Abstract
Keywords
Introduction
Large-artery atherosclerosis (LAA) is one of the main subtypes of the Trial of ORG 10172 in Acute Stroke Treatment (TOAST) classification of acute ischemic stroke (AIS), 1 which can lead to severe neurological deficits. Intravenous thrombolysis (IVT) with recombinant tissue plasminogen activator (rt-PA) is an effective treatment in the hyperacute period; however, it also increases the risk of intracranial bleeding. Hemorrhagic transformation (HT) is a common complication of IVT, with an incidence rate ranging between 10% and 48%. 2 Neurological deficits will likely be aggravated once HT occurs, which is difficult to identify because of several contributing factors.
Hemorrhagic transformation may be associated with inflammatory response and blood–brain barrier (BBB) dysfunction.3,4 Although several studies have identified the risk factors for HT after IVT, the relationship between inflammation and HT remains unclear. After brain ischemia, monocytes (Mcs) will migrate across vascular endothelial cells, activate inflammatory signaling and increase BBB permeability. 5 In addition, Mcs/macrophages (Møs) are involved in various inflammatory stages of atherosclerosis. 6 High-density lipoproteins (HDL) have anti-inflammatory, oxidative, and antithrombotic functions. 7 The monocyte-to-HDL ratio (MHR) is a novel inflammation marker that predicts pro-inflammation/anti-inflammation and thrombotic/anti-arteriosclerosis capabilities 8 and can be easily detected via routine tests and applied in clinical practice. It has already shown predictive value in atherosclerosis and cardiovascular diseases9,10; however, its predictive value in post-thrombolysis HT in AIS patients remains uncertain.
Monocytes and HDL levels are closely associated with atherosclerosis. This study aimed to investigate the association between MHR and HT after IVT in AIS patients, which could greatly help identify high HT risk.
Materials and Methods
Subjects
We retrospectively analyzed clinical data of AIS patients (September 2019 to September 2021) who accepted IVT treatment with rt-PA. All patients were hospitalized within 4.5 h from symptom onset at the Neurology department of the first affiliated hospital of Harbin Medical University, China. Diagnosis of AIS was made according to the World Health Organization criteria and confirmed by computed tomography (CT) or magnetic resonance (MRI). This study was approved by the hospital ethics committee.
Exclusion criteria were: (1) severe infection before or within 48 h after stroke; (2) suffering from blood diseases (such as mononucleosis), malignant tumors and immune diseases; (3) taking steroids, immunosuppressants and antibiotics; (4) severe motor dysfunction caused by other diseases; (5) severe heart, liver, or kidney dysfunction; (6) incomplete clinical data.
Data Collection
Data about demographics (age, gender), vascular risk factors (hypertension, diabetes mellitus, atrial fibrillation, ischemic stroke, coronary artery disease, smoking, alcohol history), baseline National Institute of Health Stroke Scale (NIHSS) score, cerebral infarction volume were collected in this study. The complete blood counts and biochemical samples, including HDL, low-density lipoprotein (LDL), triglyceride (TG), cholesterol (CHOL), and homocysteine (Hcy) were collected within 24 h of admission MHR was calculated as monocyte counts/HDL level. All patients underwent a brain CT on admission, followed by a second CT scan 24 h after IVT or when clinical symptoms worsened. Scheduled brain MRI was performed within 7 days after admission.
TOAST Classifications and HT Diagnosis
The stroke etiology was classified according to TOAST. HT was defined as the absence of intracranial hemorrhage on the initial tomography but appeared on follow-up examinations. Both TOAST and HT were independently evaluated by two experienced neurologists and disagreements were settled by further discussion.
Statistical Analysis
The data was analyzed using SPSS 19.0 statistical software. The normal distribution data was represented by mean ± SD and assessed by t-test. The nonnormal distribution data was expressed as median and quartile intervals and the comparisons were made with Mann–Whitney U test. Categorical variables were reported as frequency and percentage and compared with Pearson's χ2 tests. The variables with P < .05 from a comparison of baseline characteristics were included in further multivariate logistic regression. The optimal cut-off value, sensitivity, and specificity were determined according to receiver operating characteristics curve (ROC). P < .05 was considered as statistically significant.
Results
Baseline Characteristics of all Patients
During the study period, 444 patients with AIS were consecutively recruited; nine of them (one patient had bone metastases of prostate cancer, one patient had sepsis, one patient had severe renal failure and six patients had incomplete clinical data) were excluded and a total of 435 patients were enrolled in this study. The average age of all patients was 64.2 ± 11.7 years. Of the total, 306 patients were males (70.3%) and 52 patients (12.0%) experienced HT.
