Abstract
Objective:
The objective of this study is to evaluate the effect of autoimmune disease on the risk of venous thromboembolism (VTE) including deep vein thrombosis (DVT) and pulmonary embolism (PE) in pregnant women.
Methods:
Using the Health Care Cost and Utilization Project, Nationwide Inpatient Sample database from 2003 to 2011, the risk of developing DVT, PE, and VTE among pregnant patients with selected autoimmune diseases was estimated using unconditional logistic regression analysis.
Results:
Our study cohort consisted of 7 917 453 women of which 43 523 had underlying autoimmune diseases. Risk of VTE was high in pregnant women with systemic lupus erythematosus, dermatomyositis, rheumatoid arthritis, type 1 diabetes mellitus, ulcerative colitis, and Crohn’s disease.
Conclusion:
Most autoimmune diseases considerably increase the risk of VTE. Thromboprophylaxis may be considered in pregnancies with autoimmune disease, particularly those with systemic lupus erythematosus and dermatomyositis.
Introduction
Venous thromboembolism (VTE) is considered the primary cause of maternal morbidity and mortality in the United States, accounting for 9% of all maternal deaths. 1,2 In fact, the United States Surgeon General has declared pulmonary embolism (PE) to be the most preventable cause of death among hospitalized patients. 3 Apart from short-term mortality, VTE is associated with long-term morbidity and decreased quality of life secondary to postthrombotic syndrome. 3
A review of recent literature has shown that autoimmune diseases and immune-related disorders, both systemic and localized, are associated with a higher risk of VTE. 4 –6 Risk of thrombosis among pregnant women with antiphospholipid syndrome and systemic lupus erythematosus (SLE) has been documented and an increased risk of thrombosis in pregnant women with inflammatory bowel disease (IBD) has even been suggested. There is however little information on thrombosis risk in pregnancy in women with other autoimmune disorders. 7,8 Recognizing an increased risk of thromboembolic events in pregnant patients with autoimmune disorders could be instrumental in identifying subgroups of women who may benefit from ante- and postpartum thromboprophylaxis.
Materials and Methods
We used data obtained from the Health Care Cost and Utilization Project, Nationwide Inpatient Sample (HCUP-NIS) to carry out this retrospective population-based cohort study. The HCUP-NIS database is the largest all-payer health care database and includes demographic and clinical information on patients hospitalized in the United States, approximating 20% of all United States community hospital admissions. 9 This database classifies information used according to the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM).
We created a cohort of all delivery-related admissions from 2003 to 2011 by identifying patients with ICD-9 discharge delivery codes (V27 or 650-669). Patients were then classified as having or not having autoimmune diseases, categorized as being either systemic (labeled as “systemic disease”) or organ specific (labeled as “localized disease”). 10 Our exposed group was identified using ICD-9-CM codes 710.0, 710.2, 710.1, 710.3, 710.4, and 714.x for the chosen systemic autoimmune diseases, SLE, Sjögren syndrome, systemic sclerosis, dermatomyositis, polymyositis, and rheumatoid arthritis, respectively. Codes 571.42, 250 fifth digit 1, 579.0, 556.x, and 555.x were used to identify our chosen localized autoimmune diseases, specifically, autoimmune hepatitis, type 1 diabetes mellitus, celiac disease, ulcerative colitis, and Crohn’s disease, respectively. Prevalence of disease per 10 000 deliveries was calculated among our cohort.
Our primary end point was defined as VTE, a diagnosis of deep vein thrombosis (DVT), PE, or both using ICD-9 codes 453.4, 671.3, 671.4, 415.1, and 673.2. Crude and adjusted odds ratios (ORs) were calculated using unconditional logistic regression analysis with their respective 95% confidence intervals (CIs). Odds ratios were adjusted for age. We considered P values under .05 to be statistically significant. Our study was approved by the Medical Research Ethics Department of the Jewish General Hospital. All analyses were performed using the statistical software SAS V9.2 (SAS Institute, Cary, North Carolina).
