Abstract
Objective
During the first wave of the SARS-CoV-2 pandemic, management of anticoagulation therapy in hospitalized patients with atrial fibrillation (AF) was simplified to low-molecular-weight heparin (LMWH) followed by oral anticoagulation, mainly owing to the risk of drug–drug interactions. However, not all oral anticoagulants carry the same risk.
Methods
Observational, retrospective, and multicenter study that consecutively included hospitalized patients with AF anticoagulated with LMWH followed by oral anticoagulation or edoxaban concomitantly with empirical COVID-19 therapy. Time-to-event (mortality, total bleeds, and admissions to ICU) curves, using an unadjusted Kaplan-Meier method and Cox regression model adjusted for potential confounders were constructed.
Results
A total of 232 patients were included (80.3 ± 7.7 years, 50.0% men, CHA2DS2-VASc 4.1 ± 1.4; HAS-BLED 2.6 ± 1.0). During hospitalization, patients were taking azithromycin (98.7%), hydroxychloroquine (89.7%), and ritonavir/lopinavir (81.5%). The mean length of hospital stay was 14.6 ± 7.2 days, and total follow-up was 31.6 ± 13.4 days; 12.9% of patients required admission to ICU, 18.5% died, and 9.9% had a bleeding complication (34.8% major bleeding). Length of hospital stay was longer in patients taking LMWH (16.0 ± 7.7 vs 13.3 ± 6.5 days; P = .005), but mortality and total bleeds were similar in patients treated with edoxaban and those treated with LMWH followed by oral anticoagulation.
Conclusions
Mortality rates, arterial and venous thromboembolic complications, and bleeds did not significantly differ between AF patients receiving anticoagulation therapy with edoxaban or LMWH followed by oral anticoagulation. However, the duration of hospitalization was significantly lower with edoxaban. Edoxaban had a similar therapeutic profile to LMWH followed by oral anticoagulation and may provide additional benefits.
Background
Coronavirus disease 2019 (COVID-19) is an acute and complex disorder that, in severe cases, is characterized by the development of interstitial pneumonia and acute respiratory distress syndrome. 1 In addition, COVID-19 exposes patients to an increased risk of arterial and venous thrombosis and death. 2 Interestingly, a significant decrease in mortality has been observed with prophylactic use of direct oral anticoagulants (DOACs) or low-molecular-weight heparin (LMWH) in hospitalized COVID-19 patients.3,4 However, despite adequate thromboprophylaxis, many patients with acute respiratory distress syndrome secondary to COVID-19 develop symptomatic thromboembolic events—venous thromboembolism (VTE) in 4.4%, ischemic stroke in 2.5%, and myocardial infarction in 1.1%—suggesting that higher anticoagulation targets may be required in this population. 5 Therefore, it has been recommended that all hospitalized COVID-19 patients should receive prophylactic or full-therapeutic-intensity anticoagulation when indicated. 6
Atrial fibrillation (AF) is the most common arrhythmia in clinical practice 7 and also a frequent complication among patients with COVID-19 infection and severe pneumonia, acute respiratory distress syndrome, or sepsis. 8 Nevertheless, little is known about the optimal management of hospitalized patients with COVID-19 infection. 9 The European Society of Cardiology recently indicated that anticoagulation for the prevention of AF-related stroke or systemic embolism should be guided by the CHA2DS2-VASc score in this population. Specifically, therapeutic anticoagulation should be considered in patients (both sexes) with a CHA2DS2-VASc score ≥1/2 and is indicated in cases with a CHA2DS2-VASc score ≥2/3. 10
During the first wave of the SARS-CoV-2 pandemic, the risk of drug–drug interactions (with antiviral, antiarrhythmic, and anticoagulation agents), difficulties in controlling the international normalized ratio (INR) in hospitalized patients, and difficulties in enabling oral intake in many patients (eg, patients in intensive care, intubated patients) led some authors to suggest that oral anticoagulation with DOACs or vitamin K antagonists should be interrupted and antithrombotic treatment with LMWH started at therapeutic doses.11,12 Therefore, it is important to demonstrate that a DOAC can be used safely during COVID-19 hospitalization.
