Abstract
Venous thromboembolism (VTE) causes significant morbidity and mortality in hospitalized medical populations; however, medical patients do not currently receive thromboprophylaxis beyond their hospital stay. We reviewed the real-life occurrence of VTE-related care for 100 days post-hospitalization in Calgary, Canada. Using medical visit records with a unique patient identifier number applied throughout the city’s hospitals, 989 high-risk patients were selected for review. Almost three-quarters of the elderly patients received appropriate prophylaxis while in hospital, and only 2% received prophylaxis on discharge. Over the 100-day follow-up, 21% of the patients presented with clinically suspected VTE, of which 3.8% had confirmed VTE. Patients with multiple risk factors (≥3) had the highest frequency of confirmed VTE (≥6.1%). This study suggests that the actual rate of VTE-related follow-up care in patients post-hospitalization is high in the first 100 days, particularly among those who have multiple risk factors, warranting consideration of extended thromboprophylaxis in this population.
Keywords
Introduction
Hospitalized medical patients are at high risk of venous thromboembolism (VTE), with approximately one-quarter of all VTE events occurring in this nonsurgical population. 1 A prospective cohort study suggested a symptomatic, clinically evident VTE rate of 11% for hospitalized high-risk medical patients not receiving prophylaxis, 2 and there is strong evidence that thromboprophylaxis in this population results in fewer VTE events and reduced mortality. 1 As such, the American College of Chest Physicians recommends pharmacological prophylaxis for acutely ill hospitalized medical patients at increased risk of thrombosis, but does not recommend extending the duration of therapy beyond the period of immobilization or hospital stay. 1 The American College of Physicians (ACP) also recommends prophylaxis with heparin (or related drug) for medical patients, unless the assessed risk of bleeding outweighs the benefits of thromboprophylaxis. 3 Risk factors for VTE in hospitalized medical patients include increasing age (especially >70 years), previous VTE, known thrombophilia, immobilization for ≥3 days, hormonal medications, and history of comorbid illness (eg, cancer, heart failure, and respiratory failure). 1 Further research is needed to validate the risk factors for VTE, as prophylaxis may harm patients who are at low risk of VTE but at high risk of bleeding; yet, its underuse may result in avoidable mortality and morbidity. 1
Much of the current evidence focuses on VTE risk of medical patients during hospitalization, with some evidence to suggest the risk extends beyond hospital stay. The EXCLAIM trial examined extended-duration thromboprophylaxis with enoxaparin in hospitalized medical patients, which determined a 90-day VTE incidence of 4.4% in medical patients not receiving postdischarge prophylaxis. 4 Similarly, the MEDENOX trial suggested that VTE risk in medical patients extends for up to 3 months, with 8 VTE events (including 4 fatal pulmonary embolisms) occurring between days 15 and 110 after discharge. 5 However, both these studies were randomized clinical trials that used routine diagnostic screening in addition to clinical events to calculate the rates of VTE and had strict exclusion criteria for their study populations, which excluded risk factors for bleeding. In comparison, few “real-life” studies have been performed which identified symptomatic, clinically evident VTE events in populations not screened for risk factors for bleeding; an example is the IMPROVE observational study, which showed that 45% of the VTE events occurred after discharge in medical patients. 6 Such “real-life data” regarding the timing of VTE events in high-risk medical patients after leaving hospital are lacking, and the relationship between risk factors and VTE in this population has not been fully examined. Furthermore, data are lacking on rates of VTE in high-risk subgroups of patients, such as those with cancer. Previous retrospective cohort database studies have estimated the incidence of VTE in hospitalized patients with cancer to be between 0.6% and 5.4%, but there are few data on rates in discharged patients. 7,8 Our objective was to document the time course of symptomatic VTE events in high-risk elderly medical patients over 100 days postdischarge and to correlate the frequency of risk factors for the likelihood of VTE development. A subgroup analysis of these medical patients with cancer was also performed. These data recorded in everyday clinical practice may help to clarify the VTE risk of “real-world” medical patients and to determine the appropriateness of long-term VTE prophylaxis in high-risk elderly medical patients, including those with cancer, as well as to establish strategies regarding the frequency of clinical surveillance postdischarge in these populations.
