Abstract
Enoxaparin, a complex, biologically derived low-molecular-weight heparin, is approved for a range of clinical indications. This study was carried out to compare the potency profile and pharmacodynamic responses of branded enoxaparin (Lovenox; Sanofi, US) with a generic enoxaparin (enoxaparin sodium injection, USP). Five batches of each product were tested. Although the average molecular weight, anti-factor Xa, and anti-factor IIa potencies were similar for the two products, differences were observed in the in vitro thrombin generation and kinetics of clot formation (P = .01) and in the ex vivo pharmacodynamics regarding thrombin generation inhibition (P = .029), tissue factor pathway inhibitor release (P = .006), and inhibition of the active form of thrombin-activated fibrinolysis inhibitor (P = .023). These findings suggest that simple analytical characterization can establish good quality control in manufacturing, but they may not assure similarity in biological performance between the branded and the generic enoxaparin.
Introduction
Low-molecular-weight heparins (LMWHs; mean molecular weight 4000-5000 Da), derived from the depolymerization of heparin, exhibit a wide spectrum of biological activities associated with their antithrombotic property. They are cofactors of antithrombin (AT) and indirectly inhibit the activity specifically of factor Xa (FXa) and thrombin (factor IIa [FIIa]). 1 But their anticoagulation effect cannot be explained solely in terms of anti-FXa and anti-FIIa activity, and biological activities involving other molecules in the hemostatic process have been identified. 2 The LMWHs have multiple effects including facilitation of the release of tissue factor pathway inhibitor (TFPI); inactivation of coagulation factors IX, XIa, and XIIa; and activation of platelets, all of which impact the kinetics of inhibition of thrombin generation and clot formation.
In addition, LMWHs have multiple pleotropic effects and bind to or interact with numerous proteins, lipids, cell membranes, chemokines, and adhesion molecules, and these interactions contribute to the antithrombotic effect and immunogenic behavior of each specific LMWH.3
–6 The nature and potency of these biological effects are dependent on the specific sequence of saccharides in the oligosaccharide chains of each LMWH.7
–17 This sequencing is highly dependent on the manufacturing process because active constituents of LMWHs have not been fully characterized and the identity and structures of the polysaccharides that mediate several of the biological activities are either poorly understood or unknown. In addition to the molecular weight, the charge density, chain length, the types of disaccharides present, the presence of specific saccharide sequences, and the pattern of
Since the early 2000s when generic LMWH manufacturers applied for product registration in the United States, there was debate regarding the extent to which the set of biological markers relevant to ensure therapeutic equivalence with the originator products has been met.18 –21 The European Medicines Agency recommends that LMWHs should be compared not only by determining anticoagulant activities, such as anti-FXa and anti-FIIa but also by using other pharmacodynamic tests, such as TFPI activity, as conventional pharmacokinetic studies cannot be performed. 22 Assessment of these combined pharmacodynamic parameters provides a fingerprint of the polysaccharidic profile of an LMWH product.
This study was carried out to compare the anticoagulant activity and the potency profile of branded enoxaparin (Lovenox; Sanofi US, Bridgewater, New Jersey), with a generic version of enoxaparin sodium injection, USP (Sandoz US, Princeton, New Jersey), and to assess the pharmacodynamic responses of the two products using a number of in vitro and ex vivo analyses.
Materials and Methods
Drugs Evaluated
The physicochemical properties and biological activities of 5 batches each of the branded enoxaparin, Lovenox (batch numbers: 512107, 514947, 514956, 515234, and 515949) and of the first generic enoxaparin sodium injection, USP available in the United States (batch numbers: 914698, 914786, 914789, 915805, and 916086) were evaluated in parallel in a series of in vitro and ex vivo studies. Drugs were purchased through the Pharmacy of the Loyola University Health System (Maywood, Illinois) and other hospital systems in the United States.
Molecular Weight Profile
Gel permeation chromatography
The molecular weight distribution of the branded and generic enoxaparin product samples was determined using gel permeation chromatography (GPC)-high-performance liquid chromatography (HPLC) as reported previously. 23 In brief, 10 mg/mL solutions of each LMWH were prepared in 0.3 M Na2SO4. Each LMWH solution (20 µL) was injected into the HPLC system (Waters 845; Millipore-Waters, Lexington, Massachusetts) equipped with Millennium 2000 (Millipore-Waters, Lexington, Massachusetts) software designed for polymer analysis. The ultraviolet (UV, 234 nm) and the refractive index (RI) detectors were linked in series, with the outlet of the joint columns (TSK G3000SW and TSK G2000SW; TosoHaas, Tokyo, Japan) attached to the UV detector. Elution profiles were determined using UV and RI detection. The molecular weight profile was determined in relation to 19 narrow range heparin fractions whose molecular weights were previously determined by GPC and low-angle laser light scatter. In total, 5 batches of branded and 5 batches of generic enoxaparin were tested, and each batch of enoxaparin was tested in duplicate.