Patients with HT were older (69.6 ± 9.8 vs 63.4 ± 11.7, P < .001) and had a higher incidence of atrial fibrillation (40.4% vs 14.4%, P < .001), ischemic stroke history (51.9% vs 31.6%, P = .004), large cerebral infarction (61.5% vs 18.5%, P < .001), higher NIHSS scores (12 vs 6, P < .001), higher levels of HDL (1.27 vs 1.09, P < .001), LDL (3.34 vs 2.98, P = .01), and CHOL (5.18 vs 4.71, P = .006), and lower MHR values (0.28 vs 0.36, P = .031). There were also significant differences in TOAST classification between the two groups (P < .001). The baseline characteristics are presented in Table 1.
Comparisons of Baseline Characteristics in AIS Patients With or Without HT.

Values are n (%), mean SD, or median (interquartile range). Abbreviations: NIHSS, National Institutes of Health Stroke Scale; Mc, monocyte; HDL, high-density lipotrotein; LDL, low-dentisty lipoprotein; MHR, monocyte to high-density lipoprotein ratio; CHOL, cholesterol; TG, triglyceride; Hcy, homocysteine; LAA, large-artery atherosclerosis; SVO, small-vessel occlusion; CE, cardioembolism; ODE, other determined etiology; UE, undetermined etiology. *P < .05.
Multivariate Logistic Regressions
To further explore the correlation between MHR and HT in AIS patients, variables that were significantly different (Table 1) were entered into a subsequent logistic regression model. Since MHR is highly correlated with HDL levels (r = −0.625), we designed two independent regression models, model 1 (with MHR) and model 2 (with HDL). In addition, the baseline NIHSS score was highly correlated with large cerebral infarction (r = 0.553); therefore, large cerebral infarction was not included in the regression analysis to avoid multicollinearity.
Low MHR (Model 1: OR:0.035, 95%CI:0.003–0.390, P = .006) and high HDL levels (Model 2: OR:5.87, 95% CI:2.23–15.47, P < .001) significantly correlated with HT. Atrial fibrillation (Model 1: OR:3.96, 95%CI:1.66–9.45, P = .002; Model 2: OR:4.19, 95%CI:1.78-9.88, P = .001), history of ischemic stroke (Model 1: OR:2.21, 95%CI:1.16–4.20, P = .016; Model 2: OR:2.34, 95%CI:1.22–4.50, P = .011), and high baseline NIHSS score (Model 1: OR:1.06, 95%CI:1.01–1.11, P = .006; Model 2: OR:1.06, 95%CI:1.01–1.10, P = .01) were significant predictors of HT (Table 2).
Multivariate Logistic Regression of Risk Factors for Hemorrhagic Transformation.

Model 1 and Model 2 were adjusted for age, atrial fibrillation, ischemic stroke, NIHSS on admission, TOAST, LDL and CHOL. Abbreviations: CI, confidence interval; MHR, monocyte to high-density lipoprotein ratio; HDL, high-density lipotrotein; LDL, low-dentisty lipoprotein; *P < .05.
Baseline Characteristics in LAA Type AIS Patients
There were 123 LAA- type AIS patients with an average age of 64.4 ± 10.4 years; 21 of them (17.1%) experienced HT after IVT. Patients in the LAA type HT group had a higher incidence of atrial fibrillation (9.5% vs 0%, P = .002), history of ischemic stroke (66.7% vs 33.3%, P = .004), large cerebral infarction (71.4% vs 30.4%, P < .001), and higher NIHSS scores (12 vs 8, P = .025). In addition, TG (1.31 vs 1.65, P = .049) and MHR values (0.31 vs 0.37, P = .032) in the HT group were lower than those in the non-HT group (Table 3).
Comparisons of Baseline Characteristics in LAA-Type AIS Patients With or Without HT.
