Abstract
Objective
To assess the impact of new-onset atrial fibrillation (AF) on patients hospitalized with coronavirus disease 2019 (COVID-19).
Methods
Multicenter and retrospective study that included subjects >55 years hospitalized with COVID-19 from March to October 2020 in Spanish hospitals. Patients were divided into 3 groups (no AF, new-onset AF, and preexisting AF) and followed-up to 90 days.
Results
A total of 668 patients were included, of whom 162 (24.3%) had no AF, 107 (16.0%) new-onset AF and 399 (59.7%) preexisting AF. Compared to patients without AF, those patients with new-onset AF were older and had more comorbidities, but without differences with preexisting AF. During hospitalization, in the univariate analysis, compared to patients without AF, major bleeding and cardiovascular mortality were more frequent in patients with new-onset AF (10.3% vs 0.6%; P < .001; 2.8% vs 0.6%; P = .025, respectively), with a trend toward more stroke (1.9% vs 0%; P = .085). Outcomes were similar between AF groups, but the length of stay was greater in preexisting AF patients. Among patients with new-onset AF taking reduced doses of anticoagulant treatment was associated with higher risks of stroke and major bleeding.
Conclusions
In COVID-19 hospitalized patients, new-onset AF may be associated with worse outcomes, but influenced by the dose of anticoagulants.
Keywords
Introduction
The coronavirus disease 2019 (COVID-19) pandemic is caused by Severe Acute Respiratory Syndrome Coronavirus type 2 (SARS-CoV-2), a highly infectious virus. 1 Previous studies have shown that preexisting cardiovascular diseases increase the risk of adverse outcomes in patients hospitalized for COVID-19 infection. 2 On the other hand, SARSCoV-2 may impair the cardiovascular system through different mechanisms, including systemic inflammation, acute respiratory distress syndrome, hypercoagulability state, and direct myocardial and vascular injury, which is related to worse clinical outcomes during hospitalization.3–5
Atrial fibrillation (AF) is the most common arrhythmia in routine practice, with an estimated prevalence of 2% to 4%, and increasing due to the ageing of the population. AF increases the risk of morbidity and mortality, mainly the risk of thromboembolic complications, particularly stroke. 6 Chronic oral anticoagulant treatment is the cornerstone to decrease this risk. 7
COVID-19 patients admitted to the hospital have a higher risk of arrhythmia, increasing with the severity of COVID-19 disease, being AF the most frequent one. 8 A recent systematic review and meta-analysis of 19 studies with a total of 21 653 hospitalized patients with COVID-19 showed a pooled prevalence of AF of 11% (15% in Europe), increasing with age and severity of the disease. 9 Preexisting AF has been associated with poorer outcomes in patients hospitalized for COVID-19, including a higher risk of mortality, admission to intensive care unit, need for invasive mechanical ventilation, and longer length of in-hospital stay.9–12
Importantly, recent reports have emphasized the importance of new-onset AF in patients with COVID-19 infection, as it may be a common condition that may be associated with worse clinical outcomes after SARS-CoV-2 infection. However, most studies that have analyzed the impact of new-onset AF on the management and clinical outcomes of patients hospitalized with COVID-19 had small sample sizes, were single-center studies or analyzed only patients admitted to the intensive care unit, limiting the generalizability of the results.13–18 In addition, these studies have not focused on analyzing the impact of anticoagulant treatment on outcomes.13–18
The ACO-VID registry was a multicenter study aimed to assess the impact of AF and anticoagulant treatment on clinical outcomes (ie death, thromboembolic disease, and bleeding) in COVID-19 hospitalized patients in Spain. 19 In this study, we analyzed the impact of new-onset AF (vs preexisting AF and patients without AF) on the clinical profile and outcomes of patients hospitalized with COVID-19, in the overall population, and also according to anticoagulation status (standard vs reduced doses of oral anticoagulant treatment).
Methods
The design and characteristics of the study have been fully explained in previous manuscripts. 19 Briefly, ACO-VID was a multicenter, retrospective, noninterventional, and observational study that consecutively included subjects >55 years hospitalized with COVID-19 infection from March to October 2020 in Hospital Sagrat Cor, Fundacio Sanitaria Hospital de Mollet (Barcelona, Spain), Hospital Universitari General de Catalunya (Barcelona, Spain), Hospital Universitario de Sant Joan de Reus (Reus, Spain) and University Hospital General Santa Lucia (Cartagena, Murcia, Spain). No specific exclusion criteria were defined. The study was approved by the Ethics Committee of all participating centers. A consent waiver was requested and approved by the Ethics Committee, as this was a retrospective study, and data were collected anonymously.
