Abstract
Our study was designed to prospectively evaluate the role of the methylenetetrahydrofolate reductase (MTHFR) TT677 genotype, the prothrombin (PT) 620210A mutation, the factor V G1691A mutation in leukemic Turkish children treated according to modified BFM 95 study protocols with respect to the onset at thromboembolic events. Eighty-two consecutive leukemic children investigated had are established inherited prothrombotic risk factor: 29 children showed the MTHFR genotype (heterozygous, n = 22; homozygous, n = 7); 3 showed the heterozygous PT G20210A variant; 16 were carriers of the factor V mutation (heterozygous). In addition, combined prothrombotic defects were found in a further 11 patients the MTHFR mutation was combined with the F V mutation (n = 7), the PT G20210A variant (n = 2). In 7 (8.5%) of these 82 patients, venous thromboembolism occurred. In conclusion, the presence of inherited thrombophilia with Turkish children treated acute lymphoblastic leukemia may be useful for designing targeted primary prevention strategies.
Introduction
The TT 677 genotype of the methylenetetrahydrofolate reductase (MTHFR) and prothrombin (PT) G20210A variant of the PT gene seem to be common but probably mild risk factors for venous thromboembolism (VTE). The defects of the protein C pathway (the factor V G1691A mutation) have also been reported to be associated with an increased thrombotic risk in healthy individuals. 1 In the previous studies2,3 of healthy Turkish children, the frequency of the FV G1691A heterozygous mutation, MTHFR genotype, and PT 20210A variant were detected as 7.4%, 20%, and 2% to 4%. The reported4–6 incidence of TE in children with acute lymphoblastic leukemia (ALL) varies from 1.1% to 36.7%. There have been contradictory findings regarding the effect of prothrombotic risk factors (PRFs) on thrombosis in children with ALL in recent years.7,8 In addition, there have been no comprehensive studies investigating PRF in Turkish children with ALL. In this study, we investigated the possible effects of MTHFR TT677 genotype, PT 20210 allele, and FV Leiden mutation on venous thrombosis in Turkish children with ALL.
Patients and Methods
Inclusion Criteria
After obtaining parental consent, children 2 years of age and older who had acute onset of ALL and had been treated according to the modified BFM 95 induction/reinduction protocols between January 2007 and 2009 were included in this prospective single-center study.
Exclusion Criteria
Participants without complete remission of the disease (day 30 of the induction protocol), children <2 years of age with leukemia, children receiving heparin prophylactically, children with concomitant chronic diseases, hepatic failure, severe septicemia, and adolescents using oral contraceptives or with nicotine abuse were excluded from the study.
Patients
In all, 82 children newly diagnosed with leukemia (median age, 9.5 years; male, n = 49 and female, n = 33) who had received treatment according to the ALL-BFM 90/95 study protocols were prospectively enrolled in this study. Venous thromboemboli were diagnosed by color Doppler sonography. In addition, cerebral venous thromboses were diagnosed using magnetic resonance imaging.
Leukemia Therapy
Additional treatment included the administration of prednisone (60 mg/m2) on days 1 to 36 and weekly vincristine (1.5 mg/m2), as well as daunorubicine (30 mg/m2) on days 8 and 15 for standard-risk patients, and on days 22 and 29 for medium-risk patients. The children also received prophylactically intrathecal methotrexate on days 1, 12, 30, 45, and 59 during induction therapy. In reduction therapy, patients received Escherichia coli asparaginase (ASP) 10 000 IU/m2 on days 8, 11, 15, and 18, along with dexamethasone (10 mg/m2) on days 1 to 21, and weekly vincristine (1.5 mg/m2) and doxorobicin (30 mg/m2) on days 8, 15, 22, and 29. Medium-risk patients received a second course of this reinduction protocol. High-risk patients received E coli ASP in doses of 25 000 IU/m2 on days 6 and 11 of the high-risk -1′, -2′, and -3′ blocks in reinduction therapy.
Depending on the individual decisions by parents and our center, polychemotherapy was administered via peripheral veins or Porth catheters. Porth catheters were heparin blocked every 4 weeks with 200 IU of unfractionated heparin. Patients with relapses received FLAG-IDA protocol (fludarabine, idarubicin, and G-CSF).
Laboratory Analyses
Cells from venous blood, which was collected in EDTA sample tubes, were separated by centrifugation at 3000g for 15 minutes. The buffy-coat layer was removed and then stored at −800°C until DNA extraction was performed by standard techniques.
Ethical Aspect
This study was approved by the ethics committee of Erciyes University Faculty of Medicine. Written informed consent for their participation was obtained from the children’s parents.
