Abstract
Introduction
Venous thromboembolism (VTE) is a serious, frequent, and preventable medical complication in hospitalized patients. Although the efficacy of prophylaxis (pharmacological and/or mechanical) has been demonstrated, compliance with prophylaxis is poor at international and national levels.
Aim
To determine the indication and use of pharmacological thromboprophylaxis in hospitalized patients in Uruguay.
Methods
An observational, descriptive, cross-sectional, multicentre study involving 31 nationwide healthcare facilities was conducted. Baseline characteristics associated with hospital admission, the percentage of the population with an indication for thromboprophylaxis, and the percentage of patients receiving pharmacological thromboprophylaxis were assessed. The VTE risk was determined using the Padua score for medical patients; the Caprini score for surgical patients; the Royal College of Obstetricians and Gynaecologists (RCOG) guidelines for pregnant-postpartum patients.
Results
1925 patients were included, representing 26% of hospitalized patients in Uruguay. 71.9% of all patients were at risk of VTE. Of all patients at risk of VTE, 58.6% received pharmacological thromboprophylaxis. The reasons for not receiving thromboprophylaxis were prescribing omissions in 16.1% of cases, contraindication in 15.9% and 9.4% of patients were already anticoagulated for other reasons. Overall, just 68% of patients were “protected” against VTE. Recommendations of major thromboprophylaxis guidelines were followed in 70.1% of patients at risk.
Conclusions
Despite the progress made in adherence to thromboprophylaxis indications, nonadherence remains a problem, affecting one in six patients at risk of VTE in Uruguay
Keywords
Introduction
Venous thromboembolism (VTE), in the form of deep vein thrombosis (DVT) and pulmonary embolism (PE), is a serious and common medical complication that can occur in hospitalized patients admitted for both medical and surgical reasons. PE is the leading cause of preventable death in hospitalized patients, accounting for 5-10% of hospital deaths. VTE can result in many sequels that seriously affect the quality of life of patients, such as chronic pulmonary hypertension and post-thrombotic syndrome, both of which have high social and clinical costs. Prevention of VTE is considered a quality indicator of hospital care.1–4
For more than 20 years, both low-molecular-weight heparin (LMWH) and unfractionated heparin have been used effectively in the prevention of VTE.5–9
More recently, the benefits of direct oral anticoagulants (DOACs) have been demonstrated in major orthopedic surgery.10,11 However, there is currently insufficient evidence to support their use in other scenarios such as medical cases, nonorthopedic surgery, or nonmajor orthopedic surgery.12–16
Anticoagulants are not without risk. A correct assessment on admission and during hospital stay is required to consider the benefit. Standardized risk of VTE using clinical scores (Caprini, Padua, specific guidelines for pregnant and postpartum patients by the Royal College of Obstetricians and Gynaecologists—RCOG)17–20 are available to predict risk.
The development of international guidelines aims to facilitate the implementation of the different methods available for the prevention of VTE, which may be mechanical or pharmacological.
The adequate use of these recommendations implies the prescription of thromboprophylaxis in high-risk patients and withholding its prescription in low-risk patients.21–23
Multicentre studies conducted in Uruguay found that in 2007 only 18% of patients at high risk of VTE were prescribed pharmacological thromboprophylaxis (35% medical patients and 12% surgical patients), whereas in 2017 an improvement was noted: 55.8% of high-risk patients were receiving pharmacological thromboprophylaxis (66.7% medical patients and 41.9% surgical patients).24,25 Although these studies were multicentre, they only included hospitals in the capital city, Montevideo which accounts for 40% of the country's population (1,319,108 out of 3,286,314 inhabitants). Comprehensive health care for all citizens is guaranteed by the Integrated National Health System, a system that brings together public and private health care providers for this purpose. 26 Fifty-four percent of the population has access to private health facilities. 27 The 2007 and 2017 studies included both private and public providers.
The main objective of this study was to determine the indication and prescription of pharmacological thromboprophylaxis in hospitalized patients in a wider selection of hospitals outside of the capital city. Specific objectives were to determine the prevalence of hospitalized patients at risk of VTE, to evaluate the prescription of pharmacological thromboprophylaxis in this population, to assess the appropriate use of international guidelines and recommendations, and to compare the adherence to these recommendations in different subgroups of the population considered.
Methods
An observational, descriptive, cross-sectional, multicentre study was conducted involving 31 nationwide healthcare facilities. Data collection took place over a 3-day period between 21 and 23 July 2022 in each center by a team that could include the attending physician of the patients enrolled in the study. The study population consisted of noncritical medical, surgical, and pregnant˗postpartum patients who were hospitalized during the data collection period.
Inclusion criteria: noncritical patients admitted to medical wards (internal medicine and subspecialties such as nephrology, hematology, neurology), surgical wards, and obstetrics and gynecology wards. Exclusion criteria: patients under 18 years old. Patients admitted to intensive care units, high dependency units, or emergency departments. Patients admitted for major orthopedic surgery, cardiac surgery, and patients with a diagnosis of COVID.
Sociodemographic variables such as sex and age, and variables associated with hospital admission, including health care facility (public vs private and Montevideo vs the rest of the country), number of patients surveyed, ward of admission, and percentage of the population with an indication for thromboprophylaxis were assessed. To determine the number of patients at risk of VTE, we used validated scores: the Padua score for medical patients and the Caprini score for surgical patients.18,19 The Royal College of Obstetricians and Gynaecologists (RCOG) guidelines used for pregnant˗postpartum patients are not validated but are a set of carefully compiled guidelines for a wide range of pregnancy-related conditions. 20 The definition of indication for thromboprophylaxis according to appropriate clinical scores was defined for: medical patients as a Padua score ≥4 (range 0-20), surgical patients as a Caprini score ≥3 (range 0-74) and patients in pregnant˗postpartum period with high or intermediate risk according to RCOG guidelines. Reasons for contraindication to pharmacological thromboprophylaxis were reviewed using the NICE guidelines for risk assessment for VTE. 28
For patients at risk, we considered the percentage of patients receiving pharmacological thromboprophylaxis. For the patients who were not at risk, we recorded the cases of over-prescription of pharmacological thromboprophylaxis. Differences between the proportions of patients receiving thromboprophylaxis were evaluated using a chi-squared test with a significance level of 0.05. We used the Epi Info 3.5.4 program.
Ethical Considerations
The study was approved by the ethics committee of each participating institution. Written informed consent was obtained from each patient included in the study.
