Abstract
Background:
Previous studies have demonstrated optimized diagnostic accuracy in utilizing higher antiheparin–platelet factor 4 (PF4) enzyme-linked immunosorbent assay (ELISA) optical density (OD) thresholds for diagnosing heparin-induced thrombocytopenia (HIT). We describe the incidence of positive serotonin release assay (SRA) results, as well as performance characteristics, for antiheparin–PF4 ELISA thresholds ≥0.4, ≥0.8, and ≥1.0 OD units in the diagnosis of HIT at our institution.
Methods:
Following institutional review board approval, we conducted a single-center retrospective chart review on adult inpatients with a differential diagnosis of HIT evaluated by both antiheparin–PF4 ELISA and SRA from 2012 to 2014. The major endpoints were to assess incidence of positive SRA results, sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and accuracy at antiheparin–PF4 ELISA values ≥0.4 OD units when compared to values ≥0.8 and ≥1.0 OD units. Clinical characteristics, including demographics, laboratory values, clinical and safety outcomes, length of stay, and mortality, were collected.
Results:
A total of 140 patients with 140 antiheparin–PF4 ELISA and SRA values were evaluated, of which 23 patients were SRA positive (16.4%) and 117 patients were SRA negative (83.6%). We identified a sensitivity of 91.3% versus 82.6% and 73.9%, specificity of 61.5% versus 87.2% and 91.5%, PPV of 31.8% versus 55.9% and 63.0%, NPV of 97.3% versus 96.2% and 94.7%, and accuracy of 66.4% versus 86.4% and 88.6% at antiheparin–PF4 ELISA thresholds ≥0.4, ≥0.8, and ≥1.0 OD units, respectively.
Conclusion:
Our study suggests an increased antiheparin–PF4 ELISA threshold of 0.8 or 1.0 OD units enhances specificity, PPV, and accuracy while maintaining NPV with decreased sensitivity.
Background
Laboratory testing to aid in the diagnosis of heparin-induced thrombocytopenia (HIT) can be evaluated through an antiheparin–platelet factor (PF4) enzyme-linked immunosorbent assay (ELISA) or serotonin release assay (SRA). 1,2 The antiheparin–PF4 ELISA detects antiheparin–PF4 antibodies, quantified by the degree to which light transmittance is impeded through the reagent that is proportional to the quantity of antibody present and expressed in optical density (OD) units. A non–immunoglobulin (Ig)-specific antiheparin–PF4 ELISA detects antibodies of the IgG, IgM, and IgA subclasses, whereas the antiheparin–PF4 IgG ELISA only detects IgG, the antibodies capable of causing platelet activation, as its name suggests. 1 –3 An OD of 0.4 or greater is considered positive by the antiheparin–PF4 IgG ELISA manufacturers, as it was the upper range of normal in population studies of healthy volunteers. However, patients may develop IgG-specific antibodies that cause no or weak activation of platelets. In these cases, the SRA can be used to detect the actual activation of platelets by the antiheparin–PF4 complexes and is considered the gold standard for laboratory diagnosis of HIT.
Raschke et al have previously demonstrated a difference of 93.5% versus 99.6% sensitivity, 93.6% versus 69.3% specificity, and false-positive rates of 6.4% versus 30.7% for diagnostic antiheparin–PF4 ELISA cutoff values of 0.8 versus 0.4 OD units, respectively. 4 Chan et al have displayed similarly promising results with the use of 1.0 OD units as an antiheparin–PF4 ELISA threshold. 5 The industry standard cutoff for a positive antiheparin–PF4 ELISA value indicative of HIT is 0.4 OD units or greater, which is also the cutoff at our institution for our IgG-specific ELISA. We sought to determine the incidence of positive SRA results in patients with antiheparin–PF4 ELISA values ≥0.4 OD units when compared to patients with antiheparin–PF4 ELISA values ≥0.8 and 1.0 OD units. For our major endpoints, we describe the incidence of positive SRA results, as well as the performance characteristics of sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and accuracy, for antiheparin–PF4 ELISA thresholds of 0.4, 0.8, and 1.0 OD units, respectively.
