Abstract
Background:
Patients hospitalized with acute myocardial infarction (AMI) are often on prior single antiplatelet therapy (SAPT) or a dual antiplatelet therapy (DAPT). Whether chronic SAPT or DAPT is beneficial or associated with an increased risk in AMI is still controversial.
Methods and Results:
We prospectively enrolled 1718 consecutive patients with AMI (798 ST-segment elevation myocardial infarction and 920 non-ST-segment elevation myocardial infarction) who were divided according to their chronic APT (no APT, SAPT, or DAPT). The study primary end point was the infarct size, as estimated by troponin I peak. Incidence of major bleeding was also evaluated. Five hundred thirty-six (31%) patients were on chronic SAPT and 215 (13%) on DAPT. A graded increase in Global Registry of Acute Coronary Events (GRACE) and Can Rapid risk stratification of Unstable angina patients Suppress ADverse outcomes with Early implementation of the ACC/AHA guidelines (CRUSADE) risk scores was found going from patients without APT to those with DAPT, while a progressive smaller troponin I peak was observed with the increasing number of chronic antiplatelet agents (11.2 [interquartile range: 2-45] ng/mL, 6.6 [1-33] ng/mL, and 4.1 [1-24] ng/mL;
Conclusion:
In patients with AMI, chronic APT is associated with higher baseline ischemic and bleeding risks. Despite this and unexpectedly, they have a smaller infarct size and similar adjusted bleeding risk.
Introduction
Antiplatelet therapy (APT) is the mainstay of current acute myocardial infarction (AMI) therapy. In particular, aspirin administration is recommended immediately at hospital presentation and then indefinitely after AMI in all patients without contraindications, irrespective of the revascularization strategy. 1,2 There is general agreement on starting a P2Y12 inhibitor soon after AMI diagnosis and following elective percutaneous coronary intervention and prolonging the administration for the first year after the index event or for a longer period in high-risk patients. 3,4 Moreover, an increasing number of patients are chronically treated with APT for medical management of chronic coronary and peripheral artery disease. Thus, in the current clinical practice, a substantial number of patients with AMI may be already on single antiplatelet therapy (SAPT) or dual antiplatelet therapy (DAPT) at the time of hospital presentation.
Initial studies reported that chronic aspirin therapy is associated with higher risk in the event of AMI, possibly reflecting a kind of pharmacological “resistance.” 5,6 However, conflicting results, mainly derived from retrospective analysis of large clinical trials, have been recently reported leaving unsettled whether chronic aspirin is a risk or a protective factor for in-hospital outcome in AMI. 7 -10 This issue is even more controversial in patients with chronic DAPT. On one hand, AMI occurrence in patients on DAPT may indicate a “double resistance” to antithrombotic treatment, a high-risk condition that carry out a threat of larger myocardial infarct size and higher bleeding risk. On the other hand, chronic DAPT may protect against the thrombotic burden during the early phase of AMI due to its antithrombotic effect, limiting the extension of the necrotic area. Thus, the net risk–benefit ratio of chronic APT in patients hospitalized with AMI remains unclear. In this study, we investigated the clinical effects of chronic SAPT or DAPT on myocardial infarct size, as estimated by troponin I peak value, and major bleeding events in consecutive real-world patients with AMI.
Materials and Methods
Study Patients
This prospective, single-center study was conducted at the Centro Cardiologico Monzino between June 4, 2010, and June 25, 2016. We enrolled all consecutive patients with AMI (both ST-segment elevation myocardial infarction [STEMI] and non-ST-segment elevation myocardial infarction [NSTEMI]). Patients experiencing AMI after elective percutaneous coronary intervention (postprocedural AMI), those with documented coronary stent thrombosis, those with uncertain APT compliance, and those treated with oral anticoagulant therapy were excluded from the study.
The institutional review board of our institute approved the study (R519-CCM548). Written informed consent was obtained from all participants and the study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki.