Results
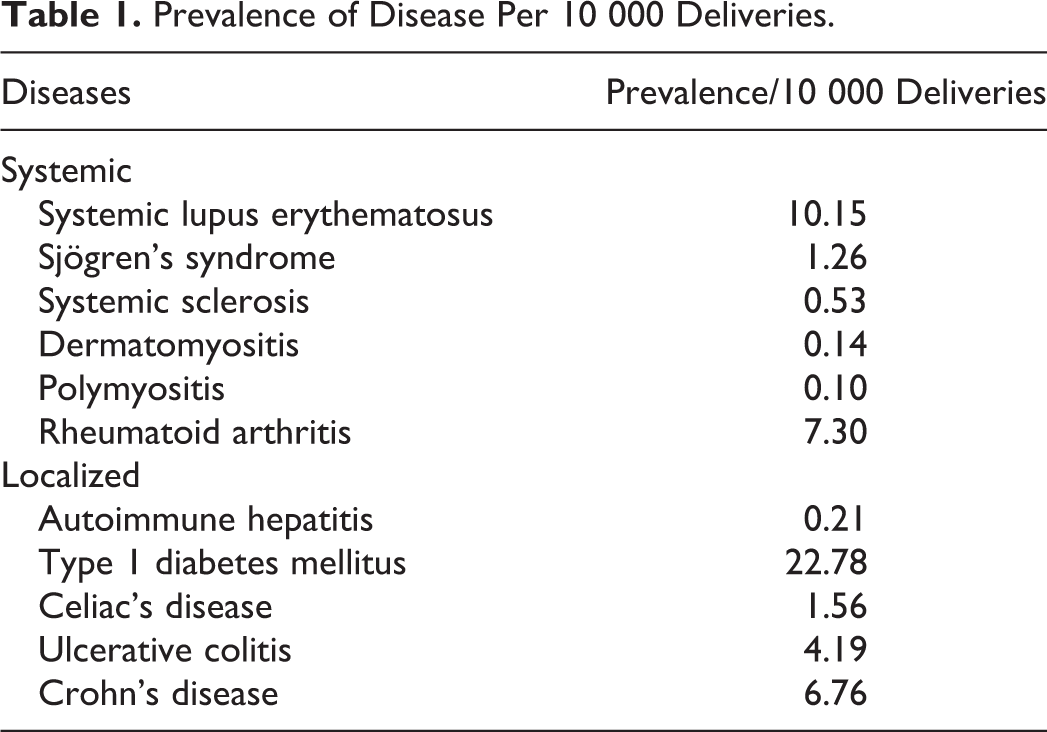
We analyzed data on 7 917 453 pregnant women over the 8-year study period. We identified 8040 pregnant women with SLE, 994 with Sjögren syndrome, 420 with systemic sclerosis, 107 with dermatomyositis, 80 with polymyositis, 5780 with rheumatoid arthritis, 163 with autoimmune hepatitis, 18 033 with type 1 diabetes mellitus, 1235 with celiac disease, 3319 with ulcerative colitis, and 5352 with Crohn’s disease (Table 1 and Figure 1).
Prevalence of Disease Per 10 000 Deliveries.
