Abstract
In patients with acute ST-segment elevation myocardial infarction, early, successful, and durable reperfusion therapy optimizes the likelihood of favorable outcomes. Fibrinolysis and primary percutaneous coronary intervention improve survival compared to no reperfusion therapy in large part by reducing infarct size (IS) and preserving left ventricular ejection fraction. There is direct correlation between IS and clinical outcomes. In this article, we will review some of the more promising pharmacological agents geared toward reduction in IS, discuss the major pathways that can lead to this desirable outcome, and evaluate the results of clinical trials performed with these and other compounds.
Introduction
In patients with acute ST-segment elevation myocardial infarction (STEMI), early, successful, and durable reperfusion therapy optimizes the likelihood of favorable outcomes. Fibrinolysis and primary percutaneous coronary intervention (PCI) improve survival compared to no reperfusion therapy in large part by reducing infarct size (IS) and preserving left ventricular ejection fraction (LVEF). Although other advantages of the “open artery hypothesis” have been proposed, including reduced arrhythmogenic potential, improved remodeling and preservation of collateral source for future myocardial infarction (MI), IS reduction correlates most directly with outcomes.
Conceptually, reducing IS can be achieved by 2 mechanisms. First, a proportion of myocardium at risk is “salvaged” by early reperfusion, and the resulting IS is inversely proportional to the salvaged area. The second mechanism reflects the hypothesis that reperfusion injury (RI) occurring during reperfusion may limit the amount of myocardium potentially salvaged, and conversely, preventing RI may enhance myocardial salvage (Figure 1). Experimental models have confirmed the existence of RI in animals. 1 Numerous mechanisms underlying RI have been proposed, some of which have been validated in human studies. Figure 2 schematically displays the interplay of these pathways, which are beyond the scope of this review. As a practical matter, however, reduction in IS after reperfusion therapy in humans is substantially less than in animal models, perhaps because of the effects of RI. It is estimated that as much as half of the final IS may be caused by RI. 2

Theoretical construct of myocardial salvage and reperfusion injury after reperfusion (reproduced with permission from 1 ).

Pathophysiology of lethal reperfusion injury (reproduced with permission from 2 ).
Determinants of IS
Four major factors affect IS. The first is the extent of the jeopardized territory, which correlates well with the vessel and location of the occlusion. Left anterior descending (LAD) artery infarcts with proximal occlusions have the largest IS in humans. 3,4 The second factor is the duration of ischemia. 5 Considering that infarction starts deep in the subendocardium and progresses toward the epicardium, longer ischemic times lead to larger infarcts and to greater transmurality, that is, a greater proportion of the thickness of the left ventricle becomes irreparably damaged. The greater the transmurality of the infarct and the more homogenous the damage, the less likely it is that viable myocardium will be present and salvaged with reperfusion. Transmural infarction also predisposes to infarct expansion, aneurysm formation, and mural thrombus. The duration of ischemia is of course affected by the time from symptom onset to first medical contact and from first medical contact to drug or device time. Modulating the impact of time to reperfusion is the baseline thrombolysis in myocardial infarction (TIMI) flow. In approximately one-third of patients, early angiography in evolving STEMI demonstrates TIMI 2 or 3 flow, 6 reflecting the often dynamic nature of coronary thrombosis in humans, perhaps affected by early pharmacotherapy. The third factor is the existence and robustness of collateral flow during coronary occlusion which can significantly influence IS. The ability of collateral flow to maintain or delay the progression of the infarct process has been shown in animal models. 7 In humans, presence of collateral channels has been associated with better outcome, irrespective of reperfusion success and total ischemic time. 8 Finally, the ability to effectively restore epicardial and myocardial perfusion has been strongly related to IS. 9 Other variables potentially affecting IS are age, gender, other comorbidities, and concomitant medications received with reperfusion. 10
From a pooled analysis of 4 randomized STEMI trials, Stone et al identified predictors of IS determined by tc-99m single-photon emission computerized tomography (SPECT) in 1199 patients within the first month after STEMI. Median IS was 10% of the left ventricular (LV) mass (interquartile range 0%-23%). The following 8 variables emerged as independent predictors: LAD artery infarct, TIMI flow 0/1 before PCI, longer door-to-balloon time, male gender (all P < .001), final TIMI flow 3 (P = .001), previous MI (P = .005), longer delay before presentation to hospital (P = .02), and failed fibrinolysis before PCI (P = .03). 11
Correlation of IS With Mortality
Reducing IS through pharmacological or device-based measures would be justified if greater myocardial salvage enhances freedom from meaningful adverse outcomes, such as death or development of heart failure with or without repeat hospitalization. Infarct size has been strongly related to mortality after reperfusion therapy in STEMI. 12 Improved survival with primary PCI compared to fibrinolysis has been correlated with greater myocardial salvage with PCI. 13 In the Enhanced Myocardial Efficacy and Removal by Aspiration of Liberated Debris (EMERALD) trial, patients with diabetes mellitus had larger IS (by SPECT: median 20% vs 11% in nondiabetics, P = .005) and greater 30-day mortality and severe heart failure (10% vs 1%, P < .0001 and 12% vs 4%, P = .02, respectively). 14 In the same trial, patients with LAD infarcts had larger IS than those with non-LAD infarcts (25.8% ± 21.8% vs 11.3% ± 13.7%, P < .0001) and demonstrated a trend toward higher 6-month mortality (5.5% vs 2.1%, P = .09) and new-onset severe heart failure (3.5% vs 1.1%, P = .08). 4 Similar results were obtained in the Intracoronary Abciximab and Aspiration Thrombectomy in Patients With Large Anterior Myocardial Infarction (INFUSE-AMI) trial with more precise measurement of IS by contrast magnetic resonance imaging (CMR). Infarct size in patients with proximal LAD occlusion was larger than in those with mid LAD occlusion (median 19.3% [9.2-25.9] vs 14.3% [6.2-18.9], P < .0001). It is notable that at 1 year, patients with smaller IS (less than median of 17.2% of LV mass seen in the entire cohort) had significantly fewer major adverse cardiac and cerebral events (1.7% vs 7.0% in those with larger infarcts) and fewer major adverse cardiac and heart failure events (0.6% vs 8.6%, respectively, P = .001, Figure 3). 15 Mortality at 30 days was also higher in the former group (4.2% vs 0.6%, respectively, P = .04). 3 Conversely, in the Acute Myocardial Infarction Study of Adenosine (AMISTAD) II trial in which the effect of adenosine was evaluated in patients with STEMI treated by primary PCI or fibrinolysis (N = 2118, of whom 243 had SPECT measurement of IS), only high-dose adenosine reduced IS compared with placebo (P = .023). This reduction in IS was not, however, associated with a significantly lower incidence of heart failure, rehospitalization, or death within 6 months. 16 Large data sets are required to more precisely evaluate the relationship between myocardial salvage achieved by active interventions and subsequent improvements in event-free survival.