Not all DOACs carry the same risk of drug–drug interactions. For instance, as the metabolism of edoxaban by CYP3A4 is less than 4%, the risk of drug–drug interactions with this agent is low. 13 Unfortunately, very little information is available on the use of DOACs in hospitalized patients with AF and COVID-19 infection.10,14,15
The present study was performed to compare the effectiveness (major thromboembolic events) and safety (bleeds) of edoxaban with that of LMWH followed by oral anticoagulation among patients with AF who had been hospitalized for COVID-19 infection in Catalonia, Spain. In addition, we analyzed length of stay, the proportion of patients requiring admission to the intensive care unit, and mortality according to anticoagulation patterns.
Methods
Patients
For this study, we included data from the ongoing Registry of thromboembolic and bleeding events in hospitalized patients with COVID-19 (ACO-VID registry) (Protocol code: EORTC1811), which collected information on hospitalized patients with COVID-19. All patients or their healthcare proxies provided written or oral consent for participation in the registry in accordance with standard ethical principles. The study was approved by the Ethics Committee for Clinical Research of the Hospital Universitario Sta. Mª del Rosell, Districts II and VIII of the Health Service of Murcia. The study analyzed data from 5 hospitals located in Catalonia, Spain.
Study Design
This was a multicenter observational study based on a retrospective analysis that consecutively included hospitalized patients with a diagnosis of nonvalvular AF who received anticoagulant treatment with LMWH followed by oral anticoagulation (INR-adjusted [range 2.0-3.0] vitamin K antagonists and DOACs) or edoxaban concomitantly with the empirical therapy used for COVID-19 infection.
Patient Selection
From March 5, 2020 to April 27, 2020, we consecutively included hospitalized adult patients with a diagnosis of COVID-19 infection and nonvalvular AF. COVID-19 infection was confirmed by reverse-transcription polymerase chain reaction assay on a nose/throat swab sample that was positive for SARS-CoV-2. Patients were receiving anticoagulant treatment with LMWH followed by oral anticoagulation (INR-adjusted [range 2.0-3.0] vitamin K antagonists and DOACs) or edoxaban.
Variables
Patient data were retrospectively recorded from the clinical history from the day of admission (also for patients transferred from another hospital) to death, hospital discharge, or last follow-up visit after discharge.
Baseline characteristics included demographics (age, sex, body weight), time to AF, thromboembolic risk (CHA2DS2-VASc), bleeding risk (HAS-BLED), cardiovascular risk factors (hypertension, diabetes), cardiovascular disease (heart failure, prior myocardial infarction, prior stroke, arteriopathy), history of bleeding, and active cancer. We also recorded the type, dose, and duration of anticoagulation therapy, treatment for COVID-19 infection, and complete blood count and biochemical parameters during hospitalization. Patients were managed according to the current clinical practice of the participating hospital (ie, treatment was not standardized).
The outcomes recorded were length of stay, proportion of patients admitted to the intensive care unit, number of bleeds (total and intracranial hemorrhage), arterial and venous thromboembolic events (stroke/systemic embolism, VTE), and death (total death, COVID-19-related deaths, and bleeding-related deaths). Follow-up ran from admission to hospital to death, hospital discharge, or last visit recorded after discharge. All bleeding events were defined according to the International Society on Thrombosis and Haemostasis classification as major, nonmajor clinically relevant, and minor. Major bleeding was defined as fatal bleeding and/or symptomatic bleeding in a critical area or organ and/or bleeding causing a fall in hemoglobin level ≥20 g/L or leading to transfusion of ≥2 units of whole blood or red cells. 16 Data were updated until May 7, 2020.
