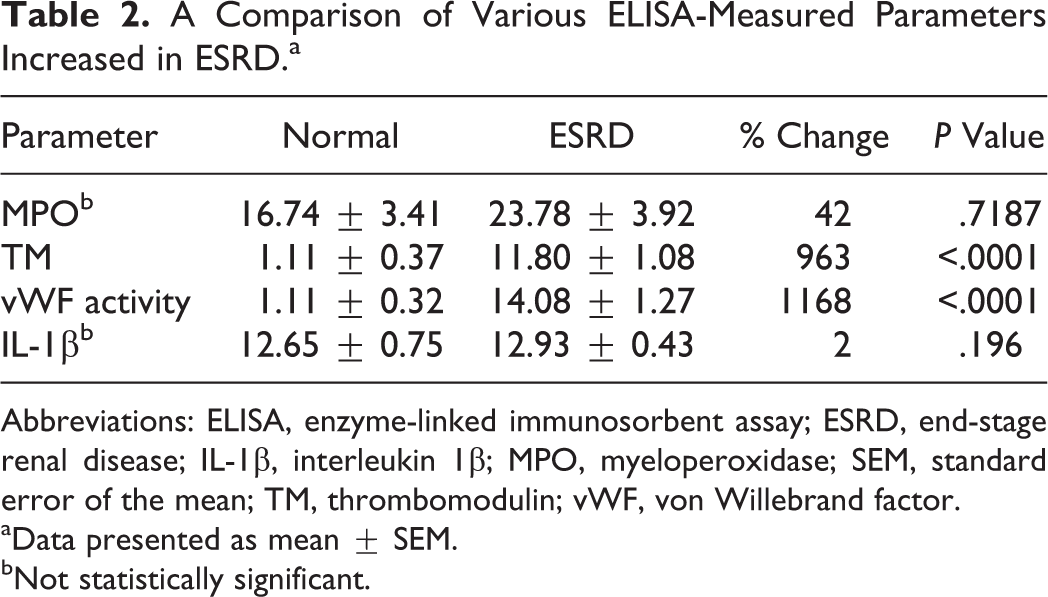
Abbreviations: ELISA, enzyme-linked immunosorbent assay; ESRD, end-stage renal disease; IL-1β, interleukin 1β; MPO, myeloperoxidase; SEM, standard error of the mean; TM, thrombomodulin; vWF, von Willebrand factor.
aData presented as mean ± SEM.
bNot statistically significant.
The various parameters measured by the Cardiac Plus Biochip microarray are illustrated in Table 3. Patients with ESRD had elevated levels of all biomarkers, except for cTnI, when compared to the control group. No significant difference was seen in the cTnI values for the ESRD and control groups. The most pronounced increase was observed in H-FABP at 1060%. Similarly, CA3 and Myo were amplified in the ESRD group with increases of 361% and 338%, respectively. In addition, CK-MB levels in the ESRD group were also higher with a change of 89% relative to the value in the normal. The slight 17% percentage of change increase seen in GPBB was not statistically significant. Figure 2 shows the composite mean concentrations of CK-MB, Myo, H-FABP, and CA3 in ESRD in comparison to the normal levels. All parameters exhibited an increase in the circulating level of the 4 analytes measured. The H-FABP and CK-MB showed marked increase, whereas the other parameters showed modest increase.