Impact of infarct size at 30 days on major adverse cardiac events at 1 year (adapted from 15 ). CMRI denotes cardiac magnetic resonance imaging; MACE, major adverse cardiac events.
Experimental Models of IS Reduction
Experimental animal models of infarct creation and attempts to limit its size have important implications to application of these attempts in humans. Many of them have shown promising results that have not been and should not have been expected to be replicated in humans for a variety of reasons. Paramount differences between animal models and humans with STEMI include the obvious facts that animals do not have atherosclerosis and spontaneous MI and that the timing of onset of ischemia in animal models is completely controlled by the investigator. Moreover, the experimental infarct is caused by external compression or ligation of the artery without the potential for embolization that exists when plaques fracture. Finally, reperfusion in experimental models is always successful and complete. Many of these concepts have been reviewed in detail elsewhere. 10,17
Other approaches to reduction in IS, such as pre- or postischemic conditioning and cooling, are beyond the scope of this review, which focuses primarily on pharmacological interventions.
Pharmacological Interventions to Reduce IS
A multitude of investigational agents have been tested for their effect on IS in patients with STEMI. Table 1 shows a partial list of those which have shown inconsistent benefit on IS (top portion) and some of the agents with more promising results that are continuing to be tested (bottom portion). The ensuing discussion reviews the mechanisms of action and data accumulated for some of these agents, by class of drugs and the efforts underway to further evaluate their potential benefit.
Inhibits Failed Attempts to Reduce Infarct Size (Partial List).

Abbreviations: TAMI 9, Thrombolysis and Angioplasty in Myocardial Infarction 9; EMIP-FR, European Myocardial Infarction ISIS-4, Fourth International Study of Infarct Survival; CALYPSO, Cylexin as an adjunct to lytic therapy to prevent superoxide reflow injury MAGIC, Magnesium in Coronaries; CORE, Collaborative Organization for RheothRx Evaluation; LIMIT AMI, Limitation of Myocardial Infarction Following Thrombolysis in Acute Myocardial Infarction; ESCAMI, Evaluation of the Safety and CArdioprotective effects of eniporide in acute Myocardial Infarction; APEX AMI, Assessment of Pexelizumab in Acute Myocardial Infarction; TRIUMPH, Tilarginine Acetate Injection in a Randomized International Study in Unstable MI Patients With Cardiogenic Shock; REVIVAL GIPS, Glycometabolic Intervention as Adjunct to Primary Percutaneous Coronary Intervention in ST-Segment Elevation Myocardial Infarction; PROTECTION AMI, Inhibition of δ-PROTEin kinase C for the reducTION of infarct size in Acute Myocardial Infarction; INFUSE-AMI, Intracoronary Abciximab and Aspiration Thrombectomy in Patients With Large Anterior Myocardial Infarction; AMIHOT, Acute Myocardial Infarction With Hyperoxemic Therapy; J-WIND, Japan-Working Groups of Acute Myocardial Infarction for the Reduction of Necrotic Damage; ANP, atrial natriuretic peptide; hSOD, human superoxide dismutase; GIK, glucose–insulin–potassium; MPTP, mitochondrial permeability transition pore; ET-1, Endothelin 1; PK, protein kinase.
Antiplatelet Agents
Platelets play a critical role in the initiation and propagation of STEMI. They accumulate rapidly at the site of plaque rupture and provide the proinflammatory and prothrombotic stimuli for the formation of the occlusive thrombus. Thus, platelet activation and aggregation are excellent candidates for pharmacological interventions geared to reduce IS.
Abciximab
Abciximab is an irreversible potent inhibitor of the glycoprotein IIb/IIIa receptor, an integrin molecule on the platelet surface which mediates platelet aggregation. It can be administered intravenously (IV), intracoronary (IC), or locally at the site of the thrombotic lesion (intralesion; IL) via dedicated balloon catheters. Although many trials have assessed the impact of abciximab on clinical end points, data regarding its effect on IS are more limited. Rakowski et al compared early (before transfer to catheterization laboratory) versus delayed (prior to PCI) use of IV abciximab in a small group of high-risk patients with STEMI and its impact on enzymatic IS. Earlier administration was associated with smaller IS (5938 ± 3949 U/L vs 8324 ± 4185 U/L, respectively, P = .04). 45 Conflicting results were obtained in other studies. 46,47 In the Controlled Abciximab and Device Investigation to Lower Late Angioplasty Complications (CADILLAC) trial in which 2082 patients with STEMI were randomized to balloon angioplasty or stenting with versus without intravenous bolus + infusion of abciximab, no significant difference in LVEF or regional wall motion was found at 12 months between the groups. 48 Such was the case also in Bavarian Reperfusion AlternatiVes Evaluation 3 (BRAVE 3) trial (Figure 4). 49

Effect of abciximab on infarct size in the BRAVE-3 trial (reproduced with permission from 49 ). BRAVE-3 indicates Bavarian Reperfusion AlternatiVes Evaluation 3.