Results
A total of 1925 patients hospitalized for medical, surgical, or pregnancy/postpartum-related admissions from 31 healthcare institutions were included. The median age of the study population was 65 years (interquartile range 50-76 years) and 52.7% (1009) were female. 73.9% (1423) of the included population were from Montevideo and 43.1% (830) were hospitalized in public health facilities. 62.8% (1208) were hospitalized for medical conditions, 31.2% (600) for surgical conditions, and 6% (117) were pregnant˗postpartum patients.
Indication for Thromboprophylaxis
According to the proposed scores, 71.9% (1385) of the patients surveyed were at risk of VTE. The indication for thromboprophylaxis by type of condition was as follows, 71.5% (864) of patients admitted for medical conditions, 80.5% (483) of patients admitted for surgical conditions, and 32.5% (38) pregnant and postpartum patients (see Figure 1).
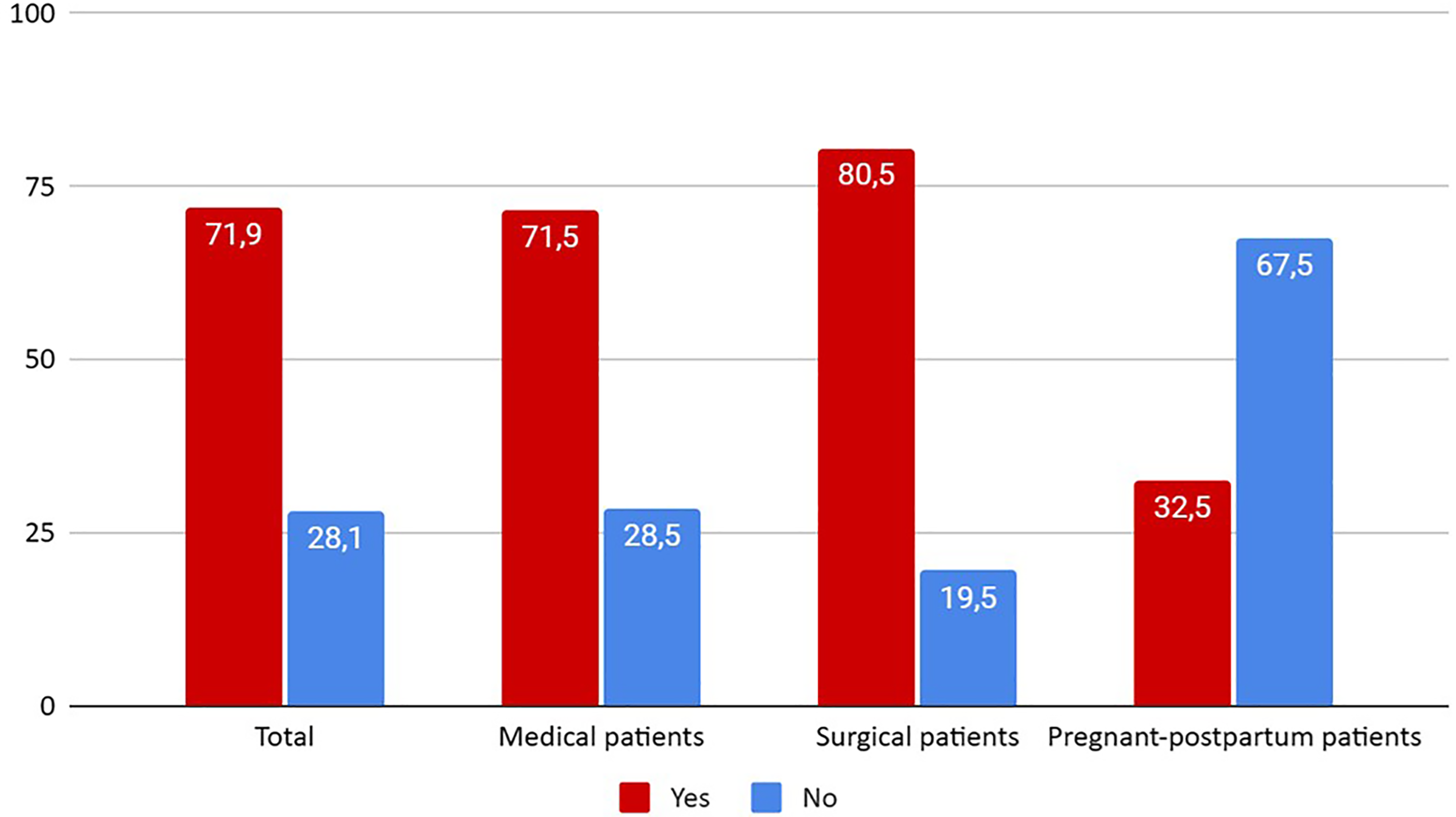
Patients at risk of VTE (%).
Adherence to Indication of Pharmacological Thromboprophylaxis
Of all patients at risk of VTE, 58.6% (811) received pharmacological thromboprophylaxis. The reasons for not administering thromboprophylaxis were omission in 16.1% (223) of cases, contraindication in 15.9% (220) and 9.4% (131) of patients were already anticoagulated for other reasons (see Figure 2). Therefore, 68% (942) of patients can be considered “protected” against VTE (percentage of patients on thromboprophylaxis and those with indication for anticoagulation). Twenty-six patients with contraindications to thromboprophylaxis still received it.