Biologic Activity Profile
In vitro evaluation of anticoagulant activity
In vitro studies (analysis of thrombin generation [see ex vivo assays for testing details] and fibrinokinetics) were performed in which varying concentrations of either branded or generic enoxaparin were added to the human plasma and the samples were then tested immediately in parallel in the assay system. Blood collection was approved by the Institutional Review Board of Loyola University Chicago, and volunteers gave written informed consent prior to venipuncture. Drug concentrations were selected to simulate those found in clinical use; modifications were made where needed based on the sensitivity of the assay system.
In vitro fibrinokinetics analysis
Fibrinokinetic analysis of clot formation was carried out in the presence of branded enoxaparin and generic enoxaparin; 5 batches of each enoxaparin product were tested. This assay assessed the initiation, kinetic rate, and strength of clot formation in human plasma supplemented in vitro with a wide concentration range of product from 0.15 to 10.0 µg/mL. A 200-µL aliquot of human plasma was added to a 96-well plate; 25 µL human thrombin (5 U/mL) and 25 µL of 0.25 mmol/L CaCl2 were then added. As the thrombin-induced clot formed, optical density recordings were taken in real time over a 30-minute period. Optical density was recorded at 405 nm on a SPECTRAmax Plus-384 plate reader (Molecular Devices, Sunnyvale, California). Testing was performed in duplicate.
In vitro antithrombotic potency: chromogenic anti-protease assays
The anti-FXa and anti-FIIa activities of the branded and generic enoxaparins were determined in an isolated biochemical buffer system supplemented with purified human AT. 24 The AT, obtained from Immuno AG (Vienna, Austria), was used at a working concentration of 1 U/mL. Purified bovine FXa and human thrombin were obtained from Enzyme Research Laboratories (South Bend, Indiana). Chromogenic substrates, Spectrozyme Xa and Spectrozyme TH, were obtained from American Diagnostica (Greenwich, Connecticut). The assays were performed on the ACL 300 Plus automated kinetic coagulation analyzer (Beckman Coulter, Fullerton, California). The concentrations of branded and generic enoxaparin that produced 50% inhibition (IC50) of FXa and thrombin in the biochemical assay system were determined. In total, 5 batches of branded and 5 of generic enoxaparin were tested, and each batch of enoxaparin was tested in triplicate.
Comparison of pharmacodynamic responses
Pharmacodynamic studies were conducted using a primate model (Macaca mulatta) to determine the effect of treatment with branded and generic enoxaparin on certain biological antithrombotic activity levels. Animal protocols were approved by the Animal Care and Use Committee of Loyola University Chicago. Following sedation with an intramuscular injection of ketamine 10 mg/kg, animals were injected subcutaneously with either branded or generic enoxaparin at a dose of 1 mg/kg. Blood samples were collected from the saphenous vein into 3.2% sodium citrate at baseline, and 1, 3, 6, 12, and 24 hours postadministration. The blood was immediately centrifuged at 1100g for 15 minutes to prepare platelet-poor plasma. The plasma was aliquoted and stored at −70°C until required for ex vivo testing of thrombin generation, TFPI, and thrombin-activated fibrinolysis inhibitor (TAFI) activity levels. Each batch of branded and generic enoxaparin was tested in 3 primates.
In vitro and ex vivo thrombin generation analysis
Thrombin generation was determined using the Technothrombin TGA assay from Technoclone (Vienna, Austria). In total, 5 batches of each enoxaparin product were tested in both the in vitro and the ex vivo studies. The coagulation cascade was activated by tissue factor and the formation of thrombin over time was monitored by a fluorogenic substrate using the BioTek FLx800 reader with Gen5 Software (Winooski, Vermont). The concentration of thrombin (nmol/L) in the plasma sample was calculated using a calibration curve of thrombin supplied in the kit. A calculation of the percentage inhibition of thrombin generated compared with normal (human for in vitro or primate for ex vivo) pooled plasma was carried out. For the in vitro studies, testing was performed in duplicate on human plasma supplemented with the product. For the ex vivo studies, testing was performed in duplicate on plasma samples collected from primates (n = 3 for each batch) following treatment with product as described above.
Ex vivo tissue factor pathway inhibitor levels
The TFPI antigen levels were determined using the Asserachrome TFPI kit (Stago, Parsipanny, New Jersey), which is a sandwich, enzyme-linked immunoassay that quantitates the concentration of TFPI in plasma samples. Tested plasmas were obtained from primates (n = 3 for each batch) treated with 5 batches of each enoxaparin product as described above. Testing was performed in duplicate. A full-length recombinant TFPI sample supplied in the kit was used as the calibration standard. Optical density readings were measured at 405 nm on a SPECTRAmax Plus-384 plate reader.
Ex vivo thrombin-activated fibrinolysis inhibitor analysis
Functional levels of TAFI were measured using the Pefakit TAFI (Pentapharm, Basle, Switzerland) chromogenic assay, which measures the amount of TAFI activated by the thrombin–thrombomodulin complex to form TAFIa. Tested plasma samples were obtained from primates (n = 3 for each batch) treated with 5 batches of each enoxaparin product as described above. Testing was performed in duplicate. A TAFI calibrator with an assigned value was supplied in the assay kit. Optical density readings were made at 405 nm on the SPECTRAmax Plus-384. A calculation was then performed to determine the percentage inhibition of the generation of TAFI to its activated form TAFIa, compared with normal (primate) pooled plasma.
