Abstract
Objectives:
Registro Informatizado de Enfermedad TromboEmbólica (RIETE) database was used to investigate whether neurosurgical patients with venous thromboembolism (VTE) were more likely to die of bleeding or VTE and the influence of anticoagulation on these outcomes.
Methods:
Clinical characteristics, treatment details, and 3-month outcomes were assessed in those who developed VTE after neurosurgery.
Results:
Of 40 663 patients enrolled, 392 (0.96%) had VTE in less than 60 days after neurosurgery. Most patients in the cohort (89%) received initial therapy with low-molecular-weight heparin, (33% received subtherapeutic doses). In the first week, 10 (2.6%) patients died (8 with pulmonary embolism [PE], no bleeding deaths; P = .005). After the first week, 20 (5.1%) patients died (2 with fatal bleeding, none from PE). Overall, this cohort was more likely to develop a fatal PE than a fatal bleed (8 vs 2 deaths, P = .058).
Conclusions:
Neurosurgical patients developing VTE were more likely to die from PE than from bleeding in the first week, despite anticoagulation.
Introduction
Venous thromboembolism (VTE) is a major cause of morbidity and mortality in the neurosurgical population. 1 –6 These patients, in particular, have the following notable risk factors for VTE: intracranial surgery, prolonged length of procedures, malignancy, weakness or paresis, and advanced age. 4,6 –9 Current guidelines from the American College of Chest Physicians recommend that patients with acute VTE be treated initially with low-molecular-weight heparin (LMWH), unfractionated heparin (UFH), or fondaparinux, followed by long-term treatment with vitamin K antagonists (VKAs). 10 However, there is uncertainty about the optimal therapy in patients with VTE who underwent recent neurosurgery, as they are often excluded from randomized clinical trials of anticoagulant therapy. 9 These patients are perceived to be at substantial risk of rebleeding if therapy with anticoagulants is prescribed, yet they are at increased risk of recurrent VTE and/or pulmonary embolism (PE) in the absence of treatment.
The Registro Informatizado de Enfermedad TromboEmbólica (RIETE; or in English: Computerized Registry of Patients With Venous Thromboembolism) initiative is an ongoing, multicenter, international (Spain, Argentina, Belgium, Brazil, Canada, Czech Republic, Ecuador, France, Germany, Greece, Hungary, Ireland, Israel, Italy, Poland, Portugal, Republic of Macedonia, Switzerland, and the United States enroll patients) observational registry of consecutive patients with symptomatic, objectively confirmed, acute VTE. 11 –15 Since its inception, RIETE was designed to gather data on the clinical characteristics, treatment patterns, and outcomes in patients with VTE. The present study utilized the RIETE cohort to investigate whether VTE is a major cause of morbidity and mortality in the neurosurgical intracranial- and spinal-operated population. The main objectives were to understand the natural history of postoperative VTE in neurosurgical patients as well as to determine the risk benefit ratio of treating these patients with anticoagulation.
Methods
We assessed the clinical characteristics, treatment details, and 3-month outcome in those who developed VTE in less than 60 days after intracranial and/or spinal neurosurgery in the RIETE database. Venous thromboembolism was confirmed by objective tests (contrast venography or full leg compression ultrasonography for suspected deep vein thrombosis [DVT]; pulmonary angiography, lung scintigraphy, or helical computed tomography scan). Patients were excluded if they were currently participating in a therapeutic clinical trial. This analysis was approved by the Institutional Review Board of NorthShore University HealthSystem (Evanston, Illinois). All patients (or their legal power of attorney if indicated) provided written consent for their participation in the registry.
Study Design
All neurosurgical patients in RIETE, with acute VTE in less than 60 days after intracranial or spinal surgery, were evaluated. The major outcomes included the development of fatal PE or fatal bleeding during the first 90 days after the diagnosis of VTE. Secondary outcomes included major bleeding and VTE recurrences. The causes of death were determined by the attending physicians. Fatal PE, in the absence of autopsy, was defined as any death appearing in less than 7 days after PE diagnosis, in the absence of an alternative cause of death. Fatal bleeding was defined as any death occurring in less than 7 days after a major bleeding episode. Bleeding complications were classified as “major” if they were overt and required a transfusion of 2 units of blood or more, were retroperitoneal, spinal, or intracranial, or if they were fatal.
Study Variables and Definitions
The following parameters were recorded when the qualifying episode of VTE was diagnosed: patient’s sex, age, and body weight; chronic heart or lung disease; additional risk factors for VTE; laboratory data on admission; anticoagulant treatment; and outcomes during the first 3 months of VTE therapy.