Methods
Following institutional review board approval, we conducted a single-center retrospective chart review on all adult inpatients with a differential diagnosis of HIT evaluated by both antiheparin–PF4 ELISA and SRA from 2012 to 2014. This time frame was chosen to include only results from an IgG antiheparin–PF4 ELISA, as our institution had transitioned from a non–Ig-specific antiheparin–PF4 ELISA prior to 2012. 6 The major endpoint was to assess the incidence of positive SRA results in patients with antiheparin–PF4 ELISA values ≥0.4 OD units when compared to patients with antiheparin–PF4 ELISA values greater than or equal to 0.8 and 1.0 OD units. We also sought to explore the ability of each antiheparin–PF4 ELISA threshold to correctly classify patients according to SRA results by calculating the sensitivity, specificity, PPV, NPV, and accuracy. Clinical characteristics, including demographics, laboratory values, clinical and safety outcomes, length of stay, and mortality, were collected. We retrospectively assessed the pretest probability of HIT using the 4Ts score, assigning 0 to 2 points for thrombocytopenia, timing of platelet decrease, thrombosis, and other causes of thrombocytopenia, to determine whether the likelihood of HIT was low, intermediate, or high. 7 We utilized diagnostic antiheparin–PF4 ELISA cutoff values of 0.4, 0.8, and 1.0 OD units in each group for comparison of our major endpoint. Patients were considered to have a diagnosis of HIT with a positive SRA result, as defined by a value greater than or equal to 20% serotonin release. Continuous data were evaluated by the Student t test, whereas noncontinuous data were analyzed by χ2 test or Mann-Whitney U test as appropriate. All P values were 2 sided and determined to be statistically significant at an α of 0.05 or less.
Results
A total of 140 patients with 140 antiheparin–PF4 ELISA and SRA values were evaluated. Of these, 23 patients were found to be SRA positive (16.4%), while 117 patients were found to be SRA negative (83.6%). Patient and clinicopathological characteristics are detailed in Tables 1 and 2, respectively, comparing SRA-positive patients with SRA-negative patients. With the exception of a longer median length of stay in the SRA-positive cohort, there were no differences in patient characteristics between the 2 groups. Most patients were exposed to unfractionated heparin (117, 83.6%), primarily for thromboembolism prophylaxis (64, 45.7%) and thromboembolism treatment (35, 25.0%). All SRA-positive patients (23, 100%) were exposed to unfractionated heparin. Thirty (21.4%) patients were exposed to heparin intraoperatively during cardiopulmonary bypass. As shown in Table 2, SRA-positive patients had higher mean 4Ts scores (5 vs 3, P < .01) and higher mean antiheparin–PF4 ELISA values (1.57 vs 0.39, P < .01). Performance characteristics of different OD unit thresholds for positive SRA results are described in Table 3. We identified a sensitivity of 91.3% versus 82.6% and 73.9% and specificity of 61.5% versus 87.2% and 91.5% at antiheparin–PF4 ELISA values ≥0.4, 0.8, and 1.0 OD units, respectively. We also identified a PPV of 31.8% versus 55.9% and 63.0%, NPV of 97.3% versus 96.2% and 94.7%, and accuracy of 66.4% versus 86.4% and 88.6% for each threshold. From our results, it is clear an increased antiheparin–PF4 ELISA threshold of 0.8 or 1.0 OD units significantly increases specificity, PPV, and accuracy while maintaining NPV.
Patient Characteristics.

Abbreviation: SRA, serotonin release assay.
aMean ± standard deviation.
bNumber (%).
cMedian (interquartile range).
Clinicopathological Characteristics of HIT.