Patients were divided into 3 cohorts according to AF status (no AF, new-onset AF, and preexisting AF). New onset AF was defined as AF that first occurred during index hospitalization and preexisting AF as having a history of paroxysmal, persistent, or permanent AF before admission. Anticoagulant treatment was prescribed according to the clinical practice of each participating center.
Data were collected from the clinical history of patients, from baseline (index hospitalization) and up to 90 days of follow-up (or the last data/visit available). The following baseline variables were recorded: Biodemographic data (age, gender), cardiovascular risk factors (hypertension, diabetes), history of stroke, or bleeding, and anticoagulant treatment at admission (low molecular weight heparin [LMWH], vitamin K antagonists, direct oral anticoagulants). The elevation of transaminases 3 x ULN at admission, day 3 and discharge was also recorded. Baseline characteristics were compared between new-onset AF and no AF patients and between new-onset AF and preexisting AF patients.
Outcomes were recorded in both at index hospitalization and during the follow-up. During hospitalization, the proportion of patients that had a stroke, arterial/venous thromboembolic disease, major bleeding (defined according to the International Society of Thrombosis and Haemostasis), all-cause death, death of cardiovascular causes or death for COVID-19 were recorded. In addition, stroke, arterial/venous thromboembolic disease, major bleeding, and all-cause death were also recorded during the entire follow-up. Outcomes were collected in the 3 cohorts of patients, and also according to the dose of the anticoagulants (standard vs reduced) and also according to the use of LMWH. Outcomes were compared between new-onset AF and no AF patients and between new-onset AF and preexisting AF patients. Standard versus reduced doses were considered as follows: LMWH 1 mg/kg every 12 h versus other doses; fondaparinux 7.5 mg/daily versus other doses; edoxaban 60 mg versus 30 mg once daily; dabigatran 150 mg versus 110 mg twice daily; apixaban 5 mg versus 2.5 mg twice daily; rivaroxaban 20 mg versus 15 mg once daily. 6 Independent variables associated with all-cause death among patients with new-onset AF (vs no AF) were also calculated.
Statistical Analysis
Qualitative variables were defined as absolute (n) and relative (%) frequencies and quantitative variables as mean and standard deviation. Qualitative variables were compared using the chi-square test or the Fisher exact test, as required and the t-test when comparing 2 means. To determine factors associated with all-cause mortality, mortality for COVID-19, and major bleeding (dependent variables) in new-onset AF population (vs no AF population), a multivariate logistic regression analysis was performed. Baseline clinical characteristics (age, sex, hypertension, diabetes, prior stroke or bleeding, elevated transaminases) and type and dose of anticoagulants were considered as independent variables. The multivariate model was constructed by including those factors with P < .10 in the bivariate analysis using automatic forward stepwise selection. Only the significant factors were finally included in the model. Statistical significance was set at P < .05 for all tests. The statistical analysis was performed using SPSS 21.0 (SPSSInc., Chicago, IL).
Results
A total of 668 patients hospitalized for COVID-19 were included in the study, of whom 162 (24.3%) had no AF, 107 (16.0%) new-onset AF and 399 (59.7%) preexisting AF. At baseline, compared to patients without AF, those patients with new-onset AF were older (77.5 ± 8.6 vs 68.8 ± 9.9 years; P < .001), had more hypertension (86.9% vs 56.2%; P < .001), prior stroke (19.6% vs 7.4%; P = .003) and history of major bleeding (19.6% vs 4.9%; P < .001). No significant differences were observed in the baseline clinical characteristics between AF cohorts. Among patients without AF, 93.8% ware taking LMWH at admission. The most common anticoagulant drug taken by new-onset AF and preexisting AF patients was edoxaban (46.7% and 48.6%, respectively) (Table 1).
Baseline Clinical Characteristics.

Abbreviation: AF, atrial fibrillation; LMWH, low-molecular-weight heparin; VKA: vitamin K antagonists; P1, new onset AF versus pre-existing AF; P2, new onset AF versus no AF.