Follow-Up
Patients were managed according to the clinical practice of each participating hospital and were not subject to any predetermined intervention. During follow-up, special attention was paid to any signs or symptoms suggesting recurrent VTE or bleeding complications. Each episode of clinically suspected recurrent DVT or PE required documentation by repeat ultrasonography, venography, lung scanning, helical computed tomography (CT) scan, or pulmonary angiography.
Data Collection and Monitoring
The primary investigator made sure that eligible patients were consecutively enrolled. Data were recorded onto a computer-based case report form at each participating hospital and submitted to a centralized coordinating center through a secure Web site. Encryption of data was used to enhance confidentiality and security. Data quality was regularly monitored and documented electronically to detect inconsistencies or errors. Data quality was also monitored by periodic visits to participating hospitals, by contract research organizations, which compared the medical records with the Web database. Patient identities remained confidential and were identified by a unique number assigned by the study coordinating center responsible for the data management.
Statistical Analysis
Mann-Whitney U test and Student t test were used to compare continuous variables. Categorical variables were compared by the Fisher exact test, and the odds ratios and corresponding 95% confidence intervals were calculated. Survival curves were constructed according to the Kaplan-Meier method. SPSS software (version 15, SPSS Inc Chicago, Illinois) was used for statistical management of the data.
Results
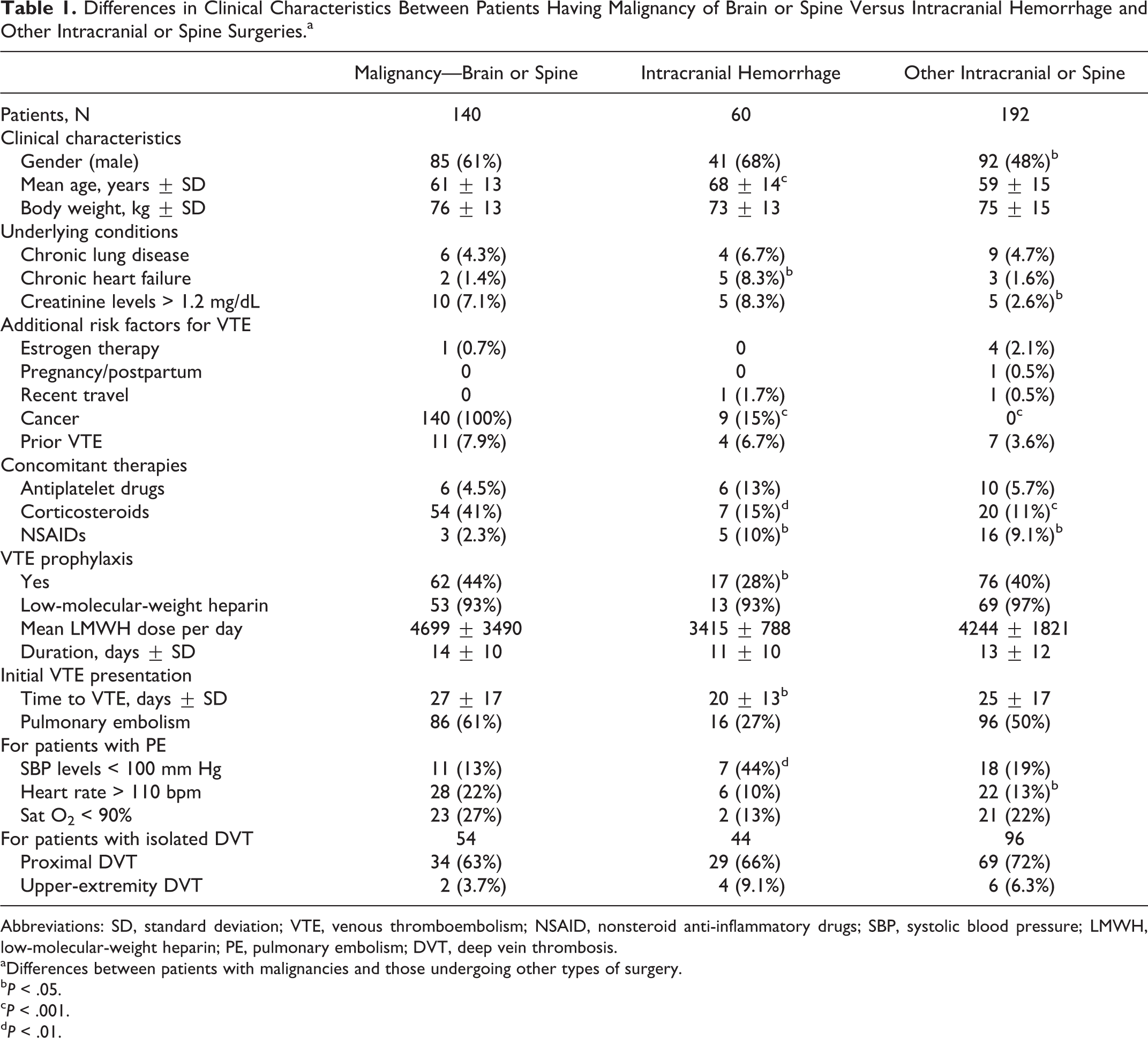