Methods
Study Design
Data from the charts of high-risk elderly medical patients hospitalized in the Calgary region and discharged between January 1, 2008 and February 28, 2008 were collected using case records and screened for eligibility. All identified patients were followed for any subsequent medical attention related to VTE. All hospitals in the region use a unique patient identifier number, enabling the tracking of subsequent patient visits to the emergency room, inpatient admissions, or outpatient visits occurring anywhere in the region’s acute care system. Data were collected on patient’s risk factors, thromboprophylaxis received in hospital or at discharge, and VTE-related events for up to 100 days postdischarge. Statistical methods used were as follows: Wilson procedure without correction for continuity was applied to calculate the proportion of patients with VTE symptoms and confirmed events, including 95% confidence intervals. Chi-square tests of association were used for assessing the association between the number of risk factors and the rates of VTE. Further details of methods can be found in the protocol in the Online Supplementary Information.
Inclusion and Exclusion Criteria
High-risk elderly medical patients were defined as aged >60 years and having at least one of the following risk factors: history of malignancy, respiratory illness, neurological illness, inflammatory bowel disease, previous VTE, acute infection, or heart failure. Medical patients with cancer were defined as those aged >60 years with a cancer diagnosis at hospital admission, had a planned cancer surgery, were receiving cancer/palliative treatment or whose cancer treatment was not specified; patients with a remote history of