All variables were compared according to the type of anticoagulant (edoxaban vs LMWH), dose of edoxaban (60 mg vs 30 mg), and dose of LMWH (prophylactic vs intermediate vs therapeutic). Enoxaparin 0.5 mg/kg daily or bemiparin 5000 IU daily was considered the prophylactic LMWH dose. For patients with body weight <120 kg and creatinine clearance >30 mL/min, enoxaparin 1 mg/kg daily, or bemiparin 5000 IU daily was used as an intermediate LMWH dose. Enoxaparin 1.5 mg/kg daily or 1 mg/kg BID or bemiparin 115 mg/kg daily was considered the therapeutic LMWH dose.
Statistical Analysis
Categorical variables were expressed as absolute frequency (n) and relative frequency (%). Continuous variables were expressed as measures of central tendency (mean and median) and dispersion (standard deviation and interquartile range). Categorical variables were compared using the χ2 test or the Fisher exact test, as appropriate. When 2 means were compared, the t test (parametric) or the Mann-Whitney test (nonparametric) was used, as appropriate, and 3 means were compared using analysis of variance (parametric) or the Kruskal-Wallis test (nonparametric), as appropriate. Time-to-event (mortality, total bleeds, and admissions to the intensive care unit) curves were constructed using an unadjusted Kaplan-Meier method. The log-rank test was used to compare outcomes according to anticoagulant therapy, dose of edoxaban, and dose of LMWH. A Cox regression model adjusted for potential confounders was constructed. Age, sex, hypertension, diabetes, prior myocardial infarction, and heart failure were included as potential cofactors, and log-likelihood was calculated. Effect sizes were presented as hazard ratio and 95% confidence interval. Statistical significance was set at 0.05 for all the tests. The data were analyzed using the statistical package IBM Statistics for Windows, Version 22.0 (IBM Corp).
Results
All patients who met the eligibility criteria (adults with nonvalvular AF, anticoagulated according to medical criteria with edoxaban or LMWH followed by oral anticoagulation, concomitantly treated with empirical therapy for COVID-19 infection during hospitalization) were included and followed up from the day of admission to death, hospital discharge, or last follow-up visit after discharge. A total of 232 patients (Edoxaban n = 118, LMWH n = 114) were included (Figure 1). At discharge, patients with LMWH switched to oral anticoagulation (apixaban [39%], edoxaban [9%], vitamin K antagonist [16%], rivaroxaban [14%], dabigatran [2%]), and the remaining patients maintained edoxaban as their anticoagulant therapy.

Study flowchart. Abbreviations: AF, atrial fibrillation; LMWH, low-molecular-weight heparin; VKA, vitamin K antagonists.
The mean age was 80.3 ± 7.7 years, 50.0% were men, the mean CHA2DS2-VASc was 4.1 ± 1.4, and the mean HAS-BLED was 2.6 ± 1.0. Most patients had hypertension and around one-third had heart failure. Regarding previous antithrombotic treatment, most patients were taking vitamin K antagonists before being prescribed edoxaban or LMWH. With respect to COVID-19 therapy during hospitalization, most were taking azithromycin, hydroxychloroquine, ritonavir/lopinavir, and ceftriaxone (Table 1).
Clinical Characteristics and Outcomes in the Total Study Population and According to the Anticoagulant Administered (Edoxaban vs LMWH).

Abbreviations: SD, standard deviation; IQR, interquartile range; VKA, vitamin K antagonist; DOAC, direct-acting oral anticoagulant; AST, aspartate aminotransferase; ALT, alanine aminotransferase; ICU, intensive care unit; CRNMB, clinically relevant nonmajor bleeding; LMWH, low-molecular-weight heparin; VTE, venous thromboembolism; AF, atrial fibrillation.
aMann-Whitney (nonparametric) test was used, as this variable did not follow a normal distribution.
Regarding outcome measures, mean length of stay was 14.6 ± 7.2 days, and mean total follow-up (since admission to last visit) was 31.6 ± 13.4 days. Furthermore, 12.9% of patients required admission to the intensive care unit, 18.5% of patients died, and 9.9% had a bleeding complication (34.8% major bleeding) (Table 1).
