Abstract
This study aimed to investigate the influence of prothrombotic risk factors on long-term outcomes of patients with perinatal arterial ischemic stroke. The study was conducted through an analysis of monitoring results that were regularly maintained for approximately 20 years at a tertiary stroke-monitoring center. The study assessed prothrombotic risk factors, radiological area of involvement, clinical presentation, treatments, clinical outcomes, and long-term outcomes of the 48 patients included in the study, with a mean monitoring time of 77.6 ± 45.7 months (range: 6-204). Our results showed that the presence of prothrombotic risk factors did not affect long-term outcomes. However, patients with middle cerebral artery infarction had the highest risk of developing cerebral palsy, whereas those with presumed stroke had the highest risk of developing epilepsy. This study suggests that prothrombotic risk factors should not be evaluated during the acute stage unless there is a strong suspicion of the patient's history, and prevention or early diagnosis of presumed stroke patients will positively impact their long-term prognosis.
Introduction
Perinatal ischemic stroke occurs once every 2300-5000 live births. Although the neonatal period corresponds to a very short part of life, strokes occur in this period at rates 10 times higher than in childhood.1–3 Despite this, the rate of mortality and recurrence in perinatal strokes is low.4,5 The important consequences of perinatal strokes are cerebral palsy and epilepsy, which are seen in long-term monitoring and affect the quality of life.1,6,7 Prothrombotic risk factors play a role in the etiology of stroke.8,9 The important question is whether the presence of hereditary thrombophilias in perinatal strokes poses a risk for the development of epilepsy and cerebral palsy in the long term.10,11 It is also important to determine which prothrombotic risk factor is more effective in perinatal stroke. This is because looking at all hereditary thrombophilias in a neonatal patient is not possible considering the total amount of blood in the patient's body.
Previous investigations have focused on radiological predictors of the long-term implications and prognosis of individuals who have experienced perinatal strokes. 12 This report highlights the fact that although the majority of patients with perinatal stroke who develop cerebral palsy are linked to the middle cerebral artery (MCA), there is no clear demarcation of a specific region for epilepsy. 13 Therefore, it is crucial to emphasize the need for additional research in this domain.
Although there are kinds of literature on perinatal stroke, the monitoring periods that demonstrate long-term prognosis are brief.1–8 At our center, the 20-year monitoring processes of pediatric stroke patients are regularly recorded. The objective of our study was to explore the necessity of screening for hereditary thrombophilias in neonatal stroke patients by assessing their long-term prognosis and patient outcomes.
Methods
This study was conducted retrospectively on the data of perinatal arterial ischemic stroke patients without hemorrhagic stroke over the age of 2 years who were monitored at the Faculty of Medicine at Ankara University between January 2002 and November 2022. Patients with (1) cerebral sinovenous thrombosis, (2) hemorrhagic stroke, (3) cardiac-related strokes, (4) presence of hypoxia, (5) born earlier than 36 weeks, and (6) bilateral stroke were excluded. Patients under the age of 2 years were excluded because of the lack of monitoring time to determine long-term prognosis. Among the 74 patients who were monitored, we excluded 10 with cerebral sinovenous thrombosis, 6 with hemorrhagic arterial ischemic stroke, and 10 who were younger than 2 years of age, had cardiac-related strokes, or were not monitored regularly. Thus, 48 patients with perinatal ischemic stroke were included in this study. Patients were monitored at the outpatient clinic at intervals of 3 or 6 months. Cerebral palsy was diagnosed in patients over the age of 3 years based on reports from the Physical Medicine and Rehabilitation Department. There were 42 patients aged > 3 years. The cranial MRIs, MR angiography, and EEGs of all patients were obtained from the patient record system. We also recorded the treatments that the patients were administered during the acute period and whether or not the patient received prophylaxis.
Ankara University is a national tertiary stroke center, and the following tests are routinely performed on all patients with stroke with family consent. All patients underwent echocardiography and hematological investigations to analyze prothrombin time and partial thromboplastin time; fibrinogen, protein C, protein S, antithrombin III, lipoprotein A, factor VIIIC (FVIII), factor IX (FIX), and homocysteine levels; and the presence of prothrombotic gene mutations (factor V G1691A [FV G1691A], prothrombin [PT G20210A], and methyl tetrahydrofolate reductase C677T [MTHFR C677T]). Blood samples obtained via peripheral venipuncture were collected into plastic tubes without any additives and into plastic tubes containing 1/10 volume of 3.8% trisodium citrate. After centrifugation, the samples were stored at 70 °C. For genetic analysis, venous blood was collected in tubes containing EDTA. The genotypes of FV G1691A, PT G20210A, and MTHFR C677T were determined via polymerase chain reaction and restriction fragment analysis, as previously reported. 14 The study protocol was approved by the Ankara University Ethics Committee, and written informed consent was obtained from the parents of each patient prior to performing any procedures.