Recently, more interest has shifted to IC and IL abciximab administration. Eitel et al compared IC abciximab to IV abciximab in 154 patients with STEMI. The IC abciximab group had significantly smaller IS at 6 months (16.7% vs 24.1%, respectively, P = .002) and a higher rate of aborted MI (16% vs 6%, P = .04). 50,51 In the Comparison of Intracoronary Versus Intravenous Abciximab in ST-segment Elevation Myocardial Infarction (CICERO) trial of 534 patients, the IC abciximab group had significantly smaller enzymatic IS than that of the IV abciximab group (AUC48 [Area-under-the-curve at 48 hours] creatine kinase-myocardial band [CK-MB] of 117 U/L vs 171 U/L, respectively, P = .006). 52 However, the largest comparison of IC and IV abciximab effect was a subset of 795 patients from the Abciximab Intracoronary versus intravenously Drug Application in STEMI (AIDA STEMI) trial that showed no difference between the 2 groups in 1-week IS assessed by CMR (16% vs 17%, respectively, P = .52). 53
A drawback of IC abciximab administration through the guide catheter is that the drug may be inadvertently delivered to the aorta or noninfarct artery, diminishing its effectiveness. This limitation may be overcome by IL abciximab administration directly at the infarct site. In the INFUSE-AMI study, 453 patients with STEMI were randomized to IL abciximab (0.25 mg/kg bolus of abciximab administered IL via the Clearway RX local drug delivery catheter) versus no abciximab. The IL abciximab group had a significant reduction in the primary end point of IS by CMR at 30 days compared to the no abciximab group (median 15.1% vs 17.9%, respectively, P = .03). 34 A larger outcomes trial is needed to confirm whether this modest reduction in IS results in clinical benefits without excessive bleeding.
Tirofiban
Tirofiban is a reversible small molecule inhibitor of the GP IIb/IIIa receptor. Intracoronary tirofiban reduced IS in comparison to IV administration in a small study of 49 patients, while earlier IV administration was better than delayed administration in a similarly sized pilot study. 54
ADP antagonists
Clopidogrel loading doses of 600 mg or 300 mg were compared in 201 patients undergoing primary PCI for STEMI. The higher dose had lower enzymatic IS (median area under the curve [AUC] CK-MB 2070 ng/mL vs 3049 ng/mL, respectively, P = .0001). 55 No studies have examined the effect of the more potent ADP antagonists prasugrel or ticagrelor on LV function.
Adenosine
There are 4 receptor types for adenosine on myocardial cells and blood vessels—A1, A2a, A2b, and A3 with various distribution patterns among various organs (heart, brain, lung, and kidney). Several hypotheses related to the cardioprotective effects of adenosine have been proposed. Prominent among them is dilation of coronary and systemic arteries which improves local myocardial blood flow and reduces cardiac oxygen demand and RI. Other potential mechanisms are simulation of pre- and postischemic conditioning mechanics, prevention of platelet and inflammatory cell activation, and generation of oxygen free radicals and cellular calcium overload. Adenosine also appears to reduce apoptosis. 56
In the randomized AMISTAD trial, IV adenosine for 3 hours reduced by 33% SPECT IS (P = .03), without effect on clinical outcomes. The benefit was more pronounced in anterior infarcts. 39 In the subsequent pivotal AMISTAD II trial, 2118 patients with STEMI undergoing reperfusion with either fibrinolysis or PCI were randomized to placebo, low-dose, or high-dose IV adenosine. Only high dose (70 µg/kg/min) reduced IS (11% vs 27% in the placebo group, P = .023) without impact on clinical outcomes (Figure 5). 16 Comparisons of IC adenosine versus placebo in 2 studies (N = 558, combined) yielded no differences in IS measured either enzymatically or by CMR. 57,58

Effect of adenosine on infarct size in AMISTAD II (reproduced with permission from 16 ). AMISTAD indicates Acute Myocardial Infarction Study of Adenosine.
Trimetazidine
Experimental studies performed in various animal models of myocardial ischemia have demonstrated the anti-ischemic and antioxidant properties of trimetazidine (TMZ). It prevents a decrease in intracellular levels of adenosine triphosphate (ATP) in response to ischemia, inhibits oxidation of fatty acids, and promotes utilization of glucose metabolism. Steg et al evaluated the benefits of TMZ as adjunctive therapy to primary PCI for AMI in a prospective, double-blind, placebo-controlled pilot trial. There was no difference in enzymatic IS between the 2 groups (P = .1). 22
Hyperoxemia
In experimental models of STEMI, the IC delivery of aqueous supersaturated oxygen (SS
Dixon et al first demonstrated that hyperoxemic therapy may be safely delivered in a pilot study.
35
In the Acute Myocardial Infarction With Hyperoxemic Therapy (AMIHOT) trial, 269 patients with anterior or inferior STEMI undergoing primary or rescue PCI within 24 hours were randomized to receive hyperoxemic reperfusion or normoxemic blood autoreperfusion for 90 minutes after reperfusion, delivered directly to the myocardial infarct zone though a catheter placed in the infarct artery. There was no significant difference in SPECT IS at 14 days between the 2 groups (median IS 11% vs 13%, respectively, P = .30). A signal for benefit was noted in patients treated within 6 hours of symptom onset, however, especially in those with anterior infarcts.
39
This observation prompted the AMIHOT II trial, in which 301 patients with anterior STEMI undergoing PCI within 6 hours of symptom onset were randomized to a 90-minute SS

Effect of supersaturated oxygen on infarct size in AMIHOT and AMIHOT II (adapted from 38 ). SSO2 denotes supersaturated oxygen; AMIHOT, Acute Myocardial Infarction With Hyperoxemic Therapy.
Sodium Hydrogen Exchange Inhibition
Sodium hydrogen (Na/H) exchange in cardiac myocytes plays a pivotal role in regulating intracellular pH, sodium, and calcium ion homeostasis. The Na/H exchanger is directly involved in myocardial necrosis, as it mediates calcium overload following myocardial ischemia and reperfusion. Buerke et al evaluated the effect of inhibition of Na+/H+ exchange with cariporide on IS in a multicenter, randomized, placebo-controlled clinical trial of patients treated with primary PCI. There was a lower AUC for CK-MB in the cariporide group, P = .047, accompanied by significant differences in LV function recovery between the 2 groups. 61
Zeymer et al studied another Na+/H+ exchange inhibitor, eniporide in the Evaluation of the Safety and CArdioprotective effects of eniporide in acute Myocardial Infarction (ESCAMI). There were no significant differences in enzymatic IS (AUC for α-HDBH [alpha-hydroxybutyrate dehydrogenase] was 41.2 ± 28.5 in the placebo group, 43.0 ± 26 in the 100 mg eniporide group, and 41.5 ± 25.9 in the 150 mg eniporide group, P = not significant [NS]). There were also no significant differences in clinical events between the groups (Table 2). 26
Clinical Outcomes in the ESCAMI Triala.