Results
Thrombotic Events
In total, 7 (male, n = 3; female, n = 4) out of 82 consecutive patients with ALL had VTE during induction (n = 4; protocol days 21-36) and relapse therapy (n = 3). The median age at thrombotic onset was 9.5 years (range = 2-16 years). The age distribution of all children was not different from the thrombosis group. Cerebral venous thrombosis was diagnosed in 2 (2.4%) patients and deep venous thrombosis (DVT) was documented in 5 (6%) patients (Table 1). In all, 7 (8.5%) out of 82 patients with leukemia with PRFs had venous thrombosis, while only 1 (1.2%) out of 82 children had no identified prothrombotic defect. In addition, 4 out of 7 children had central Porth catheters.
Clinical and Laboratory Findings of Patients With Thrombosis
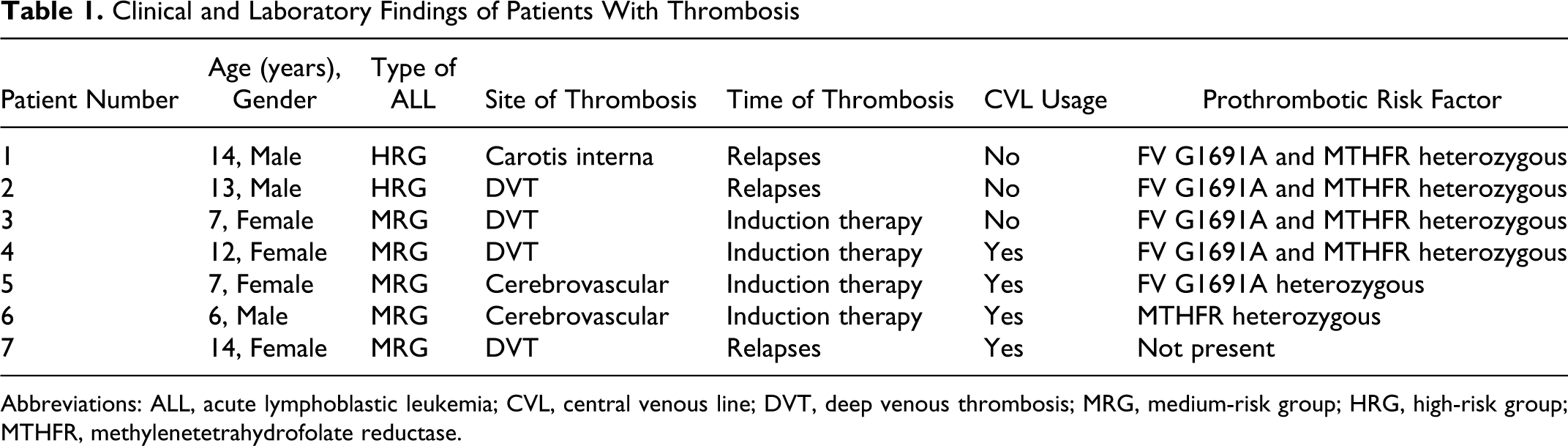
Abbreviations: ALL, acute lymphoblastic leukemia; CVL, central venous line; DVT, deep venous thrombosis; MRG, medium-risk group; HRG, high-risk group; MTHFR, methylenetetrahydrofolate reductase.
Presence of Central Lines
In 35 (42%) of the 82 children, polychemotherapy was administered through central lines. No thromboembolism-associated death occurred in these patients. In all, 11% of patients with Porth catheters developed thrombosis. There were Porth catheters in 4 out of 7 patients with thrombosis and 1 patient who had thrombosis without PRF.
Thrombosis-Associated Deaths and Location of Thrombosis
Of the 7 patients, 2 died as a result of relapse; however not related to thrombosis. A total of 4 patients had DVT, 2 had cerebrovascular occlusion, and 1 had carotis interna thrombosis.
Prevalence of Single Established PRF
All participants investigated had 1 established single PRF. The TT 677 MTHFR genotype was found in 29 children, while 2 showed the heterozygous PT G20210A variant, and 16 were carriers of the FV G1691A gene mutation (heterozygous, n = 14 and homozygous, n = 2).
Prevalence of Combined PRF
Combined PT defects were found and the heterozygous MTHFR gene mutation was combined with FV G1691A (n = 7 but 3 patients without thromboembolism) and with PT G20210A variant (n = 2; Table 2 ). The MTHFR genotype, heterozygous PT 20210A variant, and heterozygous FV Leiden mutation were found to be 35%, 2.4%, and 19.5%, respectively.
