Abstract
The aim of this study was to determine which type of prophylaxis was effective for postoperative symptomatic venous thromboembolism (VTE) in patients with gynecological malignancies. A total of 1756 consecutive patients undergoing laparotomy as first-line treatment were included. In Period 1 (2004–2009), low-molecular weight heparin (LMWH) was not available for postoperative VTE prophylaxis, but available in after Period 2 (2009–2013). In Period 3 (2013–2020), patients with pretreatment VTE could switch from LMWH to direct oral anticoagulant (DOAC) as of 2015. Preoperative VTE was screened by measuring D-dimer, followed by venous ultrasound imaging, and computed tomography and/or perfusion lung scintigraphy. Postoperative symptomatic VTE occurred with an incidence of 2.8% by the measures without prophylactic LMWH administration in Period 1. The incidence of postoperative symptomatic VTE was 0.6% in Period 2 and 0.3% in Period 3, being significantly reduced compared with Period 1 (P < .01 and < .0001). The incidences were not significantly different between Periods 2 and 3, but no patient switching to DOAC in Period 3 (n = 79) developed symptomatic VTE. Our preoperative VTE screening and postoperative selective LMWH administration were significantly preventive against postoperative symptomatic VTE.
Keywords
Introduction
Venous thromboembolism (VTE) is one of the most important postoperative complications, and pulmonary embolism (PE) may sometimes be fatal. Reviews have found that approximately 70% of deaths from acute PE occur within an hour of onset 1 and 90% of cardiac arrests occur within 2 h. 2 Should acute PE occur, the consequences may be tragic before there is time for treatment to be given, and its prevention is therefore important. Guidelines on postoperative PE prevention have for some time recommended physical therapy starting from during surgery and postoperative anticoagulant drug therapy.3,4 In the 2020 updated guidelines of the American Society of Clinical Oncology (ASCO), the answer to the Clinical Question “Should patients with cancer undergoing surgery receive perioperative VTE prophylaxis?” recommends that “Prophylaxis should be commenced preoperatively.” 5 If asymptomatic deep vein thrombosis (DVT) or PE is present preoperatively, if this is not identified and intermittent pneumatic compression (IPC) implemented or highly invasive surgery is performed, this may cause the development of iatrogenic symptomatic VTE. The American College of Chest Physicians guidelines state that even with the prophylactic use of low-molecular weight heparin (LMWH) to prevent postoperative VTE after surgery other than orthopedic surgery, fatal PE still occurs in 0.6% of cases. 3 One reason may be that this includes patients with continuing asymptomatic VTE that had already been present preoperatively.
For this reason, since November 2004, we have been screening gynecological cancer patients for VTE with D-dimer (DD) before the start of treatment, and we have previously reported that VTE was discovered in 7.3% of patients with cervical cancer, 11.5% of those with endometrial cancer, and 27.0% of those with ovarian cancer, with VTE being asymptomatic in 98.2% of these patients. 6 In areas other than gynecological cancer, VTE screening before the start of treatment reportedly identified VTE in 17 of 103 (16.5%) patients with advanced pancreatic cancer, including 3 cases of PE, and of these, 14 were asymptomatic. 7 In patients with gastric cancer, VTE was identified in 7 of 160 (4.4%) cases, all of whom were asymptomatic, 8 and asymptomatic DVT was also identified in 9 of 65 (13.9%) patients with bladder cancer. 9 When VTE has been identified before the start of treatment, we have tried to prevent the development of symptomatic VTE by responding appropriately at that point, providing physical therapy and/or anticoagulant drug therapy intraoperatively and postoperatively with patients selected according to defined criteria. VTE prophylaxis has varied over the years in light of lessons learned concerning its effectiveness and changes in health insurance drug coverage. VTE prophylaxis used in each period and the incidence of postoperative symptomatic VTE for each method of prophylaxis were analyzed, and a retrospective investigation of which type of VTE prophylaxis was effective for patients with gynecological malignancies was conducted.
Materials and Methods
Patients and Postoperative VTE Prophylaxis
This was a retrospective cohort study. The study subjects were 1756 consecutive patients with gynecological epithelial malignancies (not including carcinosarcoma) who underwent laparotomy as first-line treatment in the University of Tsukuba Hospital between November 2004 and December 2020 (Figure 1). They included the patients from among those described in our previous series of papers.6,10–12

Study flow chart. Abbreviations: VTE, venous thromboembolism; DVT, deep vein thrombosis; PE, pulmonary embolism.