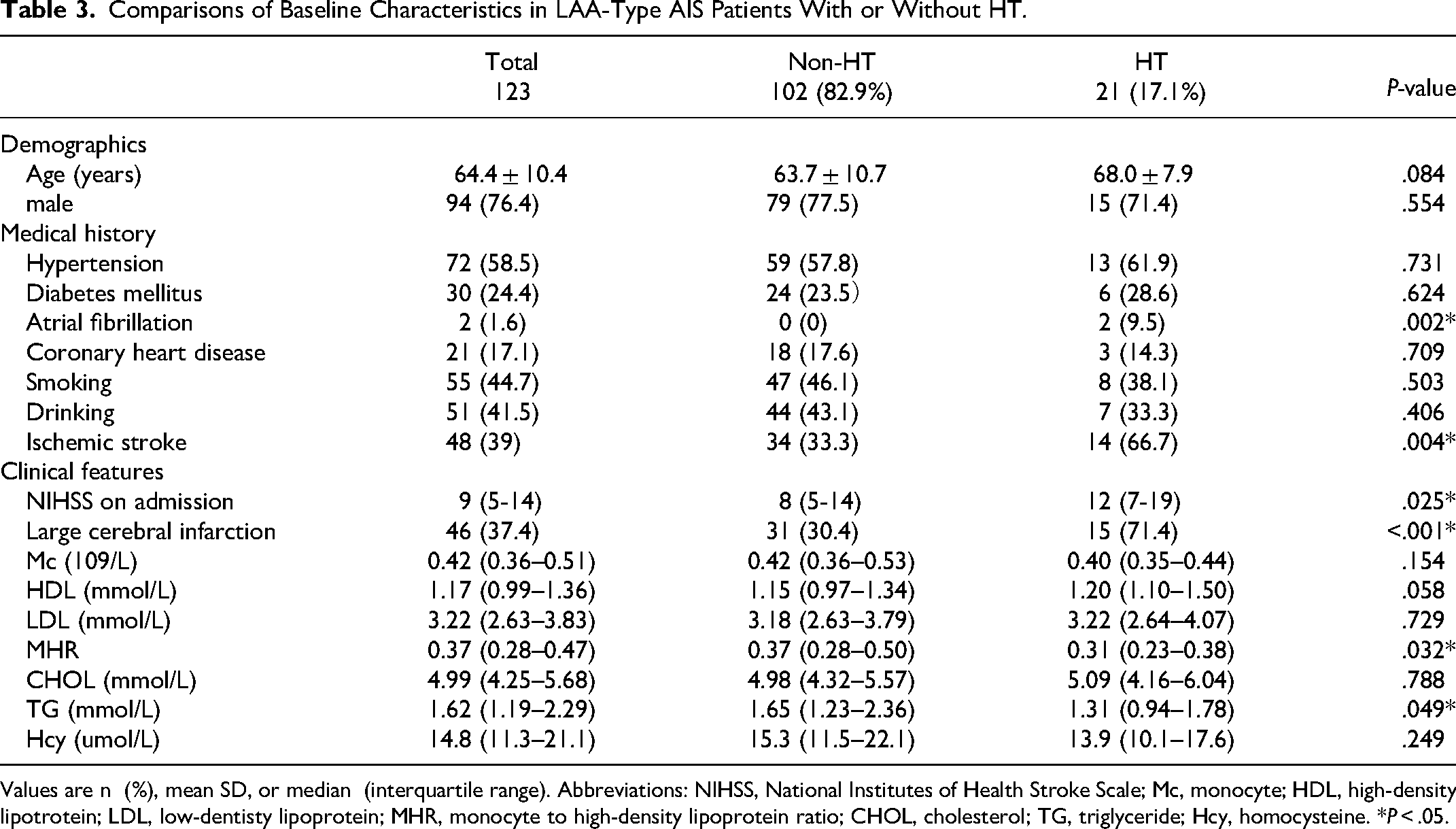
Values are n (%), mean SD, or median (interquartile range). Abbreviations: NIHSS, National Institutes of Health Stroke Scale; Mc, monocyte; HDL, high-density lipotrotein; LDL, low-dentisty lipoprotein; MHR, monocyte to high-density lipoprotein ratio; CHOL, cholesterol; TG, triglyceride; Hcy, homocysteine. *P < .05.
Correlation Analysis Between MHR and HT in Different TOAST Subtypes
There was no MHR significance in the small-vessel occlusion (SVO) and undetermined etiology (UE) types of AIS. In the cardioembolism (CE) type, MHR (0.22 vs 0.38, P = .003) was statistically significant between the HT and non-HT groups. There was only one patient with AIS of the other determined etiology (ODE) type (Table 4).
Comparisons of MHR in Different Stroke Subtypes AIS Patients With or Without HT.

