Abstract
In order to compare the effect of combined oral contraceptive (COC) and combined vaginal contraceptive (CVC) methods on the inflammation and procoagulation, we recruited female participants in 3 groups: control participants, COC users, and CVC users. We measured different blood biomarkers. The users of both COC and CVC had higher levels of C-reactive protein (P < .0001) and factor VII (P < .0001). However, CD40 ligand was only higher for COC users (P < .0001) and not the CVC users. Even though the levels of thrombin/antithrombin III were not higher for COC and CVC users, as compared to the controls, CVC users had higher levels as compared to COC users (P = .0327). As compared to the control group, we observed higher levels von Willebrand factor among CVC users but not the COC users. Longitudinal studies with larger sample size are needed to better assess the inflammatory and procoagulation response due to CVC use.
Introduction
Combined hormonal contraceptives (CHCs), particularly combined oral contraceptives (COCs), have become a common method for pregnancy prevention worldwide. 1 The CHCs utilize synthetic derivatives of naturally occurring female sex hormones (estrogen and progesterone) to inhibit ovulation and prevent pregnancy. Studies have shown that CHC use is linked to an increased risk of vascular events, including venous thromboembolism (VTE), myocardial infarction (MI), and stroke.2–7 New generations of CHCs have been developed with lower doses of estrogen, aimed at reducing the risk of vascular events. However, a meta-analysis study analyzing the effect of low-dose CHCs (containing 20-35 µg estrogen and 0.15-3.0 mg progestin) found a 2-fold increased risk of MI and stroke. 8 This may be due, in part, to changes in the level and activity of circulating coagulation factors. For example, women using oral contraceptives may become resistant to the anticoagulant action of activated protein C that not only increases the risk of VTE but may also increase the risk of MI and stroke.6,9,10
In recent years, a combined vaginal contraceptive (CVC), NuvaRing (Merck & Co, Inc, Whitehouse Station, New Jersey) has been introduced as a new form of CHC. It delivers 120 μg etonogestrel (the active metabolite of desogestrel) and 15 μg ethinyl estradiol (EE). The use of CVCs bypasses the absorption of the steroids through the gastrointestinal tract with immediate hepatic first-pass metabolic effects. The CVCs also provide a continuous release of steroids and thereby a stable concentration.11,12 The estimated risk of VTE among CVC users is reported to be about 149 per 100 000 women. 13 However, the risks of stroke and MI due to CVC use have not been established.
Increased levels of plasma markers of inflammation, such as high-sensitive C-reactive protein (hs-CRP), E-selectin, P-selectin, and CD-40L, indicate individuals with an increased risk of adverse vascular events.14–16 Similarly, alterations in the levels and activities of the coagulation proteins can also have an effect on the risk of vascular events. Our objective was to determine the differences in the levels of inflammatory markers and the coagulation cascade among CHC users that may indicate an increased risk of vascular events.
Methods
After obtaining Institutional Review Board approval at the University of Minnesota, female participants aged 19 to 30 years from the University of Minnesota student body and staff were recruited into 3 groups: (1) control participants who had not been on any hormonal contraceptive for a minimum of 6 months, (2) COC users who had been using pills for a minimum of 6 months, and (3) CVC users who had been using the vaginal ring for a minimum of 6 months. The exclusion criteria included a recent history of viral or bacterial infection, history of recent surgery, history of chronic inflammatory diseases, history of malignancies, history of pregnancy within the past 6 months, history of migraine with aura, the use of other steroids, and the use of antiplatelet/anticoagulant medications within 1 week of study participation.
With consent provided, the participants were asked to fill out a standardized questionnaire describing their medical history, family medical history, physical activities, dietary and sleeping habits, and general lifestyle choices. We recorded blood pressure (BP), pulse rate, body temperature, weight, height, and waist and hip circumferences. Body mass index (BMI) was defined as weight/height2 (kg/m2). Phlebotomy was performed to collect plasma and serum samples. All samples were appropriately aliquoted and labeled for storage at −80°C for batch analysis.