The levels of FVIII and FIX were measured using a 1-stage clotting assay, and the von Willebrand factor level was measured using an immunoturbidimetric assay. FVIII and FIX levels were accepted as high if they were above the cutoff value of 150 IU/dL. Protein C and S levels were measured using commercially available enzyme-linked immunosorbent assay kits. Lipoprotein (a) levels were measured using a particle-enhanced immunonephelometric assay (reference range:0-30 ng/mL). Following 12-h of fasting, blood samples were collected into tubes containing EDTA, and homocysteine levels were measured using an AxSYM homocysteine assay (Abbott, Wiesbaden, Germany).
Statistical Analysis
The statistical evaluation of the study was conducted blindly by members of the Department of Statistics, Faculty of Medicine, Ankara University. Statistical analyses were performed using software (Version 20 SSPSS Inc. Chicago, IL, USA) for Windows. Variables are reported as mean and standard deviation or as median, as appropriate. The association between long-term prognosis indicators (cerebral palsy, epilepsy) and both positive prothrombotic risk factors (protein C, protein S, ATIII, homocysteine, factor V-Leiden, MTHFR, prothrombin) and infarction localization (anterior cerebral artery [ACA], MCA, posterior cerebral artery [PCA], and Watershed) was assessed using binary logistic regression analysis. The relative risk (RR) with 95% confidence interval (CI) was calculated. Statistical significance was set to indicate statistical significance.
Results
Among the participants, 32 (66.7%) were female, and 16 (33.3%) male. The mean age of the patients included in the study was 6.9 ± 3.7 years (range: 2-18). perinatal ischemic strokes, 27 patients (56.3%) had neonatal ischemic stroke and 21 (43.8%) had presumed perinatal ischemic stroke (Table 1). The clinical and demographic characteristics of the patients are shown in Table 1. Maternal, fetal, and placental risk factors and family history have been added as Supplemental Tables. No placental risk factors were identified in this study.
The Demographic and Clinical Characteristics of the Study Population.
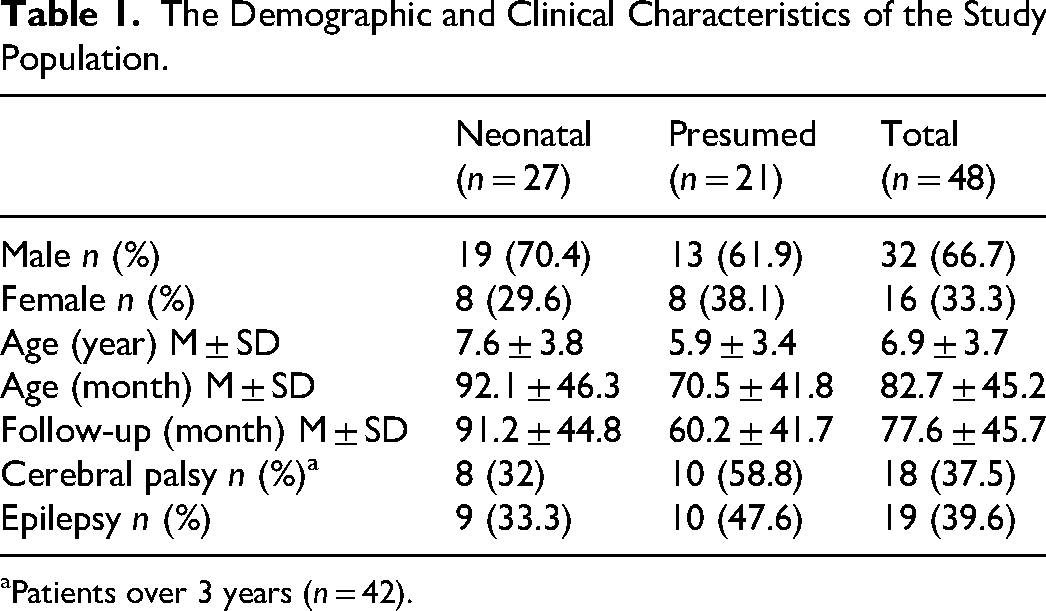
aPatients over 3 years (n = 42).
