Abstract
Our study was conducted to investigate the potential correlation between cardiac oxidative stress and inflammatory cytokine response following myocardial infarction. A total of 120 patients harboring acute myocardial infarction who underwent percutaneous coronary intervention (PCI) at our hospital were included. Their general clinical data were analyzed, and comparisons were made regarding the levels of inflammatory factors, oxidative stress markers, heart pump function, and cardiac function. The correlation between cardiac oxidative stress and inflammatory cytokine response was assessed using Pearson's linear correlation. Following treatment, significant reductions were seen in the serum levels of cortisol, thyroid-stimulating hormone (TSH), B-type natriuretic peptide (BNP), C-reactive protein (CRP), signal transducer and activator of transcription 3 (STAT3), interleukin-6 (IL-6), interleukin-8 (IL-8), tumor necrosis factor (TNF), and catalase (CAT) compared to pre-treatment levels. Conversely, the levels of growth hormone (GH), superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), total antioxidant capacity (T-AOC), cardiac output (CO), and cardiac index (CI) were significantly elevated. Serum cortisol (r = 0.481, P = .001), BNP (r = 0.437, P = .001), CRP (r = 0.542, P = .001), STAT3 (r = 0.835, P = .001), TSH (P = .001), IL-8 (r = 0.867, P = .001), TNF-α (r = 0.439, P = .001), and cardiac oxidative stress demonstrated significantly positive correlations (P < .05). Additionally, a significant negative correlation was found between GH (r = −0.654, P = .001) and immune balance (P < .05). This study evaluated the severity of myocardial infarction using indicators such as CO and CI. This study found a significant correlation between cardiac oxidative stress and inflammatory cytokines after myocardial infarction, suggesting their potential as predictors of myocardial infarction severity.
Background
At present, myocardial infarction is a prevalent cardiovascular disorder in clinical practice, with a relatively high incidence rate. If not promptly managed, it poses a significant threat to life and health. 1 Myocardial infarction is not simply a result of inadequate blood supply to the heart; it involves interconnected secondary pathological changes, such as oxidative stress and inflammation, which form a complex signal network and ultimately lead to cascading damage. The inflammatory response plays a crucial role in the progression of myocardial necrosis and ventricular remodeling following myocardial infarction. Clinical research has shown that the pathophysiology of myocardial infarction involves myocardial ischemia, which disrupts the imbalance of oxygen supply and interferes with the redox environment. 2 Hypoxia following myocardial infarction leads to myocardial cell death and triggers an inflammatory reaction. Factors influencing the severity of ischemia include the completeness or partial occlusion of blood vessels, duration of occlusion, myocardial blood supply, presence of collateral vessels, and adequacy of reperfusion after treatment. Therefore, timely treatment is crucial in preventing fatalities or long-term complications, with percutaneous coronary intervention (PCI) and thrombolysis being the primary specific treatment methods. 3 Mao Meijiao et al. reported an elevation in the serum level of tumor necrosis factor-α (TNF-α) inpatients with myocardial infarction, with an accompanying increase in pro-inflammatory cytokines and oxidative stress markers. 4 Inflammation enhances the oxidation process while reducing the antioxidant capacity of the system. Similarly, Kologrivova et al. found that inflammation induced oxidative stress during myocardial infarction, and conversely, oxidative stress triggered inflammation. However, the relationship between cardiac oxidative stress and inflammatory cytokine response after myocardial infarction remains uncertain. Therefore, our retrospective study selected 120 patients with acute myocardial infarction (AMI) to explore the correlation between cardiac oxidative stress and inflammatory cytokine response, with the aim of laying a theoretical foundation for improving the treatment efficacy of AMI in clinical practice.
Patients and Methods
Clinical Data of Patients
From July 2021 to February 2023, our hospital selected a total of 120 patients with AMI, including 73 males and 47 females, with an average age of 65.67 ± 4.48 years. The inclusion criteria include the following: 5 All participants met the diagnostic criteria for AMI. The duration of chest pain exceeds 30 min, and the ST segment elevation in two consecutive leads is ≥1 mm. The time from onset to treatment is less than 12 h. The patient is suitable for PCI surgery, with complete clinical data and signed informed consent form. The exclusion criteria are patients who need cardiogenic shock management or intra-aortic balloon pump before or during PCI; patients with severe cardiac valve stenosis, pericardial disease, cardiomyopathy, or severe pulmonary disease; pregnant women; patients with complications such as diabetes, hypertension, and immune system disease; and patients who cannot meet the research requirements. 5 The subjects have specific diseases such as pituitary adenoma, autoimmune disease, acromegaly, adrenal adenoma, or heart failure or have received or not received any other medications that may affect the results before AMI, such as corticosteroids.