Of the total 40 663 patients with symptomatic VTE in the RIETE database, 392 (0.96%) had VTE within 60 days following a neurosurgical procedure. Of these, 140 (36%) patients underwent oncologic surgery (116 with brain cancer, 9 lung, 4 breast, 4 prostate, 2 colorectal, 2 bladder, 2 kidney, and 10 other). A total of 60 (15%) patients were operated on because of cerebral bleeding and 192 (49%) patients had other reasons for surgery (34 with hernia repair, 25 meningioma, 11 other benign tumors, 13 chronic subdural hematoma, 8 lumbar channel stenosis repair, 8 arteriovenous malformation, 5 cerebral biopsy, 5 valvuloperitoneal derivation, and 75 not reported). Table 1 depicts the differences in clinical characteristics of patients having malignancy in brain or spine or intracranial hemorrhage surgeries or other surgeries. A total of 198 (51%) patients presented with PE (with or without concomitant DVT) and 194 patients presented with DVT alone.
Differences in Clinical Characteristics Between Patients Having Malignancy of Brain or Spine Versus Intracranial Hemorrhage and Other Intracranial or Spine Surgeries.a
Abbreviations: SD, standard deviation; VTE, venous thromboembolism; NSAID, nonsteroid anti-inflammatory drugs; SBP, systolic blood pressure; LMWH, low-molecular-weight heparin; PE, pulmonary embolism; DVT, deep vein thrombosis.
a Differences between patients with malignancies and those undergoing other types of surgery.
b P < .05.
c P < .001.
d P < .01.
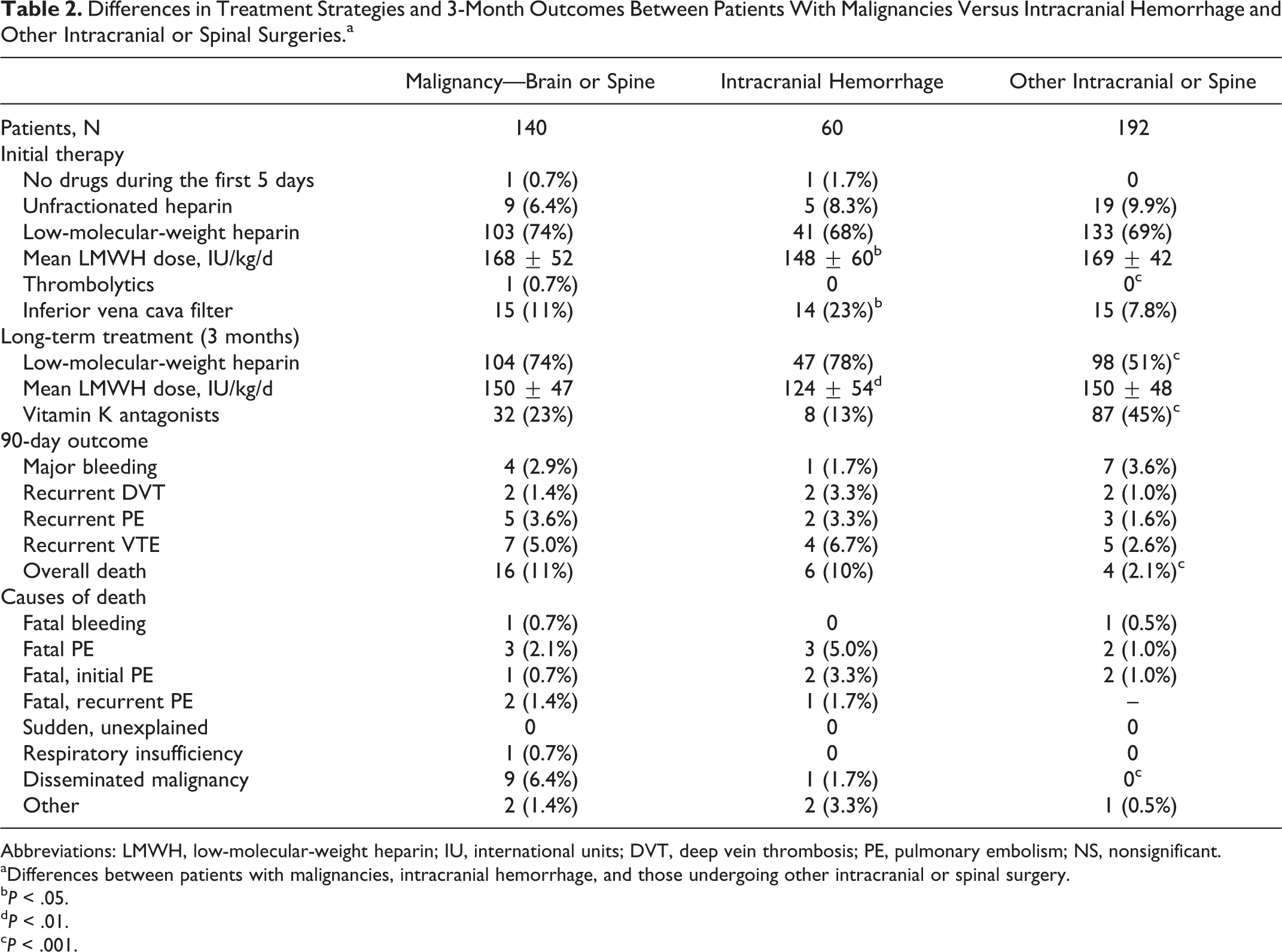
Patients with malignancies were more likely to receive corticosteroids than those in the other groups (41% vs 15% and 11%, respectively; P < .01) but less likely to receive nonsteroidal anti-inflammatory drugs (2.3% vs 10% and 9.1%, respectively; P < .05). Those with recent intracranial hemorrhage were less likely to have received pharmacological VTE prophylaxis (28% vs 44% and 44%, respectively; P < .05; Table 1). Moreover, patients with intracranial hemorrhage were less likely to present with an acute PE when compared to patients with malignancies (27% vs 61%, P < .001; or 50% in the “other brain and spinal reasons” group, P = .001). Most (89%) patients in the 3 subgroups received initial therapy with LMWH (114 of 348 