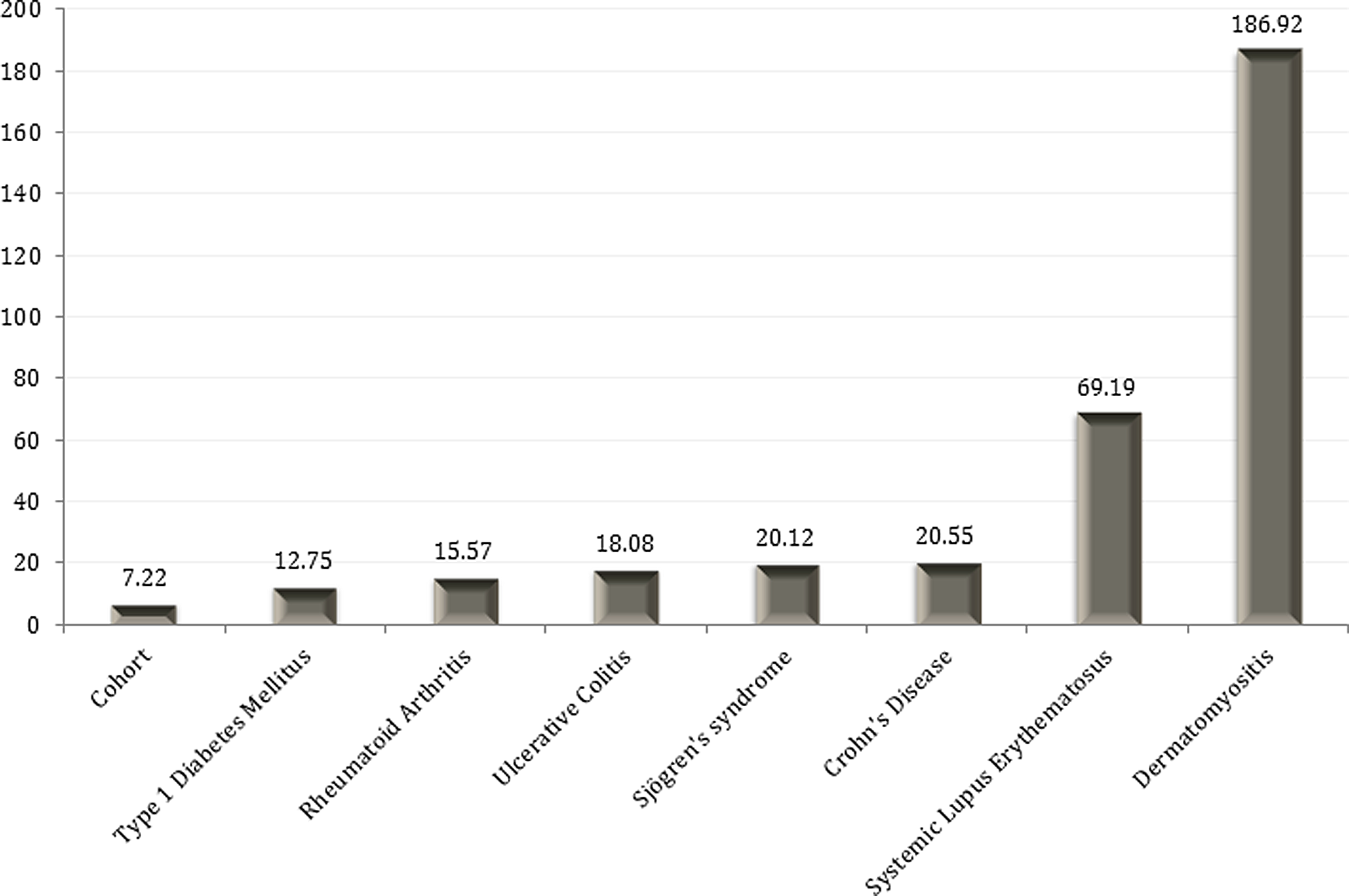
Incidence of venous thromboembolism (VTE) per 10 000 deliveries according to different autoimmune diseases.
The risk of DVT in our cohort of pregnant patients was 5.34/10 000 with a considerably higher risk among pregnant patients with the following autoimmune diseases: SLE (42 of 10 000), dermatomyositis (93 of 10 000), ulcerative colitis (18 of 10 000), and Crohn’s disease (19 of 10 000); Table 2). We noted statistically significant increases in PE events for pregnant patients with SLE (OR 9.76, 95% CI [6.13-15.55]), dermatomyositis (OR 39.71, 95% CI [5.53-284.95]), and rheumatoid arthritis (OR 3.62, 95% CI [1.50-8.70]; Table 3). The risk of PE in our cohort was 2.19 of 10 000. As for the incidence of VTE, the risk was significantly increased in women with SLE (62 of 10 000), dermatomyositis (187 of 10 000), rheumatoid arthritis (16 of 10 000), type 1 diabetes mellitus (13 of 10 000), ulcerative colitis (18 of 10 000), and Crohn’s disease (21 of 10 000; Table 4). In comparison, the risk of VTE in our general cohort was 7.22 of 10 000.