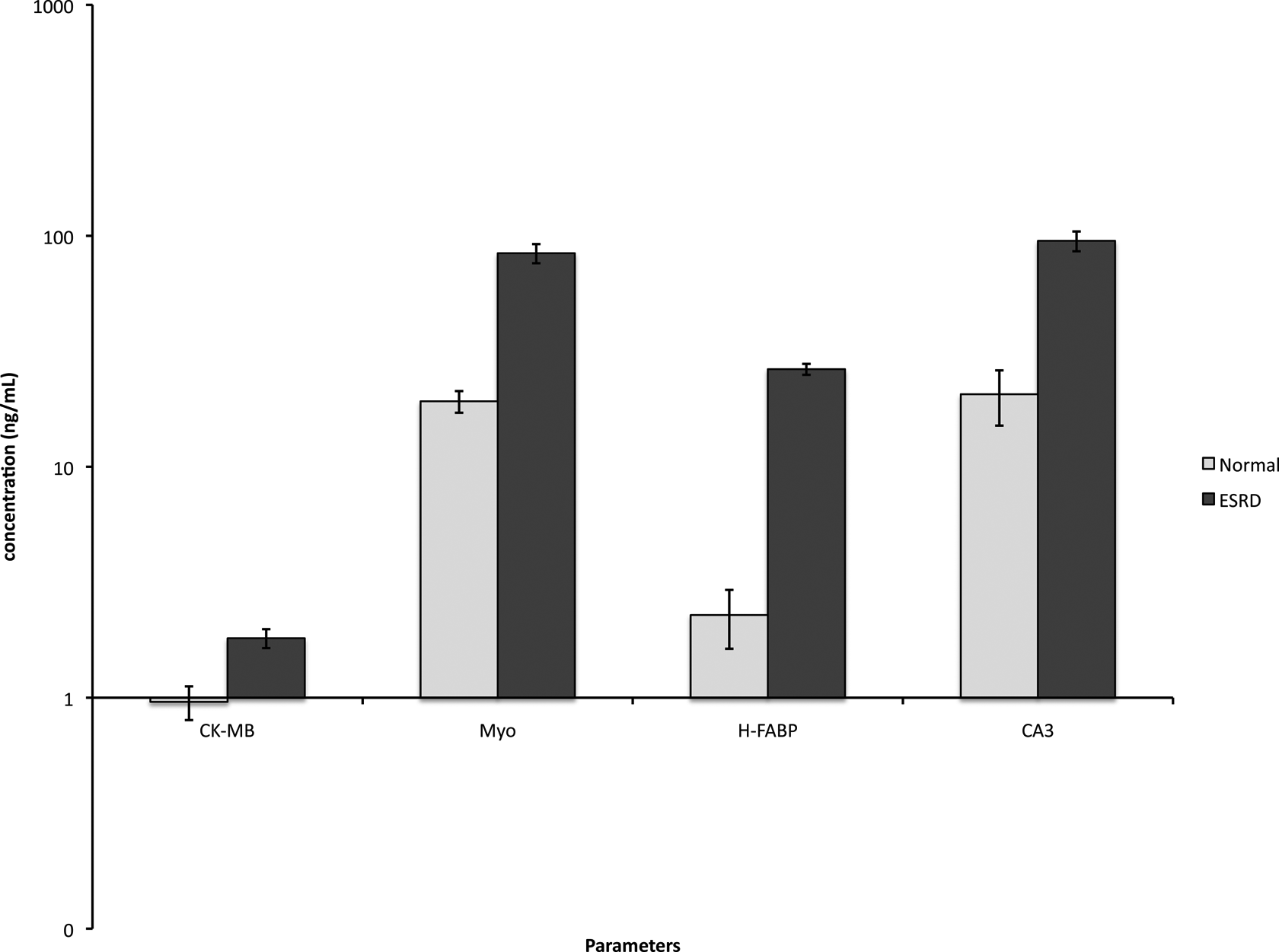
Circulating concentrations of creatine kinase MB (CK-MB), myoglobin (Myo), heart-type fatty acid binding protein (H-FABP), and carbonic anhydrase III (CA3) in the group with end-stage renal disease (ESRD) in comparison to the normals. All results represented here show the mean ± standard error of the mean (SEM).