received subtherapeutic LMWH doses <170 IU/kg/d), and a vena cava filter was inserted in 44 (11%) patients (Table 2). Patients undergoing surgery for intracranial hemorrhage received lower doses of LMWH both for initial therapy (148 ± 60 IU/kg/d vs 168 ± 52 and 169 ± 42, respectively; P < .05) and for long-term therapy (124 ± 54 IU/kg/d vs 150 ± 47 and 150 ± 48, respectively; P < .001). A total of 127 (32%) patients received long-term therapy with VKA drugs and 249 (64%) received LMWH. A larger amount of patients undergoing surgery for “other brain and spinal reasons” received long-term therapy with VKA drugs (Table 2).
Differences in Treatment Strategies and 3-Month Outcomes Between Patients With Malignancies Versus Intracranial Hemorrhage and Other Intracranial or Spinal Surgeries.a
Abbreviations: LMWH, low-molecular-weight heparin; IU, international units; DVT, deep vein thrombosis; PE, pulmonary embolism; NS, nonsignificant.
aDifferences between patients with malignancies, intracranial hemorrhage, and those undergoing other intracranial or spinal surgery.
b P < .05.
d P < .01.
c P < .001.
During the first week of VTE therapy, 10 patients died: 8 died of PE (6 died of the initial event and 2 died of recurrent PE) and 2 died of other reasons (1 due to respiratory insufficiency and 1 heart failure). No patients died of intracranial or spinal bleeding. At the 90-day follow-up, it was found that of the patients undergoing surgery for malignancy, there were 16 deaths (3 due to fatal PE and 1 fatal bleeding). Among those operated on because of prior intracranial hemorrhage, there were 6 deaths (50% of them died of PE), and among those patients undergoing surgery for “other brain and spinal reasons,” there were 4 deaths (2 with PE and 1 with intracranial bleeding). Of the 8 patients dying of PE, 4 were receiving LMWH doses > 150 IU/kg/d, 2 had an inferior vena cava (IVC) filter plus subtherapeutic (<150 IU/kg/d) LMWH, and 2 were on UFH (mean doses, 366.67 u/kg/d).