Statistical Analysis
Statistical significance was determined by 1-way or 2-way repeated-measures analysis of variance or t test, as appropriate, using SigmaStat software version 3.11 (Systat Software, San Jose, California). All pairwise multiple comparisons were made using the Student-Newman-Keuls method. P values < .05 were considered statistically significant.
Results
Molecular Profiling
No differences were observed between the branded and generic enoxaparin in terms of mean molecular weight (Table 1). Other molecular weight parameters, such as peak molecular weight, Z and Z + 1 molecular weights, and polydispersity aspects of the different batches of the branded and generic products were also not different between the products (data not shown).
Comparative Antithrombin-Mediated Anti-Factor Xa and Anti-Factor IIa In Vitro Anticoagulant Activities for Branded and Generic Enoxaparin Products.a
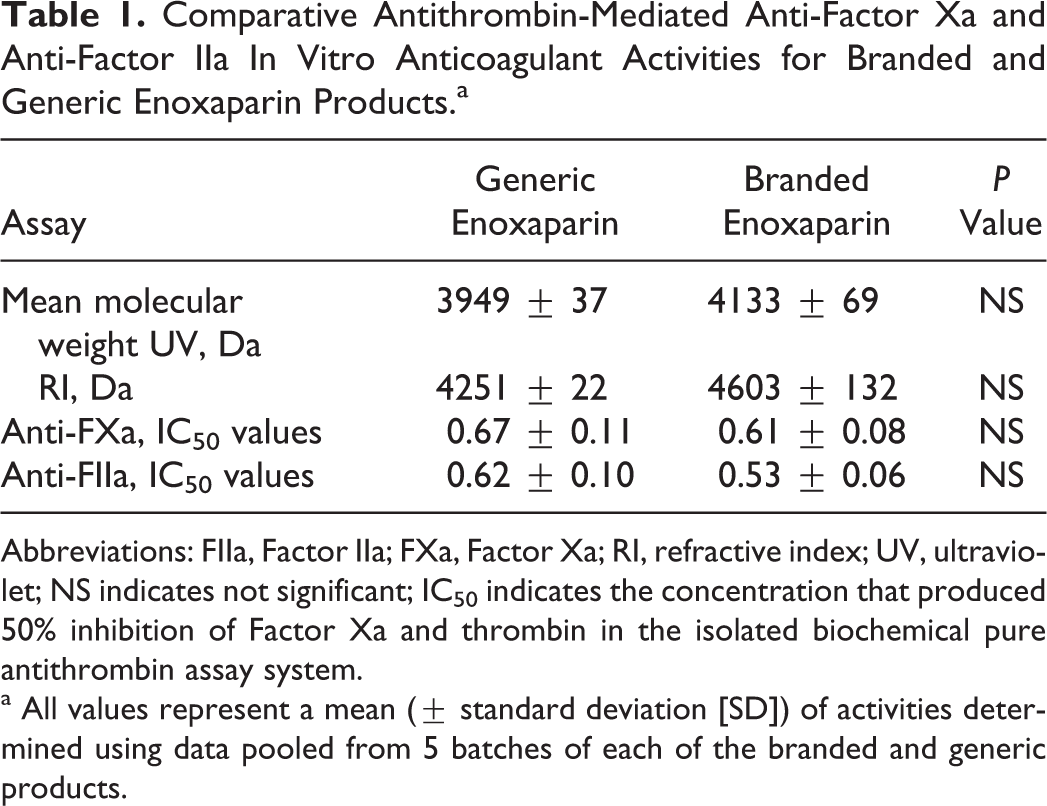
Abbreviations: FIIa, Factor IIa; FXa, Factor Xa; RI, refractive index; UV, ultraviolet; NS indicates not significant; IC50 indicates the concentration that produced 50% inhibition of Factor Xa and thrombin in the isolated biochemical pure antithrombin assay system.
a All values represent a mean (± standard deviation [SD]) of activities determined using data pooled from 5 batches of each of the branded and generic products.
In Vitro Antithrombotic Potencies
The IC50 values of the potency activities of 5 batches of each product are shown in Table 1. The potency of the products determined in biochemically defined systems was similar in the anti-FXa assay (IC50 value: 0.61 ± 0.08 vs 0.67 ± 0.11, for the branded and generic products, respectively) and in the anti-FIIa assay (IC50 value 0.53 ± 0.06 for the branded product and 0.62 ± 0.10 for the generic product).
In Vitro Anticoagulant Activities
In the thrombin generation test, a concentration-dependent anticoagulant effect was observed with both branded and generic enoxaparin products at 0.5 and 1.0 μg/mL. The branded enoxaparin was associated with greater thrombin generation inhibition than the generic product at both concentrations for all 5 batches tested. This indicates that the branded enoxaparin had higher anticoagulant activity relative to the generic version, although this study did not identify statistically significant differences (Figure 1).