cancer were excluded. Records were excluded if the patient was originally admitted for VTE, or to rule out VTE, receiving chronic anticoagulation, experiencing an acute coronary syndrome, had a hospital stay ≤ 3 days, was a surgical or orthopedic patient, or was pregnant.
Outcome Measures
Outcomes included any medical attention for VTE symptoms up to 100 days postdischarge, including emergency room admissions, outpatient visits, or readmissions. The patients were classed as confirmed VTE if they were subsequently confirmed by diagnostic testing.
Results
Study Population: High-risk Elderly Medical Patients
Of the 1134 hospital discharges in the review period, 989 (87%) were identified as consecutive high-risk elderly medical patients that met eligibility criteria and were selected for follow-up as the high-risk group. Baseline demographics are shown in Table 1. This population included all cases with risk factors, but ruled out those where a complicating factor may cloud the causation of VTE (eg, currently has/suspected of having VTE, surgical cases, and acute coronary syndromes). Data extracted from patient charts determined the incidence of medical risk factors for the study population and VTE prophylaxis prescribed (Figure 1A and B).
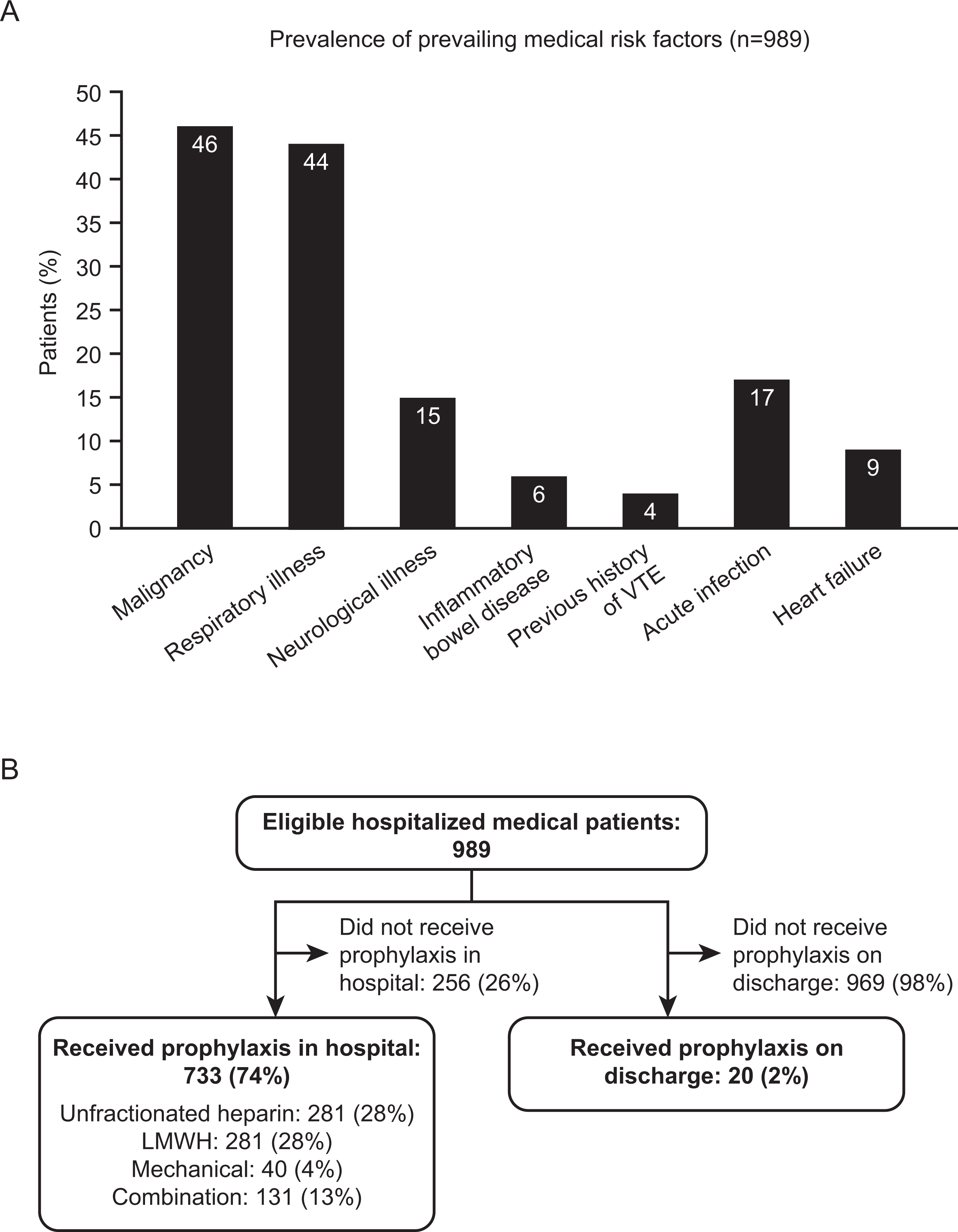
High-risk elderly medical patients study population: prevailing medical risk factors (A) and VTE prophylaxis received (B). LMWH, low-molecular-weight heparin; VTE, venous thromboembolism.
Rate and Time Course of VTE Events: All High-Risk Elderly Medical Patients Compared With Elderly Medical Patients With Cancer.a
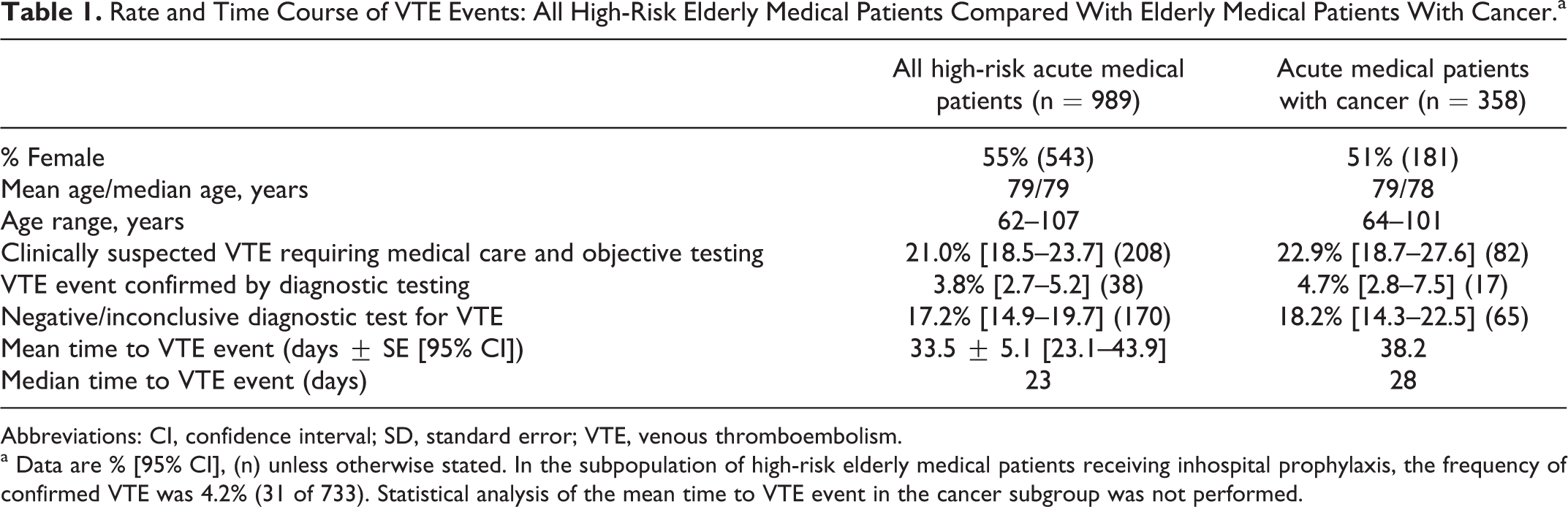
Abbreviations: CI, confidence interval; SD, standard error; VTE, venous thromboembolism.