Risk of DVT in Patients With Autoimmune Diseases.
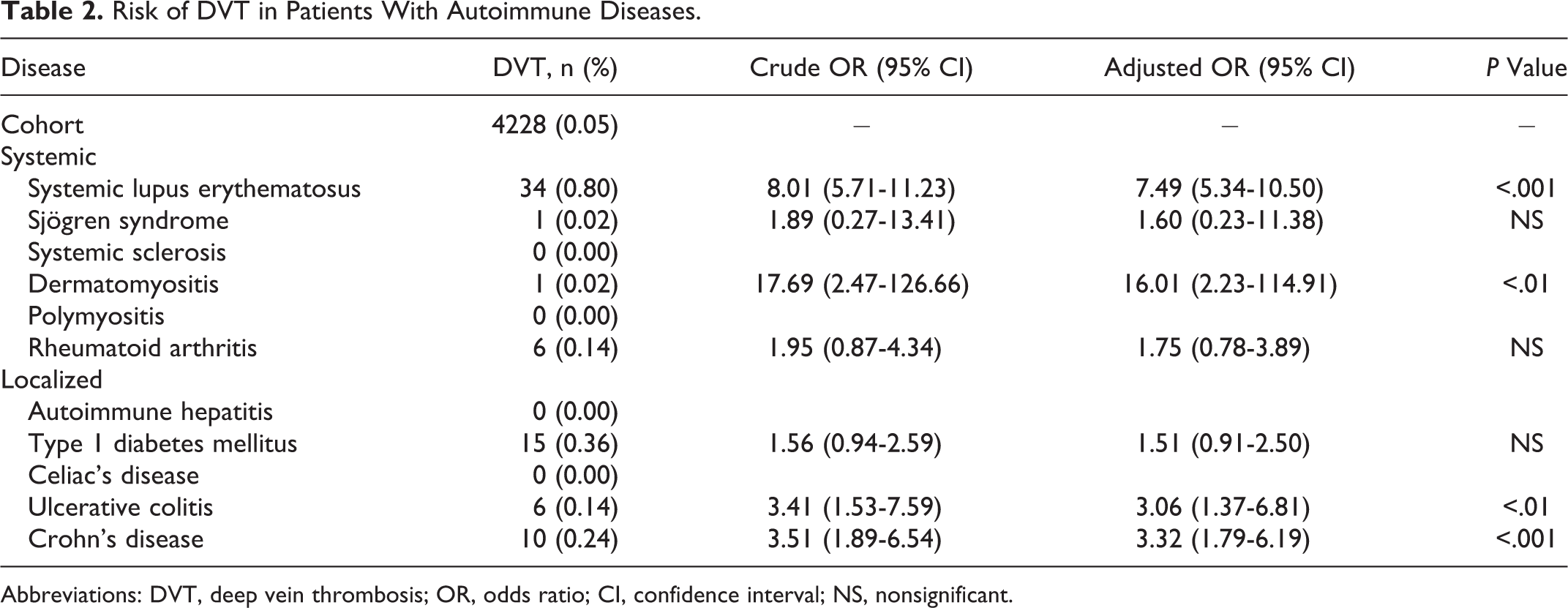
Abbreviations: DVT, deep vein thrombosis; OR, odds ratio; CI, confidence interval; NS, nonsignificant.
Risk of PE in Patients With Autoimmune Diseases.