The acute treatment criterion of our department was the treatment of patients with cardiac stroke or embolism found in more than two blood vessel perfusion territories. As cardiac stroke patients were excluded, none of the patients received acute treatment. No recurrence was observed in any patient. Patients who died were excluded from the study. Forty-two patients were over the age of three, and 18 (42.8%) were being monitored with the diagnosis of cerebral palsy. Nineteen (36.9%) were monitored for epilepsy. The clinical findings of the study population at the time of admission are shown in Table 2.
The Clinical Findings of the Study Population at the Time of Admission.

At least one prothrombotic risk factor was found in 34 (70.8%) of the 48 patients. The distribution of positive hereditary thrombophilia in relation to presumed and neonatal strokes is illustrated in Table 3.
Positive Prothrombotic Factors in the Study Population.
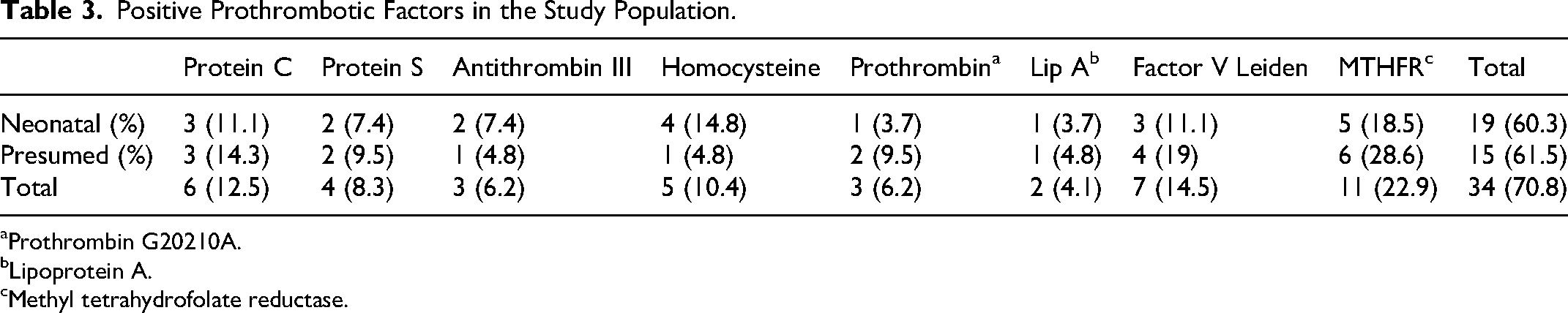
aProthrombin G20210A.
bLipoprotein A.
cMethyl tetrahydrofolate reductase.
Risk analysis was carried out by binary logistic regression analysis between whether or not cerebral palsy and epilepsy developed in patients and prothrombotic risk factors that were found positive (protein C, protein S, ATIII, homocysteine, factor V-Leiden, MTHFR, prothrombin). None had a significant relationship with cerebral palsy or epilepsy development (P > .05) (data not shown).
According to the MRI findings of the 48 patients, it was determined that 20 (41.6%) had MCA infarction (MCA), 8 (16.6%) had ACA, 4 (8.3%) had PCA, 7 (14.7%) had watershed infarction areas, and 9 (18.7%) had infarction areas that did not correspond to any perfusion territories. Table 4 displays a representation of the risk assessment for the studied population, considering both the infarction region and stroke type, and specifically examines the relationship between stroke location and the likelihood of long-term complications.
Risk Analysis of the Study Population According to Infarct Area and Stroke Type.

aMiddle cerebral artery.
bAnterior cerebral artery.
cPosterior cerebral artery.
Discussion
In the monitoring of patients who had perinatal stroke, epilepsy, motor function, language, and cognition disorders may be observed in the long term.5,6,10–17 Cerebral palsy is the most frequently encountered motor function disorder.17,18 Our study found a cerebral palsy prevalence in children with neonatal stroke over the age of 3 of 42.8%) (Table 4). Almost all of our patients had a unilateral stroke. The literature reported the rate of cerebral palsy in patients with unilateral stroke to be 30%.1,12 The rate we found in our study appears to be higher than that reported in the literature. In our opinion, the reason for this was that the number of presumed stroke patients who carry a greater risk of cerebral palsy was high in our sample. Cerebral palsy was seen most frequently in patients who corresponded to the MCA perfusion territory (64.7%, RR: 2.4) and perinatal stroke patients who presented late (58.8%, RR: 1.9).