a Data are % [95% CI], (n) unless otherwise stated. In the subpopulation of high-risk elderly medical patients receiving inhospital prophylaxis, the frequency of confirmed VTE was 4.2% (31 of 733). Statistical analysis of the mean time to VTE event in the cancer subgroup was not performed.
Symptomatic VTE Rates and Time Course Postdischarge: High-Risk Elderly Medical Patients
Rates of VTE and length of time from discharge to first confirmed VTE event for the high-risk group are shown in Table 1. The cumulative event rate over time determined that 80% of the confirmed VTE events occurred within 57 days after discharge (Figure 2).
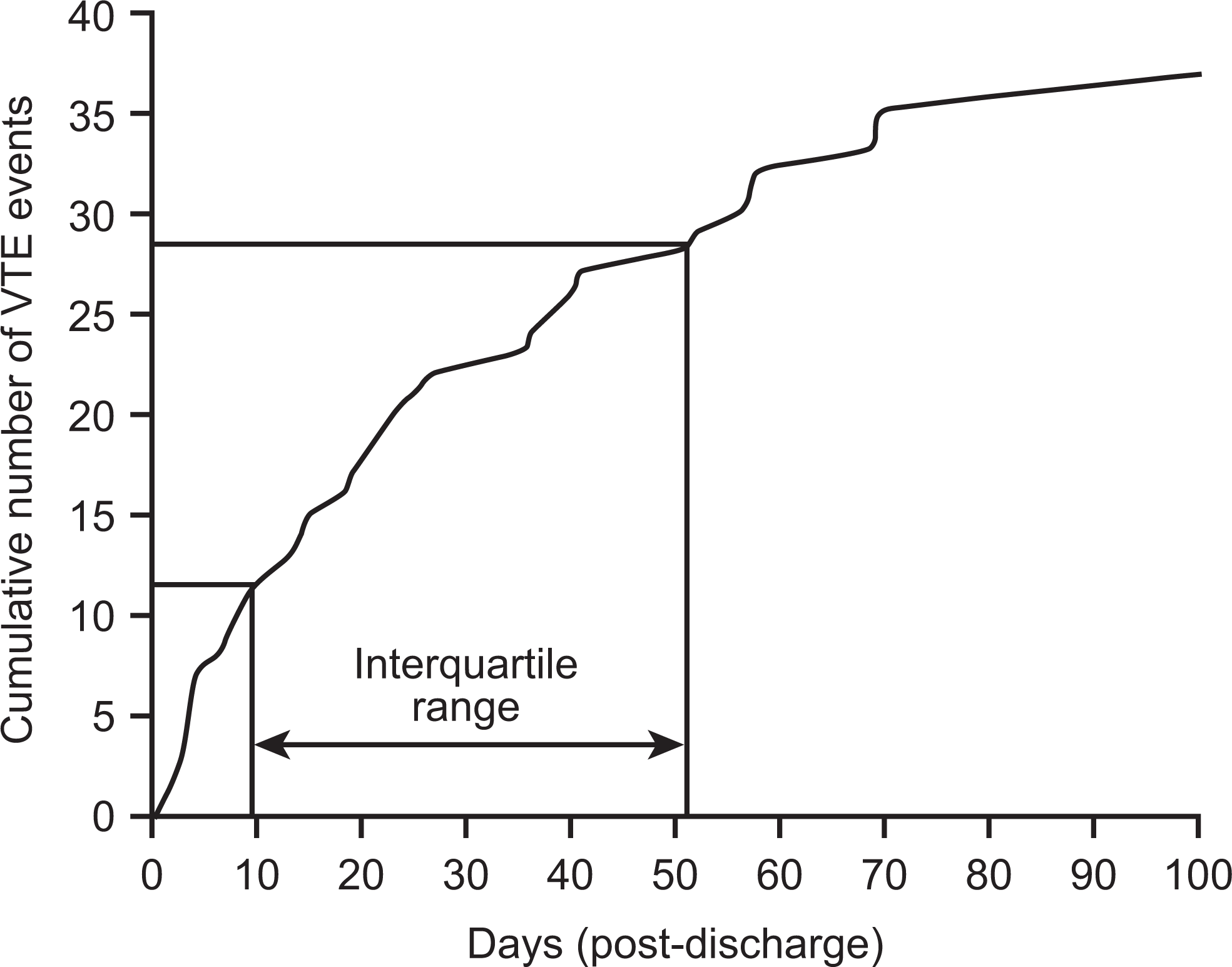
Cumulative event rate over time: high-risk elderly medical patients. VTE, venous thromboembolism.
Correlation of VTE Rates with Risk Factors: High-risk Elderly Medical Patients
Next, we analyzed the association between patients’ number of risk factors and their rates of VTE post-discharge (Figure 3). Patients with a greater number of risk factors were more likely to seek medical attention for VTE symptoms than those with fewer risk factors; patients with ≥3 risk factors had significantly more VTE events than those with ≤2 risk factors (3.2% absolute difference, P = .015). A further increase in risk was observed in patients with ≥ 4 risk factors (5.8% difference, P = 0.011 vs ≤2 risk factors), but this was not statistically significant when compared to patients with ≥3 risk factors.