Treatment Methods
Prior to PCI, patients were administered 250–325 mg of aspirin, and all patients received a loading dose of 180 mg of tigrello. Following the PCI procedure, all patients received aspirin and tigrello for 1 month (screening period). Additional postoperative treatments included atorvastatin, low-molecular-weight heparin, clopidogrel, aspirin, and other medications as part of the secondary prevention of myocardial infarction. 6 β-Blockers and nitrates were administered based on individual patient conditions. 7 The unified time for sample collection is as follows: On the second day after treatment, peripheral blood was collected for testing of various indicators. None of the subjects had complete left anterior descending branch obstruction or slight circumflex branch obstruction. Moreover, the mortality rate of the subjects was zero (no subjects died during the study).
Observation Indicators
Analysis of General Clinical Data
General data included age, gender, and history of hypertension and heart illness. On the morning following admission, fasting venous blood samples were obtained from the patients. Measurements were taken for FIB, markers of myocardial necrosis (cTnI, cTnT, Mb, and CK-MB), CHOL, triglyceride, HDL-C, LDL-C, ApoA, and ApoB.
Analysis of Peripheral Blood Inflammatory Factors
On the first day before operation and the last day before discharge, 5 mL of fasting venous blood was collected. The serum was separated by centrifugation at 3500 r/min for 10 min. ELISA was used to measure serum levels of cortisol, thyroid-stimulating hormone (TSH), growth hormone (GH), interleukin-6 (IL-6), interleukin-8 (IL-8), brain natriuretic peptide (BNP), signal transducer and activator of transcription 3 (STAT3), C-reactive protein (CRP), and TNF-α.
Analysis of Peripheral Blood Oxidative Stress Markers
One day before operation and 1 day after discharge, 5 mL of fasting venous blood was collected. The serum was separated by centrifugation at 3500 r/min for 10 min. Serum levels of superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GSH-Px), and total antioxidant capacity (T-AOC) were determined using related kits.
Analysis of Cardiac Pump Function
The cardiac pump function of the patients was assessed using a cardiac color Doppler diagnostic instrument on the first day before the operation and the last day before discharge. Evaluation indexes for cardiac pump function included cardiac output (CO), cardiac index (CI), cardiac reserve (CR), and left ventricular ejection fraction (LVEF).
Evaluation of Cardiac Function
The Killip grading system was utilized to assess the cardiac function of the patients. Grade I indicated no obvious heart failure, Grade II indicated left heart failure with lung rales < 50%, Grade III indicated acute pulmonary edema with extensive rales throughout the lungs (large, small, dry, and wet rales), and Grade IV indicated varying degrees or stages of hemodynamic changes. 7
Correlation Analysis of Cardiac Oxidative Stress and Inflammatory Cytokine Response
The correlation between cardiac oxidative stress and inflammatory cytokine response was analyzed using Pearson's linear correlation.
Statistical Analysis
All statistical analysis was carried out using SPSS 20.0 statistical software. The measurement data were presented as the mean ± standard deviation (
Results
General Clinical Data
The general clinical data of patients were shown in Table 1.
General Clinical Data of Patients.

Inflammatory Factors
The levels of serum cortisol, TSH, IL-6, IL-8, and TNF-α in the patients after treatment were significantly lower than before treatment, whereas the level of GH was evidently higher after treatment (P < .05, Table 2).
Inflammatory Factors in Peripheral Blood of Patients (

BNP, CRP, and STAT3 Levels
Following treatment, the serum levels of CRP, BNP, and STAT3 were significantly lower compared to that before treatment, with statistically significant differences (P < .05, Table 3).
Analysis of CRP and STAT3 Levels of Patients (

Oxidative Stress Markers
The activities of SOD, GSH-Px, and T-AOC capacity were significantly higher after treatment compared to before treatment, whereas the activity of CAT was significantly lower (P < .05, Table 4).