A Comparison of Various Parameters Measured by Cardiac Plus Biochip Array.a
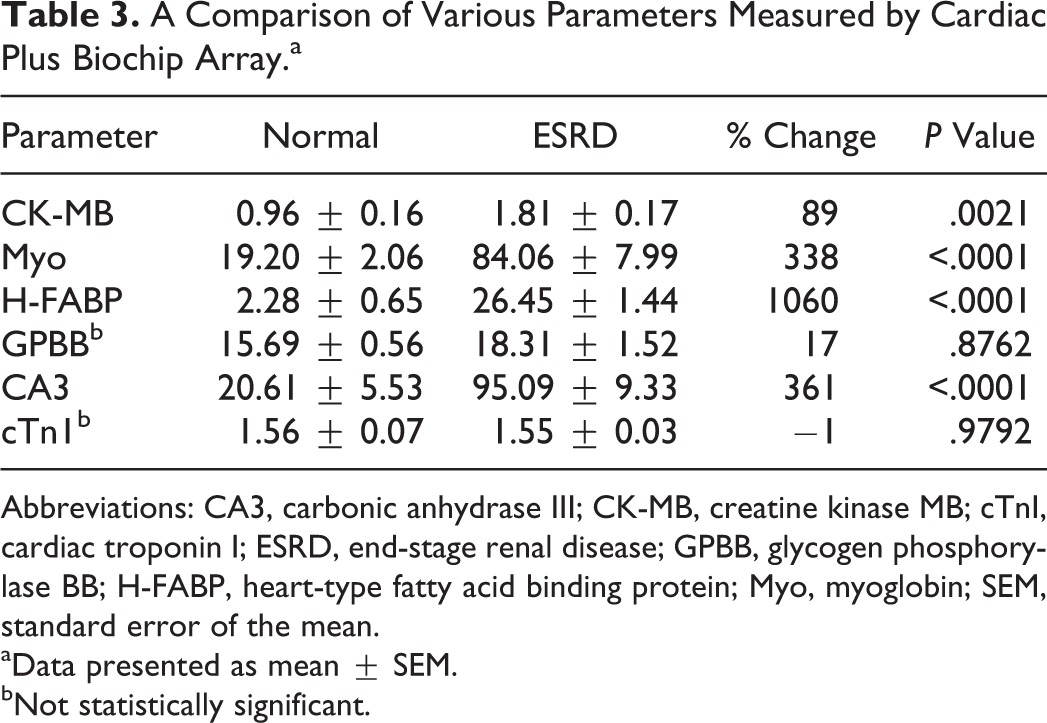
Abbreviations: CA3, carbonic anhydrase III; CK-MB, creatine kinase MB; cTnI, cardiac troponin I; ESRD, end-stage renal disease; GPBB, glycogen phosphorylase BB; H-FABP, heart-type fatty acid binding protein; Myo, myoglobin; SEM, standard error of the mean.
aData presented as mean ± SEM.
bNot statistically significant.
Table 4 displays the parameters measured by the Cerebral Array II Biochip microarray, which were all elevated