After the first week (Tables 3 and 4), 20 patients died: 2 died of intracranial hemorrhage and none died of PE (new onset or recurrent). In addition, 9 patients had recurrent VTE (5 with PE and 4 with DVT) and 9 had major bleeding (2 with fatal, 4 intracranial hemorrhage, 2 muscular hematoma, 1 gastrointestinal, 1 retroperitoneal, and 1 other). Of the 9 patients with recurrent VTE, 4 were receiving LMWH doses <150 IU/kg/d, and 2 of the 6 patients had recurrent PE, despite prior insertion of a vena cava filter. Despite the mentioned differences in initial and long-term therapy between patients in the 3 subgroups, there were no significant differences in the 90-day rate of fatal PE, recurrent VTE, or major bleeding. However, those undergoing surgery for “other brain and spinal reasons” had a lower mortality rate (2.1% vs 11% and 10%, respectively; P < .001). Overall, PE was the most common cause of death within the first week (8 deaths due to PE and no fatalities due to bleeding), despite variable doses of anticoagulation.
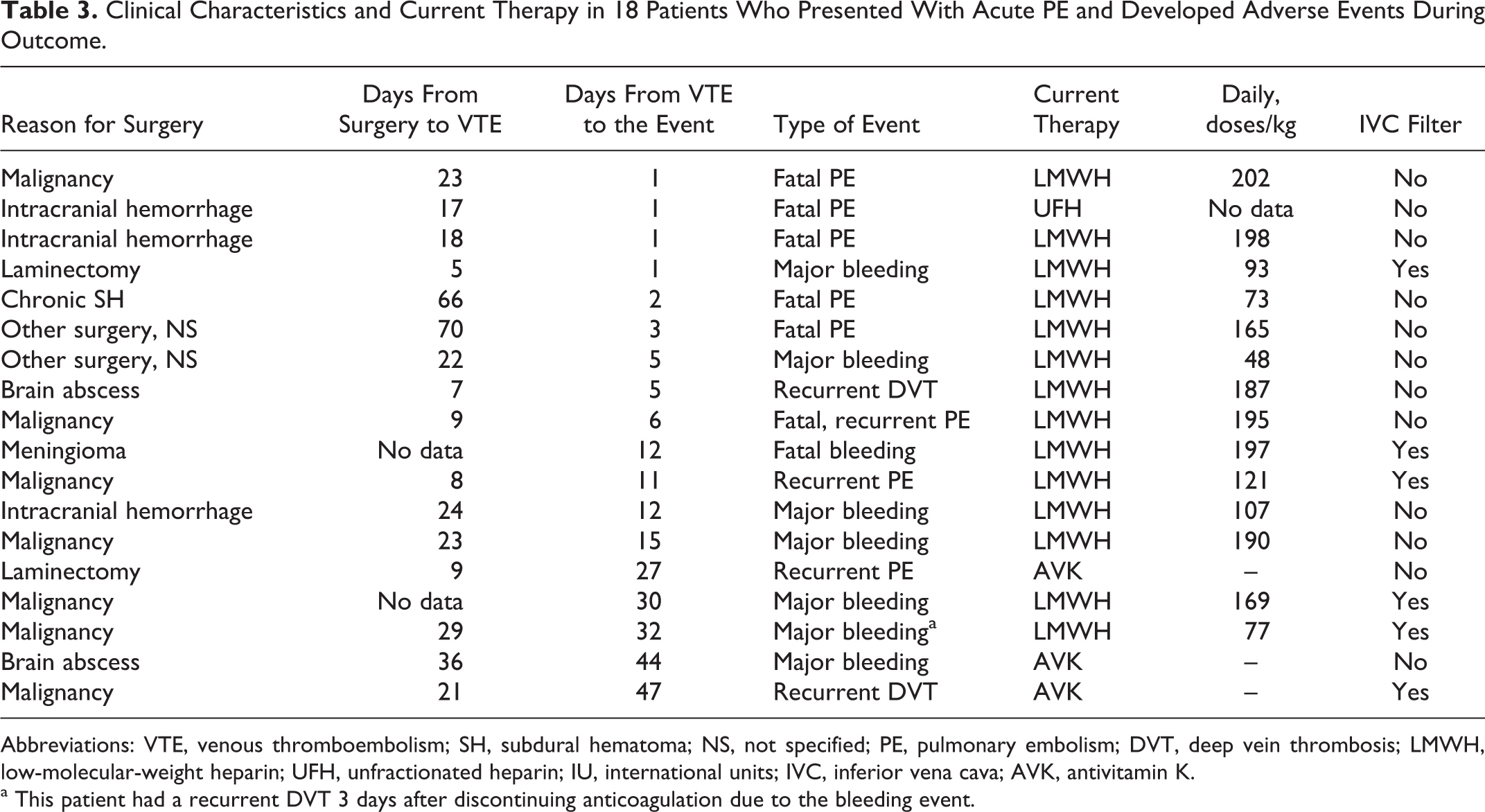
Clinical Characteristics and Current Therapy in 18 Patients Who Presented With Acute PE and Developed Adverse Events During Outcome.