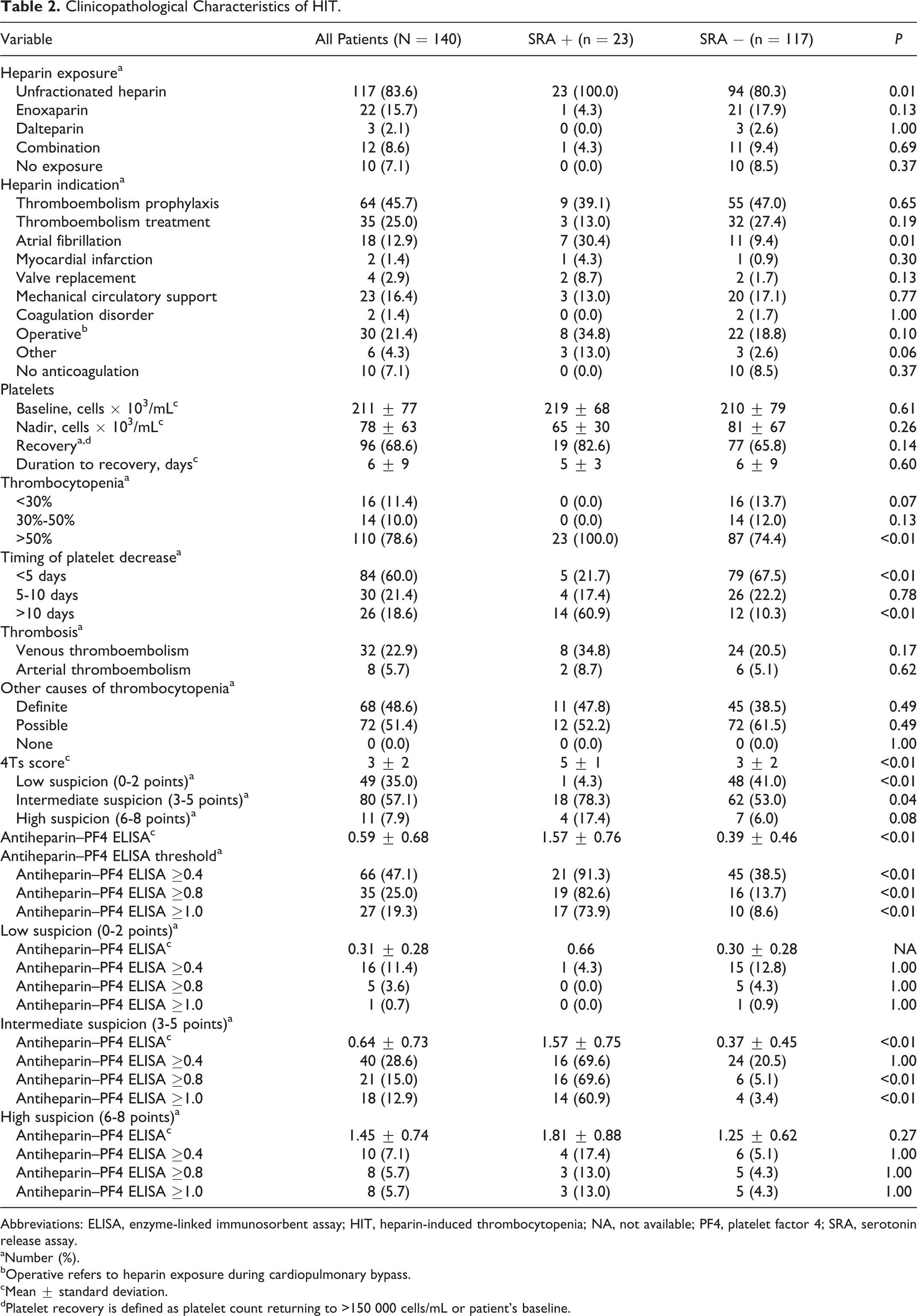
Abbreviations: ELISA, enzyme-linked immunosorbent assay; HIT, heparin-induced thrombocytopenia; NA, not available; PF4, platelet factor 4; SRA, serotonin release assay.
aNumber (%).
bOperative refers to heparin exposure during cardiopulmonary bypass.
cMean ± standard deviation.
dPlatelet recovery is defined as platelet count returning to >150 000 cells/mL or patient’s baseline.
Performance Characteristics of Different OD Unit Thresholds for a Positive SRA.