Tests for complete blood count (CBC) with differentials,
Statistical Analysis
All continuous measurements were analyzed for departure from normality. For variables with relatively normal distribution, mean and standard deviation (mean ± SD) were reported, and F tests and t tests were used for 3- and 2-group comparisons, respectively. For nonnormally distributed variables, medians and interquartile ranges were reported and Kruskal-Wallis and Wilcoxon rank sum tests were used for 3- and 2-group comparisons, respectively. For categorical variables, Chi-square or Fisher exact test was used, depending on the absence or presence of sparse data cells. In addition to study groups, all multivariate regression models were adjusted for covariates, including age, race, alcohol consumption, regular sleeping habit, and family history of cardiovascular disease (CVD) and stroke. The least square means for different groups and the P values based on the estimated regression models, were reported. For blood biomarkers a P value of <.01 was considered statistically significant. Otherwise, a P value of <.05 was considered to be statistically significant.
Results
We recruited a total of 159 participants for the study: control participants (n = 64), COC users (n = 51), and CVC users (n = 44). Among the control participants, there were 24 (37.5%) past CHC users with a median (range) last use of 13 (6-60) months. Among COC users, 19.6% used first generation (containing 0.02-0.035 mg EE and 0.4-1.5 mg norethindrone), 13.7% used second generation (containing 0.02-0.035 mg EE and 0.1-0.3 mg levonorgestrel), and 66.7% used third generation (containing 0.02-0.035 mg EE and 0.15-1.5 mg of drospirenone, norgestimate, or desogestrel). The CVC users were on NuvaRing. The median (range) of CHC use for COC and CVC users were 40 (6-156) and 24 (6-96) months, respectively. The mean age ± SD of the control, COC users, and CVC users were 21.5 ± 2.3, 22.3 ± 2.2, and 24.0 ± 3.2 years, respectively. The age difference between the control participants and CVC users was