Abbreviations: VTE, venous thromboembolism; SH, subdural hematoma; NS, not specified; PE, pulmonary embolism; DVT, deep vein thrombosis; LMWH, low-molecular-weight heparin; UFH, unfractionated heparin; IU, international units; IVC, inferior vena cava; AVK, antivitamin K.
a This patient had a recurrent DVT 3 days after discontinuing anticoagulation due to the bleeding event.
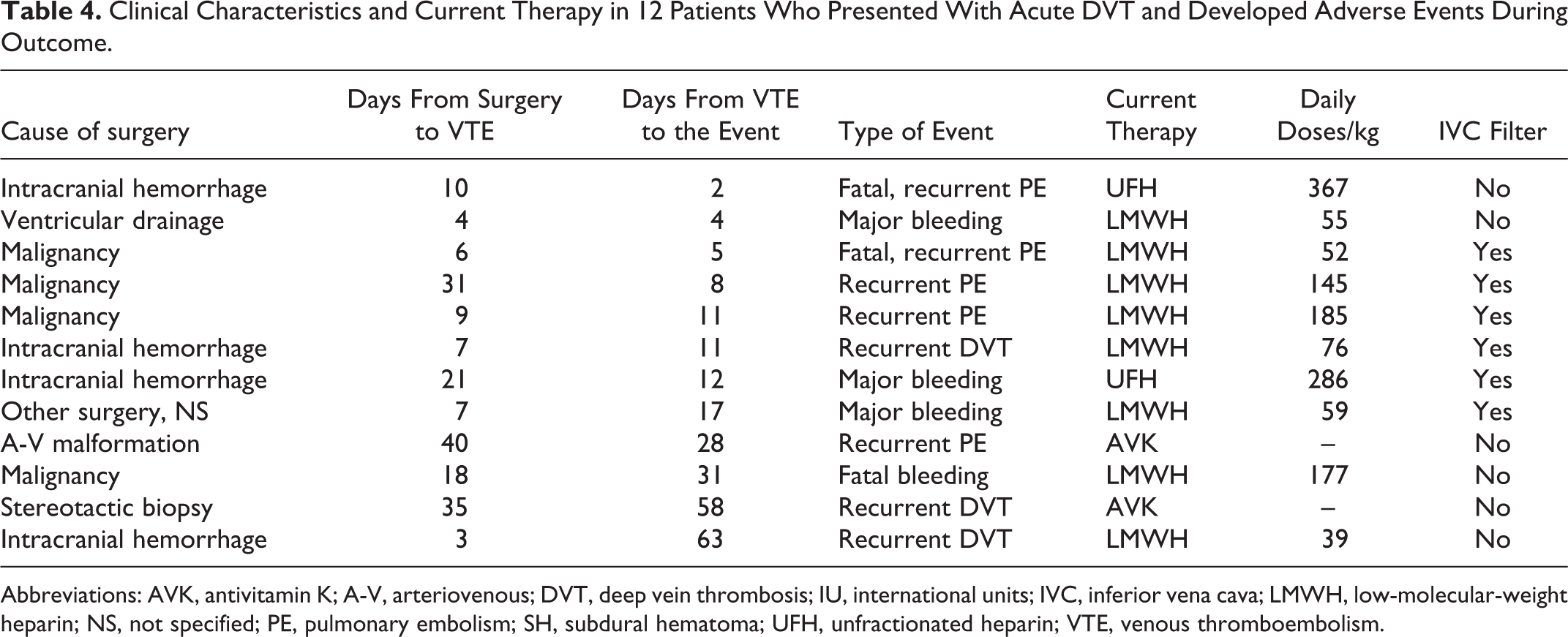
Clinical Characteristics and Current Therapy in 12 Patients Who Presented With Acute DVT and Developed Adverse Events During Outcome.
Abbreviations: AVK, antivitamin K; A-V, arteriovenous; DVT, deep vein thrombosis; IU, international units; IVC, inferior vena cava; LMWH, low-molecular-weight heparin; NS, not specified; PE, pulmonary embolism; SH, subdural hematoma; UFH, unfractionated heparin; VTE, venous thromboembolism.