in the ESRD group. The most striking increase of 1505% was seen in sTNFR1. Thrombomodulin with an increase of 478% and
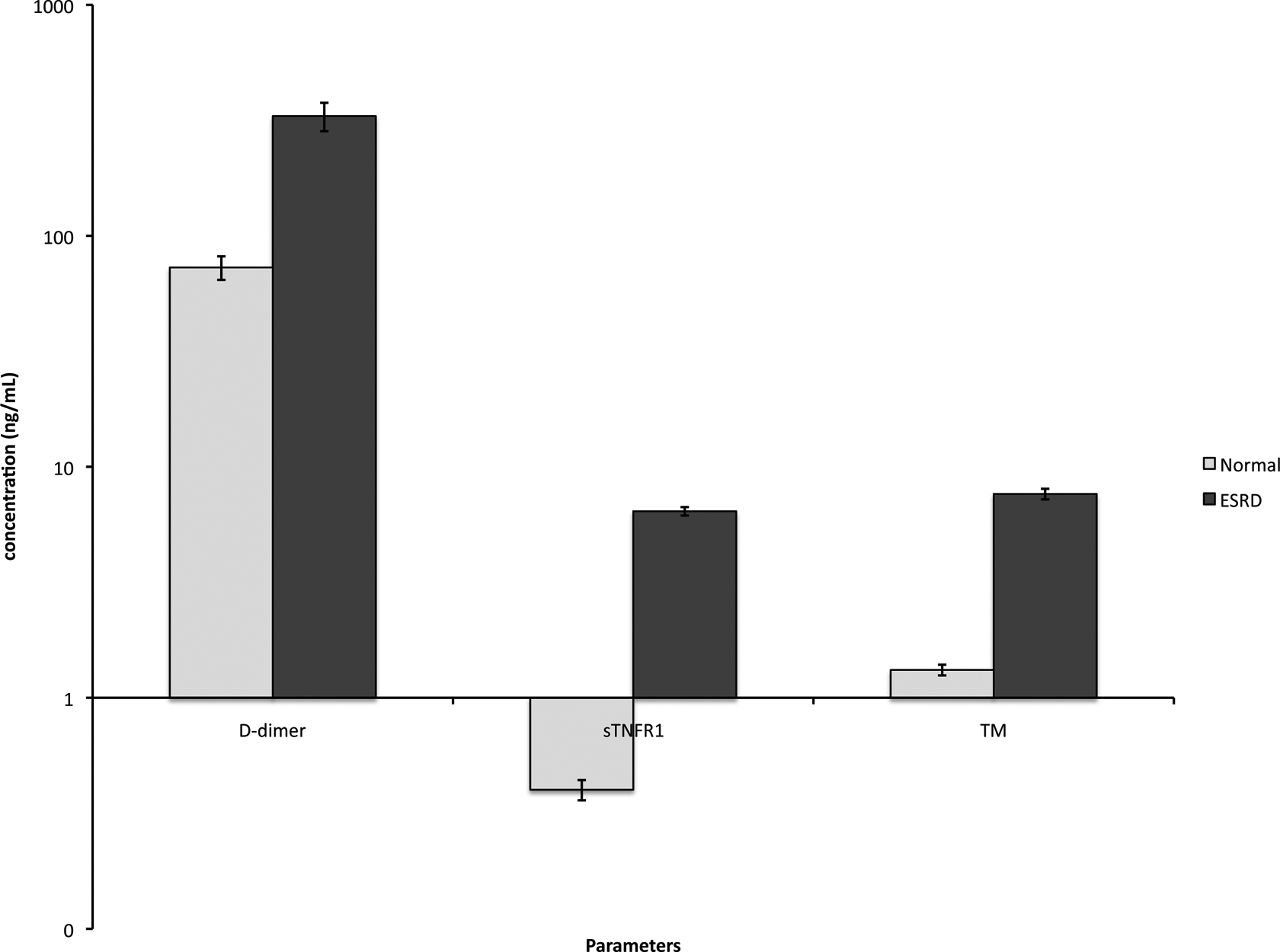
Relative increase in the circulating
A Comparison of Various Parameters Measured by Cerebral Biochip Array.a
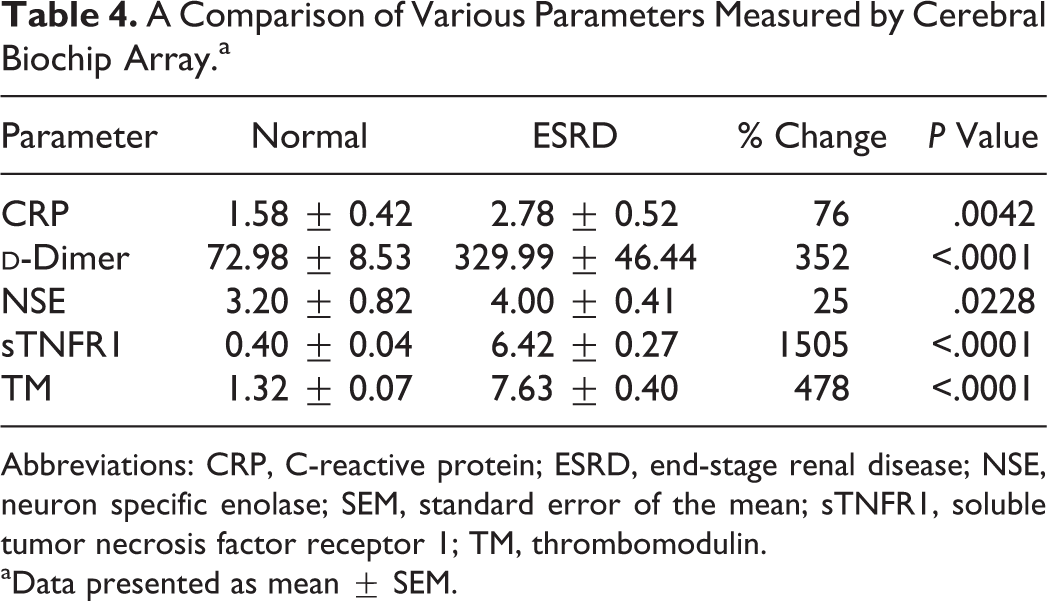
Abbreviations: CRP, C-reactive protein; ESRD, end-stage renal disease; NSE, neuron specific enolase; SEM, standard error of the mean; sTNFR1, soluble tumor necrosis factor receptor 1; TM, thrombomodulin.
aData presented as mean ± SEM.