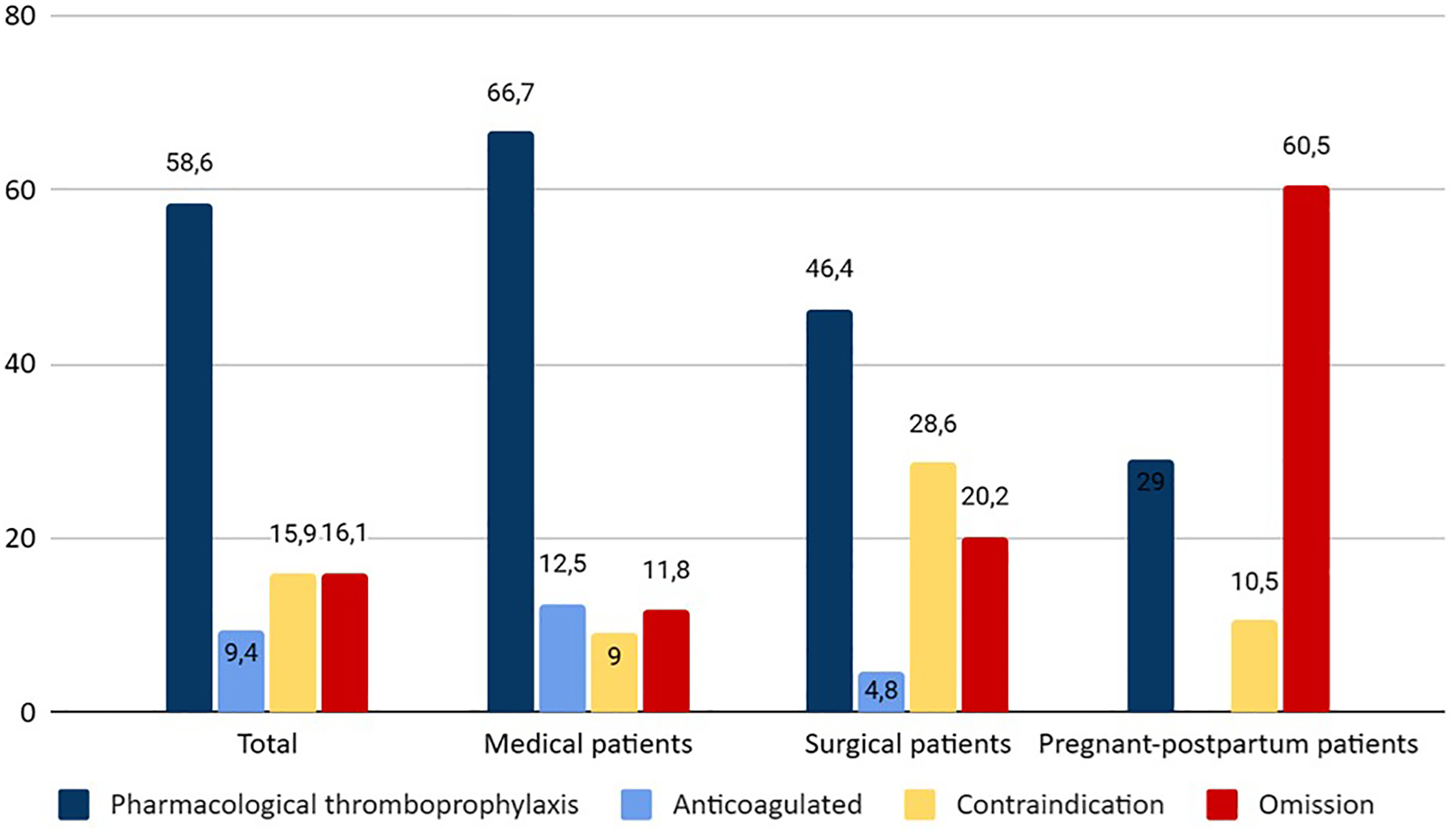
Adherence to indication of pharmacological thromboprophylaxis (%).
Medical Patients
Among medical patients who had an indication for pharmacological thromboprophylaxis (864), the compliance rate was 66.7% (576). Of those who did not receive it, 11.8% (102) were not receiving thromboprophylaxis due to prescribing omission, 9% (78) due to contraindication, and 12.5% (108) were already anticoagulated. Therefore, 79.2% of this group of patients were “protected” against VTE. The most common risk factors for VTE among all medical patients (1208) were: reduced mobility 68.8% (831), age 70 years or older 45.1% (545), infection or rheumatic disease 30.7% (371), respiratory or heart failure 28.4% (384), and active cancer 19.3% (233) (see Table 1). The most common contraindications were ongoing bleeding 5% (60), thrombocytopenia 2.6% (32), and acquired bleeding disorders 1.5% (18).
Risk Factors for VTE in Medical Patients.

Surgical Patients
Among surgical patients who had an indication for pharmacological thromboprophylaxis (483), the compliance rate was 46.4% (224). Of those who did not receive it, 20.2% (98) were not receiving thromboprophylaxis due to omission, 28.6% (138) due to contraindication, and 4.8% (23) were already anticoagulated. Therefore, 51.2% of this group of patients were “protected” against VTE. The most common risk factors for VTE in all surgical patients (600) were: age 85% (510, including all age-related categories), bed rest of more than 72 h 40.3% (242), major open surgery 21.7% (130), body mass index (BMI) greater than 25 kg/m2 18.2% (109), and planned minor surgery 17.3% (104) (see Table 2).
Risk Factors for VTE in Surgical Patients.
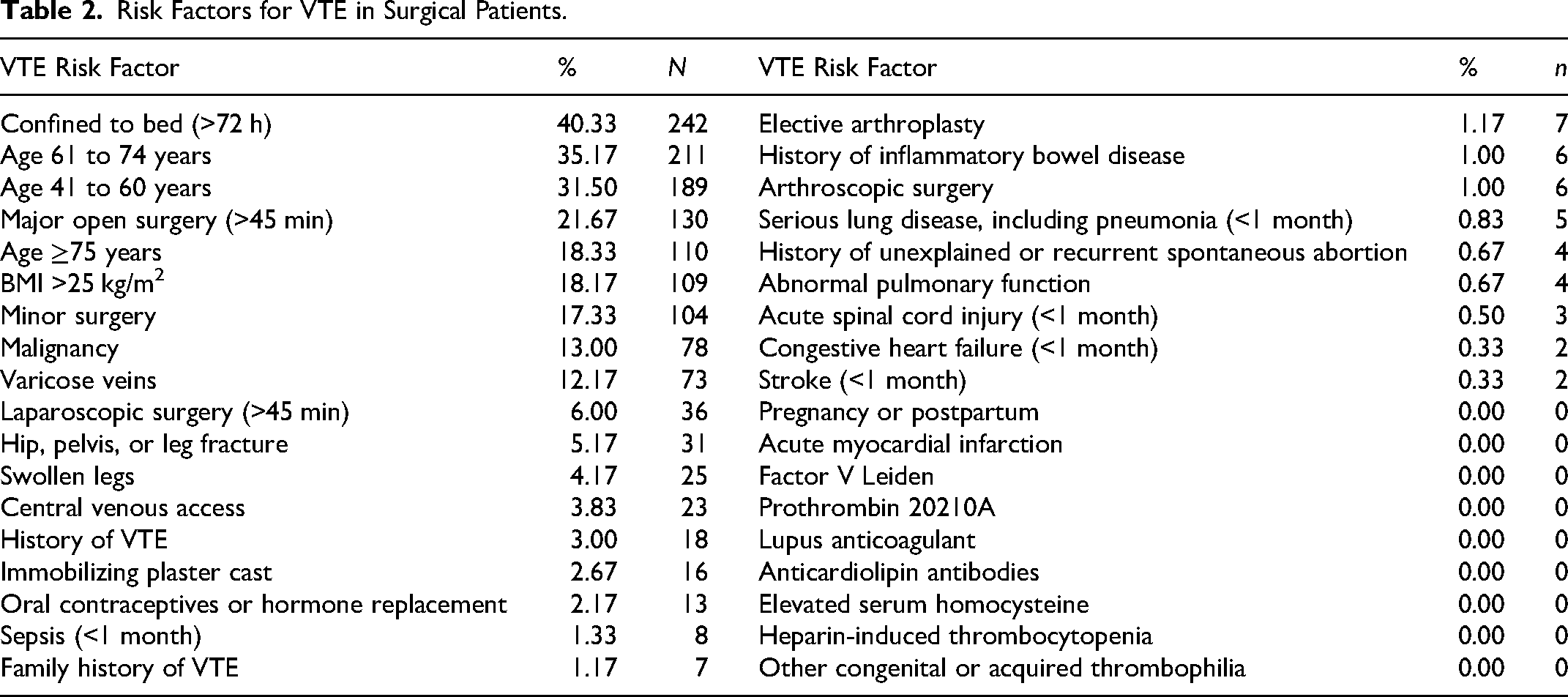
The most common contraindications were pre or postoperative period 20.3% (122) and active bleeding 8.7% (52).