Discussion
Treatment decisions in neurosurgical patients who subsequently develop VTE are often made on an empirical, individual basis, including evaluating the expected morbidity and mortality associated with different treatment options. The findings in the current study reveal that many of these patients received initial therapy at lower than recommended doses of heparin, most likely due to the concern that many physicians may feel for the risk of bleeding. Interestingly, however, we found that the rate of fatal PE exceeded the rate of fatal bleeding, particularly within the first week of therapy. All 8 patients with fatal PE in our series died during the first week after diagnosis of VTE. This included 4 patients receiving therapeutic anticoagulation and 2 patients with a vena cava filter. Although these interventions are important, deaths still occur. The absence of rebleeding or deaths from hemorrhage during the first week, despite the use of anticoagulants, is likely the most interesting finding in this study.
Unexpectedly, major bleeding was the main threat beyond the first week after VTE diagnosis. Our incidence of intracranial rebleeding during the first week of anticoagulant therapy may seem unexpectedly low, but most patients received lower than recommended doses of heparin. After the first week of therapy, there were 2 fatal bleeds and no more fatal PEs, probably because clinicians increased anticoagulation for those patients who did not bleed during the first week. Literature has repeatedly revealed a consistent finding that neurosurgical patients have a statistically significant reduction in VTE development with pharmacologic VTE prophylaxis, insignificant association of an increase in ICH, but a statistically significant doubling of “minor” bleeding. 16,17 Many of these high-risk patients have a vena cava filter placed instead of anticoagulation. In our series, we recorded 2 PE deaths with filters in place and 2 recurrent nonfatal PEs in patients with filters. According to Leon et al, the incidence of complications related to the presence of an IVC filter in high-risk patients may be as high as 22%, with some of the events resulting in significant morbidity. 18
Several studies show that pharmacologic prophylaxis is not used routinely for neurosurgical patients due to physician perception of a heightened risk of intracranial hemorrhage. 14,15,19 In our patient cohort, there were 140 patients with malignancy (53 of which received LMWH prophylaxis) and 60 patients diagnosed with an intracerebral hemorrhage (13 of which received LMWH prophylaxis). Of these patients with malignancies and intracranial hemorrhage who had received pharmacologic prophylaxis, the patients with intracranial hemorrhage were less likely to present with an acute PE (27%) than those for malignancy surgery (61%; P < .05). The patients with intracranial hemorrhage received less anticoagulation but were found to present with acute PE less than that of patients with malignancy.
There are several limitations to our study. First, this was a retrospective analysis of patients consecutively recruited, which is subject to selection bias. Second, the small sample size and limited associated outcomes do not allow for broad-based recommendations on managing these patients. Third, the types of treatment and dosing of anticoagulation were not standardized in our patient cohort. Therefore, we do not know which type and dose of anticoagulation is most efficacious in reducing adverse events such as a fatal VTE. Fourth, specifications of types and dosing of pharmacologic prophylaxis and mechanical prophylaxis were not described in the RIETE cohort of neurosurgical patients. Fifth, patients in the RIETE cohort were selected from several countries. Practice variability among different countries may affect the outcomes determined by the study. For example, the timing and dosing of prophylaxis can vary depending on the individual country’s practice pattern, underlying disease, and presence or absence of malignancy. Furthermore, the data reported from RIETE were entered into the registry by a variety of practitioners. This process may lend itself to inaccuracies in data reporting. Finally, the lack of neurosurgical data in RIETE limits the quality of this study. It would be interesting to know more details on the interventions; on how many patients underwent a new CT scan of the brain after anticoagulant treatment was started and the proportion of those with hematoma enlargement; whether there were any changes in the neurological function; and how many patients received prohemostatic agents after surgery. A further breakdown of the categories of malignancy would be of interest such as brain versus spine and within the brain as to the type of malignancy (primary vs metastatic). Also of interest would be a breakdown of patient immobility features such as hemiplegia or paraplegia and lethargy/coma predisposing to VTE.
Despite various VTE treatment plans in the patients undergoing neurosurgery of brain and spine, fatal VTEs are still prevalent. This RIETE analysis revealed that a neurosurgical patient is more likely to die of a fatal VTE, within the first week after VTE diagnosis, than a fatal bleed, particularly within the first week after VTE diagnosis. Further research is required regarding the dosage, types, and timing of pharmacologic prophylaxis and whether these variables play a role in reducing the incidence of fatal PEs in neurosurgical patients.