Clinical profile and outcomes were analyzed according to the type of anticoagulant (edoxaban vs LMWH followed by oral anticoagulation). Overall, both groups were comparable for most relevant baseline clinical characteristics. Except for length of stay, which was longer in patients taking LMWH (13.3 ± 6.5 vs 16.0 ± 7.7 days; P = .005), the remaining outcomes were similar in both groups (Table 1, Figure 2). The clinical characteristics of patients with thrombotic and hemorrhagic events are summarized in Supplemental Table 1. Outcomes remained similar between groups after adjusting for confounding factors in mortality and total bleeds. However, elderly patients were more commonly admitted to intensive coronary unit (Supplemental Table 2 and Supplemental Figure 1).

Unadjusted survival curves according to treatment (edoxaban vs LMWH followed by oral anticoagulation). Abbreviations: LMWH, low-molecular-weight heparin; ICU, intensive care unit; VKA, vitamin K antagonists.
A total of 118 patients (50.9%) were taking edoxaban. Mean age was 79.6 ± 7.1 years, mean CHA2DS2-VASc was 4.0 ± 1.5, and mean HAS-BLED 2.5 ± 1.0. With respect to the treatment of COVID-19 during hospitalization, most patients were taking azithromycin, hydroxychloroquine, ritonavir/lopinavir, and ceftriaxone. Regarding outcomes, the mean duration of hospitalization was 13.3 ± 6.5 days and the mean duration of follow-up was 32.9 ± 13.0 days. In the edoxaban group, 16.1% of patients required admission to the intensive care unit, 18.6% of patients died, and 7.6% had a bleeding complication (44.4% major bleeding). Clinical profile and outcomes were compared according to the dose of edoxaban: 55.1% of patients were taking edoxaban 60 mg and 44.9% edoxaban 30 mg. The mean CHA2DS2-VASc and mean HAS-BLED scores were higher among patients taking edoxaban 30 mg, as was the proportion of patients with creatinine clearance ≤50 mL/min. With regard to outcomes, there were more admissions to the intensive care unit and total bleeds among patients taking edoxaban 60 mg (21.5% vs 9.4% [log-rank, P = .049] and 12.3% vs 1.9% [log-rank, P = .04]) (Table 2 and Figure 3). These differences remained after adjusting for confounding factors (Supplemental Table 3 and Supplemental Figure 2).

Unadjusted survival curves according to treatment (edoxaban 60 mg vs edoxaban 30 mg). Abbreviation: ICU, intensive care unit.
Clinical Characteristics and Outcomes in Patients Treated With Edoxaban (Edoxaban 60 mg vs Edoxaban 30 mg).

Abbreviations: SD, standard deviation; IQR, interquartile range; VKA, vitamin K antagonist; DOAC, direct-acting oral anticoagulant; AST, aspartate aminotransferase; ALT, alanine aminotransferase; LMWH, low-molecular-weight heparin; ICU, intensive care unit; CRNMB, clinically relevant nonmajor bleeding; VTE, venous thromboembolism; AF, atrial fibrillation.
a Mann-Whitney test (nonparametric) was performed, as this variable did not follow a normal distribution.
A total of 114 patients (49.1%) were taking LMWH followed by oral anticoagulation. Mean age was 80.9 ± 8.2 years, mean CHA2DS2-VASc was 4.2 ± 1.3, and mean HAS-BLED was 2.6 ± 1.0. With respect to the treatment of COVID-19 during hospitalization, most patients were taking azithromycin, hydroxychloroquine, ritonavir/lopinavir, and ceftriaxone. With regard to outcomes, mean length of stay was 16.0 ± 7.7 days, and mean total duration of follow-up (since admission to last visit) was 30.3 ± 13.7 days. In the LMWH group, 9.6% of patients required admission to the intensive care unit, 18.4% of patients died, and 12.3% had a bleeding complication (28.6% major bleeding). The clinical profile and outcomes were compared according to the dose of LMWH (prophylactic, 21.9%; intermediate, 29.0%; therapeutic, 49.1%): 75.4% of patients were taking enoxaparin, 19.3% bemiparin, and 5.3% tinzaparin. The 3 groups were comparable for most relevant baseline clinical characteristics. Mortality rates were higher in patients taking the prophylactic dose of LMWH in the unadjusted analysis (36.0% vs 12.1% vs 14.3%; P = .036, log-rank P = .0026), but not after adjusting for confounders (Table 3, Figure 4, Supplemental Table 4 and Supplemental Figure 3).