statistically significant (P < .0001). We observed that 69% of the control participants, 92% of the COC users, and 91% of the CVC users were white (P = .0011). The median (range) of the days since last menstrual cycle to blood sample collection for the control, COC users, and CVC users were 19.5 (0-1263), 15 (1-81), and 11.5 (0-38) days, respectively, where difference among the 3 groups was not significantly significant (P = .1036). Information such as BP measurements, lifestyle, and medical history are shown in Table 1 . The users of CHC had a higher BP as compared to the control participants, though overall BP levels were in a relatively normal range. However, the difference in BP subsided after adjusting for the covariates. Alcohol consumption was more prevalent among CHC users (P < .0001) as compared to the control group. We also observed a more statistically significant common family history of CVD and stroke among CVC users (P = .0254). There was no statistically significant difference for the CBC differential tests among different groups (data are not shown).
Demographics, Baseline, Lifestyle, and Medical History of the Recruited Study Participants a
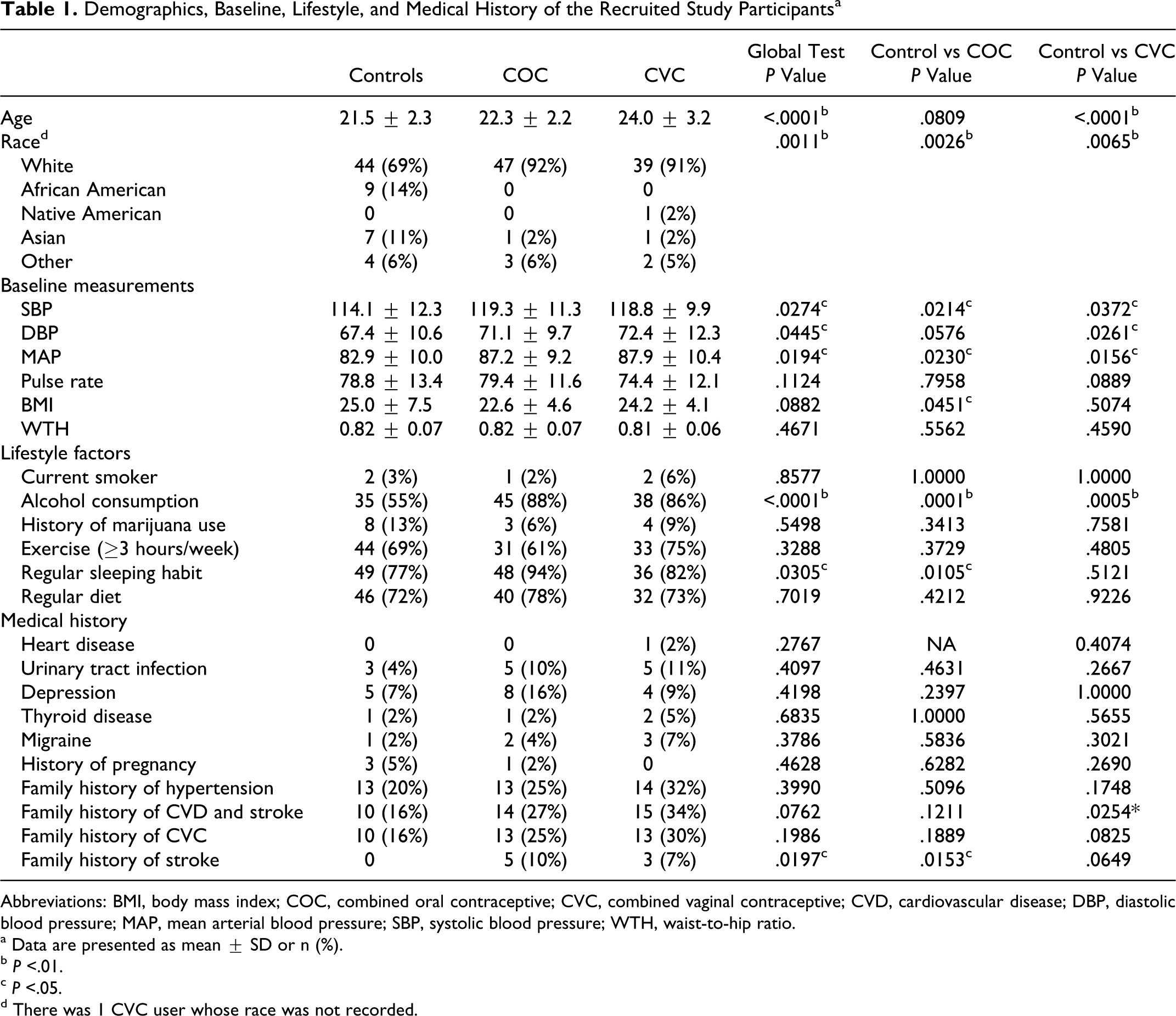
Abbreviations: BMI, body mass index; COC, combined oral contraceptive; CVC, combined vaginal contraceptive; CVD, cardiovascular disease; DBP, diastolic blood pressure; MAP, mean arterial blood pressure; SBP, systolic blood pressure; WTH, waist-to-hip ratio.
a Data are presented as mean ± SD or n (%).
b P <.01.
c P <.05.
d There was 1 CVC user whose race was not recorded.
The results for the univariate analysis are shown in Table 2 . The concentration of hs-CRP among both COC and CVC users was highly significant as compared to the control group (P < .0001 for both comparisons). We also observed a higher concentration of CD40L (P < .0001) and a lower concentration of PAI-1 (P = .0026) for COC users as compared to the control group. While FVII concentration was higher, the level of sTM was lower for both CHC groups as compared to the control group.