Mean follow-up during the study period was 65 ± 3 days. With regard to the events during the hospitalization according to AF status, in the univariate analyses, compared to patients without AF, major bleeding and cardiovascular mortality were more frequent in patients with new-onset AF (10.3% vs 0.6%; P < .001; 2.8% vs 0.6%; P = .025, respectively), with a trend towards more stroke (1.9% vs 0%; P = .085). In addition, mean length of stay was 16.3 days (95% CI 15.5-17.1), being lower in those patients without AF. During the whole follow-up, major bleeding was more common in patients with new-onset AF, compared to no AF patients (10.3% vs 3,7%), with a trend towards more stroke (1.9% vs 0%; P = .085). No significant differences were observed between AF cohorts (Table 2 and Figure 1).

Events during the total follow-up of the study according to atrial fibrillation (AF) status.
Events During the Study According to AF Status.

Abbreviations: AF, atrial fibrillation; CV, cardiovascular; P1, new onset AF versus pre-existing AF; P2, new onset AF versus no AF.
Among those patients receiving standard doses of anticoagulant treatment, no differences were observed in outcomes, except for a trend toward higher mortality for COVID-19 in patients with new-onset AF compared to patients without AF (Table 3). However, when considering those patients treated with reduced doses of anticoagulants, patients with new-onset AF had a higher risk of stroke (3.0% vs 0%; P = .045) and major bleeding (9.1% vs 0.7%; P = .005), with a trend toward a higher risk of cardiovascular mortality (3.0% vs 0.7%; P = .062) during hospitalization, and more stroke during the entire follow-up (3.0% vs 0%; P = .045). No differences in outcomes were observed when comparing reduced doses of anticoagulants between AF cohorts (Table 4). However, when considering patients taking low doses of heparin in patients with new-onset AF compared to patients without AF, these differences increased, with more in-hospital stroke (5.3% vs 0%; P = .008), major bleeding (10.5% vs 0.7%; P = .004) and cardiovascular mortality (5.3% vs 0.7%; P = .005), and overall stroke (5.3% vs 0%; P = .008) (table 5). Finally, no significant differences were observed in outcomes among those patients taking LMWH versus other anticoagulants (Table 6).
Events During the Study According to AF status among Those Patients Receiving Standard Doses of Anticoagulant Treatment.

Abbreviations: AF, atrial fibrillation; CV, cardiovascular; P1, new onset AF versus pre-existing AF; P2, new onset AF versus no AF; COVID-19, coronavirus disease 2019.
Events During the Study According to AF status among Those Patients Receiving Reduced Doses of Anticoagulant Treatment.

Abbreviations: AF, atrial fibrillation; CV, cardiovascular; P1, new onset AF versus pre-existing AF; P2, new onset AF versus no AF; COVID-19, coronavirus disease 2019.
Events During the Study According to AF status among Those Patients Receiving low Doses of Heparin.

Abbreviations: AF, atrial fibrillation; CV, cardiovascular; COVID-19, coronavirus disease 2019.
Events During the Study According to AF status among Those Patients Receiving Heparin.

Abbreviations: AF, atrial fibrillation; CV, cardiovascular; P1, new onset AF versus pre-existing AF; P2, new onset AF versus no AF; COVID-19, coronavirus disease 2019.
In the multivariate analysis, elevated transaminases (HR 4.05; 95% CI 1.59-10.32), particularly at discharge (HR 5.59; 95% CI 3.08-10.16), and elderly (>75 vs ≤75 years: HR 2.29; 95% CI 1.08-4.83) were the only variable associated with total mortality among patients with new-onset AF (vs no AF). New-onset AF increased the risk of all-cause death numerically, but this did not reach statistical significance in the multivariate analysis (Table 7). No independent variables were associated with bleeding among patients with new-onset AF (vs no AF), but elevated transaminases with mortality for COVID-19 (HR 3.27; 95% CI 2.06-5.21; P < .001), with a trend towards age (≥75 vs <75 years: HR 1.37; 95% CI 0.99-1.90; P = .06).
Factors Associated with Total Mortality among Patients with new Onset AF and no AF. multivariate Analysis.