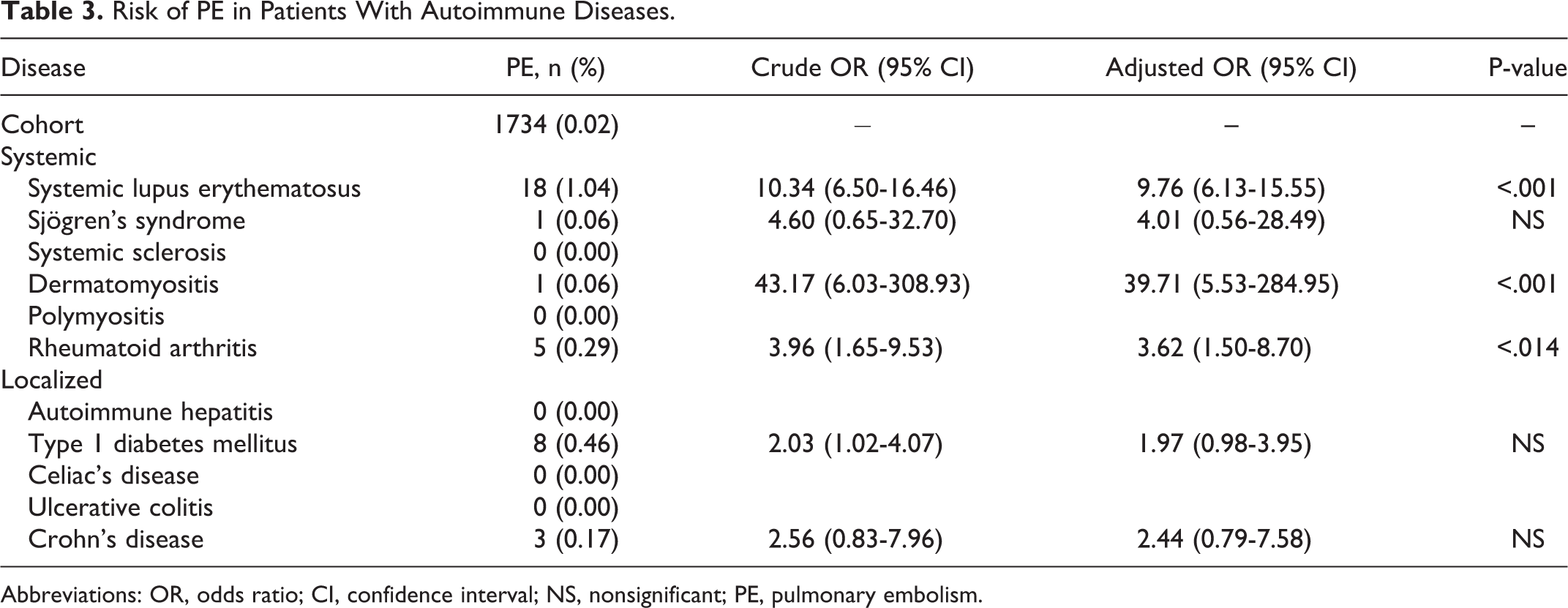
Abbreviations: OR, odds ratio; CI, confidence interval; NS, nonsignificant; PE, pulmonary embolism.
Risk of VTE in Patients With Autoimmune Diseases.