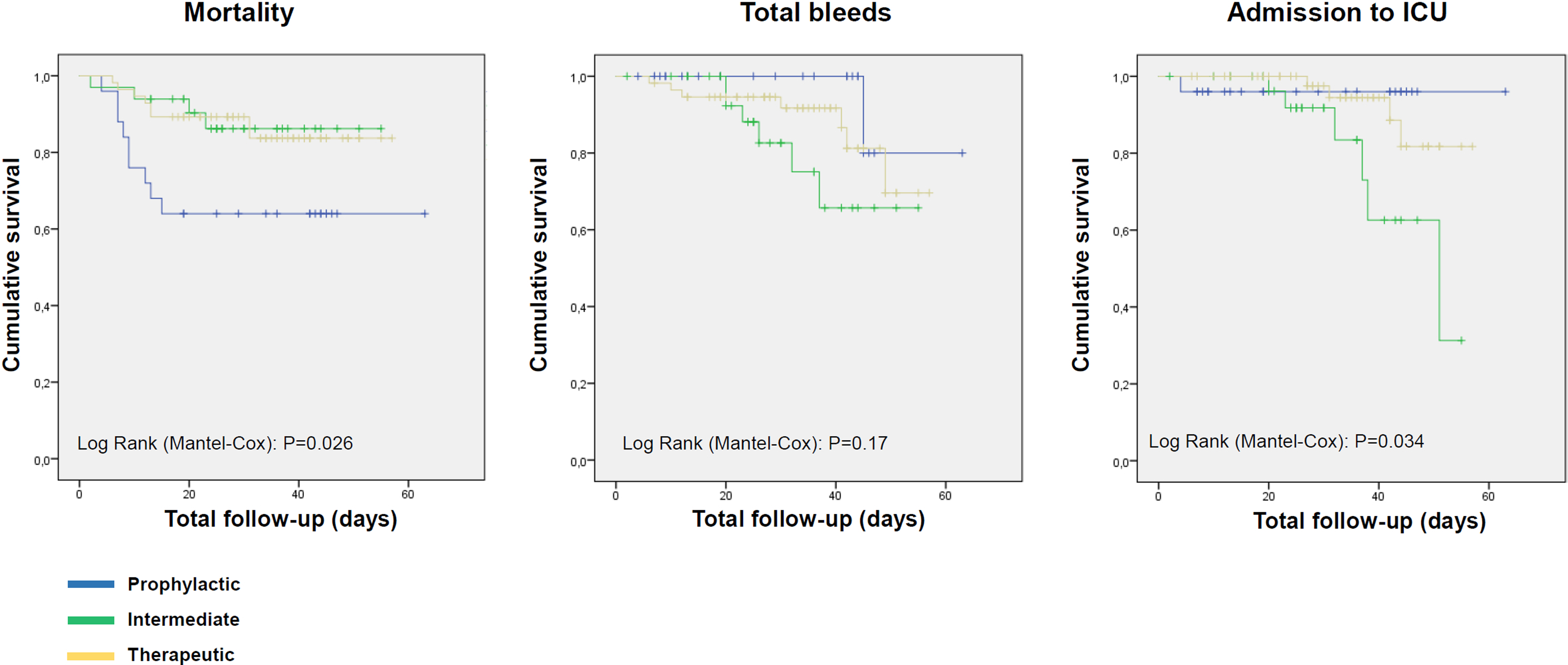
Unadjusted survival curves according to dose of LMWH (prophylactic vs intermediate vs therapeutic). Abbreviations: LMWH, low-molecular-weight heparin; ICU, intensive care unit.