The risk of later development of epilepsy in neonatal stroke patients is between 10% and 40%.12,13,19,20 This rate may increase to 67% in presumed stroke patients.13,19–23 In our study, the rate of epilepsy development in our patients was 39.6%. The risk increased 5-fold in patients with PCA infarction and 2-fold in those with watershed infarction (Table 4). Although 47.7% of late-presenting stroke patients visited with seizures, the proportion of patients who were diagnosed with epilepsy during the monitoring period was 52.6%. However, while 44.4% of stroke patients in the neonatal period visited with seizures, 33.3% of these patients were diagnosed with epilepsy in their monitoring periods of at least 2 years. Our data suggest that epilepsy development rates would be higher in stroke patients corresponding to the PCA territory (75%, RR: 5) and late-presenting stroke patients (52.6%, RR: 1.3).
Our research, in conjunction with previous studies, reveals that the prognosis for late-onset perinatal stroke is less favorable than that of neonatal arterial stroke over the long term. 22 The rates of epilepsy and cerebral palsy in the presumed stroke patients were higher.6,23–25 We believe that the main factors that affected the outcomes of prognosis were patients who were asymptomatic in the newborn period and were discovered later by various neurological signs.
Pregnancy is defined as a natural prothrombotic state. The purpose of this is to protect the mother from fatal hemorrhage complications.26,27 Therefore, strokes that occur in the neonatal period are noticeably more prevalent than other childhood strokes. Likewise, while some prothrombotic factors were high, others were low. Protein C, protein S, and ATIII levels are especially low in the neonatal period, and they only reach normal values at 1 year of age.26,27 Studies have demonstrated that prothrombotic risk factors do not increase the risk of thrombophilia in perinatal stroke patients.10,28,29 Although some studies have reported a high incidence of hereditary thrombophilias in presumed perinatal stroke, 30 which may be associated with cerebral palsy, this has not been confirmed in subsequent comprehensive studies. 11
Our study showed that the presence of prothrombotic risk factors did not increase the risk of epilepsy or cerebral palsy in long-term prognosis. Considering the body mass and amount of blood in newborns, we believe that monitoring prothrombotic factors in the early period will not change the treatment plan or long-term diagnosis. The fact that the neonatal hemostatic system differs from adult and childhood suggests that prothrombotic tests should be performed gradually. 31 Saxonhouse et al stated that it was appropriate to check prothrombin, F V-Leiden, lipoprotein A, protein S, protein C, homocysteine, and ATIII in the first stage and the other prothrombotic factors in the second stage when the child was 3-6 months old. 32 We also believe that it is correct to control prothrombotic factors gradually.
One of the most significant limitations of our study was its retrospective design. Other important limitations of the study include the small sample size and inability to provide data on the mental states of the children, as not all necessary tests can be performed on all patients (for example, IQ). Nevertheless, our mean monitoring experience of 6.5 years in perinatal ischemic stroke patients is valuable in terms of showing the long-term prognosis of patients. Our unit is one of the reference centers for childhood stroke in Turkey, which is why the data are regularly monitored.
Our study showed that in perinatal ischemic strokes, the risk of cerebral palsy in late-onset patients or those with infarctions corresponding to the MCA perfusion territory increased, whereas the risk of epilepsy increased in patients with infarctions corresponding to the PCA perfusion territory and watershed infarctions. In addition, we believe that the presence of prothrombotic risk factors does not pose a risk for the development of epilepsy and cerebral palsy in long-term monitoring, and it is not essential to check for prothrombotic risk factors in the acute period unless the history suggests a strong suspicion. Our study provides evidence that supports the notion that individuals with presumed perinatal stroke are at a greater risk of developing long-term complications. Given that timely intervention to address these complications can lead to improved outcomes, it is crucial to identify patients who have experienced presumed stroke at an early stage, as this may have a positive impact on their long-term prognosis. Therefore, we believe that future prospective studies with broader samples that will be conducted to discover presumed stroke patients at an early stage might noticeably improve long-term outcomes
Supplemental Material
sj-docx-1-cat-10.1177_10760296241231944 - Supplemental material for Investigating the Impact on Long-Term Outcomes and the Necessity of Hereditary Thrombophilia Screening in Presumed or Perinatal Arterial Ischemic Stroke
Supplemental material, sj-docx-1-cat-10.1177_10760296241231944 for Investigating the Impact on Long-Term Outcomes and the Necessity of Hereditary Thrombophilia Screening in Presumed or Perinatal Arterial Ischemic Stroke by Ömer Bektaş, MD, Özben Akinci GÖktaş, MD, Begüm Atasay, MD, and Serap Teber, MD in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