Analysis of Oxidative Stress Level of Patients (
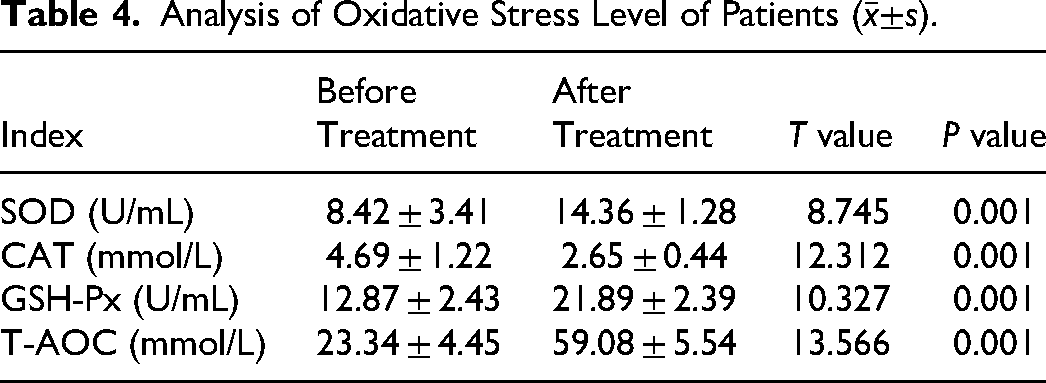
Heart Pump Ability
The CO, CI, CR, and ejection fraction of the patients after treatment were markedly higher than before treatment (P < .05, Table 5).
Analysis of Cardiac Pump Function of Patients (

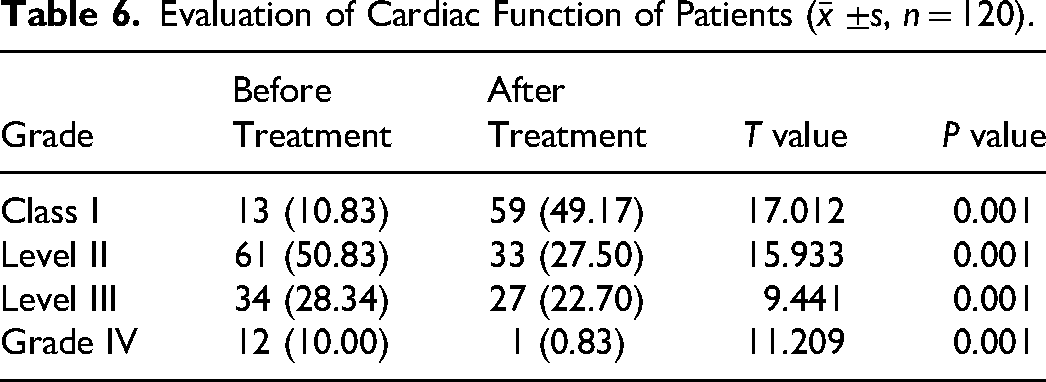
Cardiac Function
Before treatment, the percentage of patients with cardiac function Grades I, II, III, and IV were 10.83%, 50.83%, 28.34%, and 10.00%, respectively. After treatment, the percentage of patients with cardiac function Grades I, II, III, and IV were 49.17%, 27.50%, 22.70%, and 0.83, respectively (Table 6).
Evaluation of Cardiac Function of Patients (
Correlation Analysis Between Cardiac Oxidative Stress and Inflammatory Cytokine Response
There were significant positive correlations between serum cortisol (r = 0.481, P = .001), TSH (r = 0.552, P = .001), CRP (r = 0.542, P = .001), STAT3 (r = 0.835, P = .001), IL-6 (r = 0.934, P = .001), IL-8 (r = 0.867, P = .001), TNF (r = 0.439, P = .001), SOD (r = 0.652, P = .001), CAT (r = 0.710, P = .001), GSH-Px (r = 0.592, P = .001), and T-AOC (r = 0.661, P = .001). A significant negative correlation was seen between GH (r = −0.654, P = .001) and immune balance (P < .05) (Table 7).
Correlation Analysis Between Cardiac Oxidative Stress and Inflammatory Cytokine Response.