Clinical Characteristics and Outcomes in Patients Treated With LMWH.

Abbreviations: SD, standard deviation; IQR, interquartile range; VKA, vitamin K antagonist; DOAC, direct-acting oral anticoagulant; AST, aspartate aminotransferase; ALT, alanine aminotransferase; LMWH, low-molecular-weight heparin; ICU, intensive care unit; CRNMB, clinically relevant nonmajor bleeding; VTE, venous thromboembolism; AF, atrial fibrillation.
a Mann-Whitney test (nonparametric) was performed, as this variable did not follow a normal distribution.
Discussion
Our study showed that among hospitalized COVID-19 patients with AF anticoagulated with edoxaban or LMWH followed by oral anticoagulation, approximately 13% required admission to the intensive care unit, 19% died, and 10% had a bleeding complication. Although the average length of stay was higher with LMWH than with edoxaban, no other significant differences in outcomes were observed between the anticoagulant regimens. Remarkably, the results remained similar after adjustment for most relevant clinical characteristics, thus emphasizing the consistency of our data. As a result, these data suggest that although edoxaban and LMWH can be safely used in this context, once-daily edoxaban may provide additional benefits.
AF is very common in hospitalized COVID-19 patients. In a survey performed in 76 countries, 21% of respondents reported cases of AF in this population. 17 Although COVID-19 may predispose patients to arterial and venous thrombosis, the optimal preventive strategy warrants further investigation and should take patients with AF into consideration. 18 In addition, during the COVID-19 pandemic, anticoagulants were more frequently switched from warfarin to DOACs. 19 Remarkably, available information remains very scarce, even though some studies analyzing the role of edoxaban in patients with COVID-19 are ongoing. Thus, HERO-19 (Hamburg Edoxaban for Anticoagulation in COVID-19 Study, NCT04542408) is currently evaluating whether an intensive anticoagulation strategy using edoxaban (in-hospital, therapeutic LMWH dose, followed by edoxaban) is superior to standard of care (low-dose LMWH, followed by ambulatory no anticoagulation), and the CONVINCE study (CorONa Virus edoxabaN ColchicinE COVID-19, NCT04516941) has been terminated owing to the insufficient rate of patient accrual and newly available scientific evidence. Therefore, as this study was performed before large DOACs studies were conducted in COVID-19, our data are very relevant.
Our study included 232 patients with nonvalvular AF. These patients were older and had more comorbidities than patients hospitalized in China, although their characteristics were closer to those of patients hospitalized in countries similar to ours, such as Italy.8,20,21 In the ENGAGE AF-TIMI 48 trial, which compared edoxaban with warfarin among patients with nonvalvular AF, mean age was 72 years, nearly 94% of patients had hypertension, and 58% had heart failure. 22 In ETNA-AF-Europe, a multinational, multicenter, post-authorization, observational study of 13 980 patients with AF taking edoxaban, mean age was 74 years, 77% had hypertension, and 6% had heart failure. 23 These data point to marked variability in clinical profile not only among hospitalized COVID-19 patients but also compared with other AF populations taking edoxaban. Notwithstanding, patients with AF anticoagulated with edoxaban are generally older and have many comorbid conditions.