When divided by risk level according to the Caprini score, we found that 28% (135) of the patients had a moderate risk (Caprini score 3-4) and 71% (343) had a high risk (Caprini score >5). In five cases (1%), the Caprini score was not recorded and only the indication or contraindication was recorded. Among patients at moderate risk of VTE, only 37.8% (49) were receiving pharmacological thromboprophylaxis, 21.5% (31) were not receiving it due to omission, 5.2% (7) were already anticoagulated, and 35.5% (48) had a contraindication. In the high-risk group, 50.7% (174) received pharmacological thromboprophylaxis, 18.4% (63) did not receive it due to omission, 4.7% (16) were already anticoagulated, and 26.2% (90) had a contraindication (see Figure 3). In the intermediate risk group, the proportion of patients with an indication for and receiving pharmacological thromboprophylaxis was significantly lower than in the high-risk group (P = 0.039, OR 0.57, IC 95% 0.34-0.98).
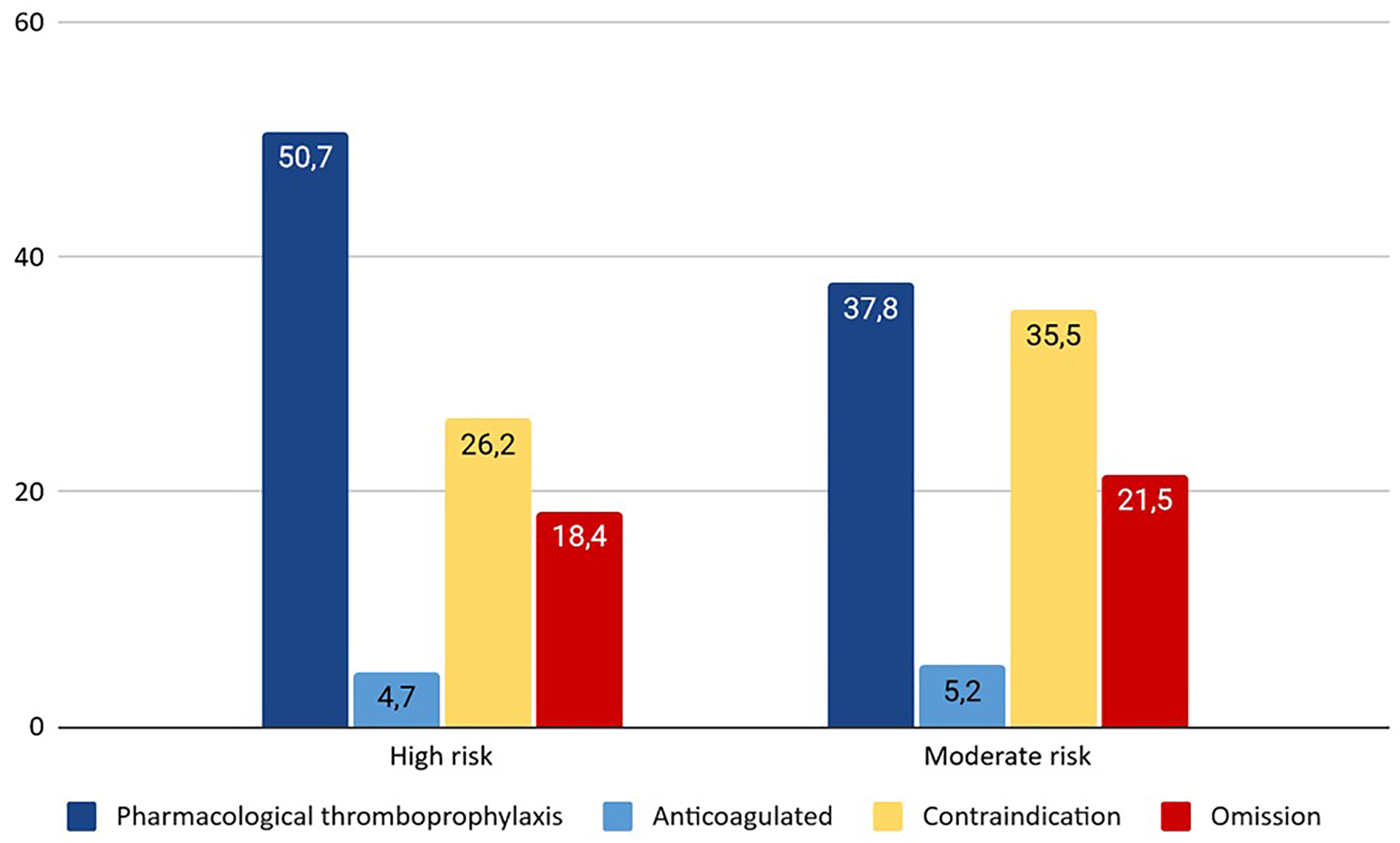
Thromboprophylaxis in moderate versus high risk surgical patients (%).