Discussion
Myocardial infarction is a life-threatening condition that predominantly occurs prior to hospital admission. If patients do not receive timely and effective treatment, it poses a significant threat to their overall safety and well-being. Oxidative stress and inflammation have gained substantial attention as crucial pathophysiological factors in myocardial infarction and the progression of heart failure. While reactive oxygen species (ROS) play a signaling role in a healthy heart, excessive and uncontrolled production of these molecules can lead to oxidative stress and damage to myocardial cells. Certain mechanisms may ultimately result in mitochondrial dysfunction and increased ROS production. This, in turn, can trigger systemic inflammation, and ROS can have detrimental effects on cell components, including mitochondria, further exacerbating the generation of free radicals and intracellular oxidative stress. This vicious cycle contributes to functional and structural maladaptation, thereby accelerating the development of myocardial infarction. 8
However, the specific mechanism underlying myocardial infarction remains unclear. 9 According to research, STAT3 has a protective function in the heart and can be activated and transcribed through both typical and atypical pathways. The STAT3 signal transduction pathway contributes to myocardial ischemic protection, as STAT3 plays a crucial role in myocardial cell function and can regulate the cardiac microenvironment. However, inflammatory factors can upregulate STAT3 expression through various signaling pathways, increasing the risk of left ventricular rupture in ischemic myocardium and enhancing the expression of CRP, an inflammatory marker associated with rupture. CRP, primarily synthesized and secreted by the liver, is a trace protein. During acute systemic inflammation, CRP can serve as a nonspecific marker to evaluate the degree of inflammatory response. If tissue damage occurs due to a disease, it stimulates an elevation in the serum or plasma CRP levels. When the body experiences acute inflammatory reactions or suffers from damage, CRP levels tend to increase. BNP, belonging to the natriuretic peptide family, is mainly synthesized by the heart. BNP is typically secreted by the atrium and ventricle. Under normal physiological conditions, healthy individuals have low levels of BNP in their circulation since the atria and ventricles synthesize and secrete minimal amounts of BNP. However, if the ventricular muscle is stretched or there is increased pressure on the ventricular wall, BNP production is mainly stimulated in the ventricles, especially the left ventricle. Acute or persistent myocardial ischemia leads to regional abnormal ventricular wall movement and left ventricular dysfunction, resulting in an increase in BNP secretion. BNP serves as an important prognostic marker and aids in the risk assessment of patients with heart failure. 10 Our results indicated that the levels of BNP, CRP, and STAT3 were significantly lower after treatment compared to before treatment.
Based on our results, the levels of serum cortisol, thyrotropin, IL-6, IL-8, TNF, and CAT were significantly lower after treatment, whereas the levels of GH, SOD, GSH-Px, T-AOC, CO, CI, CR, and ejection fraction were higher compared to before treatment (P < .05). Recent studies have indicated the crucial role of inflammation, including pro-inflammatory cytokines, in the progression of myocardial infarction. These factors contribute to the pathological mechanism of myocardial infarction, and inflammatory cytokines may, in turn, promote oxidative stress and exacerbate disease development. 11 In addition, myoglobin serves as a sensitive early indicator of myocardial necrosis, while troponin I exhibits ideal specificity. Myoglobin, found in human myocardial cells and cytosolic hemoglobin of skeletal muscle, is a newly discovered marker of myocardial necrosis. Due to its relatively small molecular weight, myoglobin rapidly rises within 2 h after myocardial injury, reaches peak concentration after 6 h, and is gradually eliminated by the kidneys within 24 h. CK-MB, as the isoenzyme of creatine phosphokinase, is primarily present in human myocardial cells. It is released from injured myocardial cells and enters the bloodstream about 5 h after the onset of myocardial infarction. CK-MB can be considered an early rising marker of myocardial necrosis after the onset of myocardial infarction and is traditionally regarded as the “gold standard” for coronary heart disease, particularly AMI.