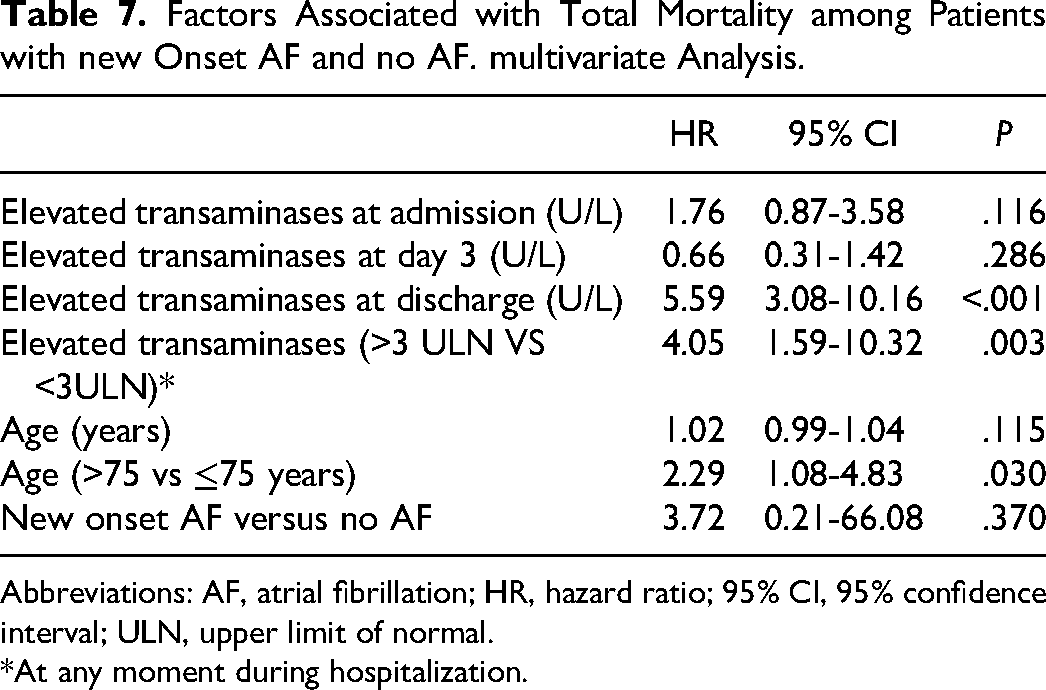
Abbreviations: AF, atrial fibrillation; HR, hazard ratio; 95% CI, 95% confidence interval; ULN, upper limit of normal.
*At any moment during hospitalization.
Discussion
Our data showed in a wide sample of patients admitted with COVID-19 in Spain, that new-onset AF was common and was associated with a higher risk of major bleeding and cardiovascular mortality, with a trend towards more stroke during hospitalization and more major bleeding during the follow-up compared to patients without AF. However, these differences disappeared in those patients receiving standard doses of anticoagulant treatment and were more marked in those taking reduced doses of anticoagulant treatment. The clinical profile and outcomes did not differ between patients with new-onset AF and those with preexisting AF. Increased age and elevated transaminases during hospitalization were independently associated with total mortality, but not new-onset AF.
AF is very common in hospitalized patients with COVID-19. 20 In our study, 16% of patients presented new-onset AF and nearly 60% preexisting AF. This high risk of AF in patients admitted with COVID-19 has been related to different mechanisms, including hypoxia (particularly in case of diffuse lung infiltration), inflammatory state, direct myocardial injury (ischemia, inflammation, etc), abnormal host immune response, or some drugs, among others..21–24 In fact, AF, including new-onset AF, has been described as the most common cardiac event associated with hospitalization for COVID-19.25–27 Although we included our patients during the first wave of COVID-19 pandemic, recent studies have shown that AF incidence is also increased in the subsequent waves of COVID-19 infection, 28 indicating that our data are current.
With regard to the clinical profile of patients according to AF status, compared to patients without AF, those patients with new-onset AF were older and had more comorbidities, without relevant differences between patients with preexisting or new-onset AF. Previous studies have shown that patients with cardiovascular disease, including AF, are complex, with many comorbidities,29,30 and this also includes patients with new-onset AF. 27 As a result, our data are consistent with previous publications.
During hospitalization, most patients without AF were treated with LMWH as thromboprophylaxis, but in nearly half of subjects with AF, either new-onset or preexisting, patients were taking edoxaban after treatment with LMWH. Of note, in our study, no differences in outcomes were observed in patients taking LMWH versus other anticoagulants. Although during the first wave, experts considered that oral anticoagulant treatment should be switched to LMWH mainly due to the risk of drug-drug interactions, leading to important changes in the efficacy or safety of oral anticoagulants,31,32 the fact is that this has not been confirmed in further studies,33,34 and except in cases in which oral ingestion is not possible (ie mechanical ventilation), the use of direct oral anticoagulants during hospitalization should be considered.