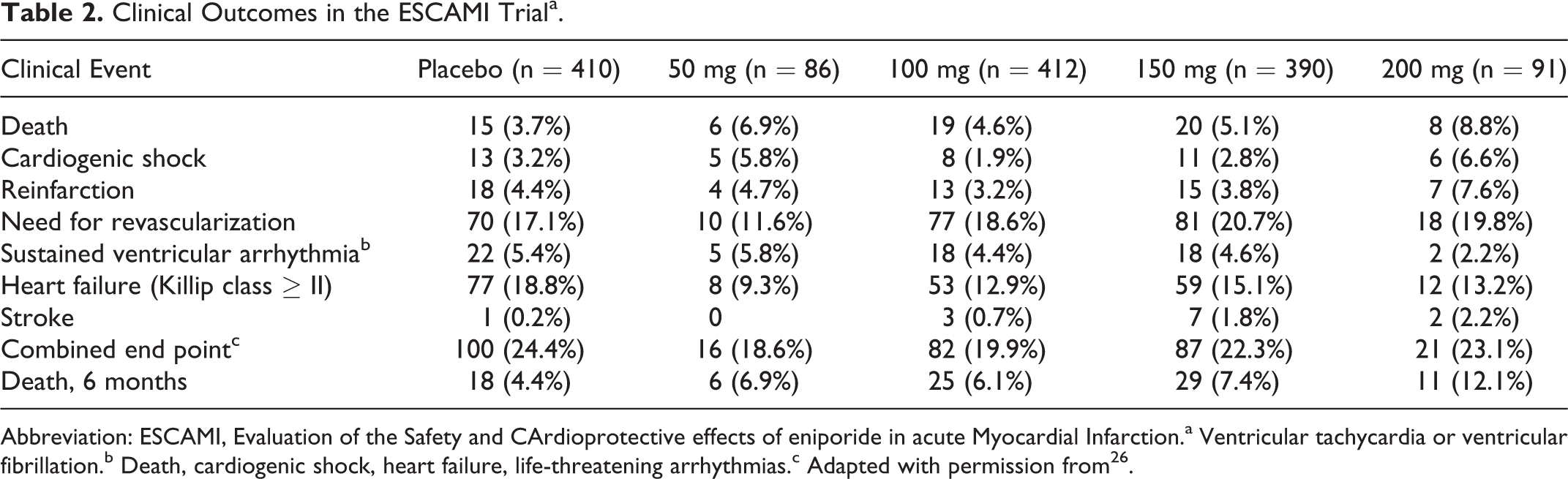
Abbreviation: ESCAMI, Evaluation of the Safety and CArdioprotective effects of eniporide in acute Myocardial Infarction.a Ventricular tachycardia or ventricular fibrillation.b Death, cardiogenic shock, heart failure, life-threatening arrhythmias.c Adapted with permission from 26 .
Atrial Natriuretic Peptide
Atrial natriuretic peptide (ANP) is secreted during myocardial ischemia or infarction and plays an important role in regulating blood pressure and volume through its natriuretic and vasodilatory effects. In experimental models, pharmacological doses of ANP administered during coronary ischemia and reperfusion reduced IS. 62 The suggested mechanisms were that ANP increased coronary collateral blood flow and also lowered end-diastolic pressure. Hayashi et al evaluated the effect of ANP compared to nitroglycerin (NTG) on LV remodeling after first anterior AMI in 60 patients. There was significant improvement in LVEF with ANP infusion compared to NTG (46.9% ± 1.0% to 54.6% ± 1.1% vs 46.8% ± 1.3% to 50.8% ± 1.3%, respectively, P< .05). 63 In the Japan-Working Groups of Acute Myocardial Infarction for the Reduction of Necrotic Damage (J-WIND) study, 569 patients with STEMI were randomly assigned to ANP or placebo for 3 days. The AUC CK was significantly lower with the ANP compared to placebo, resulting in 14.7% reduction in IS (P = .016). Correspondingly, there was significant improvement in the LVEF with ANP (44.7% vs 42.5%, respectively, P = .024). 37
Nitric Oxide
Nitric oxide and its donors may reduce RI and IS by decreasing preload, afterload, wall stress, and resultant myocardial oxygen demand, reducing coronary spasm and improving collateral blood flow. Rentrop et al randomly assigned 393 patients with AMI to IC infusion of streptokinase, NTG, or both. There was a significant increase in ejection fraction (EF) compared to baseline (3.9%) in the combined treatment arm, compared to the other 2 groups (P < .001). 64 Nitroglycerin alone did not affect EF. Jugdutt randomized 310 patients with STEMI to IV NTG therapy or placebo. Infarct size was significantly smaller in the NTG group compared to the placebo group (mean peak CK-MB 41 vs 55, P < .001). 65 Morris et al evaluated the benefits of IV isosorbide dinitrate therapy on IS in 292 patients with AMI receiving fibrinolytic therapy. There was no significant difference in IS between the 2 groups as assessed by α-hydroxybutyrate dehydrogenase (α-HBDH) release. 66
Large randomized studies of patients with AMI from the fibrinolytic era also failed to show any benefit of oral nitrate therapy on clinical outcomes. 67 However in a recent large registry of patients with ACS, patients on chronic nitrate therapy, irrespective of clinical presentation (STEMI or non-STEMI), had significantly lower levels of median peak CK-MB (41 U vs 101 U; P < .0001) and troponin than nitrate-naive patients. Unfortunately, this effect did not translate to reduced mortality. 68
A different nitric oxide donor, sodium nitrite, was tested in the NIAMI trial and found to have no significant effect on IS, compared to placebo, when administered just before reperfusion. 69
Neutrophil Inhibitors
Neutrophils play an important role in the development of RI by releasing oxygen free radicals, proteases, and proinflammatory mediators. These mechanisms further increase the infiltration of neutrophils into the ischemic myocardium. 70 Targeted attempts at depletion of neutrophils, direct inhibition of neutrophils, and inhibition of cell adhesion molecules on neutrophils have been studied. Although experimental studies showed promise, none of the human trials demonstrated favorable outcomes.