Univariate Analysis of the Blood Biomarkers
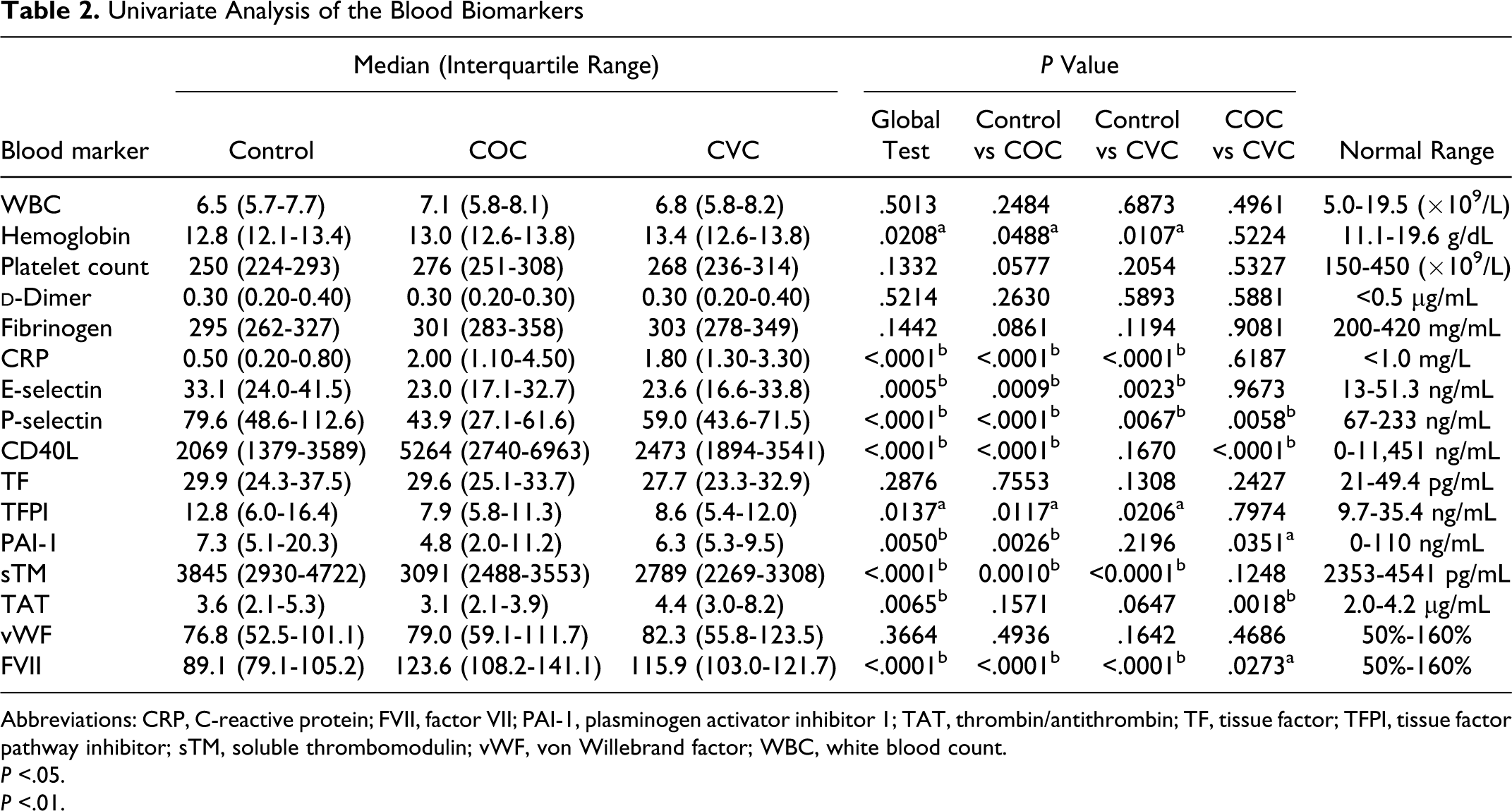
Abbreviations: CRP, C-reactive protein; FVII, factor VII; PAI-1, plasminogen activator inhibitor 1; TAT, thrombin/antithrombin; TF, tissue factor; TFPI, tissue factor pathway inhibitor; sTM, soluble thrombomodulin; vWF, von Willebrand factor; WBC, white blood count.
P <.05.
P <.01.
As compared to the control group, the levels of hs-CRP and FVII for both CHC groups and CD40L for the COC users only remained significantly higher (P < .0001) after adjusting for age, race, alcohol consumption, sleeping habit, and family history of CVD and stroke (shown in Figure 1 ). While the levels of TF were not different among the groups, TFPI levels were significantly lower among CHC users compared to the control group. The difference between other biomarkers such as sE-selectin, sP-selectin, and sTM remained the same as in the univariate analysis (see Table 3 for complete results). Smoking and BMI did not significantly alter the levels of sE-selectin or sP-selectin in the univariate regression or the multivariate regression.