Study Design
Patients were interviewed at admission on APT type, duration, and compliance with prescription before the index event. Information about chronic aspirin and/or thienopyridine use, defined as therapy taken regularly in the previous 30 days before admission, was collected and recorded in the case report form. Demographic, clinical, biochemical, echocardiographic, and angiographic data were also obtained. Troponin I (Beckman Coulter, Fullerton, California) was measured every 6 hours from hospital admission to 24 hours after reaching the peak value. Left ventricular ejection fraction (LVEF) was measured by echocardiogram in all patients within 24 hours from hospital admission. At hospital admission, ischemic and bleeding risks were assessed using the Global Registry of Acute Coronary Events (GRACE) and Can Rapid risk stratification of Unstable angina patients Suppress ADverse outcomes with Early implementation of the ACC/AHA guidelines (CRUSADE) scores, respectively. 11,12
All patients received standard medical treatment and underwent coronary revascularization on the basis of the current standards of care recommended by published guidelines. The primary end point of the study was the infarct size, as estimated by troponin I peak value. Major bleeding, defined as the need for blood transfusion (performed when hemoglobin value was ≤8 g/dL), or hemorrhagic stroke was evaluated as secondary end point. Moreover, a composite clinical end point of in-hospital mortality, acute pulmonary edema, cardiogenic shock, and major bleeding was also considered.
Statistical Analysis
A sample size of 1700 patients was calculated under the assumption of a 25% difference in mean troponin I peak value going from patients without APT to those with chronic SAPT and from them to those with chronic DAPT. This sample size allowed a 90% statistical power to deem as significant (α error of .05).
Continuous variables are presented as mean (standard deviation, SD). Variables with a skewed distribution are presented as median and interquartile range. Categorical data are presented as n (%). Trends across groups (no APT, SAPT, and DAPT) were assessed by Analysis of covariance and Mantel-Haenszel χ2, as appropriate.
The association between troponin I peak and chronic APT was assessed by general linear model analysis, adjusted for potential confounders. The variables included in the model were selected according to the so-called epidemiological approach by choosing those recognized to have potential influence on enzymatic infarct size (baseline risk profile, as assessed by the GRACE risk score, AMI type [STEMI vs NSTEMI], AMI location [anterior vs nonanterior], time to presentation, prior statin, and prior β-blocker use). Results are expressed as adjusted geometric means with 95% confidence intervals (CIs).
The association between bleeding and chronic APT was assessed by multiple logistic regression analysis, which was adjusted for the CRUSADE bleeding risk score. Results are presented as odds ratio (OR) with 95% CI.
All tests were 2 tailed, and
Results
A total of 1718 consecutive patients with AMI (mean age: 67 [13] years, 1263 men) with STEMI (n = 798) and NSTEMI (n = 920) were included in this study. Information on chronic APT was available in all patients. In particular, 536 (31%) patients were on chronic SAPT and 215 (13%) were on chronic DAPT before hospital admission.
Table 1 shows the baseline characteristics of study patients according to prior APT. Patients with chronic APT were older, had higher incidence of comorbidities (diabetes mellitus, hypertension, hyperlipidemia, and known coronary artery disease), and were more likely to have STEMI, lower estimated glomerular filtration rate, and LVEF. A progressive increase in GRACE and CRUSADE risk scores was found going from patients with no APT to those with SAPT and ending with those with DAPT. In-hospital clinical complications are reported in Table 2. In particular, in-hospital mortality and composite clinical end point rates were similar in the 3 groups.
Baseline Characteristics of Study Patients According to Their Prior Antiplatelet Therapy.

Abbreviations: ACE, angiotensin-converting enzyme; AMI, acute myocardial infarction; APT, antiplatelet therapy; ARB, angiotensin II receptor blocker; CABG, coronary aortic bypass graft; CRUSADE, Can Rapid risk stratification of Unstable angina patients Suppress ADverse outcomes with Early implementation of the ACC/AHA guidelines; DAPT, dual antiplatelet therapy; eGFR, estimated glomerular filtration rate; GRACE, Global Registry of Acute Coronary Events; hs-CRP, high-sensitivity C-reactive protein; LAD, left anterior descending coronary artery; LVEF, left ventricular ejection fraction; MI, myocardial infarction; PCI, percutaneous coronary intervention; SAPT, single antiplatelet therapy; STEMI, ST-segment elevation myocardial infarction.
a
bIn STEMI patients only.