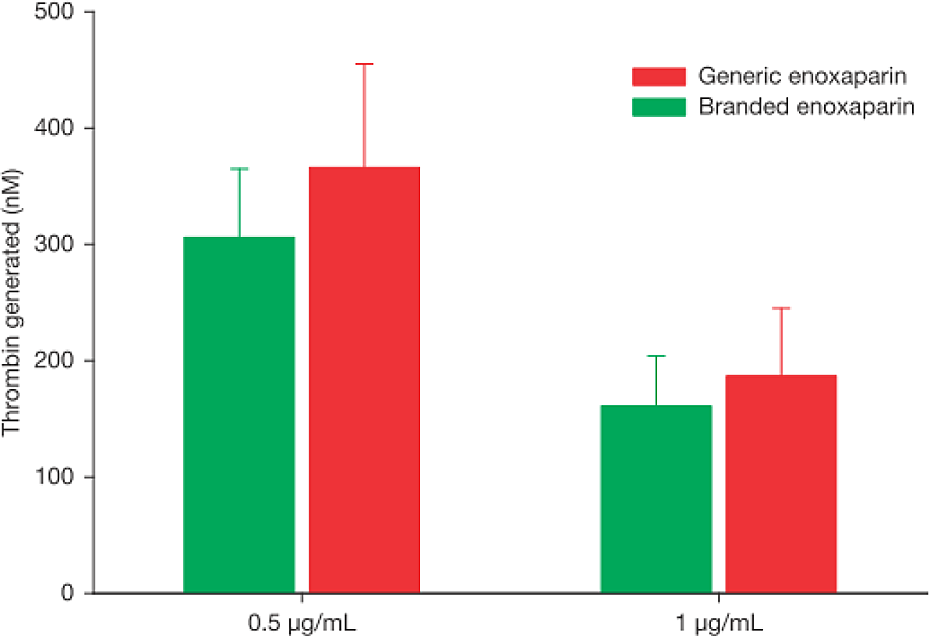
The level of in vitro thrombin generation induced by tissue factor in human plasma following the addition of either branded or generic enoxaparin. Mean (± standard deviation) thrombin levels for pooled data from the 5 batches of each enoxaparin product are shown.
A significant difference in the effect of the branded enoxaparin compared with the generic enoxaparin on fibrinokinetics was observed throughout the entire period of clot formation, and this difference was stable across all drug concentrations tested and in all batches of test products (P < .01; Figure 2). Branded enoxaparin consistently exhibited a more potent anticoagulant effect demonstrated by slower clot formation with a weaker final clot structure.
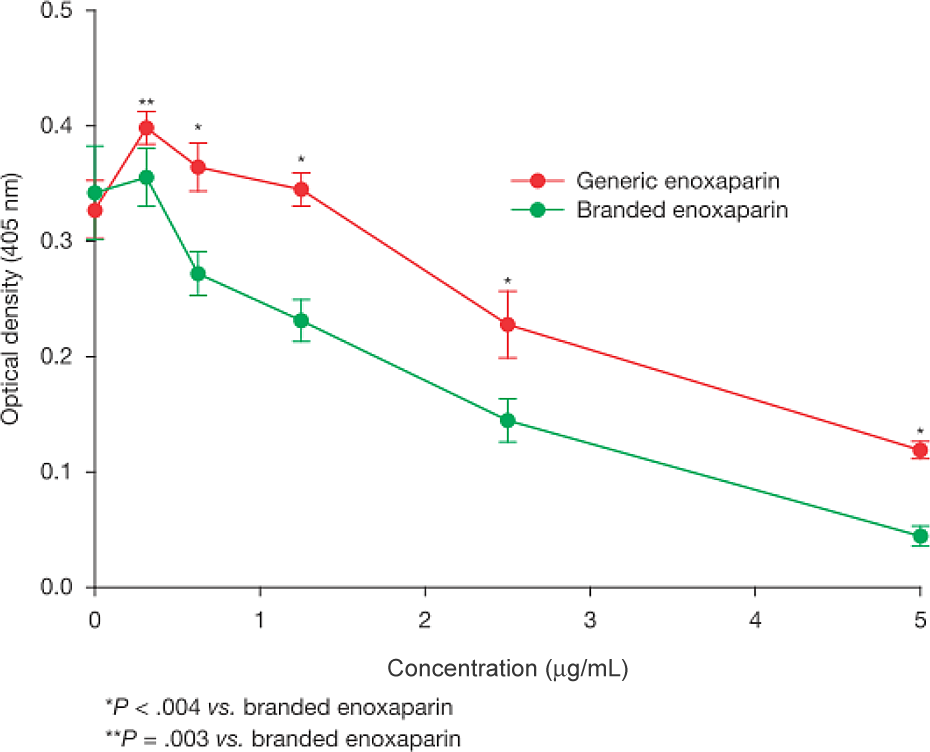
In vitro fibrinokinetic assay depicting the optical density readings taken at the 30-minute point in fibrin clot formation. Mean (± standard deviation) values for pooled data from 5 batches of branded and 5 batches of generic enoxaparin tested at multiple concentrations are presented.