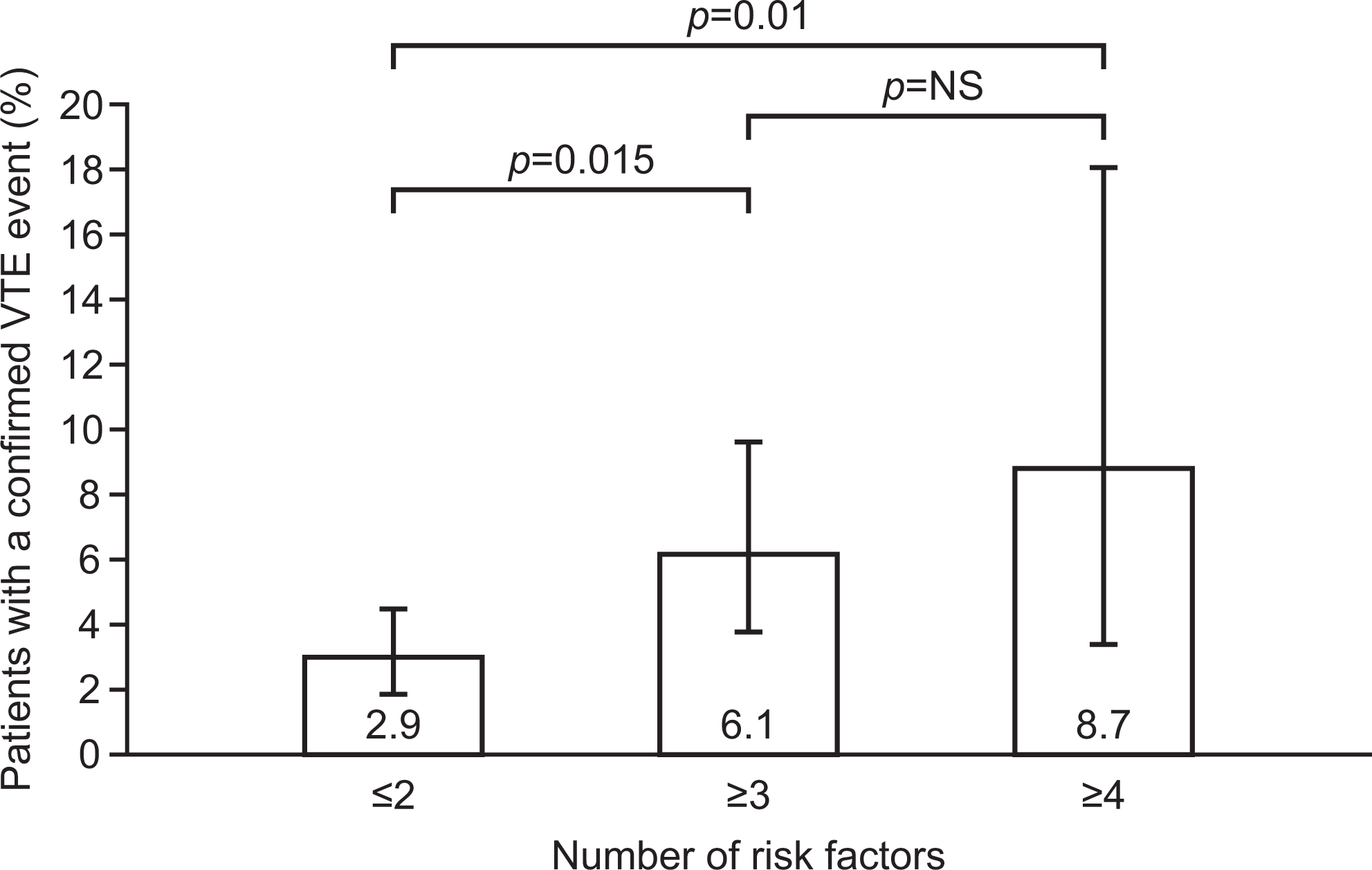
Influence of risk factors on rates of VTE. Data are the percentage of patients with confirmed VTE, according to the presence of risk factors for VTE. Error bars represent 95% CIs. CI, confidence interval; VTE, venous thromboembolism.
Subpopulation Analysis: VTE Rates and Time Course in Patients With Acute Cancer
Of the 1134 hospital discharges in the review period, 358 (32%) patients met the criteria for inclusion as medical patients with cancer in the subpopulation analysis. In all, 73% (261 of 358) of the subgroup received mechanical or pharmacological prophylaxis in hospital, whereas only 2% of all the patients received anticoagulation prophylaxis after discharge. The results of the subpopulation analysis of patients with cancer are shown in Table 1.
Interpretation
We selected the records of high-risk elderly medical patients, including a subset of elderly medical patients with cancer, discharged from hospital in Calgary during a 2-month period in 2008 and followed them for 100 days for subsequent VTE events. Almost three-quarters of the patients in both the groups received appropriate prophylaxis while in hospital and only 2% received prophylaxis on discharge, in keeping with current ACP guidelines not to prescribe prophylaxis on discharge. 3 Throughout the entire follow-up, a considerable proportion of the study population presented with clinically suspected VTE, requiring medical care and objective testing (21% of the entire study population and 23% for the cancer subgroup), resulting in rates of confirmed VTE diagnoses of 4% for the entire study population and 5% in the cancer subgroup. The cumulative event rate over time revealed that the majority of these cases occurred just over 1 month after discharge (mean time to VTE event: 34/38 days in the high-risk/cancer subgroup). In the subgroup population with cancer, it was noted that 23% sought out medical attention for VTE symptoms, with 5% having a confirmed VTE diagnosis. Furthermore, this population followed the same pattern of VTE event rates with a mean time to VTE of 38 days. As expected, patients with a greater number of risk factors had a higher prevalence of VTE: 6% of hospitalized medical patients with ≥3 risk factors developed symptomatic VTE confirmed by diagnostic testing, which increased to 8.7% for those with ≥4 risk factors. Only 2.9% of the patients with ≤2 risk factors developed VTE.