Single and Combined PRFs Found Among Symptomatic/All Children With ALL (Symptomatic Thromboembolic Patients/All Children)
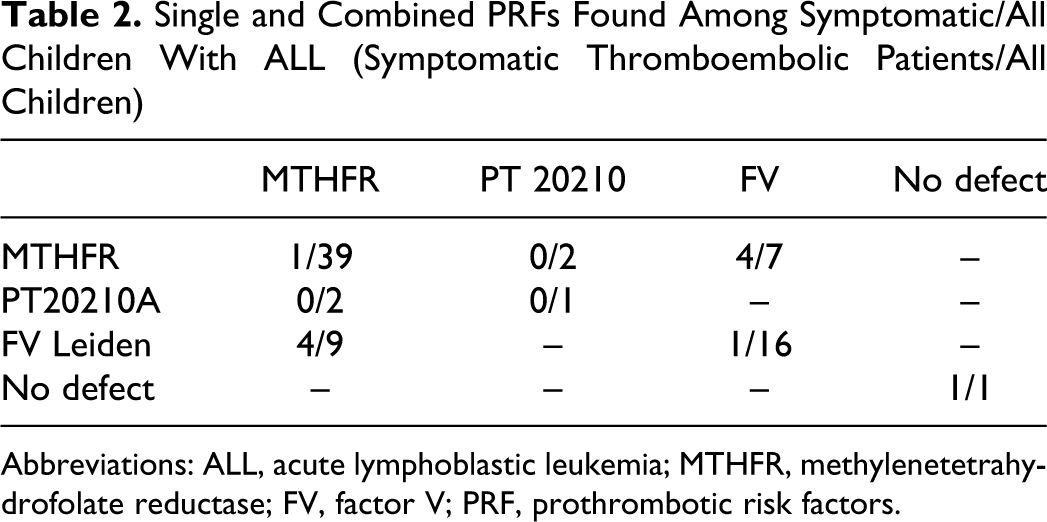
Abbreviations: ALL, acute lymphoblastic leukemia; MTHFR, methylenetetrahydrofolate reductase; FV, factor V; PRF, prothrombotic risk factors.
The thrombosis-free cumulative survival analysis was not performed because there were no thrombosis-related deaths among the patients. Single and combined condition predisposition to thrombosis was found in 7 of 82 children with thromboembolic leukemia and during the ALL-BFM study protocols. Of 7 the symptomatic children 1 did not develop thromboembolism.
Discussion
The present prospective study focused on the role of PRF in consecutively admitted newly diagnosed children with ALL presenting with single and combined PRF. The rate of thrombotic events reported here (8.5%) was found to be within the range treated with BFM-ALL 95. The TT 677 genotype (35%), PT 20210 variant (2.4%), and heterozygous FV G1691A mutation (19.5%) were found within the total patient group in this study.
Previous studies carried out in Turkey showed an 8% to 10% prevalence of FV Leiden mutation and 2% to 4% of PT 20210 variant.2,9 The homozygous and heterozygous MTHFR prevalence were found to be 13% and 33%, respectively. 9 However, we have shown here that VTE occurred in 14.6% of children with leukemia with a diagnosed PRF (7 of 48). It is now evident that different protocols for ALL can lead to different rates of thrombotic complication; differences in drugs (sources, dosages, time, and administration) could explain the variation.10–12 In the PARKAA studies, 13 the TE rates reached 29% and 36.7%. These studies failed to show any correlation between the presence of FV Leiden, PT mutation 20210A, and the development of thrombosis.
Another multicenter prospective study of children with leukemia on modified BFM 90/95 evaluated the risk of thrombosis in patients who had at least 1 identifiable prothrombotic defect at diagnosis. 1 It has been reported that the risk was higher in patients with a prothrombotic defect (46.5% vs 2.2%; P < .001) and was cumulative; patients with multiple defects had a significantly higher risk compared to those with a single defect (P = .009). A meta-analysis 14 concluded that inherited thrombophilia increased the thrombotic risk in children with ALL by approximately 8-fold.
In the present study, we found that the risk of thrombosis increased particularly in the presence of 2 PRFs. The risk of TE in our patients was affected by their treatment regime, the usage of a central catheter, and the activation state of the disease.15,16 Besides the involvement of thrombophilia, additional factors such as steroid and E coli-ASP administration and endothelial cell injury (with CVL) may function as the trigger mechanism.10,11 For this reason, particularly children with ALL and children at high risk of developing TE, such as those who replaced CVL and received
Nowak-Göttl et al reported that the use of dexamethasone instead of prednisone concomitantly administered with ASP in patients with leukemia of the same living population treated according to the BFM-adapted protocols during induction therapy, significantly reduced the symptomatic onset of VTE in the children investigated. 7 Recently, Giordano et al investigated a number of hemostatic and inflammatory markers at diagnosis and at different time points during chemotherapy for the remission induction in order to identify alterations in the plasma levels of prothrombotic markers before and during the course of chemotherapy. After starting induction therapy, endothelial activation markers significantly increased, suggesting an insult by chemotherapy on the vascular endothelium and steroid administration. 17
Although the probability of thrombosis was found to be high in the presence of 2 genetic PRFs in this study, there were some limitations, for example, the study group came from a single-treatment center and the number of the group was limited. Moreover, this study only looked at the genetic factors, whereas additional risk factors such as protein C/S, antithrombin III, factor V, factor VIII, homocysteine levels, antiphospholipid-anticardiolipin antibodies, lipoprotein A, and von Willebrand factor were not evaluated.
Further prospective larger multicenter studies are recommended to establish adequate anticoagulant treatment during polychemotherapy of Turkish children with ALL, carrying combined hereditary PRF.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