Ex Vivo Responses From the Pharmacodynamic Model
Analysis of blood samples collected from primates treated subcutaneously with 1 mg/kg of either branded or generic enoxaparin showed that the generic enoxaparin was associated with a more sustained anticoagulant effect in terms of thrombin inhibition (P = .023; Table 2). The anti-FIIa area under the curve (AUC) over a period of 24 hours for the branded enoxaparin samples was 91 ± 20 μg*hrs/mL; whereas for the generic enoxaparin samples the AUC was 135 ± 28 μg*hrs/mL. There was no difference between branded enoxaparin and generic enoxaporin in terms of inhibition of FXa. The anti-FXa AUC over a period of 24 hours for the branded enoxaparin samples was 185 ± 21 μg*hrs/mL, whereas for the generic enoxaparin samples the AUC was 178 ± 26 μg*hrs/mL.
Ex Vivo Pharmacodynamic Analyses of Blood Samples Taken From Primates Treated With a Subcutaneous 1 mg/kg Dose of Either Branded or Generic Enoxaparin.a
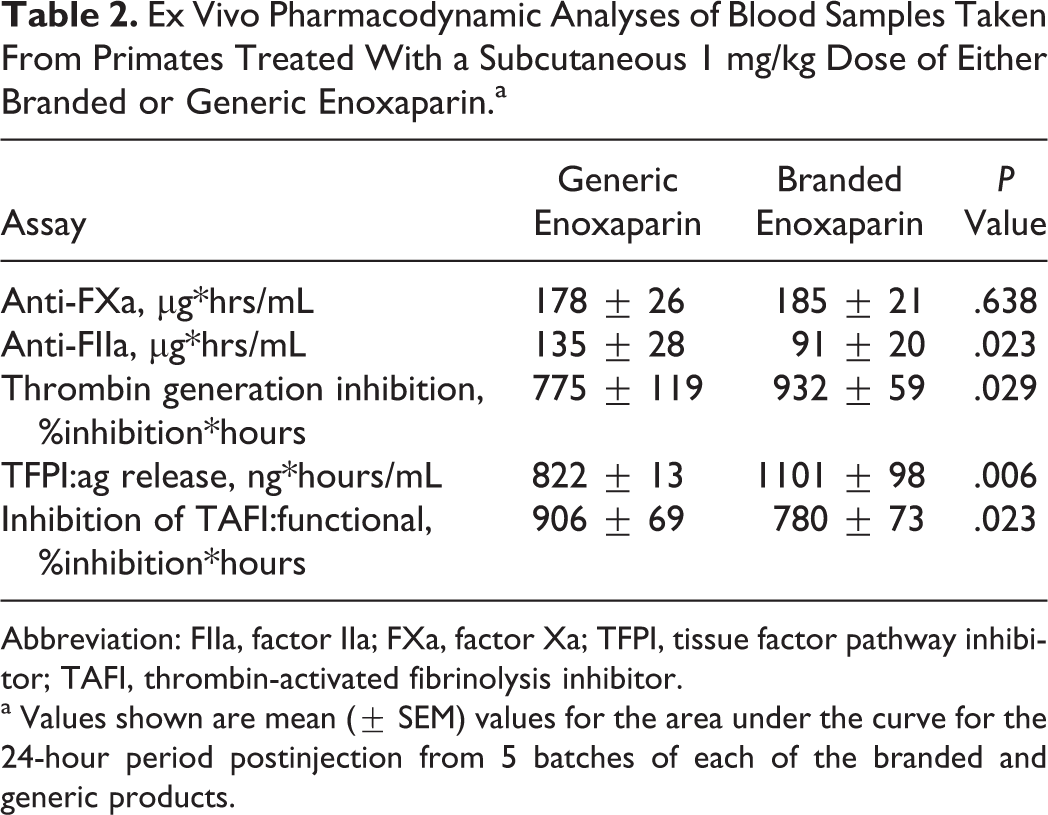
Abbreviation: FIIa, factor IIa; FXa, factor Xa; TFPI, tissue factor pathway inhibitor; TAFI, thrombin-activated fibrinolysis inhibitor.
a Values shown are mean (± SEM) values for the area under the curve for the 24-hour period postinjection from 5 batches of each of the branded and generic products.
Analysis of blood samples collected from primates treated subcutaneously with 1 mg/kg of either branded or generic enoxaparin showed that the branded enoxaparin was associated with a significantly stronger and more sustained anticoagulant effect in terms of inhibition of thrombin generation (Figure 3). The AUC over a period of 24 hours for the branded enoxaparin samples was 932% ± 59%inhibition*hours, whereas for the generic enoxaparin samples the AUC was 775% ± 119%inhibition*hours (P = .029; Table 2).