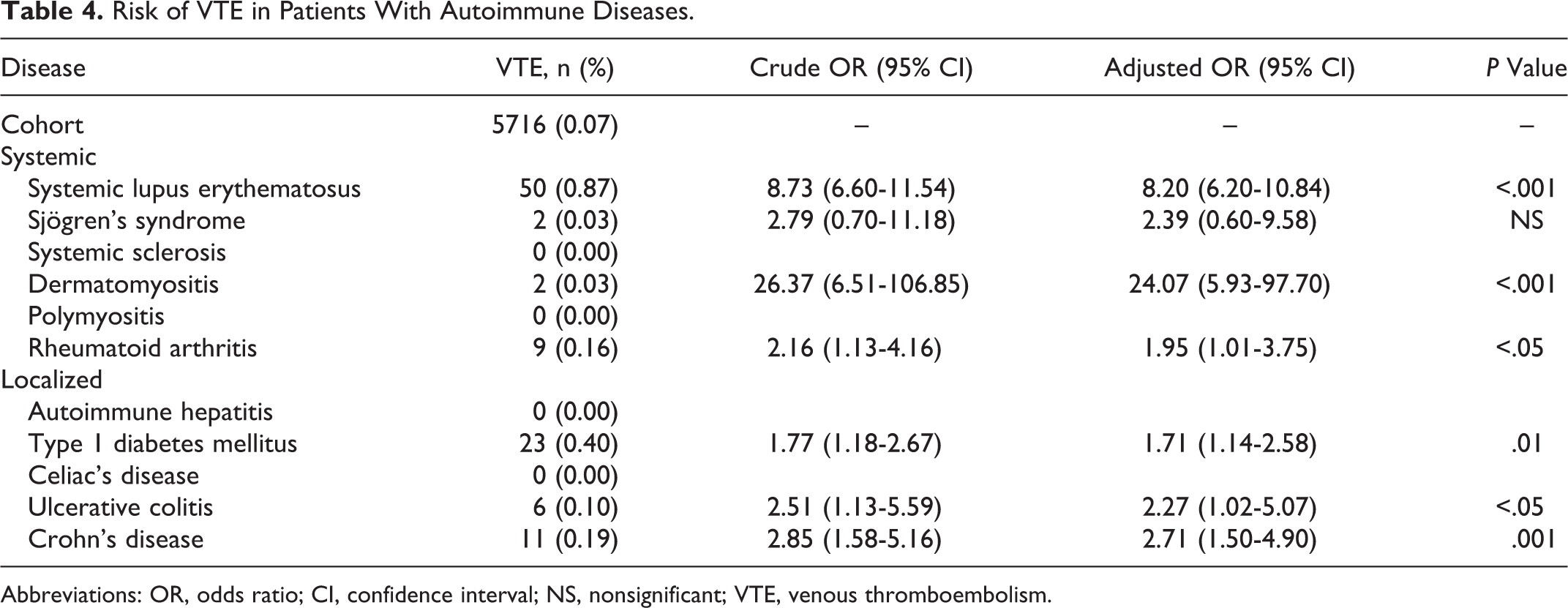
Abbreviations: OR, odds ratio; CI, confidence interval; NS, nonsignificant; VTE, venous thromboembolism.
Discussion
The risk of VTE was found to be significantly increased for many of the systematic and localized autoimmune diseases. The risk of VTE was greatest for patients with pregnancies complicated by dermatomyositis, followed by patients with SLE, Crohn’s disease, ulcerative colitis, rheumatoid arthritis, and type 1 diabetes mellitus.
Given the rarity of autoimmune diseases in pregnancy and the low frequency of thrombotic episodes, large administrative databases are required to have sufficient power to detect clinically relevant associations. One of the main limitations inherent in the use of an administrative database is coding accuracy. We used codes for VTE that were both pregnancy specific and general similar to a study by Abbasi et al. 11 Findings from a previous study found that ICD-9 pregnancy-specific codes for VTE in pregnancy identified only 31% of VTEs, whereas standard codes for VTE identified 80% of VTEs. 11 The validity of ICD-9 coding has been assessed for many autoimmune diseases. Moores and Sathe reviewed methods for identifying patients with SLE in the general population using ICD-9 code 710.0 and found a sensitivity and specificity of 98.2% and 72.5%, respectively. 12 Liu et al used ICD-9 codes 555.x and 556.x to identify patients with IBD and calculated a positive predictive value of 81%. 13 A systematic review for the validation of coding for diabetes was done by Chen et al and found sensitivities ranging from 91.2% to 95.6% and specificities ranging from 92.8% to 97.6%, although they did not differentiate between type 1 and type 2 diabetes mellitus. 14 Chung et al did a similar review for patients with rheumatoid arthritis and found positive predictive values ranging from 34% to 97% with the highest positive predictive value in studies limited to rheumatology clinics. 15 Unfortunately, no ICD-9 coding differences exist between history of disease and flare of disease, and as such these two entities could not be differentiated in our study. Furthermore, not all diseases such as Graves’ and Addison’s disease are coded individually and therefore were not included in this study.