In the Limitation of Myocardial Infarction Following Thrombolysis in Acute Myocardial Infarction (LIMIT-AMI) study, the safety and efficacy of a recombinant, humanized, monoclonal antibody to the CD18 subunit of the β2 integrin adhesion receptors (recombinant humanized monoclonal antibody against CD18 [rhuMAb CD18]) in reducing IS in 394 patients having STEMI treated with a thrombolytic agent was studied. There was no significant impact of rhuMAb CD18 on IS as assessed by SPECT at ≥120 hours. 71 Faxon et al evaluated the impact of Hu23F2G (LeukoArrest), an antibody to the CD11/CD18 integrin receptors, on SPECT IS in 492 patients undergoing primary PCI for an AMI. There was no significant difference in IS between the groups (mean 16% with placebo vs 17.2% and 16.6% in the Hu23F2G 0.3 mg/kg and 1.0 mg/kg groups, respectively; P = NS). 24 Further concern is raised by the potential for higher rates of infection with these treatments.
Complement System Inhibitors
Inhibition of the complement complex may decrease the inflammatory cascade activated by myocardial necrosis and RI. The C5b-9b inhibitor pexelizumab failed to demonstrate a reduction in 90-day mortality in the large Assessment of Pexelizumab in Acute Myocardial Infarction (APEX AMI) study. 25 In a small subset of the study population, pexelizumab-treated patients had smaller median IS at day 3 (10.5% vs 16.2%, P = .02) and day 90 (5.9% vs 12.4%, P = .015) and higher LVEF compared with placebo-treated patients. The median peak CK in the pexelizumab group was also significantly lower than placebo (Table 3). However, after multivariable correction for differences in baseline features, pexelizumab was no longer associated with IS reduction. 72 It is notable that in a previous trial (COMplement inhibition in Myocardial infarction treated with Angioplasty [COMMA]), pexelizumab decreased mortality without substantially affecting IS. Potential explanations for this dissociation included an effect on apoptosis and a lack of activation of the complement system, which is more likely with fibrinolysis. 73
Effect of Pexelizumab on Infarct Size in APEX-AMI Trial.a
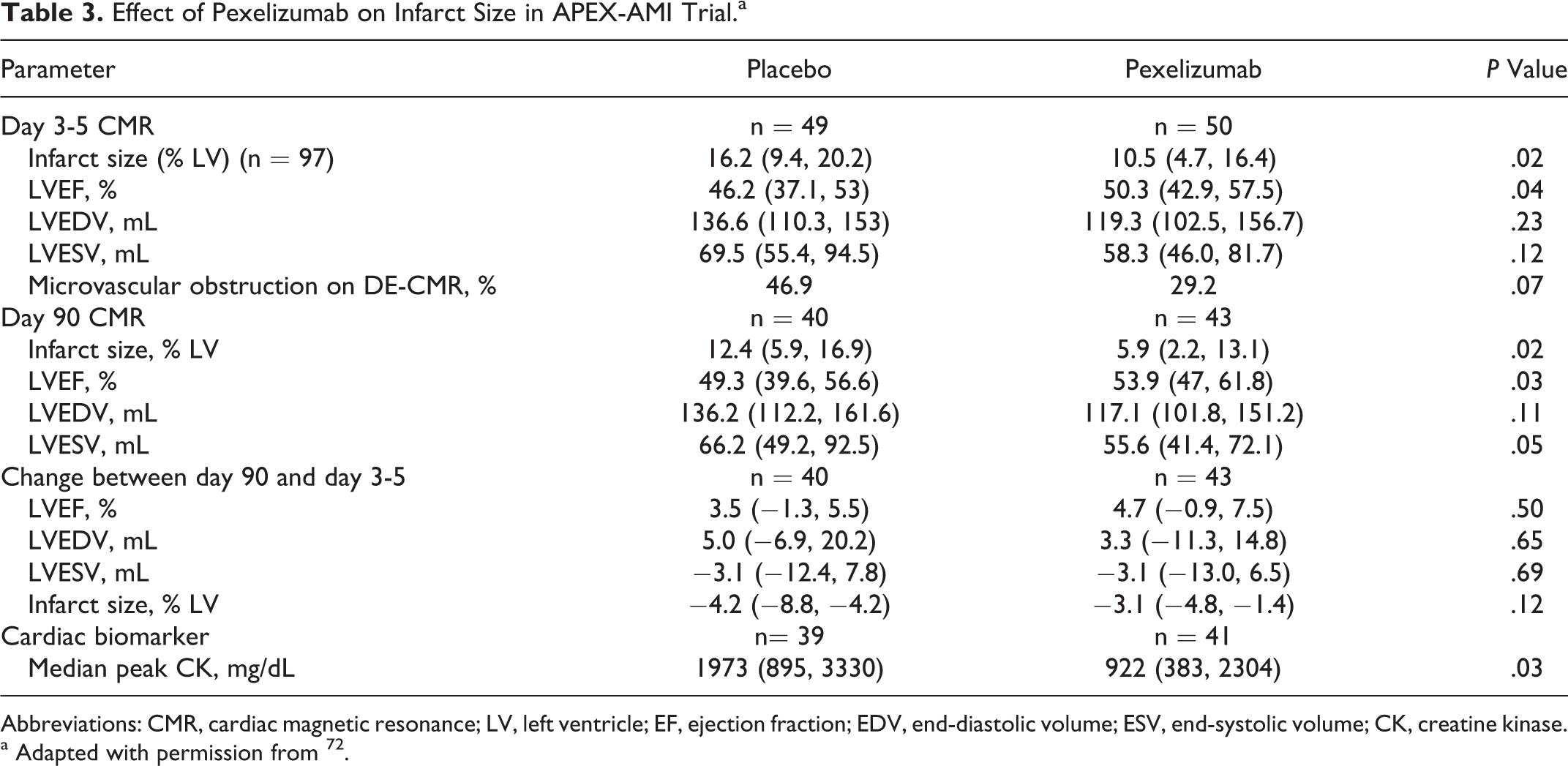
Abbreviations: CMR, cardiac magnetic resonance; LV, left ventricle; EF, ejection fraction; EDV, end-diastolic volume; ESV, end-systolic volume; CK, creatine kinase.
a Adapted with permission from 72 .