Pregnant-Postpartum Patients
Among pregnant-postpartum patients who had an indication for pharmacological thromboprophylaxis (38), compliance was 29% (11). Of those who did not receive thromboprophylaxis, 60.5% (23) did not receive it because of omission and 10.5% (4) because of contraindication. The most common risk factors for VTE in pregnant-postpartum patients (117) were: age >35 years 22.2% (26), elective cesarean section 20.5% (24), BMI >30 kg/m2 16.2% (19), parity >3 12.8% (15), and smoking habit 12% (14) (see Table 3). The most common contraindications were postsurgery 10.3% (12) and active bleeding 2.6% (3). When divided by risk level according to the RCOG guidelines, we found that 86.8% (33) were at intermediate risk of VTE and 13.2% (5) were at high risk of VTE. Of the intermediate-risk patients, only 24.3% (8) were receiving pharmacological thromboprophylaxis, 66.7% (22) were not receiving it due to omission and 9% (3) had a contraindication. Among high-risk patients, 60% (3) received pharmacological thromboprophylaxis, 20% (1) did not receive it due to omission, and 20% (1) had a contraindication (see Figure 4). There is a higher proportion of patients at high risk of VTE who had an indication for thromboprophylaxis and consequently received it compared to the intermediate risk group, with a P-value of 0.052.
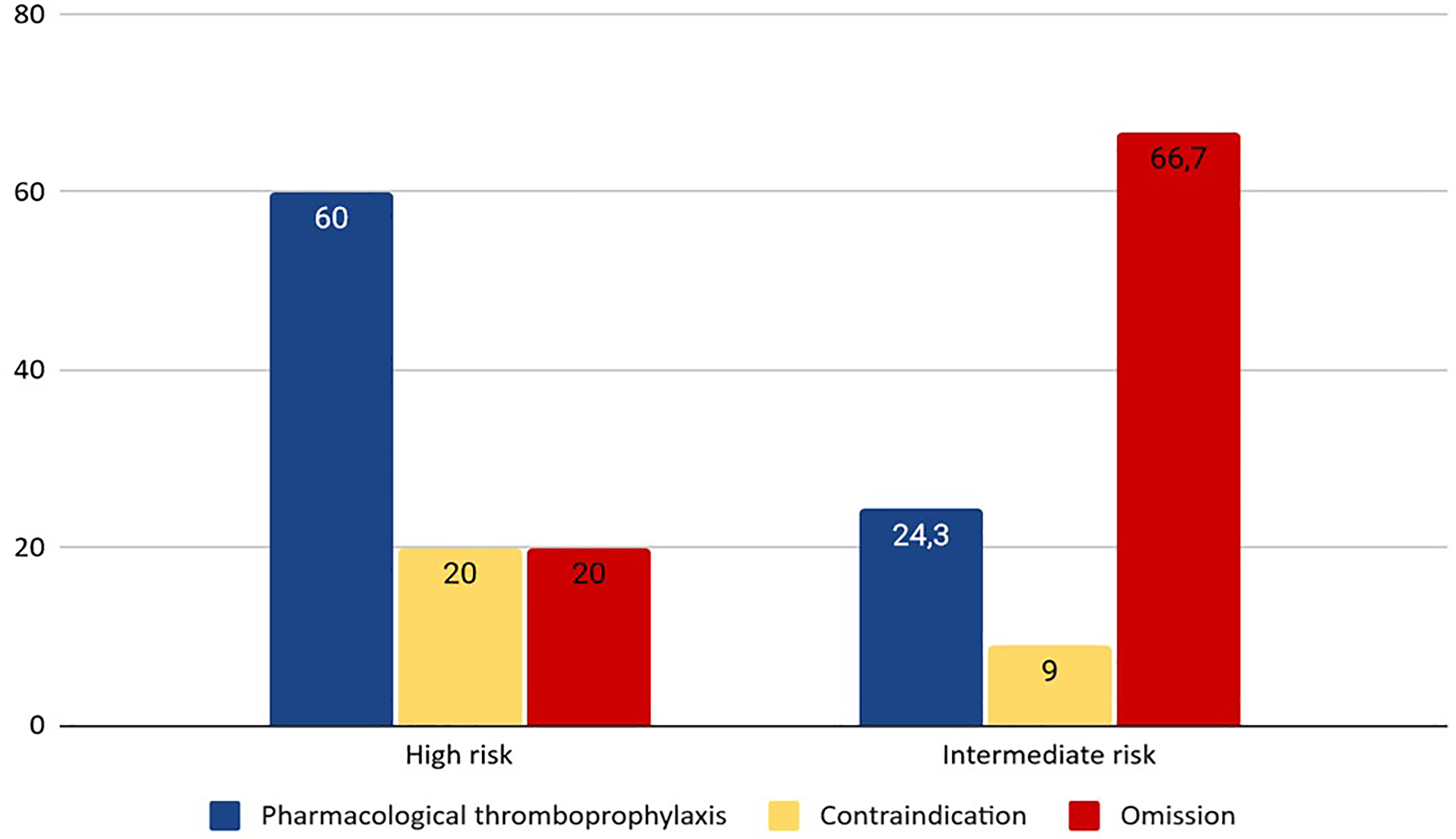
Thromboprophylaxis in intermediate versus high risk pregnant-postpartum patients (%).
Risk Factors for VTE in Pregnancy-Postpartum Patients.