During hospitalization, major bleeding and cardiovascular mortality were more frequent in patients with new-onset AF, with a trend towards more stroke compared to patients without AF, with similar outcome rates between patients with new-onset AF or preexisting AF. In addition, length of stay was longer in patients with AF. Although some studies have shown that patients with AF have a higher risk of major adverse cardiovascular events,35,36 the majority of publications have reported that new-onset AF is associated with an increased risk of a variety of events, such as acute respiratory syndrome (with higher requirements of intensive care and invasive ventilation), hemorrhage, stroke and particularly in-hospital mortality.13–15,27,37 These studies have also shown that the risk of adverse outcomes is in general similar between new-onset and preexisting AF patients,13–15,35–37 or even worse in patients with new-onset AF. 27 Consequently, although one might think that new-onset AF is a consequence of decompensation from COVID-19 infection, the reality is that it has important prognostic implications. In fact, new-onset AF implies a situation of increased inherent risk, which is similar to preexisting AF. Therefore, in this context, a more intensive approach must be carried out in order to reduce the risk of presenting possible complications.
Although the number of patients was limited in some subgroups, another important point in our study was that the risk of outcomes differed according to the dose of anticoagulant treatment. Thus, whereas among those patients receiving standard doses of anticoagulant treatment, there was only a trend toward a higher mortality for COVID-19 in patients with AF (new-onset and chronic AF) compared to patients without AF, in those patients treated with reduced doses of anticoagulants, compared to patients without AF, patients with new-onset AF had a higher risk of stroke and major bleeding, with a trend towards a higher risk of cardiovascular mortality during hospitalization, and more stroke during the entire follow-up. Of note, although some patients taking low doses of oral anticoagulants were properly anticoagulated, this did not occur when taking low doses of heparin. In fact, in these patients, the differences in outcomes increased. In addition, in the multivariate analysis, although new-onset AF increased the risk of total mortality, this was not statistically significant. As a result, it seems that in fully anticoagulated patients, new-onset AF is more a marker of risk (ie worse clinical profile), rather than a risk factor by itself, but on the contrary, a risk factor in those patients receiving reduced doses of anticoagulants.19,26 As a result, our data strongly suggest that anticoagulant treatment can modulate the risk of adverse events. Therefore, both new-onset AF and preexisting AF, should not be considered as a simple by stander of the in-hospital COVID-19 course and proper anticoagulant treatment should be promoted in these patients. 38
Finally, elevated transaminases were independently associated with an increased risk of total mortality and mortality for COVID-19 among patients with new onset AF (vs no AF). First, severe COVID-19 infection have an increased risk of hepatotoxicity, due to a direct effect of SARS-CoV-2. Although uncommon, drug-induced liver injury has been associated with the use of antiviral drugs.39,40 These facts may explain the higher risk of mortality shown in our study. However, some oral anticoagulants, such as dabigatran or edoxaban, have shown a lower risk of hepatotoxicity, as metabolism by cytochrome P450 is absent or low, respectively. 6 In this context, these oral anticoagulants, with a lower risk of hepatotoxicity could provide an added benefit in this clinical setting. 33
This study has some limitations that should be commented. This registry had a retrospective design. Consequently, only data available in the electronic clinical history of patients could be collected. However, 3 independent centers participated in this study and the number of patients was relatively high, reducing this potential bias. Finally, our results can be extended only to those patients with a similar clinical profile and healthcare system.
In conclusion, in patients hospitalized with COVID-19, patients with new-onset AF were older and had more comorbidities than patients without AF, but similar to patients with preexisting AF. In addition, in the univariate analysis, new-onset AF (vs no AF) was associated with a higher risk of major bleeding and in-hospital cardiovascular mortality, but not in the multivariate analysis. This could be related to the dose of the anticoagulants, as no differences in outcomes were observed in those patients receiving standard doses of anticoagulant treatment, but were more marked in those taking reduced doses of anticoagulants.
Footnotes
Acknowledgements
Content Ed Net provided medical writing and editorial support, which was funded by the Fundación para la Formación e Investigación Sanitarias de la Región de Murcia.
Author Contributions
All authors contributed extensively to the work presented in this paper. All authors have contributed significantly to the conception, design, or acquisition of data, or analysis and interpretation of data. All authors have participated in drafting, reviewing, and/or revising the manuscript and have approved its submission. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Conflicts of Interest
The author(s) declare not other conflict of interests related to this article that those state in funding source section.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fundación para la Formación e Investigación Sanitarias de la Región de Murcia, though an unrestricted grant from Daichii Sankyo (DS) Spain. DS had no role in the design, analysis, interpretation and publication of the registry.