In summary, the clinical impact of VTE in the patients undergoing neurosurgery of brain and spine is considerable. During the first week after detecting VTE, the main threat is fatal PE (Figure 1, P value .005); beyond the first week, the main threat is bleeding. Future randomized controlled trials may clarify the benefits of different therapeutic options in this population.
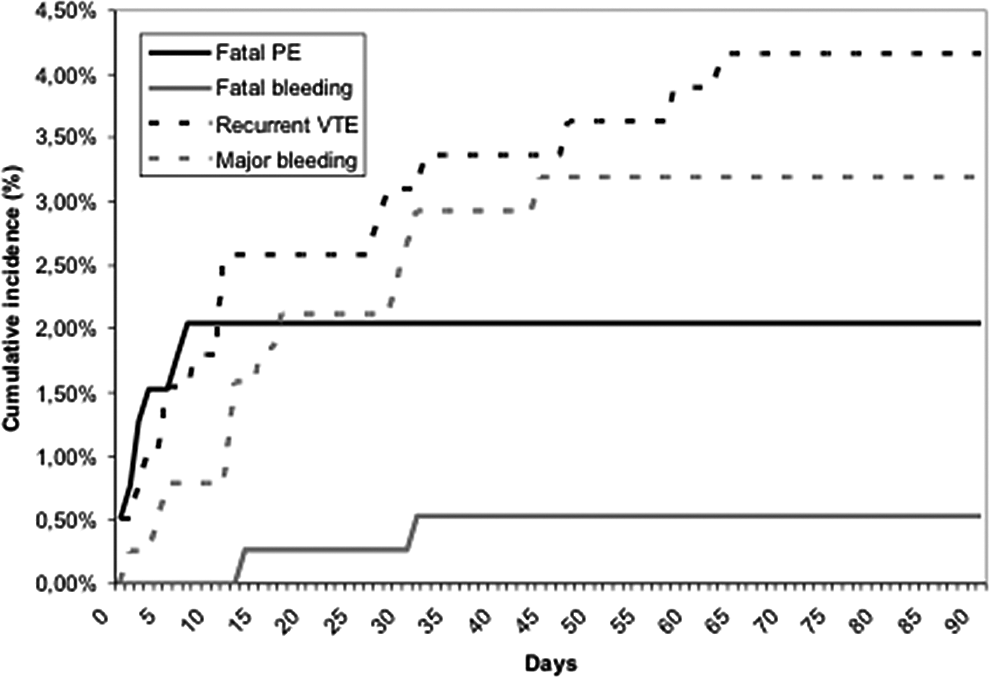
Cumulative incidence of events. P value of fatal pulmonary embolism (PE) within the first 7 days after venous thromboembolism (VTE) diagnosis, compared to fatal PE after 7 days, was .005. P value of fatal PE compared to fatal bleeding was .433. There was no difference in the P value of fatal PE compared to fatal bleeding. P value of recurrent VTE compared to major bleeding was .058. There was a trend found when comparing recurrent VTE to major bleeding.
Footnotes
Appendix
Acknowledgments
We express our gratitude to Sanofi Spain for supporting this Registry with an unrestricted educational grant. We also express our gratitude to Bayer Pharma AG for supporting this Registry. Bayer Pharma AG’s support was limited to the part of RIETE outside Spain, which accounts for a 18.86% of the total patients included in the RIETE Registry. We also thank the RIETE Registry Coordinating Center, S & H Medical Science Service, for their quality control, logistic, and administrative support and Professor Salvador Ortiz, Universidad Autónoma de Madrid and Statistical Advisor S& H Medical Science Service, for the statistical analysis of the data presented in this article.
Authors’ Note
Coordinator of the RIETE Registry: Dr Manuel Monreal (Spain). RIETE Steering Committee Members: Dr Hervè Decousus (France); Dr Paolo Prandoni (Italy); Dr Benjamin Brenner (Israel). RIETE National Coordinators: Dr Raquel Barba (Spain); Dr Pierpaolo Di Micco (Italy); Dr Laurent Bertoletti (France); Dr Sebastian Schellong (Germany); Dr Manolis Papadakis (Greece); Dr. Inna Tzoran (Israel); Dr Abilio Reis (Portugal); Dr Marijan Bosevski (R.Macedonia); Dr Henri Bounameaux (Switzerland); Dr Radovan Malý (Czech Republic). RIETE Registry Coordinating Center: S & H Medical Science Service. A full list of the RIETE investigators is given in the appendix.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