Abbreviations: LAA, large-artery atherosclerosis; SVO, small-vessel occlusion; CE, cardioembolism; ODE, other determined etiology; UE, undetermined etiology. *P < .05.
Regression analysis was performed on patients of each TOAST subtype; we found that MHR was independently associated with HT in LAA-type AIS (Unadjusted: OR:0.02, 95%CI:0.001–0.76, P = .035; Adjusted: OR:0.01, 95%CI:0.00–0.62, P = .031). In patients with CE, MHR was associated with HT when confounders were not controlled (Unadjusted: OR: 0.00, 95% CI: 0.00–0.20, P = .009), but no correlation was observed after adjusting for confounders (P > .05). MHR was not associated with HT in AIS patients with SVO or UE (Table 5).
Multivariate Logistic Regression Analysis Between MHR and HT in Different Stroke Subtypes.

Adjustment for age, NIHSS on admission, LDL, CHOL, atrial fibrillation, ischemic stroke. Abbreviations: LAA, large-artery atherosclerosis; SVO, small-vessel occlusion; CE, cardioembolism; ODE, other determined etiology; UE, undetermined etiology. *P < .05.
The ROC curve and area under the curve (AUC) of MHR related to HT in the LAA-type AIS are shown in Figure 1. The AUC was 0.649 (95% CI: 0.532–0.766, P = .032). The optimal cut-off value was 0.41 with a sensitivity of 85.7% and a specificity of 43.1%

Receiver operating characteristic curve to assess the correlation between MHR and HT in LAA-type AIS. Abbreviations: MHR, monocyte to high-density lipoprotein ratio; HT, hemorrhagic transformation; AIS, acute ischemic stroke.
Discussion
High neutrophil-to-lymphocyte ratio (NLR) 11 and systemic inflammatory response index (SIRI) level 12 have been proven to be related to the poor outcome of AIS after recanalization. Reportedly, Chinese patients are more likely to have LAA (25.4% vs 14.7%) and have a lower incidence of CE (15.8% vs 25.7%) compared with that of the Caucasian stroke patients. 13 MHR was more relevant to LAA-type AIS compared with NLR and SIRI. This study was one of the few to investigate the correlation between MHR and HT risk of in LAA-type AIS. The results showed that a lower MHR was independently associated with HT after IVT, and this association was observed only in LAA-type AIS.
Inflammatory responses and BBB disruption play essential roles in both atherosclerosis and HT.14,15 Monocytes can migrate to the subendothelial space, transform into macrophages and further differentiate into foam cells after oxidized LDL uptake Foam cells secrete cytokines, chemokines, and tissue factors that contribute to atherosclerosis progression. 16 In addition, Mcs/Møs are involved in stroke-induced inflammation. 17 HDL can stabilize plaques through the following mechanisms: (1) inhibiting the activation and migration of monocytes; (2) exerting immunomodulatory effects by affecting the innate immune sensors 18 ; (3) reversing the transport of CHOL from plaques and inhibiting the oxidation of LDL; (4) protecting the endothelium by activating nitric oxide synthase.19,20
Monocyte-to-HDL ratio, a novel marker of inflammation and oxidative stress, has a greater advantage in predicting atherosclerosis and related diseases than monocytes or HDL, and has been proven to be valuable in coronary atherosclerotic heart disease in recent years. Further, MHR is an independent predictor of severity and future cardiovascular events in patients with acute coronary syndrome.10,21 Significant association was observed between MHR and slow blood flow or no reflow after percutaneous coronary intervention in patients with non-ST-segment elevation myocardial infarction. 22 However, few studies on the correlation between MHR and HT in ischemic stroke have been reported. A high MHR is an independent predictor of 30-day mortality in ischemic stroke patients, 23 and a cohort study based on rural Chinese populations suggested a higher risk of ischemic stroke in groups with high levels of MHR. 24 Xia et al. 25 analyzed 340 patients who underwent IVT and observed that the MHR was higher in patients with HT than in those without HT.