Erythropoietin
Erythropoietin (EPO) is secreted from the kidney in response to hypoxic stimuli. Erythropoietin receptors are expressed by hypoxic-ischemic vascular endothelial cells and myocytes. Through these receptors, EPO mediates potential endocrine and autocrine/paracrine actions, such as antiapoptosis and proangiogenic properties, which may reduce IS. Prunier et al randomized 110 patients with STEMI undergoing primary PCI to receive standard therapy (n = 57) or combination therapy with IV administration of 1000 U/kg of EPO-β immediately after reperfusion (n = 53). One of the primary study end points was that CMR imaging assessed IS at 3 months. Although EPO significantly decreased the incidence of microvascular obstruction (43.4% vs 65.3% in the control group, P = .03) and reduced LV volume and function impairment at 5 days (P < .05), there was no significant difference in IS between the 2 groups (median IS 17.5 g in the EPO group vs 16.0 g in the control group; P = .64). 74 In the largest randomized study to date involving 222 patients with STEMI, Najjar et al evaluated the effect of 60 000 units of IV EPO-α, administered within 4 hours of reperfusion, on CMR-measured IS. In the efficacy cohort of the study (n = 138), IS did not differ between groups at either 2 to 6 days (15.8% ± 10.3% vs 15.0% ± 10.0%, P = .67) or at 12 ± 2 weeks (10.6% ± 8.6% vs 10.4% ± 7.6%, P = .87). Moreover, in patients aged ≥70 years (n = 21), mean IS within the first week was larger in the IV EPO-α arm than in the placebo group (19.9% ± 9.9% vs 11.7% ± 7.2%, P = .26); there was also a higher incidence of the composite end point of death, MI, stroke, or stent thrombosis in the IV EPO-α group (4.0% vs 0.0%, P = .04). The very high doses of EPO administered were thought to be responsible for the poor outcomes in these trials. 30 Taniguchi et al evaluated the effect of low-dose IV EPO (3 doses of 6000 IU—during PCI, 24 and 48 hours post-PCI) on SPECT-derived IS in 35 patients with STEMI. The EPO group had significantly lower IS at 6 months compared to acute phase (reduced by 38.5%, P = .003), while the control group did not have favorable remodeling. 75 Larger studies are required to confirm these observations.
Antidiabetic Medications
Exenatide, a glucagon-like-peptide analog, via activation of receptor-mediated survival pathways, increased glucose uptake, inhibition of apoptotic factors, and/or activation of cyclic adenosine monophosphate, and cyclic guanosine monophosphate may reduce RI. 76 Lonborg et al evaluated the cardioprotective effects of exenatide administered at the time of reperfusion in 172 patients with STEMI undergoing primary PCI. The infusion was commenced 15 minutes before intervention and continued for 6 hours postintervention. The primary endpoint was CMR-derived salvage index measured at 90 ± 21 days after MI. A significantly larger salvage index was found in the exenatide group than in the placebo group (0.71 ± 0.13 vs 0.62 ± 0.16, P = .003). The absolute IS between the 2 groups was not significantly different (13 ± 9 g vs 17 ± 14 g, respectively, P = .11). Treatment with exenatide in patients with a shorter delay to PCI was associated with smaller IS (9 vs 13 grams in the placebo group, P = .008). 38
Pioglitazone increases insulin sensitivity and has pleiotropic and anti-inflammatory properties. Experimental studies have demonstrated that pioglitazone may prevent RI. 77 Kataoka et al evaluated the effects of pioglitazone on enzymatic IS in 319 diabetic patients with STEMI. There was a strong trend favoring the pioglitazone group for lower mean peak CK levels (2041 IU/L vs 3207 IU/L; P = .06). Pioglitazone was also associated with a higher incidence of myocardial blush score ≥2 (71% vs 38%; P = .04) as well as better ST-segment resolution. 78
Metformin, a biguanide, enhances phosphorylation of adenosine monophosphate-activated protein kinase and may improve systolic and diastolic functions. The Glycometabolic Intervention as Adjunct to Primary Percutaneous Coronary Intervention in ST-Segment Elevation Myocardial Infarction (GIPS) III trial tested its utility in patients having STEMI without diabetes. At 4 months, there was no difference in CMR EF between the placebo (N = 135) and metformin (N = 136) groups (P = .10), and there was also no difference in incidence of major adverse cardiac events (P = .16). 31
Statins
Several possible mechanisms explaining the cardioprotective effects of statins have been proposed, including improved endothelial function, enhanced plaque stability, and induction of pharmacological ischemic preconditioning via opening of mitochondrial ATP-sensitive channels. Decreased endothelial adhesion molecule receptor expression, increased nitric oxide availability, and decreased production of reactive oxygen species may also improve endothelial function. Additionally, statins promote plaque stabilization by decreasing lipid oxidation, inflammation, and cell death. 79
Kiyokuni et al evaluated the effect of chronic statin therapy on IS. Among 310 patients with STEMI who received fibrinolysis within 12 hours from symptom onset, 39 were on chronic statin therapy and 271 were not. Enzymatic IS was significantly lower in the statin group (AUC CK 55 972 ± 45 934 vs 84 195 ± 84 276 IU, P = .04). 80 However, these findings could not be demonstrated in randomized studies. Hahn et al randomized 173 patients with STEMI undergoing primary PCI within 12 hours after symptom onset to atorvastatin (80 mg before PCI continued for 5 days after PCI) or a control group (10 mg daily after PCI). The SPECT-derived IS measured at 5 to 14 days was not significantly different between the 2 groups (median 19.0% in the atorvastatin group vs 18.0% in the control group, P = .76). 81
Glucose–Insulin–Potassium
Glucose–insulin–potassium (GIK) infusion may provide cardiac protective effects through several mechanisms. 82 High-dose glucose can improve the efficiency of myocardial energy production during acute ischemia by becoming the preferred fuel for the myocardium. Exogenous insulin administration suppresses circulating levels of free fatty acids and decreases their myocardial uptake. Ischemia causes depletion of potassium levels from the myocytes because of local acidosis. Exogenous potassium increases intracellular levels, thereby raising the threshold for ventricular arrhythmias. The effect of GIK infusion in patients with STEMI has been controversial. Pilot studies have demonstrated that GIK infusion is safe and may improve myocardial perfusion and LV remodeling after primary PCI. Meta-analysis of smaller studies, comparing GIK infusion versus control, in almost 5000 patients suggested a reduction in mortality with GIK infusion therapy (hazard ratio = 0.82 [0.68-0.98], P = .03). 83 However, these outcomes were not replicated in large randomized studies. In the Clinical Trial of MEtabolic Modulation in Acute Myocardial Infarction Treatment Evaluation-Estudios Cardiologicos Latinoamerica (CREATE-ECLA) trial, 20 201 patients with STEMI were randomized to receive GIK IV infusion for 24 hours plus usual care or to usual care alone. There was no difference in 30-day mortality between the groups (9.7% vs 10%, P = .45). 84 Diaz et al also reported the absence of a survival benefit at 30 days and 6 months with GIK in a randomized trial of 2748 patients with STEMI. 85 Data on IS were not reported from these studies.