From November 2004 to July 2009 (Period 1), during which LMWH could not be used as postoperative VTE prophylaxis because it was not covered by health insurance, there were 495 study subjects. From August 2009 to July 2013 (Period 2), LMWH was covered by health insurance for use as postoperative VTE prophylaxis, and there were 501 study subjects during this period, with those to whom LMWH was administered either until discharge or until postoperative day 14, whichever was earlier. From August 2013 to December 2020 (Period 3), there were 760 study subjects, and if anticoagulant drug therapy was used, LMWH was then administered until at least postoperative day 14. In Period 3, the treatment and prevention of recurrence of venous thromboembolism (DVT and PE) were added to the health insurance indications for direct oral anticoagulants (DOACs). For this reason, from February 2015, patients found to have pretreatment VTE were allowed to switch from LMWH to DOAC on postoperative day 3 or 4 if they were sufficiently capable of oral intake. There were 184 study subjects from August 2013 to January 2015 and 576 from February 2015 to December 2020.
The numbers of histologically confirmed cervical cancer patients in Periods 1, 2, and 3 were 133, 118, and 176, respectively; the numbers of histologically confirmed endometrial cancer patients were 229, 233, and 324, respectively; and the numbers of patients with suspected ovarian cancer before the start of treatment in whom ovarian cancer (not including borderline malignant tumors), fallopian tube cancer, or peritoneal cancer (included in ovarian cancer figures below) was confirmed clinically and histologically after surgery were 133, 150, and 260, respectively. Patients’ characteristics are shown in Table 1. The occurrence of symptomatic VTE was evaluated until postoperative day 21.
Patients’ Characteristics (n = 1756).
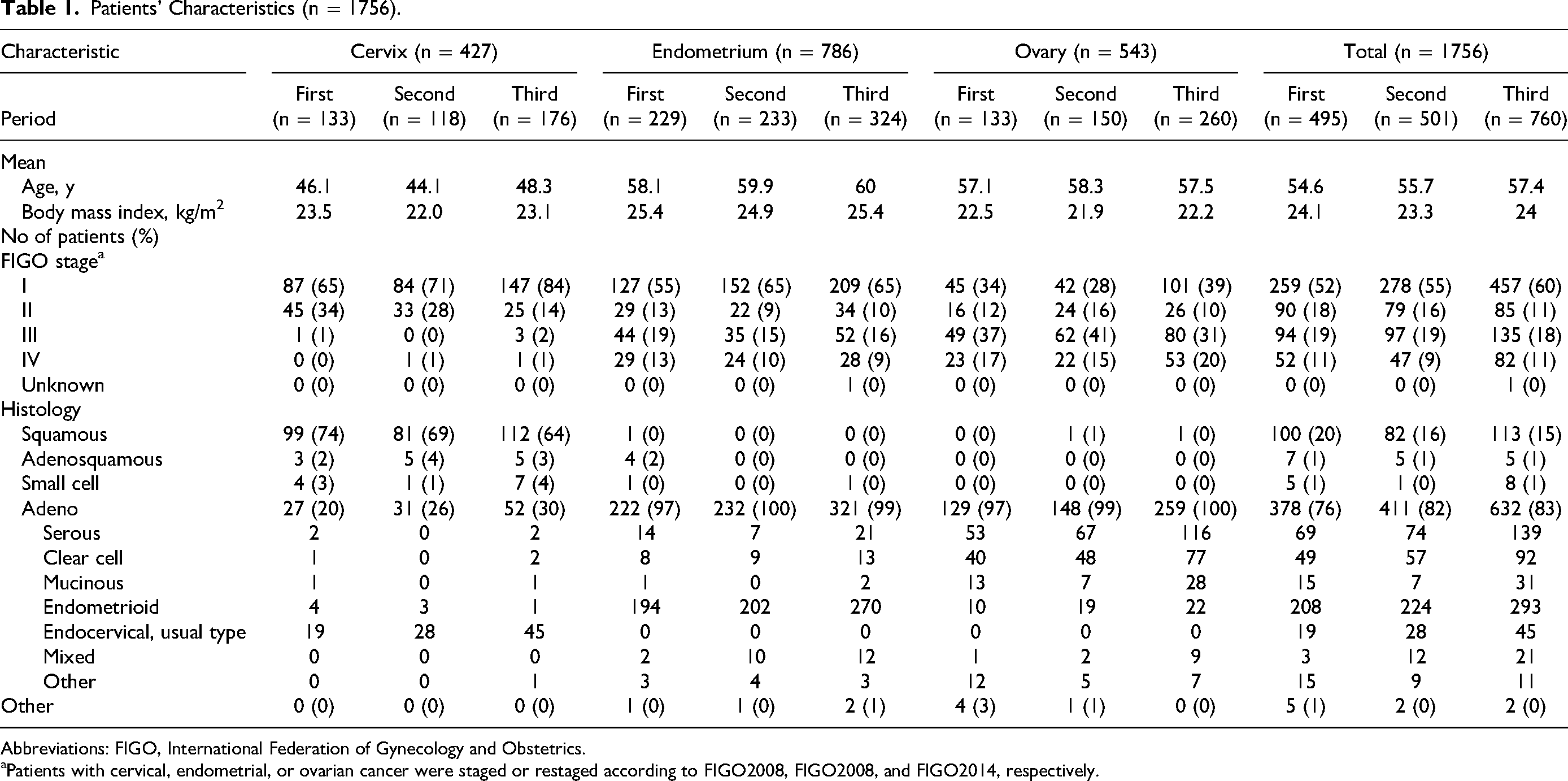
Abbreviations: FIGO, International Federation of Gynecology and Obstetrics.
Patients with cervical, endometrial, or ovarian cancer were staged or restaged according to FIGO2008, FIGO2008, and FIGO2014, respectively.
Detection of VTE and PE Before Surgery
DD was measured in all study subjects, although the method of measurement varied in different periods, and if the result exceeded our institution's reference value, then venous ultrasound imaging (VUI) was conducted to screen for DVT.10–14 For ovarian cancer patients, VUI was conducted in all cases irrespective of the DD level, with the objective of calculating the sensitivity, specificity, and negative and positive predictive values of DD. 10 Preoperative contrast-enhanced computed tomography (CT) was also performed, primarily to look for lymph node and distant metastases, and asymptomatic VTE was also sometimes discovered in this process. If DVT was identified, PE screening was conducted; during Period 1, this was done by either perfusion lung scintigraphy or contrast-enhanced CT, and from Period 2, by contrast-enhanced CT in almost all cases.