Cohort entry for our study included women with a delivery diagnosis to avoid overrepresentation of disease incidence, given that these women are more likely to have multiple admissions in pregnancy but only one admission for the actual birth. As a result, the true incidence of disease in pregnancy may be underestimated, as only cases in which the diagnosis was coded for in the delivery admission were included in the analysis. Similarly, the outcome was only coded if present during the delivery admission and this may overlook some patients diagnosed or treated as outpatients during periods outside their delivery admission. As such, it was not possible to take into account all events occurring in the critical 6-week postpartum period subsequent to discharge from hospital after delivery. Although these factors may in part compromise the true point estimate, this approach has successfully been used in different contexts with comparable results to the established literature. 11,16 –18 We only adjusted for age in our analysis, as other potential confounders such as previous DVT, obesity, mode of delivery, postpartum hemorrhage, and known thrombophilia may be elements in the causal pathway and as such not variables that should be adjusted for. 19 –21 As well, it may have been interesting and informative to compare outcomes of women having received anticoagulation therapy with those who did not. Unfortunately, although ICD-9 codes do exist for this entity, it is impossible to determine whether the administration would have been as prophylaxis or as treatment (before or after the event occurred). Furthermore, much of the thromboprophylaxis used in pregnancy is stopped before admission for labor and, as such, coding for them will likely not have occurred in the delivery admission. 22
We found the risk of DVT, PE, and VTE to be significantly increased in our cohort of patients with SLE. In fact, this finding was expected as many studies have shown an increased risk of thrombosis in these patients outside pregnancy. 23 Additionally, a review of the literature by Zoller et al reported that thrombosis occurs in 10% to 26% of patients with SLE. 4 Current recommendations for SLE in pregnancy are based on empirical data and suggest low-dose aspirin therapy during pregnancy and low-molecular-weight heparin (LMWH) postpartum. 24
As for patients with Sjögren syndrome, we found an increased risk of DVT, PE, and VTE although nonstatistically significant. There is currently limited literature on thrombosis risk in patients with this disease. An Italian study of 36 patients with Sjögren syndrome which resulted in 45 pregnancies and 40 deliveries looked at variables influencing pregnancy and fetal outcome. Less than half of patients received some form of treatment during their pregnancy, including 11% of who received LMWH and 13% of who received low-dose aspirin. No differences were observed in outcome between the groups of patients, although risk of VTE was not studied. 25 Although it may be possible that an increased risk actually exists, our data do not allow us to draw any conclusions.
We did not identify any cases of DVT, PE, or VTE among pregnant women with systemic sclerosis. A possible explanation could be that we identified very few patients with this disease (n = 420). As well, this diagnosis is often made after childbearing age, and until recently, pregnancy was not recommended for these patients, as many case report series published in the 1970s and 1980s demonstrated an extremely high-maternal mortality rate. 26 –31
With regard to pregnancies complicated by dermatomyositis, we found a 16-fold increased risk of DVT, a 39-fold increased risk of PE, and 24-fold increased risk of VTE, all of which were statistically significant. Few studies have been done on dermatomyositis in pregnancy, and none of those have looked at VTE as a main outcome. 32 –34 A large Swedish cohort study by Zoller et al found an increased risk of PE in hospitalized nonpregnant patients with dermatomyositis without prior history of VTE. 6 The risk of VTE found in our study outweighs the risk of most current indications for thromboprophylaxis. 1,35 As such, we suggest that thromboprophylaxis be considered in cases of dermatomyositis.
No events were identified for pregnant patients with polymyositis and autoimmune hepatitis. Given these diseases are extremely rare in pregnancy, the number of cases included in our study was low. Further research is needed to examine whether or not an increased risk of thromboembolism exists among this patient population.