Van der Horst et al evaluated the effect of GIK infusion on enzymatic IS in a randomized controlled trial of 940 patients with STEMI treated with primary PCI. There were no differences in IS between the 2 groups (peak CK-MB level was 249 ± 228 U/L vs 240 ± 200 U/L, P = NS). Mean LVEF at hospital discharge was also similar in the GIK and control groups (43.7% + 11.0% vs 42.4% + 11.7%, respectively, P = .12). 32
Cyclosporine
Multiple animal models have suggested that cyclosporine administered at the time of reperfusion may significantly reduce IS through inhibition of mitochondrial permeability transition pore (MPTP) formation. The MPTP appears to form in the early stages of reperfusion in response to calcium overload and reactive oxygen species generation and allows free passage of molecules <1.5 kDa into the mitochondrial matrix, causing osmotic swelling and destruction of the mitochondria. 86 Piot et al randomly assigned 58 patients with STEMI to receive either an IV bolus of 2.5 mg/kg of cyclosporine or normal saline immediately before PCI. The release of CK was significantly reduced in the cyclosporine group (median 138 053 units vs 247 930 units, P = .04). Infarct size measured by CMR was significantly reduced in the cyclosporine group (median 37 vs 46 grams, P = .04). These benefits on IS persisted 6 months later (29 ± 15 g in the cyclosporine group vs 38 ± 14 g in the control group, P = .04). 40
A similarly acting inhibitor of MPTP, Bendavia, is currently studied in the phase 2a EMBRACE STEMI (Evaluation of Myocardial Effects of Bendavia for Reducing Reperfusion Injury in Patients With Acute Coronary Events) study, enrolling ˜300 patients with first anterior STEMI treated with primary PCI. 87
Nicorandil
Nicorandil acts both as a nitric oxide donor and as a potassium channel opener. 88 It counteracts free radicals and inhibits neutrophil activation which may improve myocardial blood flow and reduce IS. The opening of potassium channels in the ischemic myocardium decreases the action potential, which in turn decreases sodium calcium exchange and calcium overload, potentially reducing RI. In the J-WIND randomized trial, 545 patients with STEMI were assigned to receive either IV nicorandil or matching placebo. There was no difference in the total mean CK activity between the 2 groups (70 520 IU/mL/h with nicorandil vs 70 852 IU/mL/h with placebo, P = .94). 37 Fukuzawa et al studied 62 patients with AMI who were randomly assigned to receive IV nicorandil or placebo. Nicorandil was cardioprotective only in patients with preexisting angina. 98
Renin–Angiotensin System Inhibitors
Renin–angiotensin system (RAS) inhibition may limit IS through several mechanisms. Renin–angiotensin system inhibition reduces angiotensin II levels and has favorable effects on calcium overload, inotropy, diastolic dysfunction, coronary vasoconstriction, free radical production, and degradation of bradykinin and neurohormonal modulation. Together these effects may reduce RI and associated infarct expansion. Several experimental studies have established the positive effects of angiotensin-converting enzyme inhibition (ACEI) and angiotensin receptor blockade on IS reduction. 89 In a large collaborative review of several randomized trials, early administration of an ACEI after MI reduced 30-day mortality by 7% compared to placebo. The benefit was noticeable during the first few days after AMI, suggesting mechanisms other than those associated with LV remodeling. 90 In a retrospective analysis, the effect of pretreatment with RAS inhibitors on enzymatic IS in patients presenting with first STEMI was studied: Compared to patients not taking RAS inhibitors, patients who were taking RAS inhibitors had significantly smaller IS (peak troponin I 79 ng/dL in the RAS inhibitor group vs 120 ng/dL in the no RAS inhibitor group, P = .016). This association persisted even after adjusting for baseline characteristics and treatment. 43
Antioxidants
Generation of oxygen free radicals is believed to be a key contributor to RI. Oxygen radicals react with cellular phospholipids and proteins, causing lipid peroxidation with subsequent alteration in membrane ultrastructure and protein dysfunction. 91 Several oxygen free radical scavengers or antioxidants have been tested in animal studies, including superoxide dismutase, the H2O2 degrading enzyme catalase, the xanthine oxidase inhibitor allopurinol, the iron chelator desferoxamine (DFO), and nonspecific antioxidants including N-acetylcysteine (NAC), vitamin E, and vitamin C. Only a few of these were studied in human trials.