We found that 13% of patients required admission to the intensive care unit, 19% of patients died, and 10% had bleeding complication. These figures are much higher than those reported in the edoxaban 60 mg arm of the ENGAGE AF-TIMI 48 trial (death, 4%/year; any bleeding, 2.75%/year) 22 but lower than those reported in the first publications of hospitalized COVID-19 patients,19,20 even though the patients in our study had a worse clinical profile. A review of various studies showed that in patients with COVID-19 infection who had not received anticoagulation therapy or were receiving prophylactic doses, VTE, and stroke occurred in approximately 20% and 3%, respectively. 24 Other authors reported that despite adequate thromboprophylaxis, symptomatic VTE occurred in 4.4% of patients and ischemic stroke in 2.5%. 5 In our study, these percentages were 1.7% and 0.9%, respectively, possibly because patients in our study received better treatment, particularly with full-dose anticoagulation. This observation is in line with the results of a recent study showing that among elderly patients with COVID-19, those on long-term oral anticoagulant treatment for AF may have a lower risk of all-cause mortality than those not receiving anticoagulation therapy. 25
During the first wave of the SARS-CoV-2 pandemic, some authors recommended switching from oral anticoagulation to LMWH in hospitalized COVID-19 patients.11,12,15,26,27 In the case of DOACs, this recommendation was based mainly on the risk of drug–drug interactions.15,27 Furthermore, it has been reported that heparin can decrease inflammatory biomarker levels, thus diminishing the inflammatory response to COVID-19. 28 In addition, some data suggest that heparin may reduce mortality among hospitalized COVID-19 patients.5,28 By contrast, some studies have reported that oral factor Xa inhibitors have a role in factor Xa-dependent enhancement of fibrinolysis. 29 In our study, the peak D-dimer was numerically lower in the edoxaban group, suggesting that edoxaban has a role in fibrinolysis and that this could have a positive clinical impact. Of note, the length of hospital stay was significantly lower with edoxaban than with LMWH followed by oral anticoagulation, likely owing to a simplification of treatment. This is important, as a lower length of stay may lead to less excessive caseloads and costs. 30 Furthermore, taking an oral medication (edoxaban) may reduce exposure of health care practitioners when compared to administering LMWH. 31
Remarkably, since the start of the COVID-19 pandemic, the management of pneumonia has changed over time, particularly since the very beginning. In fact, some drugs, such as hydroxychloroquine, azithromycin, and lopinavir/ritonavir are no longer being used, whereas other drugs, including dexamethasone, tocilizumab, baricitinib, remdesivir, and even new antivirals, such as molnupiravir or nirmatrelvir plus ritonavir (Paxlovid), are now being prescribed to affected patients. However, as the risk of drug–drug interactions with edoxaban seems low, the main conclusions of our study remain in effect, despite changes in the current therapeutic approach to COVID-19 pneumonia.13,32–35 Additionally, as the metabolism of edoxaban by CYP3A4 is marginal, the risk of drug–drug interactions with edoxaban is low and even lower than with other DOACs, thus potentially adding value in this clinical context.10,13–15
As a result, although edoxaban and LMWH followed by oral anticoagulation can be used safely in hospitalized COVID-19 patients with AF, edoxaban may provide an additional benefit. In line with our study, observational studies have shown that in patients with COVID-19-associated interstitial pneumonia, long-term DOAC intake may be associated with a decrease in mortality risk.36,37
We also compared the clinical profile and outcomes according to the dose of edoxaban. Remarkably, the CHA2DS2-VASc and HAS-BLED scores were higher among patients taking edoxaban 30 mg than among those taking 60 mg. In the ETNA-AF-Europe registry, patients taking edoxaban 30 mg were older and had a higher thromboembolic and bleeding risk. 23 Mortality rates were similar among patients taking edoxaban 60 mg and those taking 30 mg. However, there were more total bleeds among patients taking edoxaban 60 mg, with no significant differences in the severity of the bleeds. In order to ensure adequate protection with edoxaban, the dose should be adjusted according to the summary of product characteristics. 13
Clinical profile and outcomes were also compared according to the dose of LMWH followed by oral anticoagulation. Nearly half of the patients were taking the therapeutic dose of LMWH, although, remarkably, differences in clinical profile were detected according to the dose of LMWH, namely, age was younger in patients taking the therapeutic dose and mortality rates were higher in patients taking the prophylactic dose. Considering that anticoagulation has been associated with reduced mortality in hospitalized COVID-19 patients owing to the high risk of thrombosis and that some authors recommend higher therapeutic doses of LMWH than prophylactic doses, only full anticoagulation doses of LMWH should be recommended in patients with AF unless contraindicated.1,5
This study is subject to a series of limitations. First, it was not a randomized clinical trial, but an observational study. Although randomized clinical trials are the best format for providing information about differences between 2 therapies, patients were consecutively included in our study, thus reducing this potential bias. In fact, not many significant differences were observed in the clinical profile of patients taking edoxaban or LMWH followed by oral anticoagulation. Furthermore, total mean duration of follow-up reached 32 days. Therefore, it is uncertain whether the results observed in our study would remain valid over time. Studies with longer follow-ups are warranted. In addition, while the potential heterogeneity between centers was not specifically analyzed, the impact of the COVID-19 pandemic and the management protocols was similar between hospitals. Finally, our results should only be applied to patients whose clinical profile and therapeutic management are similar to those presented here.