As for pregnancies complicated by rheumatoid arthritis, we found an increased risk of DVT (nonstatistically significant), PE, and VTE (statistically significant). This finding is not unexpected, given an increased risk has also been reported outside pregnancy. 4 Many studies have reported that rheumatoid arthritis tends to go into remission during pregnancy but flares up in the postpartum period. 7 Including readmissions in our study may have strengthened this association.
As for pregnancies complicated by type 1 diabetes mellitus, we found an increased risk of DVT (nonstatistically significant), PE (nonstatistically significant), and VTE (statistically significant). Three studies have demonstrated an increased risk of VTE outside pregnancy. 4,5,36 In a large retrospective cohort study, Won et al failed to find an association between gestational diabetes and VTE risk. 37 However, as opposed to type 1 diabetes mellitus, gestational diabetes mellitus is not generally considered an autoimmune disease. 38
No events were identified in patients with celiac disease. This could be explained by the fact that if these patients were well controlled by their diet, their circulating autoantibody count greatly decreases, perhaps decreasing the effect of autoimmunity and inflammation. 39 Our study therefore does not allow us to draw any conclusions for the thrombotic risk with this disease.
Finally, we looked at risk associated with pregnancies complicated by inflammatory bowel disease (IBD), ulcerative colitis and Crohn’s disease. Studies in nonpregnant patients have demonstrated an increased VTE in patients with IBD, especially when hospitalized and with active disease. 4 A study of IBD in antenatal and postnatal patients using the NIS database also found an increased risk of VTE events. 8 This correlates with our findings of increased VTE risk for these patients in pregnancy.
Two studies have estimated the risk of venous thrombosis and PE to be 4 to 6 times higher during pregnancy. 40,41 This can be explained by physiological changes that occur during pregnancy. Well known is the Virchow triad consisting of vascular stasis, endothelial trauma, and hypercoagulability, all of which are affected by pregnancy. In fact, decreased venous return from the lower extremities secondary to compression of pelvic veins and inferior vena cava by the gravid uterus, decreased mobilization, endothelial cell injury from delivery, and increased synthesis of clotting factors during pregnancy are some of the many mechanisms that have been implicated in this cascade. 42 When we take into account the additional risk that autoimmunity seems to bring to this equation, this risk seems amplified. In fact, of the autoimmune diseases that we studied, dermatomyositis, followed by SLE, was associated with the highest increase in risk of venous thrombotic event. Not only were systemic autoimmune diseases identified as being at higher risk of VTE but organ-specific disease as well. Type 1 diabetes mellitus, ulcerative colitis, and Crohn’s disease have all been shown to increase this risk but to a lesser degree. A possible explanation can be found in a study by Zoller et al, in which it is theorized that all autoimmunity increases inflammation, shifting normal homeostasis in the direction of thrombogenicity. 4
The American College of Obstetricians and Gynecologists (ACOG) has issued recommendations that include few indications to consider or order thromboprophylaxis with LMWH in pregnancy. These are limited to patients undergoing cesarean section with additional risk factors; including obesity, patients with a history of thrombosis with low-risk thrombophilias or patients with a history of thrombosis related to a transient risk factor that is either estrogen related or pregnancy related, patients with a prior history of idiopathic thrombosis, patients with a history of 2 or more events, and patients with significantly high risk of acquired or inherited thrombophilias. 1,43,44 The ACOG also recommends the use of pneumatic compressive devices placed preoperatively in all patients undergoing a cesarean section, the risk of venous thrombosis stated as being twice as elevated. 1 Therapeutic anticoagulation is recommended for patients with a thromboembolic event that occurred during the present pregnancy or in patients with mechanical heart valves. Although thromboprophylaxis ante- and postpartum should remain individualized, our results suggest that thromboprophylaxis also be considered in pregnant women with autoimmune disease, as the risk is comparable or greater than those of other indications. Consideration should be given to patients with dermatomyositis and systemic lupus erythematosus.
Our results suggest that many autoimmune diseases are associated with higher risk of venous thrombosis. As such, we believe that autoimmunity should be considered a risk factor for VTE and that appropriate thromboprophylaxis may be considered.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was obtained from our institution.