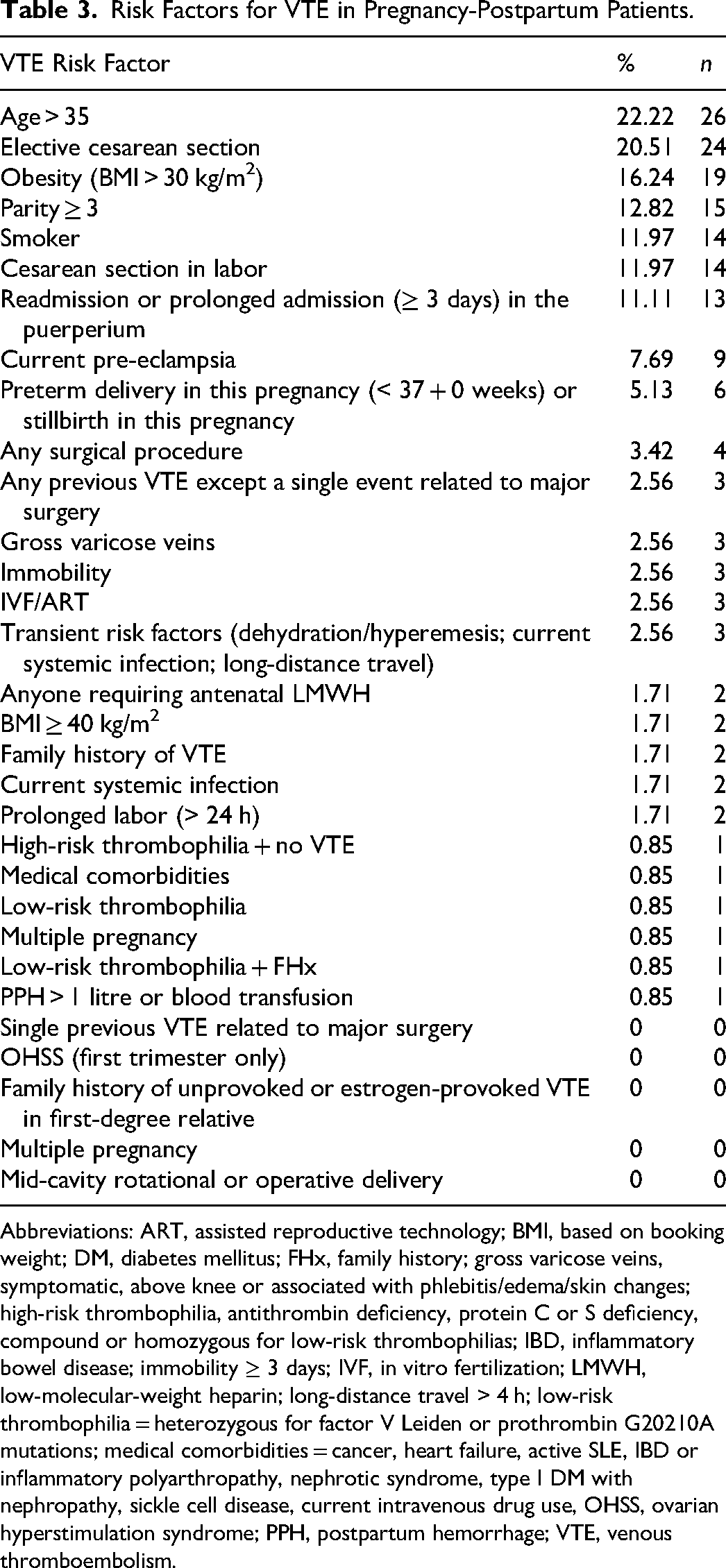
Abbreviations: ART, assisted reproductive technology; BMI, based on booking weight; DM, diabetes mellitus; FHx, family history; gross varicose veins, symptomatic, above knee or associated with phlebitis/edema/skin changes; high-risk thrombophilia, antithrombin deficiency, protein C or S deficiency, compound or homozygous for low-risk thrombophilias; IBD, inflammatory bowel disease; immobility ≥ 3 days; IVF, in vitro fertilization; LMWH, low-molecular-weight heparin; long-distance travel > 4 h; low-risk thrombophilia = heterozygous for factor V Leiden or prothrombin G20210A mutations; medical comorbidities = cancer, heart failure, active SLE, IBD or inflammatory polyarthropathy, nephrotic syndrome, type I DM with nephropathy, sickle cell disease, current intravenous drug use, OHSS, ovarian hyperstimulation syndrome; PPH, postpartum hemorrhage; VTE, venous thromboembolism.
Patients at Low Risk of VTE or Without Indication for Thromboprophylaxis
Of the 535 patients without an indication for pharmacological thromboprophylaxis, 23.9% (129) received it. Of the 341 medical patients without an indication for thromboprophylaxis, 29.3% (100) received it, as did 23.7% (22) of the 115 surgical patients. Of the 79 pregnant-postpartum patients without an indication, 7.6% were receiving pharmacological thromboprophylaxis.
Comparison Between Groups
The proportion of medical patients with an indication for and receiving pharmacological thromboprophylaxis was significantly higher than the proportion of surgical or pregnant-postpartum patients combined (P < 0.0001, OR 1.27, CI 95% 1.18-1.38 for the entire group). There were no significant differences in the prescription of pharmacological thromboprophylaxis in patients at risk when compared by location, funding, number of patients, or proportion of patients at risk.
Compliance with International Guidelines
Overall, 70.1% (1349/1925) of patients were treated according to international guidelines for pharmacological thromboprophylaxis (sum of the percentage of patients at risk who received pharmacological thromboprophylaxis and the percentage of patients not at risk who did not receive it).
Limitations
Thirty patients surveyed did not want to participate in the study.
Discussion
This study, with 1925 patients, is the largest cross-sectional study of the use of pharmacological thromboprophylaxis in hospitalized patients at risk of VTE according to international guidelines utilized in Uruguay giving a clear picture of current clinical practice.
The total number of nonintensive care beds available in Uruguay is 7346; therefore, approximately 26% of all hospitalized patients were included, 27 making it one of the largest nationwide surveys carried out (see Table 4), when you take into account the percentage of hospital beds included.
Comparison of Percentage of Available Hospital Beds Surveyed in International Studies.