In-Hospital Adverse Events and Troponin I Peak Value of Study Patients According to Prior Antiplatelet Therapy.
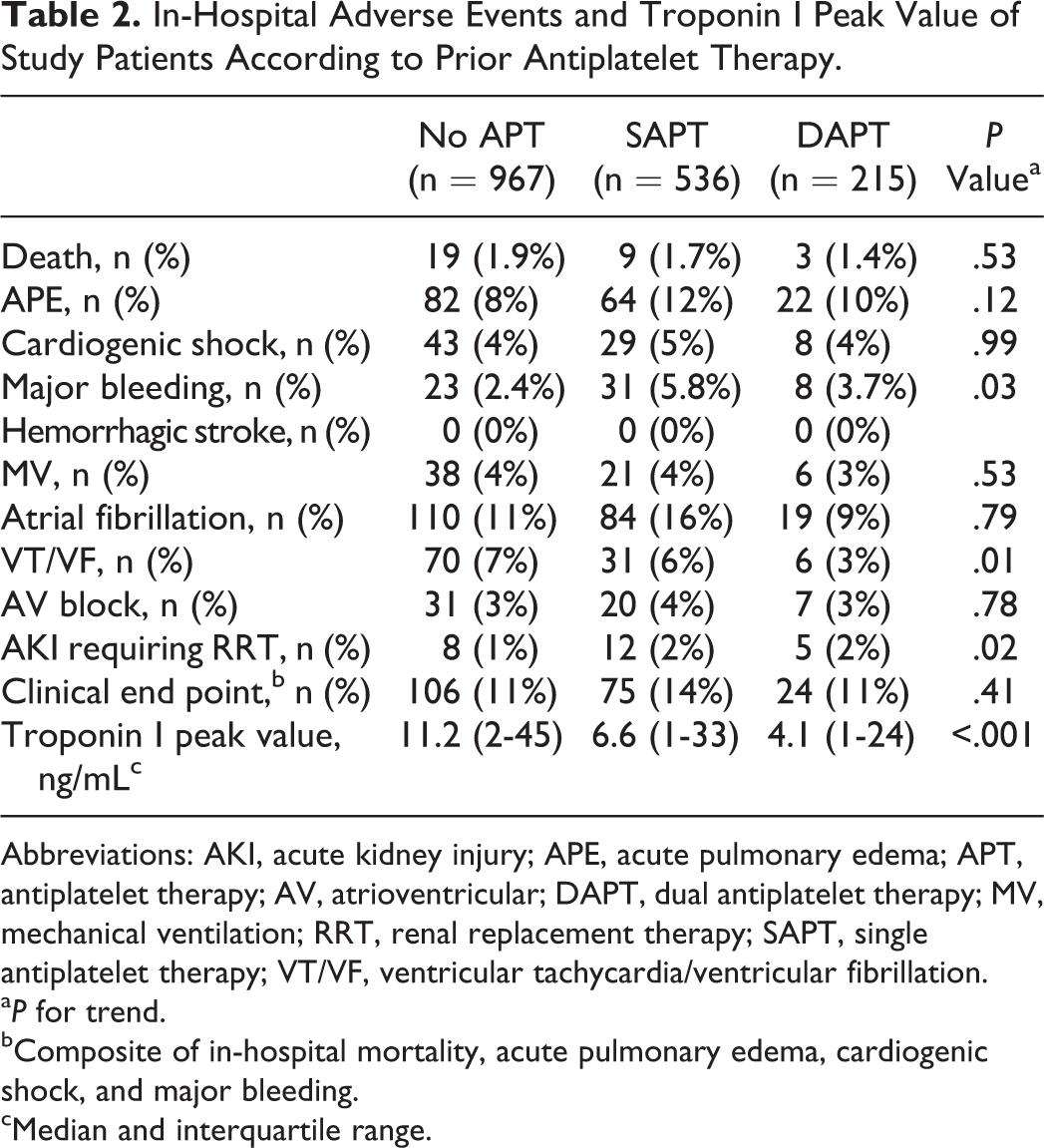
Abbreviations: AKI, acute kidney injury; APE, acute pulmonary edema; APT, antiplatelet therapy; AV, atrioventricular; DAPT, dual antiplatelet therapy; MV, mechanical ventilation; RRT, renal replacement therapy; SAPT, single antiplatelet therapy; VT/VF, ventricular tachycardia/ventricular fibrillation.
a
bComposite of in-hospital mortality, acute pulmonary edema, cardiogenic shock, and major bleeding.
cMedian and interquartile range.