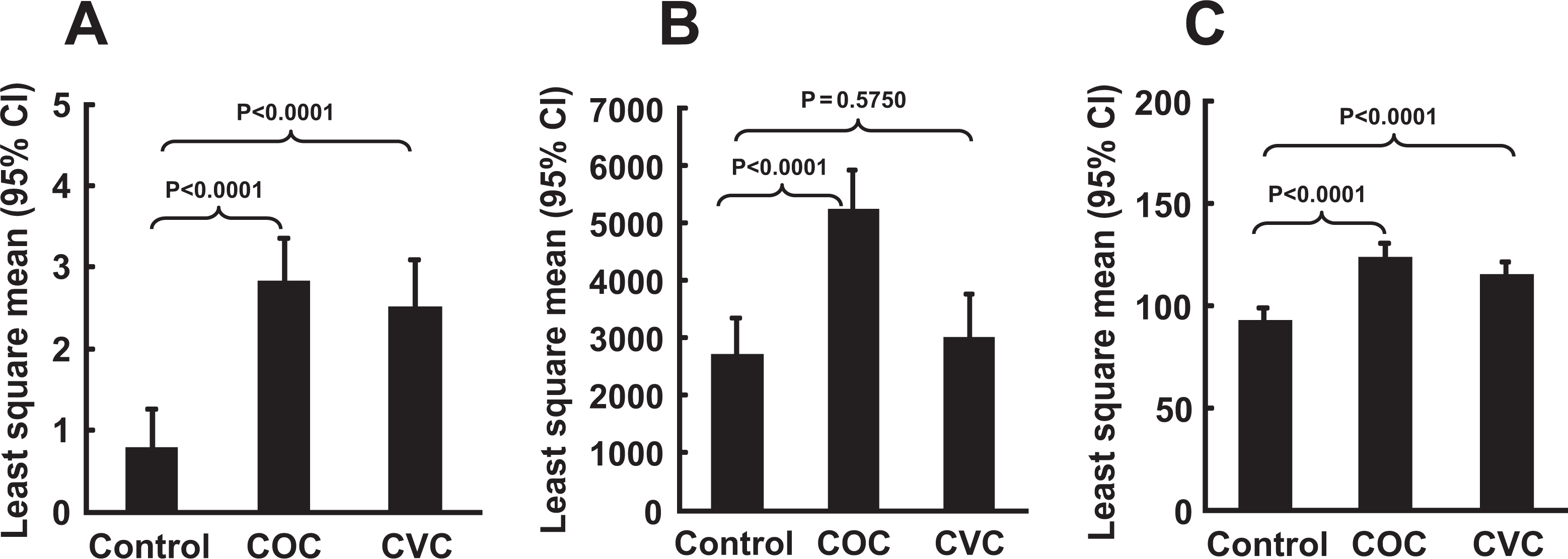
Multivariate analysis for biomarkers (A) CRP, (B) CD40L, and (C) FVII levels among the 3 studied groups. CRP indicates C-reactive protein; CD40L, CD40 ligand; FVII, factor VII.