Whether in the acute phase after myocardial infarction or during chronic cardiac remodeling, oxidative stress and inflammation are closely intertwined. Ischemia–reperfusion injury increases the generation of ROS. Indeed, ROS recruit circulating inflammatory cells and fibroblast progenitor cells through various mechanics. Moreover, the elevated ROS levels in dysfunctional myocardial cells induce severe DNA oxidative damage, subsequently triggering the activation of ribozyme polyribose polymerase. The over-activation of this enzyme disrupts numerous cell metabolic pathways and promotes the expression of inflammatory mediators, which are critical for cardiac remodeling and failure. 12 Our findings revealed significant positive correlations between serum cortisol (r = 0.481, P = .001), thyrotropin (r = 0.552, P = .001), BNP (r = 0.437, P = .001), CRP (r = 0.542, P = .0001, P = .001), IL-8 (r = 0.867, P = .001), TNF-α (r = 0.439, P = .001), and cardiac oxidative stress (P < .05). Conversely, there was a significant negative correlation between GH (r = −0.654, P = .001) and immune balance (P < .05). Several cytokines have been detected in the serum of patients with early myocardial infarction, with TNF and IL-6 accounting for 74.53% and 67.98%, respectively, in the serum of patients with myocardial infarction. TNF acts as the primary pro-inflammatory cytokine by directly activating sensory neurons through receptors, stimulating the synthesis of other pro-inflammatory cytokines, and inducing myocardial cell damage. 13
Myocardial infarction is characterized by a systemic pro-inflammatory state. The overexpression of TNF damages mitochondrial DNA, inhibits antioxidant factors, alters the activity of mitochondrial complex III, and consequently increases the production of ROS. On the contrary, inhibiting TNF in vivo in animal models of heart failure can stabilize the oxidative imbalance and reduce cell apoptosis. 14 As for IL-6, the findings in animal models have been inconclusive. While IL-6 is a negative prognostic marker of myocardial infarction, a study discovered that IL-6-induced mitochondrial targeting signal transducer and transcription activators were overexpressed in myocardial cells, leading to the inhibition of electron supply from complexes I to II and a reduction in ROS production during ischemia. On the other hand, other research groups reported an IL-6-dependent increase in intracellular ROS increase, which not only affects redox regulation but also impairs mitochondrial function and inhibits antioxidant factors. As a result, the inflammatory response promotes the generation of mitochondrial ROS and contributes to the progression of ROS-mediated fibrosis leading to heart failure. Moreover, oxidative stress is one of the triggering factors for myocardial infarction, as ROS can induce structural and functional damage of myocardial cells. 15 It was reported that patients with myocardial infarction exhibited increased levels of serum nitrite, which serves as a biochemical marker for the stable decomposition of NO. Furthermore, several end products of intracellular oxidative damage following oxidative stress have been identified. The level of serum MDA was elevated in individuals with myocardial infarction, and plasma oxidative stress parameters were higher while plasma antioxidant parameters were lower compared to the healthy control group. Oxidative stress leads to the formation of various complex compounds that interfere with the normal functioning of myocardial cells.
The damage of oxidative stress to myocardial cells is mainly manifested in the following aspects: Oxidative stress can cause oxidative damage to biological molecules such as proteins, lipids, and nucleic acids in myocardial cells, leading to abnormal cell function and death. Oxidative stress can affect the calcium ion balance within myocardial cells, leading to an increase in intracellular calcium ion concentration, which in turn leads to a decrease in myocardial cell contractility and arrhythmia. Oxidative stress can affect mitochondrial function within myocardial cells, leading to a decrease in mitochondrial membrane potential and damage to mitochondrial respiratory chain, leading to abnormal energy metabolism and cell death. Oxidative stress can affect the antioxidant system within myocardial cells, leading to a decrease in antioxidant enzyme activity and a decrease in free radical scavenging ability, thereby exacerbating the degree of oxidative stress.
In conclusion, there existed a certain correlation between cardiac oxidative stress and inflammatory cytokines following myocardial infarction, which may serve as predictors of the severity of the condition. However, it is important to note that the sample size in this study was relatively small, and there was a lack of follow-up for patients after treatment. Therefore, future studies should aim to expand the number of subjects to further investigate these relationships and outcomes. The limitation of this study is that the number of subjects included is relatively small, and it is a single-center study, lacking regional representation.
Footnotes
Availability of Data and Material
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Authors’ Contributions
D.D. and Y.F. contributed to the conception and design of the study; T.F., L.Z., L.L., M.G., and H.W. performed the experiments and collected and analyzed data. D.D. wrote the manuscript. All authors reviewed and approved the final version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
The study protocol was approved by the Ethics Committee of Affiliated Hospital of Hebei Engineering University. All methods were carried out in accordance with relevant guidelines and regulations. Informed consent was obtained from all the study subjects before enrollment.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