In the entire population, a close and direct relationship between troponin I peak levels and GRACE risk score was detected (Figure 1). A progressively lower troponin I peak value was observed with the increasing number of chronic antiplatelet agents (Figure 2). At general linear model analysis, the significant trend in infarct size reduction among groups was maintained after adjustment for patient’s baseline risk profile, as estimated by the GRACE risk score, AMI type, AMI location, time to presentation, and chronic treatment with statins and β-blockers. The adjusted geometric mean values for troponin I peak were 8.2 (95% CI: 7.3-9.3) ng/mL, 6.7 (95% CI: 5.7-7.9) ng/mL, and 5.0 (95% CI: 3.8-6.6) ng/mL in patients with no APT, SAPT, and DAPT, respectively (
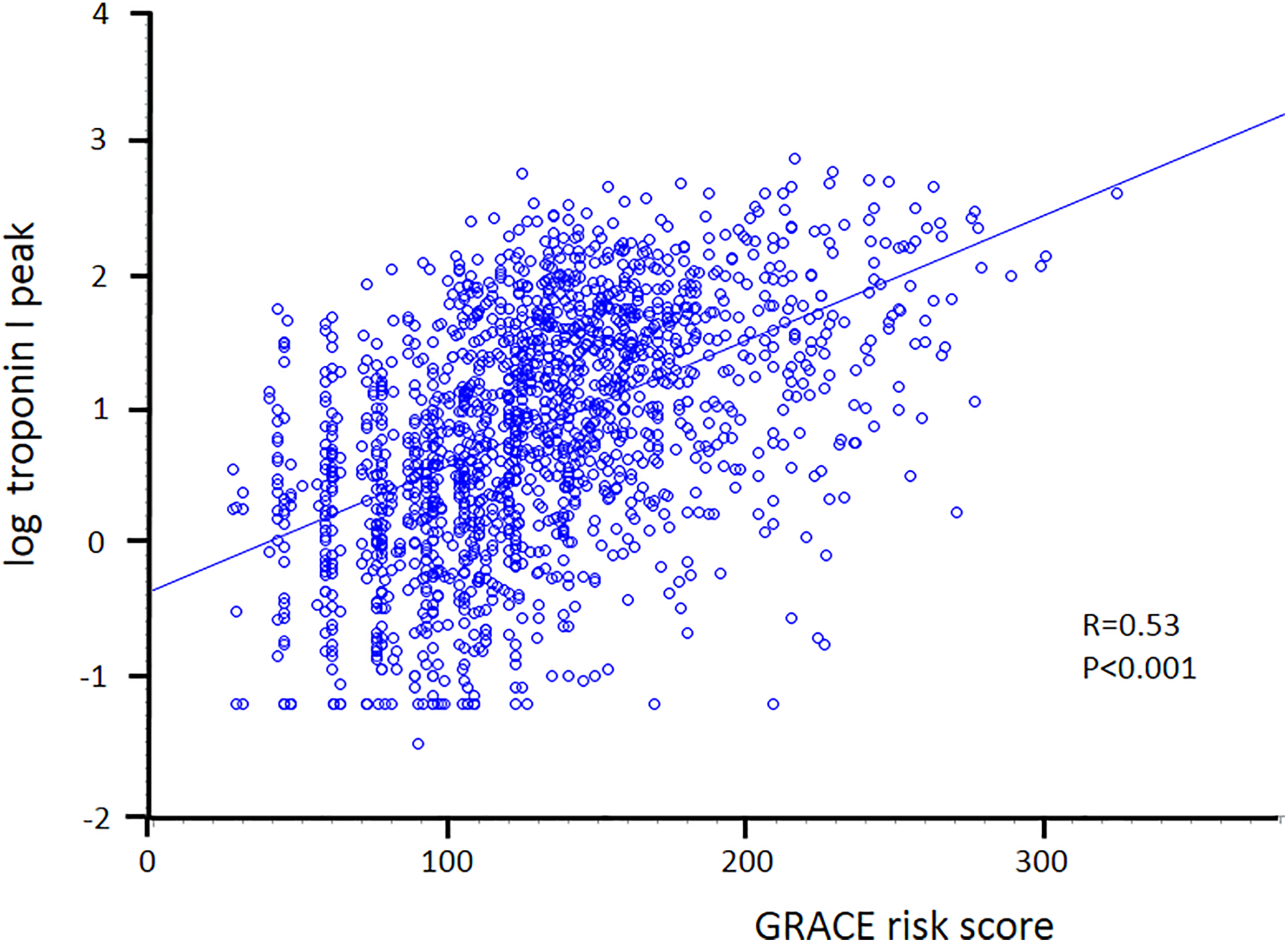
Correlation of troponin I peak value (log-transformed) with Global Registry of Acute Coronary Events (GRACE) risk score in the entire study population.