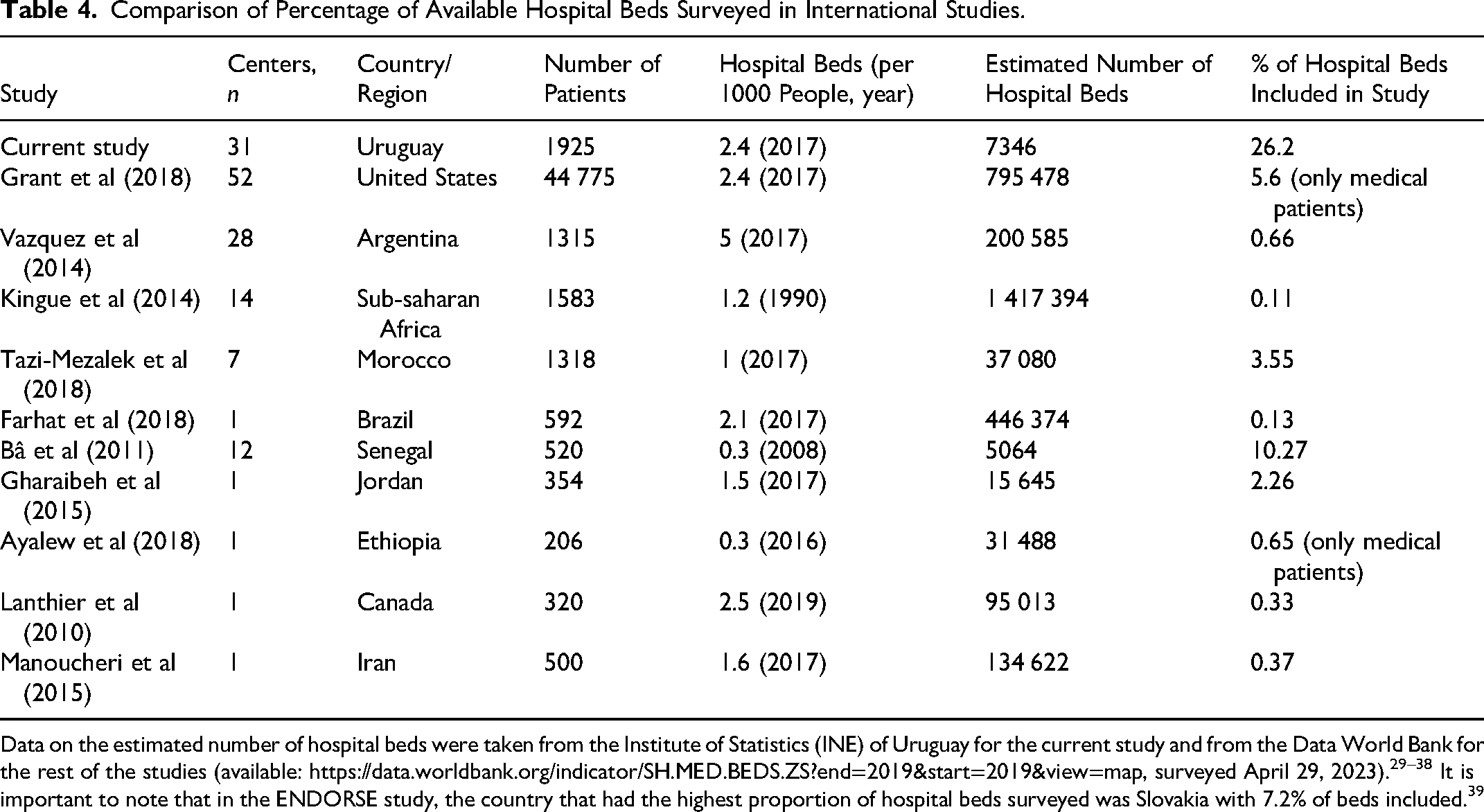
Data on the estimated number of hospital beds were taken from the Institute of Statistics (INE) of Uruguay for the current study and from the Data World Bank for the rest of the studies (available: https://data.worldbank.org/indicator/SH.MED.BEDS.ZS?end=2019&start=2019&view=map, surveyed April 29, 2023).29–38 It is important to note that in the ENDORSE study, the country that had the highest proportion of hospital beds surveyed was Slovakia with 7.2% of beds included. 39
The findings are consistent with previous national and international studies showing that the risk of VTE is present in a high percentage of hospitalized patients (between 60% and 80%).25,40,41 This study reports that 71.9% (1385) of the total study population was at risk of VTE, with a higher risk in surgical patients 80.5% (483) than in medical patients 71.5% (864). The risk in pregnant-postpartum patients is 32.5% (38), as expected given the characteristics of these patients.
Pharmacological venous thromboprophylaxis was used in 58.6% (811) of the eligible population studied. Thromboprophylaxis was not given in 16.1% (223) of cases because of prescribing omission, in 15.9% (220) because of contraindication, and in 9.4% (131) of patients because they were already anticoagulated. Special consideration should be given to anticoagulation as a contraindication to thromboprophylaxis, as these patients are also protected against VTE. For this reason, we believe it is correct to include this specific group of patients in the population considered to have received correct thromboprophylaxis. We can conclude that 68% of patients were protected against VTE.
At a national level, there has been an increase in the use of pharmacological thromboprophylaxis. Comparing the three multicenter studies carried out to date (2007, 2017, current study), the first with 316 patients, the second 427 patients, and the current study of 1925 patients, adherence to thromboprophylaxis recommendations increased from 18%, 55.8%, and 58.6%, respectively.24,25
On a regional (South American) level, two studies conducted in Brazil in the last decade33,42 showed that 21.6% and 63.7% of patients at risk of VTE actually received pharmacologic thromboprophylaxis. Both studies excluded anticoagulated patients from the analysis, but in this study, these patients were included as they can be considered to be “protected” against VTE. Excluding anticoagulated patients from those at risk, 811 of the 1254 (64.7%) of nonanticoagulated patients correctly received pharmacological thromboprophylaxis, above the South American average.
On a global level, the meta-analysis by Forgo 43 included 137 288 medical patients from 20 countries, of whom 50.5% had an indication for thromboprophylaxis and 54% actually received it (excluding anticoagulated patients). This represents a moderate improvement compared with the 2008 ENDORSE study, in which 42% of medical patients were at risk of VTE, but only 40% received thromboprophylaxis. 39 Our study, after excluding anticoagulated medical patients, shows a compliance rate of 76.2%, above the global average. The meta-analysis by Forgo et al showed considerable geographical differences, with an average compliance rate of 68.8% in North America and 66.8% in Europe, compared with 37% in Asia, 44.9% in Africa, and 58% in Latin America. Our study shows higher compliance rates to those found in North America and Europe.
It is important to note that the population at high risk of VTE includes a proportion of patients with contraindications to pharmacological thromboprophylaxis, 15.9% of the population (9% of medical patients, 28.6% of surgical patients, and 10.5% of pregnant-postpartum patients). For these patients, we did not ask whether they used any form of mechanical thromboprophylaxis, but we know that the availability of intermittent pneumatic compression devices is low. In the study by Martinez et al, which looked at this issue, there were no reported cases of patients using intermittent pneumatic compression devices when they had a contraindication to pharmacological thromboprophylaxis. 25
We believe that much remains to be done to ensure that intermittent pneumatic compression devices are made available to patients who may need them and that they are correctly prescribed.
When analyzed by patient type, compliance is higher in medical patients (66.7%) compared to surgical patients (46.6%) and pregnant˗postpartum patients (29%). When analyzed according to moderate or high risk of VTE, compliance improves in surgical and pregnant˗postpartum high-risk patients. In the high-risk surgical patients, this improvement in compliance is statistically significant and in the pregnant˗postpartum patients, it is almost statistically significant (P = .052), possibly due to the limited number of patients in this group.