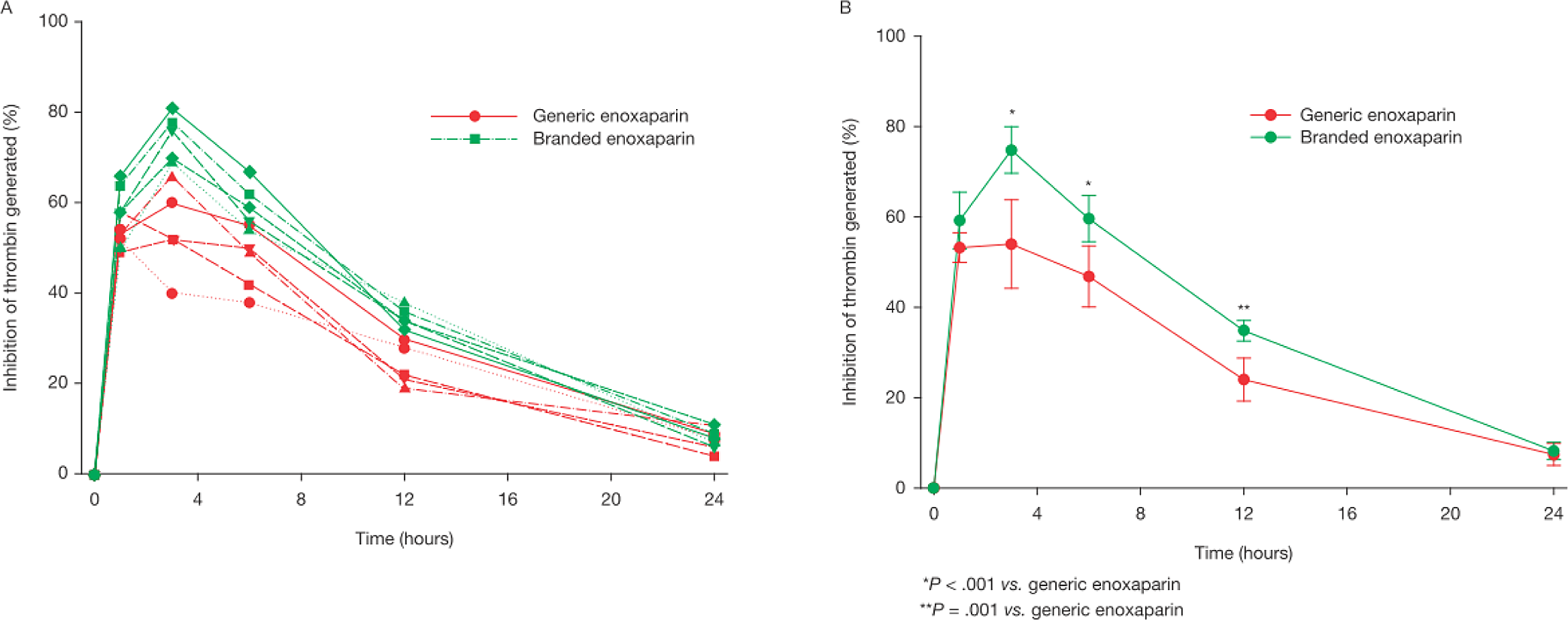
The comparative ex vivo anticoagulant effects shown as the percentage inhibition of generated thrombin over a 24-hour period after administration of a 1 mg/kg subcutaneous dose of branded or generic enoxaparin to primates. A, Results for individual batches. B, Results are shown as mean values (± SEM) for all treated primates for the branded and generic enoxaparin products.
Analysis of blood samples collected from primates treated with 1 mg/kg of either branded or generic enoxaparin revealed a more potent and sustained activity for the branded product in terms of TFPI release (Figure 4). The AUC for the 5 batches of branded enoxaparin over a period of 24 hours was 1101 ± 98 ng*hours/mL, whereas for the 5 batches of generic enoxaparin the AUC was 822 ± 13 ng*hours/mL (P = .006; Table 2).
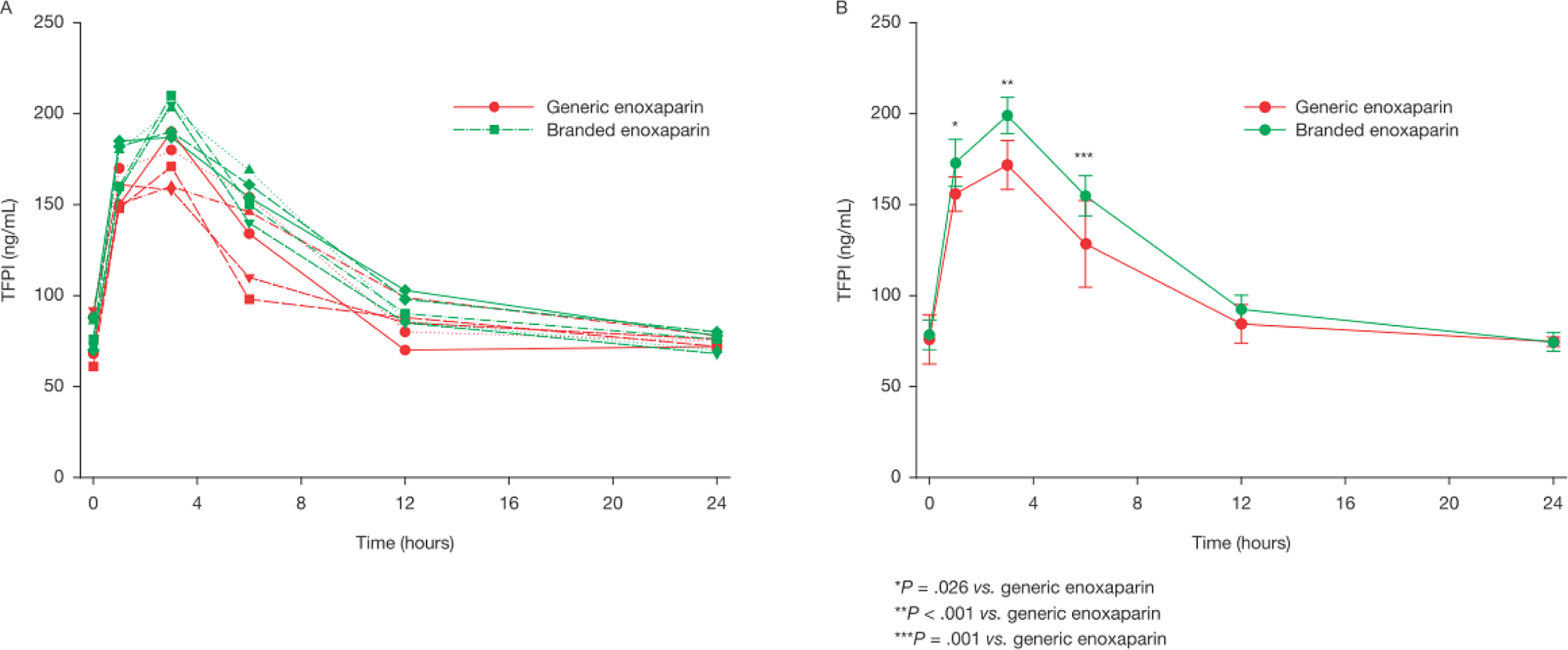
The level of tissue factor pathway inhibitor release over a 24-hour period following the administration of a 1 mg/kg subcutaneous dose of branded or generic enoxaparin to primates. A, Results for individual batches. B, Results are shown as mean values (± SEM) for all treated primates for the branded and generic enoxaparin products.