Treatment for VTE Before Surgery
If VTE was identified, until January 2015, in principle, all patients were admitted and given anticoagulant therapy with unfractionated heparin (UFH). From February 2015, if the thrombus was localized in the soleus vein and no floating thrombi were present, outpatient management by DOAC was also allowed if the thrombus was located distal to the popliteal vein, organized or subacute, and asymptomatic. However, patients with suspected ovarian clear cell carcinoma, which produces tissue factors and is thought to have a high incidence of VTE,14,15 as well as patients with VTE considered to be due to the accumulation of ascites or a large tumor, were admitted and treated with UFH anticoagulant therapy out of concern that the VTE might become exacerbated before surgery.
In addition, for patients with central DVT, floating thrombi, or PE, the Department of Cardiology was consulted on the need for inferior vena cava filter (IVCF) placement, but in fact this was required for only two patients who had already had an IVCF inserted at another hospital and two in whom insertion was performed at our hospital, and all those patients underwent initial treatment before 2010.
In terms of cancer treatment, if the risk that preoperative VTE might develop into symptomatic VTE as a result of surgery was predicted to be low, including patients in whom DVT was localized in the soleus vein or exhibited organizing tendencies, the standard surgical procedure was chosen. For patients with advanced disease that was nevertheless expected to respond well to preoperative chemotherapy, a common scenario in ovarian high-grade serous carcinoma, neoadjuvant chemotherapy (NAC) was chosen, and a strategy of treating VTE during NAC to reduce the risk of postoperative symptomatic VTE after interval debulking surgery (IDS) was adopted. In the case of tumors believed to contribute directly to the occurrence of VTE, including clear cell carcinoma and bulky tumors, the strategy adopted was to avoid the standard surgical procedure because its high level of invasiveness meant that it might cause postoperative symptomatic VTE; instead, limited surgery to remove the primary lesion alone was conducted as initial surgery, and the standard surgery was performed as secondary surgery after the VTE had improved or resolved. IPC was not used either intraoperatively or postoperatively for patients with preoperatively identified VTE. Elastic stockings were worn by the patients.