Multivariate Analysis of the Blood Biomarkers
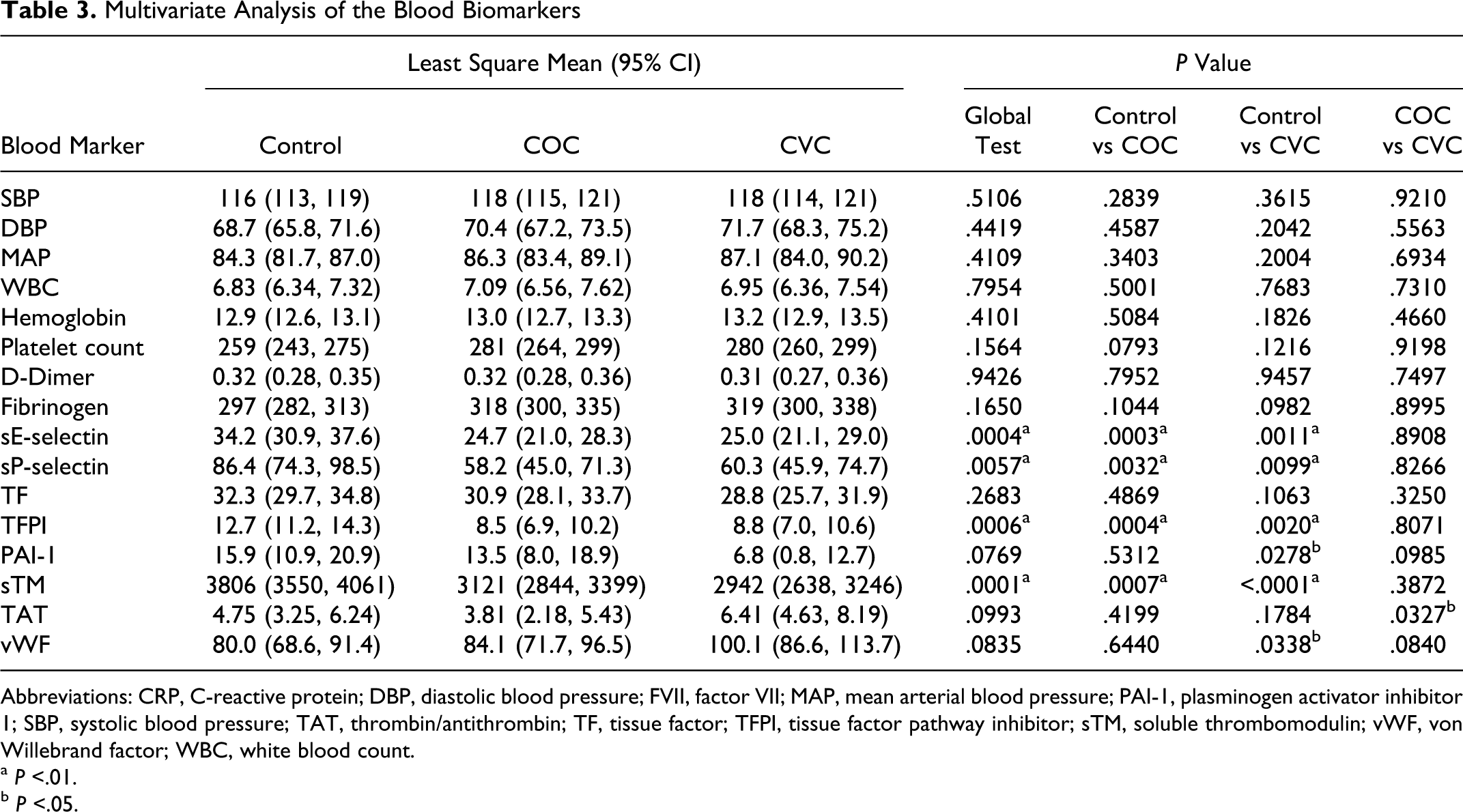
Abbreviations: CRP, C-reactive protein; DBP, diastolic blood pressure; FVII, factor VII; MAP, mean arterial blood pressure; PAI-1, plasminogen activator inhibitor 1; SBP, systolic blood pressure; TAT, thrombin/antithrombin; TF, tissue factor; TFPI, tissue factor pathway inhibitor; sTM, soluble thrombomodulin; vWF, von Willebrand factor; WBC, white blood count.
a P <.01.
b P <.05.
We compared “never users” with “past users” of CHCs that were included in the control group. We only observed a statistically significantly higher level of sE-selectin among past users (P = .0066; data are not shown), suggesting activation of the endothelium. Among different COC generations, biomarkers that were significantly different (ie, hs-CRP, sE-selectin, sCD40 L, TFPI, sTM, and FVII) lost their significant difference for the users of the second-generation COC as compared to the control group. Among first-generation COC users, the difference in sP-selectin and TFPI levels was no longer statistically significant as compared to the control group (data are not shown).