Geometric mean and 95% confidence interval of troponin I peak value in the study population stratified according to chronic APT. APT indicates antiplatelet therapy; DAPT, dual antiplatelet therapy; SAPT, single antiplatelet therapy.
The incidence of major bleeding was higher in patients with chronic APT than in those without APT (5.2% vs 2.4%;

Odds ratio (OR) and 95% confidence intervals for major bleeding in patients with chronic single antiplatelet therapy (SAPT) and dual antiplatelet therapy (DAPT). Patients without chronic antiplatelet therapy were considered as the referent group. Analyses were also adjusted for the Can Rapid risk stratification of Unstable angina patients Suppress ADverse outcomes with Early implementation of the ACC/AHA guidelines (CRUSADE) risk score.
Figure 4 summarizes the progressive increase in baseline ischemic and bleeding risks, as estimated by the GRACE and CRUSADE scores, respectively, in patients with SAPT and DAPT, compared to those without APT, and the similar OR for the composite clinical end point among the 3 groups.
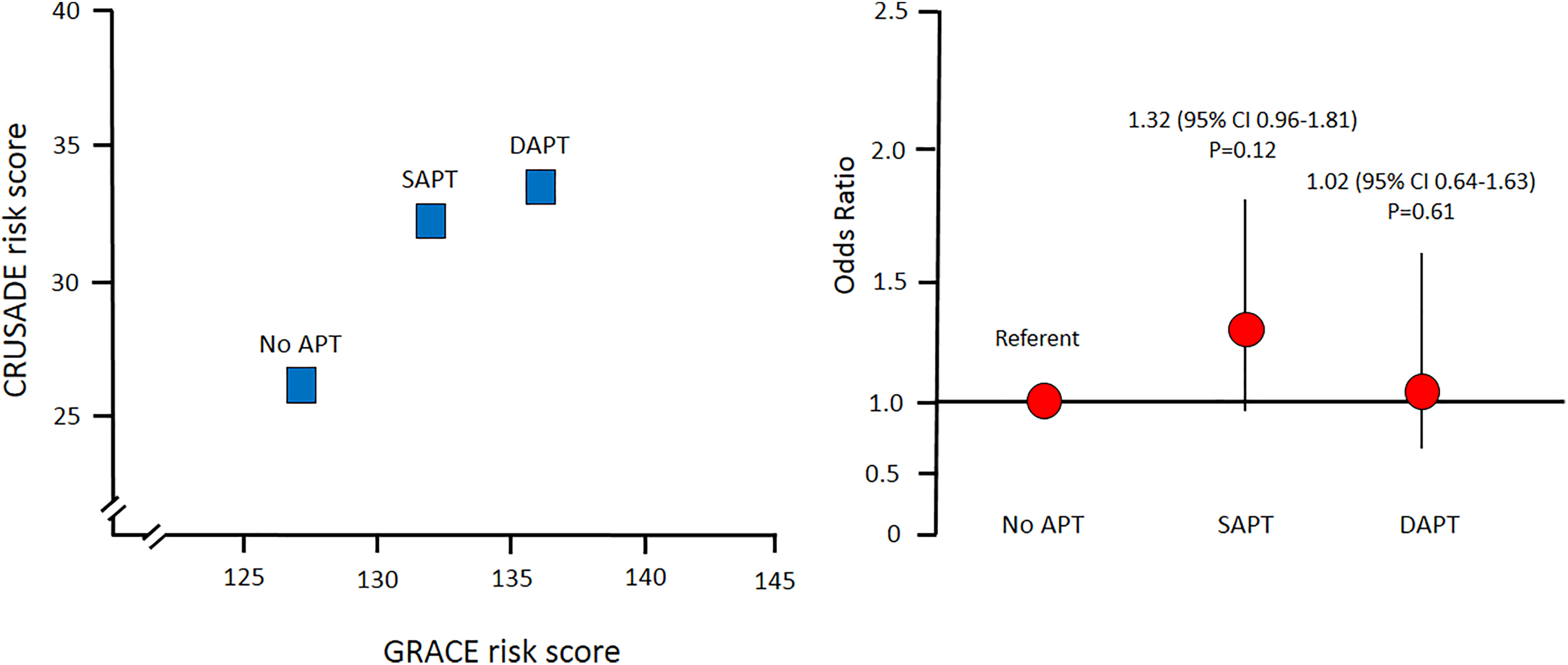
Left panel: Mean Global Registry of Acute Coronary Events (GRACE) and Can Rapid risk stratification of Unstable angina patients Suppress ADverse outcomes with Early implementation of the ACC/AHA guidelines (CRUSADE) risk scores in patients without chronic antiplatelet therapy (APT), with single antiplatelet therapy (SAPT), and dual antiplatelet therapy (DAPT). Right panel: Odds ratio and 95% confidence intervals (CI) for the composite of in-hospital mortality, acute pulmonary edema, cardiogenic shock, and major bleeding (clinical endpoint) in the 3 study groups.
Discussion
The primary finding of this study was that patients on chronic APT hospitalized with AMI have a smaller myocardial infarct size, despite they have higher baseline ischemic risk than patients without chronic APT. This was not associated with an increased risk of major bleeding, when adjusted for the underlying bleeding risk profile.