Anticoagulant Therapy
Preoperative anticoagulant therapy was not administered unless preoperative VTE was identified. When VTE was identified preoperatively, for patients who had been admitted for continuous UFH and scheduled to undergo surgery, the UFH was discontinued 12 h preoperatively. For patients who had not been admitted but were being managed as outpatients with oral anticoagulant drug therapy, and for those who had been taking oral anticoagulants in the interval between NAC and IDS or between limited surgery and the standard surgical procedure was conducted, therapy was switched to UFH 7 days preoperatively if they had been taking warfarin or 3 days preoperatively if they had been taking a DOAC, and the UFH was then discontinued 12 h preoperatively. In the case of DOAC, the reason for switching 3 days before was to determine the postoperative starting dose of UFH.
In principle, UFH for pretreatment VTE was resumed at 8–12 h postoperatively and continued until the patient was fully ambulatory, generally after 3–4 days, after which it was switched to warfarin. In after Period 2, LMWH was covered by the health insurance to include “Prevention of venous thromboembolism in abdominal surgery patients with high risk of venous thromboembolism,” and was therefore administered from 24 h postoperatively, not only to patients with pretreatment VTE, but also to those who underwent lymphadenectomy, with a BMI ≥28 kg/m2, with a history of VTE, or with factors predisposing them to thromboembolism. From February 2015, patients found to have pretreatment VTE were allowed to switch from LMWH to DOAC, and in this case, they were instructed to take it for at least 6 months.
Statistical Analysis
The incidences of VTE between the study periods were compared by two-sided Fisher's exact test using R version 4.0.5 (https://www.r-project.org/). P values less than .05 were considered as statistically significant.
Results
As described above, preoperative prophylaxis for postoperative symptomatic VTE was consistent throughout all three periods, but the postoperative treatment was different in each period. In order to identify the effective type of prophylaxis for postoperative symptomatic VTE in patients receiving laparotomy for gynecological malignancies, methods of prophylaxis and postoperative symptomatic VTE events and their incidences were investigated and compared among the study periods.
In Period 1, preoperative VTE was identified in 67 patients, of whom 47 had DVT alone, 19 both DVT and PE, and 1 PE alone (Figure 1). Postoperative UFH resumption was delayed in some cases for reasons including intraoperative massive hemorrhage or vascular damage. In Period 1, a total of 14 patients developed postoperative symptomatic VTE (Figure 1 and Table 2). VTE had been identified before the start of treatment in five of these patients, all of whom developed symptomatic DVT and PE, but there were no fatalities. Of the 428 patients in whom preoperative VTE had not been identified, 3 developed symptomatic DVT and PE and 6 developed symptomatic DVT alone, with one death from PE (Figure 1). In this period, the incidence of postoperative symptomatic VTE was 2.8%, and that of symptomatic PE was 1.6% (Table 3). Four of the five patients who developed postoperative symptomatic PE after having been identified to have preoperative VTE had experienced major intraoperative bleeding for reasons such as vascular damage, and the resumption of UFH had been delayed until 18, 18, 19, and 23 h postoperatively due to the risk of recurrent hemorrhage. The nine patients who developed postoperative symptomatic VTE despite preoperative VTE not having been identified had all undergone pelvic and/or para-aortic lymphadenectomy. The patient who developed symptomatic PE on postoperative day 1 had a body mass index (BMI) of 38.5 kg/m2. The patient who developed symptomatic DVT on postoperative day 2 had a BMI of 28.9 kg/m2 and a history of VTE. In the remaining seven cases, onset occurred on postoperative day 12–21 (median day 12), and no factors that might have caused VTE other than lymphadenectomy were apparent.
Patients Who Developed Symptomatic Venous Thromboembolism After Surgery Within 21 Postoperative Days.

Abbreviations: BMI, body mass index; DD, D-dimer; DVT, deep vein thrombosis; PE, pulmonary embolism; N, negative; P, positive; NE, not examined; POD, postoperative day; IVCF, inferior vena cava filter; AT, anticoagulant therapy.