Discussion
The major findings of this study were the differential levels of inflammatory markers and coagulation cascade proteins among CHC users. Specifically, levels of hs-CRP and FVII were higher in CHC users and levels of sE-selectin, sP-selectin, TFPI, and sTM were decreased in CHC users. A significant increase in sCD40 L levels was seen in COC users but not in CVC users, suggesting in vivo platelet activation among COC users. These findings may provide mechanistic insight into the increased risk of vascular events associated with CHC use.
We observed elevated levels of hs-CRP in CHC users which is in agreement with the findings of previous publications.17–20 Silvestri et al 19 suggested that the elevated levels of hs-CRP among postmenopausal women on hormone replacement therapy may be due to metabolic hepatic activation and not an acute response. This hypothesis was based on the fact that the levels of other proinflammatory markers, such as interleukin 6 (IL-6) and E-selectin, were reduced in the same population studied. In another study, van Rooijen et al 20 arrived at a similar conclusion by showing higher levels of hs-CRP with unaffected levels of IL-6 and tumor necrosis factor-α among COC users. However, CVCs are not hepatically cleared due to their vaginal administration which warrants further studies to assess underlying pathophysiology of increased hs-CRP among CVC users. In another study 21 on the effect of hormone therapy among postmenopausal women, hs-CRP and IL-6 levels were negatively correlated among the conjugated equine estrogen-only group, while a positive correlation was observed among progestin-containing groups.The use of COCs may not influence vascular risk by affecting the oxidative modification of LDL but instead by promoting the atherosclerotic process and endothelial cell inflammation directly. 20 Numerous studies comparing transdermal estrogen and CRP among postmenopausal women have shown that transdermal estrogen replacement therapy does not increase CRP significantly compared to oral estrogen replacement therapy. 22 There is only 1 published study on CRP levels in premenopausal woman using transdermal CHC versus COC, and it showed a similar increase in CRP for each. 23 We cannot assume that the effects of hormone replacement therapy in postmenopausal women will be the same as the effects of hormonal contraception in premenopausal women, nor can we assume that transvaginal delivery will be identical to transdermal delivery.
For both COC and CVC users, we observed lower levels of endothelial and platelet activation molecules (sE- and sP-selectin) as compared to the control group. Other published studies have also reported lower levels of sE- and sP-selectin due to the use of COCs.20,24 However, in yet another study, the reduction in the levels of E- and P-selectin between CHC users was not statistically significant. 25 P-selectin is a glycoprotein that is stored in the Weibel-Palade bodies of endothelial cells and in the alpha granules of platelets. Upon activation of endothelial cells and platelets, P-selectin is expressed on the cell surface. E-selectin, on the other hand, is expressed only on endothelial cells responsible for recruiting leukocytes to the site of inflamed vessel walls. Measuring soluble forms of E- and P-selectin therefore is a marker of endothelial and platelet activation. Lower levels of sE- and sP-selectin among CHC users suggest a nonendothelial activation pathway causing a rise in hs-CRP levels. 20
Compared to the control group, we observed a significantly elevated level of sCD40L for COC users but not for CVC users. Serum levels of sCD40L correlate with platelet activation 26 that play an important role in the pathophysiology of acute coronary syndromes. 15 There are numerous studies looking at the association of CD40L and cardiovascular events, with some showing an association whereas others showing no effect. 27 The CD40L has been shown to directly activate platelets and result in atherosclerosis in experimental models, and blocking it, or mutating its receptor, results in decreased thrombosis and atherosclerosis.28,29
There is a significantly increased risk of developing future MI and stroke among healthy women and patients with a history of atrial fibrillation who have a high concentration of sCD40L.14,16 Higher levels of sCD40L concentration among COC users may be due to a higher level of lymphocytes observed among COC users, since sCD40L is also found on the surface of B cells. The fact that the sCD40L levels rose only in COC users may indicate that these agents pose greater cardiovascular risk than CVC.