The age criterion of >60 years was chosen due to the significantly increased rates of VTE in older patients compared with younger patients, 9 which ensured enough events would occur to achieve statistical significance. The high-risk criteria chosen for this study are supported by risk factors identified in the EXCLAIM study, where patients >75 years were identified as a key group to target for prophylaxis, being at a greater risk of VTE than bleeding. 4 As such, the majority of the discharged population were classified as high risk in our study, but this may not reflect the demographics of all centers or those of other countries. Also, this study had a high proportion of patients with malignancy (46%) when compared to a previous analysis (9%), 10 probably due to 1 of the 3 hospitals chosen being a major cancer treatment center. Notably, the rate of high-risk elderly medical patients seeking out medical attention for VTE symptoms in our real-life review (21%) was higher than rates observed in clinical studies; in MEDENOX, only 17% of the patients in the placebo group experienced VTE up to 110 days after discharge. 5 Despite inhospital prophylaxis, our real-life study showed dramatically higher symptomatic VTE rates postdischarge than the LIFENOX study: only 0.7% in the placebo group. 11 This result was not unexpected, since routine screening for thrombosis is often performed in clinical trials, but not in everyday clinical practice, so it is thought symptomatic VTE rates are underestimated in clinical trials compared to real-life settings; screening and/or treating for asymptomatic VTE in clinical trials reduces the potential for these thrombi to progress and become symptomatic. 1 Also, LIFENOX studied a patient population discharged with elastic stockings, which is unrepresentative of Canadian practices. 11 In our study, 4% of the high-risk elderly medical patients had a VTE confirmed by diagnostic testing in the postdischarge follow-up period, a total of 38 events. This is consistent with EXCLAIM, which reported a VTE incidence of 4.4% in a 90-day period in medical patients not receiving postdischarge prophylaxis. 4 Our 100-day follow-up was chosen to ensure the plateau in the time course of VTE events was captured in order to identify an optimal time to consider prophylaxis; indeed our design was successful, identifying the plateau of events, with the number of events reducing after day 50. Previous clinical trials studying postdischarge VTE rates in medical patients have shorter follow-ups (ADOPT: 30 days) 12 ; our study suggests these follow-up periods are not long enough for a complete evaluation of the benefits of prolonged prophylaxis.
As expected, patients with a greater number of risk factors had a higher prevalence of VTE events than those with fewer risk factors. This correlation is in agreement with risk assessment models of similar at-risk groups, both in hospitalized patients 2 and in those post-discharge. 6 However, are the rates of VTE in a high-risk older population sufficient to outweigh the risks of bleeding? Our data, showing a VTE risk of over 6% in patients with ≥ 3 risk factors, increasing to 9% in those with ≥4, suggests targeting prophylaxis to these high-risk subgroups has a more favorable risk-benefit profile than observed in the overall medical populations examined in previous studies, such as EXCLAIM, ADOPT, and MAGELLAN. 4,12,13 Patients with ≤2 risk factors had a lower rate of VTE (2.9%). Our results need to be balanced with the outcomes of EXCLAIM, which while not supporting a general need for extended thromboprophylaxis in a hospitalized medical population, identified high-risk subgroups where the benefit did outweigh the risks. 4 Prolonged use of enoxaparin prevented 6 fewer symptomatic VTE cases per 1000 patients but at a cost of 5 more major bleeding events per 1000; however, the benefits of additional prophylaxis outweighed the risks of bleeding in high-risk subgroups: women, patients >75 years and those without bathroom privileges. 4 Our real-life study shows that a subset of high-risk patients >60 years may also benefit from extended use of thromboprophylaxis. In particular, patients >60 years with cancer may benefit, since approximately 1 in 20 patients experienced a confirmed VTE event in our review post-discharge. Our data support the use of clinical risk factors at hospital admission to predict VTE risk in medical patients and suggest that the short courses of prophylaxis currently prescribed predischarge may not adequately provide coverage for those at highest risk.