Analysis of blood samples collected from primates treated with 1 mg/kg subcutaneous injection of either branded or generic enoxaparin revealed an inhibitory effect on TAFI conversion to TAFIa (Figure 5). The generic product produced more inhibition of TAFI conversion to TAFIa than the branded product. The AUC over a period of 24 hours demonstrated a significant difference at 780% ± 73%inhibition*hours for the branded enoxaparin and 906% ± 69%inhibition*hours for the generic enoxaparin (P = .023; Table 2).
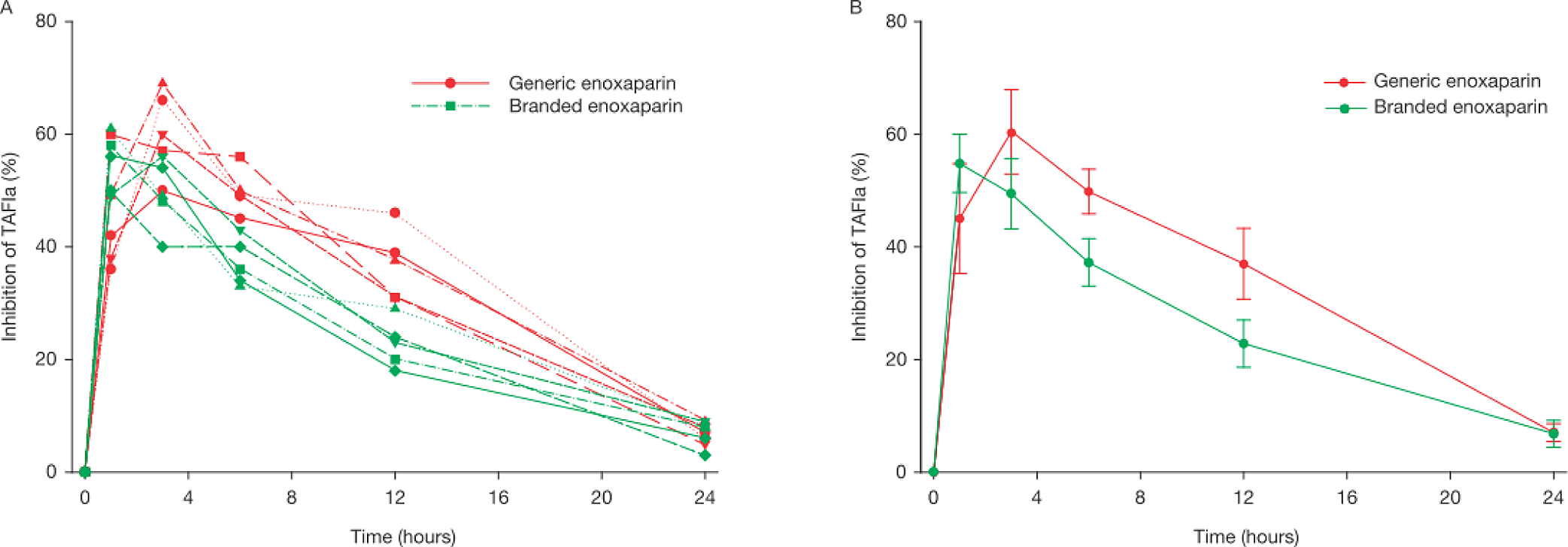
The percentage inhibition of the conversion of thrombin-activated fibrinolysis inhibitor (TAFI) to the active form (TAFIa) over a 24-hour period after administration of a 1 mg/kg subcutaneous dose of branded or generic enoxaparin to primates. A, Results for individual batches. B, Results are shown as mean values (± SEM) for all treated primates for the branded and generic enoxaparin products.