Discussion
Patients with ESRD undergoing maintenance hemodialysis for treatment have elevated mortality rates with a large proportion of these deaths resulting from cardiovascular events or cardiovascular disease. This study determined sTNFR1 to be the most profoundly increased biomarker in the plasma samples of patients with ESRD compared to the normal. Soluble TNFR1 is a 55-kDa protein receptor that triggers intracellular signaling by recruiting adaptor proteins. It plays a role in proinflammatory (antiapoptotic) responses and proapoptotic responses and has also been associated with myocardial infarctions. 11,12 The amplified sTNFR1 levels are most likely the result of increased cellular damage and release of this receptor protein from cell membranes. Considering the profound elevation in sTNFR1 levels in patients with ESRD, this biomarker could potentially be used as a risk-stratification tool to decrease the sequelae of cardiovascular disease in this patient population.
Additionally, the level of H-FABP and vWF activity was also drastically increased in the ESRD group compared to levels seen in the control group. The H-FABP is a 15-kDa cytosolic protein that is highly specific for cardiomyocytes. In fact, a recent study illustrated H-FABP to be statistically more sensitive than cTnI and CK-MB in the detection of myocardial injury. 13 Our data further support this theory, given that CK-MB was only mildly elevated in patients with ESRD and no difference was seen in cTnI levels between the 2 groups. However, the increased amount of H-FABP in patients with ESRD was evident. Despite extensive evidence supporting the association of elevated cTnI levels with cardiac and all-cause mortality in asymptomatic hemodialysis patients, H-FABP may prove to be a more reliable biomarker in these patients. 14,15 Conceivably, H-FABP may detect subclinical myocardium injury prior to the onset of cTnI elevation in patients with ESRD. In addition, vWF is a protein that promotes the attachment of platelets to areas of injury in blood vessels. 16 Various studies have established the connection between high vWF levels and increased risk of arterial thrombosis including, but not limited to, myocardial infarction and ischemic stroke. 17 The amplification of vWF activity seen in this study further elucidates the role hemostatic processes play in ESRD and their effect on the increased susceptibility of this patient group to the consequences of cardiovascular disease.
Thrombomodulin, CA3,
The amplification of 11 of the 16 biomarkers with statistical significance revealed in the ESRD group lends further support to the multifactorial pathophysiology present in ESRD.
The relative increase in various biomarkers in patients with ESRD is compared in terms of percentage of change to the normal levels. In addition, the data are also represented for each of these markers in terms of absolute concentrations. Although both of these approaches provided differences in the levels of these parameters, percentage of change revealed greater differences in the ESRD compared to the normal. Thus, percentage of change may portray the true difference in the circulating levels of these biomarkers.
Limitations of this study include the small sample size and that the patients with ESRD were not epidemiologically matched to individuals in the control group. The results of this study warrant further investigation and an analysis of variables such as age, sex, BMI, and smoking status to determine their affect, if any, on biomarker levels. A future direction of this study would be the comparison of patient outcomes to the statistically relevant biomarkers to determine clinical correlations. If such correlations are revealed then specific biomarkers could become highly predictive in the population with ESRD and their cardiovascular complications.
Conclusion
Our study illustrates the complex nature of the pathogenesis of ESRD, which has yet to be fully understood. The results obtained are consistent with previous studies demonstrating that the activation of both hemostatic and inflammatory processes plays a role in the polypathologic process that is ESRD. The substantial increase in vWF activity proposes its potential regulatory role in the hemostatic processes apparent in ESRD. Likewise, such remarkable elevations in H-FABP and sTNFR1 suggest these biomarkers may have a central role in the overall inflammatory process in the pathogenesis of ESRD.
Footnotes
Acknowledgments
The authors are grateful to Dean Linda Brubaker, MD, for providing this opportunity and support to complete this study. The authors are also grateful to the staff of the Loyola Dialysis unit for facilitating the procurement of the blood samples used in this study and the staff of the Hemostasis and Thrombosis Laboratory for their expert help in generating the data on biomarkers. We are also thankful to Mr Rajneesh Mathur of Randox laboratories for helpful suggestions and advice related to the technical issues.
Authors’ Note
This study was a part of the STAR research program completed for the requirement of MD Honors in Research Degree at the Loyola University Stritch School of Medicine.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by internal funds of the Department of Pathology.