Strengths and Limitations
Traditionally, chart review data are seen as weak due to their many limitations; however, our study design has unique strengths, making it a more robust study. The Calgary region has a single, integrated information system used across acute care sites, so tracking of all subsequent visits was possible using the unique Calgary patient identifier. Also, the location is geographically isolated, minimizing the likelihood of patients seeking medical attention elsewhere, maximizing the chances of patient follow-up. However, there are inherent limitations of chart reviews; unlike clinical trials, our review was an uncontrolled, epidemiological study carried out in everyday clinical practice, not designed to measure efficacy or safety. As safety outcomes were not measured, a comprehensive risk-benefit analysis was not possible. Also, as our study relied on data accuracy in the patients’ charts, incomplete documentation may be a limitation. Our study relied on the patient to seek out medical attention for VTE symptoms to determine event rates, so patients not seeking attention, who died or went to alternative care providers would not be captured. Finally, the prophylaxis practices and the patient population in the Calgary region may not be representative of, or be applicable to, other centers and/or countries.
Conclusion
Our study suggests that in a real-life setting, cumulative risk factors (including cancer) result in an increased risk of VTE events post-discharge in a hospitalized medical population. A mean prevalence of 4% confirmed VTE (increasing to 6% and 9% in patients with ≥3 or ≥4 risk factors, respectively) was recorded, with patients continuing to seek medical attention for VTE symptoms up to 100 days postdischarge, with 34 days being the mean time to first event. Evidence-based guidelines do not currently support extended prophylaxis for medical patients, 1 yet our data show that the risk extends well beyond a typical hospital stay. The 4% VTE rate was greater than expected for a real-life study and may change the perception of a low benefit-to-risk ratio of prophylaxis upon discharge. Coupled with the results of the EXCLAIM study, the VTE rates observed here may suggest that prolonged VTE prophylaxis may be of benefit in high-risk medical populations, such as older patients. These data also highlight the problem of early discharge before adequate prophylaxis can be administered. 14 However, such extended prophylaxis would increase the cost of therapy and may increase the bleeding risk of certain patients. Indeed, recent trials have failed to show an increased benefit-to-risk ratio of novel oral anticoagulants over traditional subcutaneous (sc) thromboprophylaxis; the MAGELLAN study determined an increased bleeding profile of oral rivaroxaban compared with sc enoxaparin, 13 whereas the ADOPT study found increased bleeding and no superior efficacy with oral apixaban compared with sc enoxaparin. 12 Therefore, a patient’s individual benefit-risk assessment must always be taken into account 1 when considering prophylaxis. Further, large-scale clinical trials and real-life studies are warranted to confirm the relative risks and benefits of extended thromboprophylaxis in high-risk elderly medical patients postdischarge.
Footnotes
Authors’ Note
This study was presented orally at the annual meeting of the American Society for Hematology (ASH) in Orlando, FL, December 2010.
Authors’ Contribution
R.D. Hull had the original idea for the study, provided guidance and input into all drafts, and is the guarantor of the study. J. Liang performed the statistical analyses and reviewed and approved all drafts. A.L. Stevenson interpreted data, wrote the first draft, and approved the final draft. T. Merali developed the study protocol, supervised data collection, data management, analysis, and reviewed all drafts. A. Mills provided clinical assistance in reviewing the results, provided input into the drafts, and reviewed all drafts.
Declaration of Conflicting Interests
The author(s) declared a potential conflict of interest as follows: R.D. Hull has received grants/research support from Bayer Pharmaceuticals Corp, LEO Pharma Inc, and Sanofi-Aventis; been a consultant for Bayer Pharmaceuticals Corp, LEO Pharma Inc, Pfizer Inc, GlaxoSmithKline, and Wyeth Pharmaceuticals; and sat on advisory boards for Bayer Pharmaceuticals Corp, Pfizer Inc, and Sanofi-Aventis. A. Stevenson is an employee of Watermeadow Medical, which received payment from the Medical Continuing Education Fund, University of Calgary for work on this manuscript. Editorial assistance was provided by Watermeadow Medical. T. Merali is an employee of Drug Intelligence, which received payment from the Thrombosis Research Group, University of Calgary for work on this study. J. Liang and A. Mills have no conflicts of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or publication of this article: This work was funded by Sanofi-Aventis, Canada.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