Comparison of Postoperative Symptomatic VTE Incidences Among Study Periods.

Abbreviations: VTE, venous thromboembolism; DVT, deep vein thrombosis; PE, pulmonary embolism; DOAC, direct oral anticoagulant.
P value †<.01 and ††<.0001 by Fisher's exact test.
In Period 2, preoperative VTE was identified in 73 patients, of whom 56 had DVT alone, 15 both DVT and PE, and 2 PE alone (Figure 1). Learning from the results of Period 1, care was taken to ensure that UFH was resumed at 8–12 h postoperatively for all those patients in whom VTE had been present before the start of treatment. In Period 2, three patients developed postoperative symptomatic VTE (Figure 1 and Table 2). None of them had been identified as having pretreatment VTE, and of the 428 patients in whom preoperative VTE had not been identified, 2 developed symptomatic PE, and 1 developed symptomatic DVT alone. In this period, the incidence of postoperative symptomatic VTE was 0.6%, and that of symptomatic PE was 0.4% (Table 3). Compared with Period 1, the incidence of symptomatic VTE was significantly lower (P < .01; Table 3). One of the patients who developed PE was discharged on postoperative day 7 and so had received LMWH for only 7 days before developing PE after discharge on postoperative day 11. The other two patients developed PE on day 20 and DVT on day 21, respectively, after having taken LMWH for 14 days.
In Period 3, preoperative VTE was identified in 111 patients, of whom 91 had DVT alone, 16 both DVT and PE, and 4 PE alone (Figure 1). Because one of the patients in Period 2 had developed symptomatic PE having discontinued LMWH after 7 days, in principle, patients who were treated with LMWH continued taking it for 14 days. In Period 3, two patients developed postoperative symptomatic VTE (Figure 1 and Table 2). None of them had been identified as having pretreatment VTE, and of the 649 patients in whom preoperative VTE had not been identified, 1 developed symptomatic DVT and PE both, and 1 developed symptomatic DVT alone (Figure 1). In this period, the incidence of postoperative symptomatic VTE was 0.3%, and that of symptomatic PE was 0.1% (Table 3). Compared with Period 1, the incidence of symptomatic VTE was significantly lower (P < .0001; Table 3), but there was no statistically significant reduction compared with Period 2 (P = .39). The one patient who developed PE did so on day 21, after having taken LMWH for 14 days. The other patient, who developed DVT on postoperative day 14, had a sheet-shaped metastatic lymph node surrounding a vein that could not be removed and was left in place. The patient who developed symptomatic DVT had received anticoagulant drug therapy. None of those patients who were taking DOACs in Period 3 developed symptomatic VTE, but with no significant reduction (n = 79; Table 3). The occurrence of Grade ≥3 postoperative hemorrhage was not increased by taking DOACs within Period 3 (0.4% [3/681] vs 0% [0/79]).
Our current perioperative symptomatic VTE prophylaxis for gynecological malignancies is summarized in Figure 2.

Our current perioperative symptomatic VTE prophylaxis for gynecological cancer patients. For patients without ②③④⑤, elastic stockings and IPC are used during and after surgery to prevent postoperative VTE, with no administered anticoagulant. UFH is administered from 8–12 h to until 24 h after end of surgery for patients with ③④⑤, then UFH is changed to LMWH. LMWH is administered for the shorter period of “until discharge” or “14 days after surgery” in patients with ②③④⑤. Patients having ④⑤ can switch from LMWH to DOAC, usually 3–4 days after surgery, then DOAC is given until 6 months at least.
Discussion
We first started screening patients with gynecological malignancies for pretreatment VTE when we treated a 72-year-old patient with endometrial clear cell carcinoma who had asymptomatic DVT and PE. 16 Having administered UFH and inserted an IVCF, we did not conduct retroperitoneal lymphadenectomy, but performed limited surgery comprising simple hysterectomy and bilateral adnexectomy, and treatment was completed without the occurrence of symptomatic VTE.
Today, the concept of cancer-associated thrombosis is well established, but we were unaware of it at the time. Nevertheless, we started screening in November 2004 because we considered that the presence of asymptomatic VTE from before the start of treatment might be a factor in the occurrence of postoperative VTE or of VTE during chemotherapy or radiotherapy.