Usually, patients experiencing AMI while on chronic aspirin therapy are considered a high-risk group, as previous studies have shown that chronic aspirin use is an independent predictor of worse short-term ischemic outcomes. 5,6,13,14 In agreement with these observations, use of aspirin has been included among the risk factors of the widely adopted Thrombolysis in Myocardial Infarction Risk score. 6 Indeed, in the Antman et al’s study, 6 the authors found that chronic aspirin use was associated with an OR of 1.74 of having an ischemic event at 14 days. Notably, this risk was even higher than that of elevated cardiac enzymes. The negative association between chronic aspirin use and worse outcome may be amplified in patients with AMI who are on chronic DAPT, possibly reflecting a sort of “double” hyporesponsiveness to APT. However, recent data questioned these results, suggesting a short-term nondetrimental effect or even a potential benefit associated with chronic APT. 7 -10 Notably, although the clinical benefit of antiplatelet agents in AMI has always been attributed to their antithrombotic action, experimental and clinical studies have demonstrated that some of these agents may reduce infarct size by cardioprotective mechanisms that go beyond their antiplatelet effect. 15 -18 Thus, whether chronic APT represents a risk or a protective factor is still unclear. The issue is relevant because a growing number of patients hospitalized with AMI are already treated with aspirin, both for primary or secondary prevention, and some of them are on DAPT. 19 Moreover, the current treatment of AMI is based on the combined use of potent antithrombotic agents that may expose patients already on APT to a higher bleeding risk. Therefore, we investigated the potential association between chronic SAPT and DAPT and both myocardial infarct size, as assessed by troponin I peak value, and major bleeding risk in patients with AMI treated with contemporary pharmacological and interventional treatments.