In conclusion, approximately 1 in every 5 hospitalized COVID-19 patients with AF anticoagulated with edoxaban or LMWH followed by oral anticoagulation died and 1 out of 10 developed a bleeding complication during hospitalization. No significant differences were found between patients treated using these approaches in terms of mortality rates, arterial and venous thromboembolic complications, and bleeds. However, the duration of hospitalization was significantly lower with edoxaban than with LMWH followed by oral anticoagulation. Therefore, while both drugs are safe in this population, edoxaban may provide an additional benefit.
Supplemental Material
sj-pdf-1-cat-10.1177_10760296231180865 - Supplemental material for Edoxaban Versus Low-Molecular-Weight Heparin in Hospitalized COVID-19 Patients With Atrial Fibrillation
Supplemental material, sj-pdf-1-cat-10.1177_10760296231180865 for Edoxaban Versus Low-Molecular-Weight Heparin in Hospitalized COVID-19 Patients With Atrial Fibrillation by Pável Olivera, César Velásquez-Escandón, Desirée Campoy, Katia Flores, Tania Canals, Erik Johansson, María José Herranz, Laia Martínez, Juan José Cerezo-Manchado and Ramón Salinas in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-pdf-2-cat-10.1177_10760296231180865 - Supplemental material for Edoxaban Versus Low-Molecular-Weight Heparin in Hospitalized COVID-19 Patients With Atrial Fibrillation
Supplemental material, sj-pdf-2-cat-10.1177_10760296231180865 for Edoxaban Versus Low-Molecular-Weight Heparin in Hospitalized COVID-19 Patients With Atrial Fibrillation by Pável Olivera, César Velásquez-Escandón, Desirée Campoy, Katia Flores, Tania Canals, Erik Johansson, María José Herranz, Laia Martínez, Juan José Cerezo-Manchado and Ramón Salinas in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
Writing and editorial assistance were provided by Content Ed Net (Madrid, Spain).
Authors’ Note
We report the results of a subanalysis of the ongoing evaluation of Registry of thromboembolic and bleeding events in hospitalized patients with covid-19 (ACO-VID registry) (Protocol code: EORTC1811). This research was performed in accordance with standard ethical principles and approved by the Ethics Committee for Clinical Research of Hospital Universitario Sta. Mª del Rosell, Districts II and VIII of the Murcian Health Service. All patients or their healthcare proxies provided consent for participation in the registry. Neither the patients nor the public was involved in the design, or conduct, or reporting, or dissemination of our research. All data relevant to the study are included in the article or uploaded as supplementary information. Data are available upon reasonable request from the corresponding author.
Authors’ Contributions
All authors participated substantially in the conception and the design of the work and in the acquisition, analysis, and interpretation of the data and have revised and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PO reports honoraria for participation on advisory boards and consulting/speaker fees from Bayer, Pfizer, Bristol-Myers Squibb, Boehringer Ingelheim, Amgen, Daiichi Sankyo, and Techdow Pharma Spain. DC reports honoraria for participation on advisory boards and consulting/speaker fees from Bayer, Boehringer Ingelheim, and Daiichi Sankyo. JJCM reports honoraria for participation on advisory boards and consulting/speaker fees from Pfizer, Bristol-Myers Squibb, Boehringer Ingelheim, and Daichii-Sankyo. He has also received financial support to pay for publishing costs from Boehringer Ingelheim.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Writing and editorial assistance was funded by Daiichi Sankyo.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