Our pretreatment handling of patients with preoperatively identified VTE remained unchanged from Period 1 to Period 3, and we intend to continue the same policy in the future. However, anticoagulant therapy is the mainstay of VTE treatment, and the use of IVCFs is limited to cervical cancer and endometrial cancer patients with severe bleeding who are unable to take anticoagulants. 17
In Period 1, some patients with preoperatively identified VTE developed PE soon after surgery, and the possibility that this was caused by the VTE that had already been present prior to treatment cannot be excluded (Table 2). The 2007 ASCO guidelines state that “Prophylaxis should be commenced preoperatively, or as early as possible in the postoperative period,” and the delayed postoperative resumption of UFH may have been a causative factor 18 (Table 2). In terms of the characteristics of those patients in whom preoperative VTE was not identified who developed postoperative symptomatic VTE, all nine had undergone a surgical procedure including lymphadenectomy, and in seven of the nine, it developed at a late stage, more than 10 days postoperatively. One cause of this late-onset may have been venous displacement by lymphocysts as suggested in some reports19,20 including ours where all 4 of 132 (3.0%) patients with no pretreatment VTE developed symptomatic VTE on 12, 12, 13, and 31 days after surgery with formation of lymphocysts, 12 although this has not been confirmed in the present study. Not only did both the patients with endometrial cancer who developed it on postoperative days 1 and 2 have a BMI ≥28 kg/m2, but the patient who developed it on day 2 also had a history of thromboembolism, and these may also have been causative factors, keeping in line with our previous report where obesity was found as an independent and significant risk factor for posttreatment VTE in endometrial cancer. 11 In Period 1, the duration of postoperative UFH administration was designated as until the patient was fully ambulatory (normally 3–4 days). The ASCO guidelines state that “Prophylaxis should be continued for at least 7 to 10 days postoperatively. Prolonged prophylaxis for up to 4 weeks may be considered in patients undergoing major abdominal or pelvic surgery for cancer with high-risk features such as residual malignant disease after operation, obese patients, and those with a previous history of VTE,” 18 and the short administration period may also have been a factor contributing to the development of VTE in Period 1.
In Period 2, VTE prophylaxis was provided with enoxaparin sodium, a form of LMWH. In view of the guidelines, an administration period of 4 weeks was proposed, 18 but because the package insert of the LMWH that is covered by Japanese health insurance states that “The efficacy and safety of administration for 15 days or more have not been investigated in Japanese clinical trials,” a maximum 14-day period was adopted. Had it been possible to continue administration for 4 weeks in Period 2, in line with the ASCO guidelines, the cases of symptomatic VTE that occurred on days 20 and 21 after the patients had taken LMWH for 14 days might have been prevented. The case of VTE that occurred on day 11 after the patient had only taken LMWH for 7 days because she had been discharged on postoperative day 7 might also have been prevented by an administration period of 14 days.
The incidence of symptomatic postoperative VTE in Period 1 was significantly reduced both in Periods 2 and 3, while there was no significant difference between Periods 2 and 3 (Table 3). As described in the Materials and Methods section, the differences in measures among the study periods are mainly as follows: Periods 2 and 1, postoperative LMWH instead of no anticoagulant for VTE prophylaxis and postoperative LMWH instead of UFH for pretreatment VTE; Periods 3 and 1, postoperative LMWH instead of no anticoagulant for VTE prophylaxis and postoperative LMWH instead of UFH for pretreatment VTE as well as perioperative DOAC instead of warfarin for pretreatment VTE; Periods 3 and 2, the duration (≥ or ≤ 14 days) of postoperative LMWH for VTE prophylaxis as well as DOAC instead of warfarin for pretreatment VTE. Accordingly, LMWH is suggested to be effective for preventing postoperative symptomatic VTE occurrence compared with no anticoagulant and for preventing postoperative symptomatic VTE recurrence compared with UFH, while the effect of LMWH duration or DOAC instead of warfarin is undetermined.