The serum levels of TF stayed relatively constant among different groups in our study. However, we noted a reduction in TFPI levels for both CHC user groups when compared to the control.30–32 The TFPI is a glycoprotein produced by endothelial cells to inhibit coagulation by preventing TF from binding to FVII. Thus, lower levels of TFPI are indicative of a hypercoagulable state. Although we noted a reduction in PAI-1 levels for both CHC user groups, with higher reduction in CVC users compared to the control, the reduction was not statistically significant. The difference between the 2 CHC user groups was also not significant, in agreement with another published study comparing levels of PAI-1 between COC and CVC users. 33 Plasminogen activator inhibitor 1 is a glycoprotein produced by both the liver and endothelial cells. Lower levels of PAI-1 indicate higher fibrinolysis activity among CHC users. Other published data have confirmed our finding.25,34,35 We also observed a statistically significant reduction in sTM levels among both CHC user groups. Thrombomodulin is a transmembrane glycoprotein with antithrombogenic properties and is expressed by endothelial cells acting as a receptor for thrombin. Binding of TM to thrombin causes the loss of thrombin’s procoagulant activity. 36 An increased level of sTM is considered a surrogate marker for endothelial injury.
Even though there was no statistically significant difference in TAT levels between each of the CHC user groups and the control, the CVC user group had a higher level of TAT. The observed TAT level, among CVC users, was toward the upper limit within the normal range. Another published study reported no difference in TAT between NuvaRing and COC users; however, the number of participants was small. 33 We did not observe any significant correlation between age and TAT level (TAT increased 0.19 per 1 year increase in age, P = .30). The same was observed for the effect of exercise hours/week (≥3 hours) on TAT. We did not observe any statistically significant change in levels of vWF among the 3 groups in our study. There are published data regarding the use of COCs and their impact on vWF level that confirm our finding.25,37
The FVII activity levels were higher among both CHC user groups, as compared to the control group. The CVC users in our study had a lower level of FVII than the COC users (though not statistically significantly lower). This differs from a published study by Magnusdottir et al 33 showing a higher level of FVII activity among NuvaRing users at cycle 6 as compared to the second-generation COC users. It is important to mention that all the published studies looking at surrogate blood biomarkers either lacked a sufficient sample size or studied the biomarkers within three to six 28-day cycles of use.11,33,38,39
Our study was limited in various ways. The presented results should be interpreted cautiously and the results not considered definite as the sample size was small, and we did a relatively large number of tests. For instance, including different generations of COCs may have negatively influenced the results. It has been suggested that different generations of COCs may impose different levels of risk of developing thrombi, leading to the development of VTE.40,41 However, the comparison between COC generations in our study may be inadequate due to the small sample size in each group. Nonetheless, including different generations of COCs creates a more realistic scenario of today’s hormonal contraceptive practice and use. Due to the predominately white racial/ethnic makeup of the students on the University of Minnesota campus, we were not able to recruit more participants from minority groups. Further studies are warranted to investigate of the effect of hormonal contraceptives among different race/ethnic groups, particulary among the never-user participants who plan to start using hormonal contraceptives. This would ultimately provide better information for prescribing a safer and convenient hormonal contraceptive regimen tailored for each individual. Furthermore, the clinical significance of the changes in the markers studied here can only be assessed in a randomized longitudinal study.
We examined differences in levels of various serum biomarkers related to endothelial and platelet function, inflammation, and coagulation due to use and types of CHC among young, healthy women with no history or genetic predisposition for vascular events. The most striking finding was a differential effect of COC use on sCD40L levels not observed with CVC use. Whether this renders COC users an increased risk of vascular events compared to CVC users is a question for future longitudinal study. Also, as newer contraceptive methods such as vaginal rings, implants, and progestin-only vehicles become increasingly popular, further studies will be necessary to determine their effects on inflammatory markers, the coagulation cascade, and the risk of vascular events.
Footnotes
Acknowledgment
The authors express their gratitude to Eliza W. Hartley for her help in preparing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Office of the Vice President for Research and Minnesota Medical Foundation at the University of Minnesota, Minneapolis, MN.