However, in our study, we found that lower MHR and higher HDL were independently correlated with HT in patients with AIS who received IVT, which was opposite to the conclusions of above studies. Wang et al reported that low levels of MHR were associated with HT in acute infarction within 24 h of onset in a study of 974 patients without thrombolysis treatment. 26 A meta-analysis (n = 3573) of nine studies showed that HDL levels in patients with HT were significantly higher than in the non-HT group (95% CI: 0.01–0.09, P = .008), 27 suggesting that high HDL levels are a potential predictor of HT, which was consistent with the results of this study; its possible mechanisms are follows: (1) HDL increases the bleeding risk by enhancing the antiplatelet effects and the activity of antithrombin C 28 ; (2) moderate CHOL level is essential for maintaining normal membrane fluidity and integrity of blood vessels; whereas, 29 HDL can reduce CHOL levels in the vascular wall, leading to BBB destruction. In addition, the discrepancy in the findings of this study may be related to the functions of different monocyte subsets. 30 Mc/Mø have both pro-inflammation and anti-inflammation effects. 31 It has been shown that bone marrow-derived Mc/Mø can prevent HT in a mouse model of ischemic stroke and show a protective effect. Immature Ly6chi monocytes can rapidly migrate to the infarct boundary, differentiate into mature Ly6clo macrophages in the lesion area and prevent post-infarction HT by releasing TGF-β1. 32 However, the specific roles of monocytes and HDL in HT remain unclear.
The subjects of the above studies included all types of AIS, and there was no association analysis between MHR and HT in the five different types of strokes. We observed that lower MHR was only independently associated with HT in LAA-type AIS patients in a further logistic model, which is the first study to investigate the association of MHR with HT in LAA-type AIS. A retrospective study of 1090 patients showed that high MHR was only strongly associated with severity and poor prognosis of 90 days in LAA-type acute cerebral infarction within 24 h of onset. 33 At the same time, in another study of 316 patients with LAA-type infarction, patients with high-level MHR had a 2.52-fold poorer prognosis compared to those with low-level MHR. 34 These conflicting conclusions may be related to the different times of onset, races, geographies, risk factors, and sample sizes, as well as the distinct Mc subpopulations and HDL functions in different ischemic stroke stages. Further analysis showed that MHR was not significantly correlated with HT after IVT in other TOAST subtypes after adjusting for confounders, which further clarified the close association between HDL, MHR, and atherosclerosis. In LAA-type AIS, patients in the HT group had a higher incidence of atrial fibrillation, history of ischemic stroke, large cerebral infarction, and higher NIHSS score on admission.
Our study explored the association between MHR and HT in AIS patients who received IVT from a new perspective, but this study has several limitations. First, this was a single-center retrospective analysis, and the patients were all from the northern cities of China. Second, the MHR levels were not dynamically measured. Third, other inflammatory factors, such as C-reactive protein and erythrocyte sedimentation rate, were not included as confounding factors. Finally, only clinical patient data were analyzed, animal models were not studied, and the identification analysis and mechanism of monocyte subsets were not discussed. In the future, more multicenter, large-sample studies and basic experiments are required to confirm this conclusion.
Conclusion
A low MHR level was independently associated with an increased risk of HT among patients with AIS who received IVT, and this conclusion only existed in the LAA-type. MHR may serve as a promising marker to help clinicians identify high risk of HT patients and choose appropriate treatment to avoid potential bleeding. Further studies are needed to clarify the effects of inflammatory response in the pathophysiology of HT after IVT.
Supplemental Material
sj-xlsx-1-cat-10.1177_10760296231167849 - Supplemental material for Low MHR Is Associated with Hemorrhagic Transformation in Acute Large Artery Atherosclerosis Ischemic Stroke Patients with Intravenous Thrombolysis
Supplemental material, sj-xlsx-1-cat-10.1177_10760296231167849 for Low MHR Is Associated with Hemorrhagic Transformation in Acute Large Artery Atherosclerosis Ischemic Stroke Patients with Intravenous Thrombolysis by Delong Meng, MM, Yan Li, MM, Ting Ju, MM, Wei Huo, MD, and Mingfei Wang, MD in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
We thank our colleagues for investing their time, expertise, and efforts to produce valuable contributions.
Author Contributions
LY and HW designed the study. LY and MD collected the data. JT and WM conducted the statistical analysis and manuscript writing. LY and MD contributed equally to this work and shared first authorship. All authors read and approved the final manuscript.
Data Availability Statement
The original contributions presented in the study are included in the Supplemental material, further inquiries can be directed to the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
The studies involving human participants were reviewed and approved by Ethics Committee of the First Affiliated Hospital of Harbin Medical University. Written informed consent for participation was not required for this study in accordance with the national legislation and the institutional requirements.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Scientific Research Foundation of the First Hospital of Harbin Medical University (2019B06).
Publisher's Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