N-acetylcysteine has antioxidant properties and inhibits neutrophil aggregation. Orhan et al evaluated the effect of NAC in limiting myocardial ischemia RI in a randomized trial of 20 patients undergoing coronary artery bypass grafting (CABG). Patients receiving NAC had significantly lower CK-MB release after CABG (39 vs 47 IU/mL at 6 hours, P = .02; 31 vs 44 IU/mL at 12 hours, P = .01). 92 Chan et al investigated the effects of DFO administered to patients with STEMI prior to PCI on CMR IS. Sixty patients were assigned to receive an IV bolus of DFO (500 mg) immediately before PPCI (Primary Percutaneous Coronary Intervention) followed by a 12-hour infusion or placebo. There was no difference in CMR-determined IS (DFO, 17.4% ± 10.8%, vs placebo, 18.6% ± 10.2%, P = .73). Nor were differences in myocardial salvage index or area under the curve for CK or Tn present. 93
Ranolazine
The mechanisms responsible for the cardioprotective effects of ranolazine are unclear. One suggested mechanism is that ranolazine partially inhibits fatty acid oxidation, shifting metabolism during ischemia toward glucose oxidation with increased efficiency of oxygen use. Ischemia increases late sodium current, which is then exchanged for intracellular calcium via the sodium–calcium exchanger. Calcium overload in myocytes leads to mechanical dysfunction and cell death. Ranolazine may prevent this late sodium current. 94
Pelliccia et al performed a randomized study to evaluate the effect of pretreatment with ranolazine prior to elective PCI on periprocedural myocardial damage. Seventy patients with stable angina were assigned to ranolazine (1000 mg twice daily for 7 days before the procedure) or placebo. Ranolazine-treated patients had significantly fewer periprocedural MIs (6% vs 22%, P = .04). Peak CK-MB and peak troponin I were also significantly lower in the ranolazine group. 41
Endothelin Receptor Antagonists
Endothelin 1 is a potent vasoconstrictor peptide abundantly released during ischemia and reperfusion. Endothelin 1 enhances neutrophil adhesion, reactive oxygen species generation, fibroblast proliferation, and apoptosis of cardiomyocytes. Endothelin receptor antagonists (ETAs) oppose these effects and thus may prevent RI and reduce IS. 95 In a randomized, double-blind, placebo-controlled trial, Adlbrecht et al enrolled 57 patients with STEMI to receive IV BQ-123 or placebo, at the onset of PCI. The ETA group had significantly smaller median CK release (1365 U/L vs 2132 U/L, P = .01). The ETA group also had significantly better recovery of LVEF (63% vs 59%, P = .047). 42
Calcium Channel Antagonists
Diltiazem may benefit patients with ischemia and myocardial injury by improving vascular reactivity, reducing shear stress, and by promoting favorable hemodynamic conditions. Theroux et al evaluated the effects of IV followed by oral diltiazem on IS in a pilot study of patients with AMI. There were no significant differences in enzymatic IS between the 2 groups (median peak CK 2751 IU/L in the diltiazem group vs 2336 IU/L in the placebo group, P = NS). There were also no significant differences in LVEF recovery assessed at 6 weeks. However, the diltiazem group unexpectedly had reduced rates of death, reinfarction, or recurrent ischemia at 35 days (13% vs 41%, respectively, P = .02). 96 Pizzetti et al studied the effects of diltiazem, administered before coronary reperfusion on IS, residual myocardial viability, and recovery of LV function. Ninety patients with AMI admitted within 3 hours of symptom onset were randomized to IV diltiazem (n = 43) or placebo (n = 47) and subsequently treated with tissue plasminogen activator. The peak CK release was significantly higher in the placebo group (2931 ± 2456 vs 1726 ± 1004 IU/L, P < .05). Correspondingly, residual myocardial viability and recovery of LVEF were significantly higher in the diltiazem group. 44 Larger studies are required to confirm these observations.
Protein Kinase Inhibitors
Protein kinase C inhibitors (PKCs) prevent the transduction of signals from cell membrane to intracellular locations. As such, inhibition of PKC may result in enhanced recovery of intracellular acidosis, preservation of mitochondrial function, and restoration of cellular energy stores, thereby reducing myocyte and endothelial cell injury after an ischemic insult. There are at least 10 isoforms of PKC and inhibition of the ∊ isoform seems to promote preconditioning. The δ-PKC inhibitor, KAI-9803, prevents localization of δ-PKC to the mitochondria and may restore cellular energy stores and reduce acidemia. It was tested in a dose-escalation study of patients with anterior STEMI undergoing primary PCI. There were encouraging signals in the overall active drug cohort with respect to enzymatic IS and STR (ST-segment resolution), while toxicity was tolerable. Yet, there was no clear dose–response relationship in this study. 97
Delcasertib, also a mitochondrial δ-PKC inhibitor, which may inhibit apoptosis, counteract free radical species, and prevent RI, was tested in escalating doses in the Inhibition of δ-PROTEin kinase C for the reducTION of infarct size in Acute Myocardial Infarction (PROTECTION AMI) study. Among 908 patients with anterior MI randomized to placebo or delcasertib at 50, 150, and 450 mg/h infusion for 2.5 hours after primary PCI, there was no difference in mean CK-MB AUC between the groups (P = .34) or in mean peak CK-MB (P = .72); there was also no difference in time to stable ST segments in infarct area by continuous 24-hour Holter monitoring or in EF measured by scintigraphy at 3 months. Similar results were noted in a smaller cohort of patients with inferior STEMI randomized to placebo or delcasertib 450 mg/h infusion. 33
Ongoing Investigations for IS Reduction
Among the many approaches to reduction in IS mentioned earlier, some are under intense and advanced clinical investigation. Larger ongoing clinical studies with cyclosporine are eagerly anticipated (CYCLosporinE A in Reperfused Acute Myocardial Infarction [CYCLE]—NCT01650662 and Does Cyclosporine ImpRove Clinical oUtcome in ST Elevation Myocardial Infarction Patients [CIRCUS]—NCT01502774). A large supplemental oxygen study (DETermination of the Role of OXygen in Suspected Acute Myocardial Infarction [DETOX2-AMI], NCT01787110) is currently underway evaluating mortality and surrogate end points in 6600 patients. The PRESERVATION 1 study (NCT01226563; IK-5001 for the Prevention of Remodeling of the Ventricle and Congestive Heart Failure After Acute Myocardial Infarction) tests the utility of a combination of sodium alginate and calcium gluconate to prevent unfavorable remodeling. The Nitric Oxide for Inhalation in Myocardial Infarction Size (NOMI) study (NCT01398384) tests the effects of inhaled nitric oxide on IS. Larger studies with exenatide are currently ongoing (Myocardial Protection of Exenatide in AMI [EMPIRE], NCT01580514). Finally, the microvascular obstruction (MVO) study (NCT01747174) evaluates IC adenosine or nitroprusside on IS.
Conclusions
The beneficial effects of reperfusion therapy are mitigated by RI, which increases IS. Despite intensive research into strategies for reducing RI and IS in patients with STEMI (and after dozens of successful approaches in animals), there is as of yet no clear pharmacological intervention that has been shown to enhance myocardial salvage in a consistent and clinically relevant fashion. Favorable avenues for future research include cyclosporine, adenosine analogs, exenatide, SS
Footnotes
Author Contributions
S. Sattur contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring itegrity and accuracy. S. J. Berner contributed to conception and design; contributed to acquisition, analysis, and interpretation; critically revised the manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring itegrity and accuracy. G. W. Stone contributed to conception and design; contributed to acquisition and analysis; critically revised the manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring itegrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