In our study, 44% of patients with AMI were on chronic APT, with 31% receiving SAPT while 13% receiving DAPT. These percentages are in agreement with recent studies reporting that a similar proportion of patients hospitalized with AMI are on chronic APT. 10,20,21 As expected, the baseline cardiovascular risk profile of patients on chronic APT was worse than that of patients who were not taking APT. In particular, GRACE risk score progressively increased going from patients not taking APT to those on SAPT and ending with those on DAPT. However, despite higher baseline ischemic risk, patients on chronic SAPT or DAPT had a 42% and 62% lower peak value of troponin I, respectively, compared to patients without APT. This result remained unchanged also after adjustment for major confounders, including GRACE score, AMI type and location, time to presentation, and chronic β-blocker and statin therapy. 22 -25 Notably, because we found a close correlation between GRACE risk score and troponin I peak value, a larger infarct size could be anticipated in patients with chronic APT. Thus, we can speculate that infarct size reduction associated with SAPT and, at a greater extent with DAPT, could have been even more relevant. Concordant with this hypothesis, we found similar in-hospital mortality and complication rates among the 3 groups, despite they were supposed to have different outcomes based on the GRACE score.
We also investigated the relationship between chronic APT and bleeding risk. As expected, we found a higher incidence of major bleeding in patients on chronic APT. However, when the bleeding risk was adjusted for the baseline CRUSADE score, no significant differences were found among the 3 groups. This indicates that in-hospital major bleeding events were mainly related to the baseline risk profile of our patients, rather than to chronic APT. Future studies are warranted to confirm our observations and to clarify their potential clinical implications. Indeed, there is still some uncertainty about the safety and efficacy of antithrombotic therapies in patients hospitalized with AMI who are on chronic SAPT or DAPT. Nonetheless, in aggregate, our data suggest a possible net clinical benefit associated with chronic APT (Figure 4).
The strength of our study, when compared to those published thus far, is the inclusion of a prospectively recent and real-world cohort of consecutive patients with AMI. Indeed, most of previous studies are retrospective analyses of large clinical trials with restricted inclusion criteria. 5,7 -9,26,27 Moreover, we focused our investigation on the potential impact that both SAPT and DAPT may have on myocardial infarct size and bleeding risk, adjusted for the patient’s baseline risk profile. Specifically, we considered peak troponin concentration as our primary end point because it is closely correlated with long-term myocardial function and infarct size, 28 and ATP selectively targets coronary thrombus burden in AMI.
In interpreting our data, some limitations should be considered. First, this is a single-center study whose observational design does not allow establishing a cause–effect relationship between chronic APT and infarct size. Indeed, although the multivariable analysis was adjusted for major confounders, we cannot exclude the presence of unmeasured parameters. Second, determination of chronic APT use was based on patient self-reporting only. Thus, compliance to APT cannot be fully established. Third, our study was not powered to detect any significant difference in mortality that should be the focus of larger prospective studies. Finally, we acknowledge the lack of standardized bleeding classifications in our study. However, in addition to hemorrhagic stroke, we chose blood transfusion because, in the setting of AMI, it reflects bleeding and provides an additional independent risk of short-term mortality. 29 Moreover, our prediction of bleeding risk might have been influenced by the fact that the CRUSADE score does not consider several critical variables associated with bleeding, such as periprocedural antithrombotic therapy and arterial access site (radial vs femoral).
Conclusion
In patients hospitalized with AMI, chronic APT is associated with higher baseline ischemic and bleeding risk profile. Despite this and unexpectedly, they have lower troponin I peak value, which is inversely correlated with the number of chronic antiplatelet agents, and similar adjusted bleeding risk.
Footnotes
Acknowledgments
The authors appreciate the assistance of Eleonora Ecca in data collection.
Authors’ Contribution
Jeness Campodonico, Nicola Cosentino, Antonio L. Bartorelli and Giancarlo Marenzi contributed to conception and design, contributed to analysis and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Valentina Milazzo contributed to conception, contributed to acquisition, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Mara Rubino contributed to design, contributed to acquisition, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Monica De Metrio contributed to acquisition and analysis, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Ivana Marana, Marco Moltrasio, Marco Grazi, Gianfranco Lauri, and Emilio Assanelli contributed to acquisition, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Alice Bonomi contributed to conception, contributed to analysis, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Fabrizio Veglia contributed to analysis, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Elisa Chiorino contributed to acquisition and interpretation, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