A recent randomized trial examining safety and efficacy of 28-day apixaban versus enoxaparin for preventing postoperative VTE, screened by clinical criteria or standard symptoms, in gynecological cancer patients reported that there were no differences in rates of VTE events (1.0% vs 1.5%; OR, 1.57; 95% CI, 0.26–9.50; P = .68) as well as bleeding events.21,22 The authors concluded that apixaban was a safe alternative to LMWH with equivalent efficacy of postoperative thromboprophylaxis. The VALERIA study, another recent prospective, randomized trial comparing 30-day rivaroxaban versus enoxaparin in VTE prophylaxis after major gynecological cancer surgery, reported that symptomatic VTE was 2.63% versus 1.75% (relative risk, 1.50; 95% CI, 0.25–8.80; P = .6535) and major/clinically relevant nonmajor bleeding was 0% versus 2.63% (hazard ratio, 0.14; 95% CI, 0.007–2.73; P = .1963). 23 The authors concluded that their results support the hypothesis that DOACs might be an attractive alternative strategy to LMWH to prevent VTE after major gynecological cancer surgery. As regards the current study, the incidence of postoperative symptomatic VTE was 0.6% in Period 2 and 0.3% in Period 3 (Table 3), both of which were lower than those by LMWH and DOAC in the two trials.21,23 Although the prophylactic effect of the measures in Period 1 is undetermined due to the absence of publications on the postoperative symptomatic VTE incidences without prophylaxis, it is possible that our preoperative VTE screening contributes to effective prevention of postoperative symptomatic VTE in all the study periods. Furthermore, none of the patients with preoperative VTE who switched to DOAC in Period 3 developed symptomatic VTE with no increase in Grade ≥3 postoperative hemorrhage (Table 3). In addition to appropriate pretreatment screening and treatment for VTE, should it be possible to provide long-term postoperative anticoagulant drug therapy by expanding the indications for health insurance coverage of DOAC use to include “Prevention of venous thromboembolism in abdominal surgery patients with high risk of venous thromboembolism” as with LMWH, it might be possible to reduce the incidence of symptomatic VTE following surgery for gynecological malignancy to close to zero. Future trials are warranted to test our hypothesis.
In the patient who underwent intraoperative venous suturing and the patient in whom a large metastatic lymph node surrounding a vein was left in place, DVT may have developed due to venous wall damage. In light of this, we are considering administering LMWH to patients with possible venous damage. Our intended perioperative symptomatic VTE prophylaxis for gynecological malignancies going forward is summarized in Figure 2.
This was a retrospective study in a single institution, and as such includes a number of biases. The anticoagulants used perioperatively for symptomatic VTE prophylaxis varied over time. Comparing the study periods may not exactly correspond to the comparison of actual prophylactic measures. Because during the study period of more than 15 years, there were changes in the patient's lifestyle such as eating habits, progression of late marriage and late birth, and the spread of oral contraceptives/low-dose estrogen progestin.
Conclusion
We herein investigated which type of VTE prophylaxis was efficacious for patients with gynecological malignancies by comparing the incidences of postoperative symptomatic VTE among the study periods of different prophylactic methods. The incidences in Periods 2 and 3 were both significantly lower than Period 1, and no patient who switched to DOAC in Period 3 developed symptomatic VTE. Our preoperative VTE screening and postoperative selective LMWH administration were effective for preventing postoperative symptomatic VTE. Because of the small number of events, postoperative symptomatic VTE would be difficult to investigate in a prospective, confirmatory study, but the emergence of high-quality evidence from prospective studies are awaited in order to validate our findings.
Footnotes
Author Contributions
Takeo Minaguchi and Toyomi Satoh conceived the idea of the manuscript and supervised the project. Nobutaka Tasaka, Kaoru Fujieda, Takeo Minaguchi, and Toyomi Satoh analyzed, and interpreted data and wrote the manuscript. Ayumi Shikama, Azusa Akiyama, and Sari Nakao helped with statistical analyses. Asuka Suzuki, Asami Suto, and Hiroya Itagaki collected data. All authors discussed the results and commented on the manuscript.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Toyomi Satoh received speaker's fees from Daiichi Sankyo Co, Ltd. The remaining authors declare no conflict of interest for this article.
Ethics Approval
Ethical approval to report this study was obtained from the Institutional Ethics Committee at University of Tsukuba Hospital (H30-45).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by JSPS KAKENHI Grant Number JP18K09218 (Grant-Aid for Scientific Research (C)).
Informed Consent
Informed consent for patient information to be published in this article was not obtained because the institutional ethics committee waived its requirement due to the opt-out approach in accordance with national regulations.