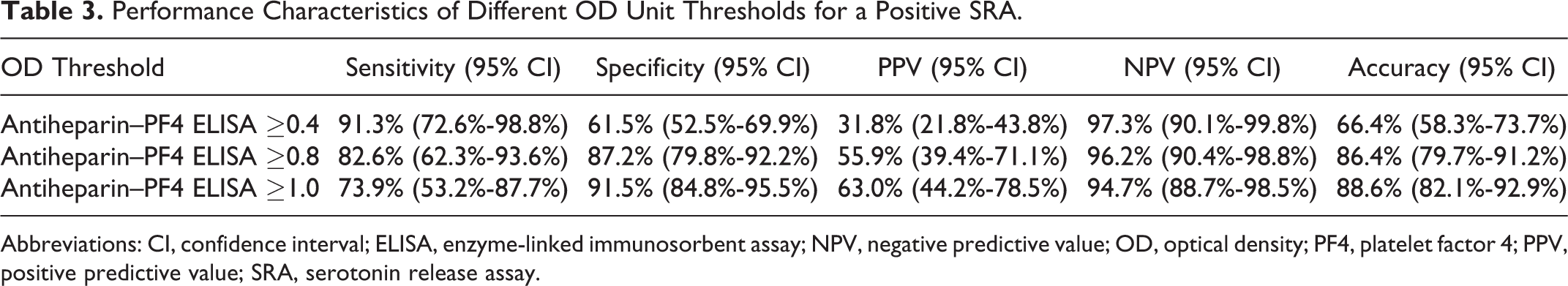
Abbreviations: CI, confidence interval; ELISA, enzyme-linked immunosorbent assay; NPV, negative predictive value; OD, optical density; PF4, platelet factor 4; PPV, positive predictive value; SRA, serotonin release assay.
Discussion
In this study, we identified increased specificity (61.5% vs 87.2% and 91.5%), increased PPV (31.8% vs 55.9% and 63.0%), and increased accuracy (66.4% vs 86.4% and 88.6%) with increased antiheparin–PF4 ELISA thresholds of 0.8 and 1.0 OD units, respectively. Although a previous study by Chan et al also demonstrated these findings, their results did not display the decreased sensitivity (91.3% vs 82.6% and 73.9%) we found. 5 In our study, we utilized a definition of >20% release of serotonin for a positive SRA result, whereas Raschke et al utilized a definition of >50% for a positive SRA result. 4 As a possible explanation, in 2 of these cases, the antiheparin–PF4 ELISA was drawn within 5 days of heparin exposure, which may have been too early for antiheparin–PF4 antibody development and may have resulted in a higher numerical antiheparin–PF4 ELISA value if taken at least 5 days after heparin exposure. However, a previous study by Warkentin et al demonstrated a small percentage of positive SRA results in patients with antiheparin–PF4 ELISA results between 0.4 and 1.0 (2.7%). 8 Although Whitlatch et al did not examine performance characteristics of the antiheparin–PF4 ELISA in their study, they noted a significantly higher antiheparin–PF4 ELISA value in patients with HIT when compared to those without (1.04 vs 0.63 and 0.60 OD units in patients with questionable and negative HIT, respectively). 9 Along with our results, this suggests the sensitivity of an increased antiheparin–PF4 ELISA diagnostic threshold would not be perfect but could aid clinicians in correctly identifying HIT as the most likely diagnosis. Raschke et al developed a decision support model algorithm for suspected HIT through utilization of the 4Ts score and confirmatory SRA testing based on antiheparin–PF4 ELISA results. 4,7 Our data support the approach of thoughtful consideration of the clinical picture through the 4Ts score, as well as important laboratory results of antiheparin–PF4 ELISA and SRA values, in real-world clinical practice to identify and treat the clinicopathological diagnosis of HIT. Although Chan et al displayed 100% sensitivity of increased antiheparin–PF4 ELISA diagnostic cutoffs, our results demonstrate we would have missed the diagnosis of HIT in 2, 4, and 6 patients at 0.4, 0.8, and 1.0 OD units, respectively, and these results exhibit a false-negative rate of 8.7%, 17.4%, and 26.1% at each threshold. 5 Our data represent a small sample size overall, although, and focus retrospectively on a single center’s practice. In combination with clinical discretion, increasing the diagnostic threshold for a positive antiheparin–PF4 ELISA may decrease unnecessary laboratory tests, including SRA, as well as limit the risks associated with empiric anticoagulation with a direct thrombin inhibitor while HIT is ruled out.
Conclusion
Our study suggests that an increased antiheparin–PF4 ELISA threshold of 0.8 or 1.0 OD units enhances specificity, PPV, and accuracy while maintaining NPV with decreased sensitivity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