Discussion
To date, there are no published head-to-head comparisons of branded and generic LMWHs. Our study shows that simple analytical and pharmacokinetic/pharmacodynamic characteristics do not allow assuring similarity or dissimilarity between a branded and a generic enoxaparin. Both the branded and the generic products presented a good homology from batch to batch, indicating that both manufacturers used established and quality controlled processes. There are no significant differences in the average molecular weight profile and in the in vitro anti-FXa and anti-FIIa assay of antithrombotic potencies of the two enoxaparin products. However, many factors influence the biologic activity of clot formation; clot stability is also multifactorial and dependent on the amount of coagulation activators and inhibitors present in vivo. Thrombin generation plays a pivotal role in the development of a thrombus and inhibition of thrombin generation is the main determinant of the mechanism of action of enoxaparin.
Our analyses show that there are differences in the inhibition of thrombin generation and the dynamics of the inhibition of clot formation performed in vitro in plasma-based systems. The branded enoxaparin produced a strong inhibition of both thrombin generation and the progression of clot formation, whereas the generic product produced relatively weaker inhibitory effects and allowed for a faster progression of clot formation. These assays provide insight into the activation of the coagulation cascade, the rate of thrombin generation, the rate of fibrin clot formation, and the type and quality of fibrin clot eventually formed. They detect a combination of the direct anti-FIIa (thrombin inhibition) activity of enoxaparin as well as its inhibitory effect on the generation of thrombin (anti-FXa activity).
With regard to the studies in treated subjects (ex vivo), the inhibition of thrombin generation with the branded enoxaparin produced a consistently stronger effect than the generic enoxaparin across all batches tested, supporting the findings of the in vitro studies. The branded drug releases more TFPI, an important factor mediating the antithrombotic actions of LMWHs. Endothelial cells release TFPI, an inhibitor of tissue factor, when heparin is present. One of the effects of thrombin inhibition by a heparin/LMWH is to reduce the conversion of TAFI to TAFIa. The TAFIa plays a role in clot stability via an antifibrinolytic effect. In primates treated with equigravimetric doses of branded and generic enoxaparin, the generic product produced more inhibition of TAFIa in comparison with the branded product. Activation of TAFI is dependent on thrombin–thrombomodulin complex. However, there is suggestive evidence that with LMWHs this process is more complex, involving nonthrombin pathways with possible binding of certain oligosaccharides to the TAFI molecule. The TAFI activation may not, therefore, necessarily be directly proportional to the AT activity of the LMWH.
These findings underscore the importance of measurements beyond anti-FXa and anti-FIIa activities performed in purified AT-based biochemical systems for LMWHs. The differences exemplified between the branded and the generic products are not the mere consequence of variability but of real differences in the manufacturing process. These biologic drugs are complex, and they have multiple effects on different stages of the coagulation cascade and the hemostatic process which cumulatively produce their anticoagulant effect. There is no simple in vitro assay that can compare the anticoagulant efficacy of branded and generic LMWHs. 18 In fact, it has been stated that “the relationship between anti-FXa levels and clinical outcomes is not clear-cut”. 10
The observed variations between branded and generic enoxaparin could be due to compositional differences in the oligosaccharide components of the drugs. Specific oligosaccharide sequences within the branded and generic enoxaparins bind to plasma proteins and cell membranes, and the resultant effects on activation or inhibition of biological pathways could be relevant to these findings. Differences in the component oligosaccharide sequences in blood circulation at a given time may also be pertinent. In branded enoxaparin, only 30% of the oligosaccharide chains have been shown to have direct anticoagulant activities (evaluated by the anti-FXa and anti-FIIa assays),25,26 whereas the remainder have the so-called non-anticoagulant effects or are not yet characterized. This large component of the LMWH cannot be ignored for this injectable drug since the majority of the oligosaccharide chains of an LMWH may mediate its biologic effects, depending on the structure of the saccharides in the chains.
How these found differences translate in terms of clinical efficacy in patients is unknown. However, the data of this study imply that there is a need to measure a broader range of anticoagulant properties in order to understand the similarity or dissimilarity between LMWHs. Clinical trials performed in appropriate settings will aid to assure the prescriber the clinical similarity between a branded and a generic enoxaparin.
Footnotes
Acknowledgment
Editorial support in the preparation of this article (limited to editing for style, referencing and figure editing) was provided by Marinella Calle, PhD of Excerpta Medica, funded by Sanofi. The sponsors had no role in study design, data collection and data interpretation, and the decision to submit for publication. The authors are fully responsible for all content and editorial decisions related to the development of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a research grant to Dr. Jeanine Walenga from Sanofi (Paris, France).