An important fact to consider, common to all the multicenter studies carried out in our country, is the greater compliance with the indication for pharmacological thromboprophylaxis in medical patients compared to surgical patients. This differs from the experience reported in the ENDORSE study or in regional studies on the subject.30,33,39,42 An explanation could be the exclusion of orthopedic patients in the studies in our country since orthopedic patients are a population in which systematic use of thromboprophylaxis has been achieved.
The low adherence to thromboprophylaxis in pregnant-postpartum patients, especially those at intermediate risk of VTE, is in line with the results of a recent study in post-cesarean section patients. 44 This is a point in which national policies will require review and promotion.
In contrast, 23.9% (128/535) of the patients with no indication of pharmacological thromboprophylaxis were receiving it. This phenomenon was more frequently seen among medical patients 29.3%, followed by 23.7% of surgical patients and 7.6% of pregnant˗postpartum patients. Also, 26 patients with a formal contraindication for thromboprophylaxis were receiving it. This highlights the importance of applying thromboprophylaxis guidelines to correctly evaluate the prescription of thromboprophylaxis to avoid overprescription.
In our study, in 1349 of the total 1925 cases included, 70.1% prescribers correctly complied with international thromboprophylaxis guidelines—CHEST guidelines.1,45 The cases included in this percentage are cases in which doctors correctly prescribe thromboprophylaxis to patients with an indication (high risk of VTE) and do not prescribe it to patients with low risk. The correct indication of thromboprophylaxis as defined above is based on the use of “the Padua or Caprini scores and the RCOG recommendations.”1,17–20
A limitation of our study is the bias that may result from the fact that the treating physician could participate in the data collection. However, this was a key factor in allowing simultaneous data collection at the national level.
All current international guidelines recommend pharmacological or mechanical prevention of VTE in hospitalized patients at risk.1,20,45,46 There are no arguments against the cost-effectiveness of this approach, which has been repeatedly shown to be less expensive than the treatment of this serious complication with high mortality and morbidity. 47
In conclusion, this study analyzed data from 26% of hospitalized patients in Uruguay. It adequately represents the compliance with thromboprophylaxis recommendations in Uruguay. The recommendations of the main thromboprophylaxis guidelines were applied in 70.1% of patients. Despite the progress made in adherence to thromboprophylaxis, nonadherence remains a problem (affecting one in six patients at risk of VTE) and is consistent with published data worldwide.
Footnotes
Acknowledgements
Roberto Gutierrez, Claudina Fajardo Romina Millar, Patricia Gómez, Dary Guillín, Cristian Lemus, Leonardo Stratta, Federico Bonavita, Karen Arias, Ana Buschiazzo, Mariana Gilardoni, Santiago Moyano, María Noel Spangenberg, Sabrina Ranero, Agustina Santa Cruz, Ximena Cuba, Catherin García, María Isabel Da Rosa, Lorena Vargas, Alfonsina Rodríguez, Jota Martínez, Diego Pedraza, Florencia O Neil, Mariana González, Erika Rojas, Marlene Sica, Claudia Solé, Lourdes Casanello, Laura Betancor, Leslye Camejo, Alejandra Sosa, Florencia Lavega, Valentina Tais, Alicia Izaguirre, Patricia Larramendi, Ignacio Vales, Vania Teixeira, Jessica Imbriago, Gabriela Capurro, Joaquín Ferreira, Bruna Cocco, Martín Dorelis, Carolina Ibañez, Matias Lista, Sebastián Maglione, Romina Urrutia, Melanie Isern, Sergio Cáceres, Valentina Moreira, Matías Banega, Federico García, Silvia Monti, Guillermo Fontes, Mikaela Tor, Joaquina Díaz, Andrea Queirolo, Romina Castillo, Fernando Tabares, Leticia Esnal, Marcela La Camera, Analía Cánepa, Diego Fleitas, Noralia Santiago, Mónica Erlich, Andrea Santacruz, Andrea Simonetti, Lucía Pommies, Daniela Refreschini, Johana Ramón, Lorena Nicassio, Verónica Ventura, Ana Bruner, Maia Demasi, Ivette Lacroix, Flavio Sosa, Victoria Eroza, Luciano Romillo, Noemí Sosa, Horacio Sosa, Florencia Rodriguez, Lucía Musto, Rocío Góngora, Marcela Dutra, Sebastián Varona, Valeria Minetti, Yasir Chara, Jeanine Ordoñez, Ignacio Borgia, Stephanie González, Danilo Magallanes, Lorena Silva, Eliana Silveira, Daniel Castro, Lucia Romero, Federico Paradiso, Rogelio Blanc, Analía Aguiar, Valeria Perez, Jessica Montaño, Yamibel Agrinzones, María Isabel Da Rosa, Lorena Vargas, Luis Fuentes, Luciana Nario, Cintia García, Mónica Gutiérrez, Valentín García, Camila Cabrera, Maite Aguirre, Amalia Archondo, Carolina Santos, Marilina Leal, Luis Florio, Jeniffer Brunetto, Micaela Bica, Valentina Olivares, Sofía Aurucci, Adriana Peixoto, Daniella Mussio, Paula Negrín, Silvana de León, Jimena Ravera.
Author Contributions
Valentina Mérola contributed to conception and design of the study, acquisition of data, analysis and interpretation of data, drafting the article, and revising it critically for important intellectual content and final approval of the version to be submitted. Ana Carina Pizzarossa contributed to conception and design of the study, acquisition of data, analysis and interpretation of data, drafting the article, and revising it critically for important intellectual content and final approval of the version to be submitted. Maynés López contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Franco Peverelli contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Gustavo Bruno contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Mariana González contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Federico Roca contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Leticia Dentone contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Gerardo Pérez contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Lucía Da Silveira contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Lilián Díaz contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Josefina Tafuri contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Rosario Cuadro contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Martín Zaquiere contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Guillermina Bartaburu contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Franco Pacello contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Carina Celio contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. María José López contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Marcelo Viana contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Laura Fraga contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Valeria Blanco contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Paula Chalart contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Daniel Leal contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Xilef Rodríguez contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Laura Teti contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Carolina Goñi contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Eduardo Infante contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Alejandra Prícoli contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Victoria Altieri contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Cecilia Guillermo contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted. Rosario Martínez contributed to conception and design of the study, acquisition of data, and revising it critically for important intellectual content and final approval of the version to be submitted.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
