Abstract
Peripheral artery disease (PAD) is a common disease affecting over 200 million people worldwide. PAD is associated with significant limb and cardiovascular morbidity and mortality which is reduced by antiplatelet and antithrombotic therapy. However, the optimal type, dose, and time of antithrombotic therapy is still uncertain.We searched 4 electronic databases from January 1, 1990, to June 1, 2020, for randomized controlled trials of patients who received oral anticoagulant and antiplatelet therapy for PAD. The primary outcome was a composite of acute limb ischemia, major amputation, myocardial infarction, ischemic stroke, death from cardiovascular events, or death from any cause. Secondary outcomes included major bleeding, fatal bleeding, and intracranial hemorrhage events.We identified 3 studies that satisfied inclusion and exclusion criteria. Compared with antiplatelet alone, oral anticoagulant plus antiplatelet therapy improved acute limb ischemia (p < 0.00001), stroke (p = 0.005), and major amputation events (p = 0.11). However, oral anticoagulant plus antiplatelet therapy was not effective for prevention of myocardial infarction (p = 0.23), death from cardiovascular events (p = 0.65), or death from any cause (p = 0.66). Additionally, a significant increase in major bleeding events was demonstrated (p < 0.00001). There was no significant difference in fatal bleeding (p = 0.16) or intracranial hemorrhage events (p = 0.43). This meta-analysis showed that oral anticoagulant plus antiplatelet therapy for PAD may improve acute limb ischemia and major amputation or stroke risk compared with antiplatelet therapy alone, but could increase the risk of major bleeding events. On the other hand, measuring myocardial infarction, death, fatal bleeding, or intracranial hemorrhage risk remains controversial.
Keywords
Introduction
Peripheral arterial disease (PAD) is associated with a substantial risk of disease progression, cardiovascular morbidity and mortality, and secondary prevention is recommended for most patients. 1–2 Evidence suggests the use of antiplatelet therapy for secondary prevention in patients with PAD to reduce the threat of cardiovascular diseases. 1–2 Moreover, antiplatelet and antithrombotic therapy may increase the patency of revascularized vessels after interventions, and result in reduced acute limb ischemia risk, reintervention rates, and possibly reduced cardiovascular complications. 3 These advantages should be weighed against the risk of bleeding complications associated with anticoagulant and antiplatelet medicines. We therefore performed a meta-analysis of 3 randomized controlled trials (RCTs). 4–5 comparing oral anticoagulant plus antiplatelet therapy with antiplatelet alone for the treatment of PAD to resolve this discrepancy and provide evidence to clinical physicians.
Methods
Literature Search
Using PubMed, Embase, Web of Science, and Cochrane Library, we searched literature published between January 1, 1990, and June 1, 2020. The following search terms were included: anticoagulation (vitamin K antagonists or non-vitamin K antagonist oral anticoagulants (Direct oral anticoagulant DOACs)) or/and antiplatelet and PAD; and/or comparative studies or RCTs or cohort studies or retrospective or prospective studies. Inclusion criteria were (1) studies comparing oral anticoagulant plus antiplatelet (experimental group) with antiplatelet (control group) and (2) effectiveness of intact clinical data(include RCTs, intact patient demographics, follow-up time, outcomes). There were no language restrictions. We identified 3 studies that met our criteria (Figure 1).

Flow chart of literature review.
Two investigators (Tang and Li) independently extracted data utilizing a data abstraction tool: number of patients in the experimental (oral anticoagulant plus antiplatelet) and control antiplatelet) groups, study quality, time of follow-up, and primary and secondary PAD outcomes. The primary outcome was a composite of acute limb ischemia, major amputation, myocardial infarction, ischemic stroke, and death from cardiovascular or any cause events, and the secondary outcome was a composite of acute limb ischemia, major amputation, myocardial infarction, ischemic stroke, and death from cardiovascular or any cause events.
Data Extraction and Quality Assessment
Publication details, inclusion and exclusion criteria, enrolled patient demographics, interventions used, and outcomes (primary and secondary outcomes) were collected. Risk of bias in RCTs (including masking of participants, method of sequence generation and allocation concealment, intention-to-treat analysis, incomplete or unclear data, time to follow-up, and loss to follow-up) was assessed. Study quality was assessed using the Modified Jadad scale. 6 Any Disagreements between reviewers were resolved by consensus.
Statistical Analysis
Statistical analysis was performed using Review Manager (version 5.3; Cochrane Collaboration software). We used fixed-effects models for primary outcomes and secondary outcomes. Statistical heterogeneity was assessed by I 2 . The level of heterogeneity was distinguished as low (I 2 = 25%-49%), moderate (I 2 = 50%-74%), and high (I 2 ≥75%) heterogeneity. Primary and secondary outcomes were analyzed using odds ratios, with a 2-sided statistical significance level of 5%.
Results
Study Characteristics and Quality
The initial search strategy identified 23 full-text articles, and 20 citations were initially screened. Of these, 3 trials met the appropriate criteria for inclusion in the review (Figure 1). The 3 RCTs 3 –5 included experimental groups that received oral anticoagulant plus antiplatelet therapy for PAD and control groups that received antiplatelet therapy for PAD. The quality of RCTs was evaluated by modified Jadad score. 6 Table 1 shows the baseline characteristics for each study.
Baseline Characteristics of Included Clinical Trials.

Abbreviations—AC: anticoagulant, ABI: ankle brachial index, CAD: coronary artery disease, RCT: randomized controlled trial, NA: not available.
Primary Outcome
Primary and secondary outcomes are shown in Table 2. The 3 studies 3 –5 included results of acute limb ischemia, myocardial infarction, stroke, death from cardiovascular events, and death from any cause. Results of major amputation events were included in 2 studies. 4–5 Meta-analysis indicated that oral anticoagulant plus antiplatelet therapy for PAD reduced the occurrence of acute limb ischemia (p < 0.00001;I 2 = 43%), stroke (p = 0.005;I 2 = 33%), and major amputation events (p = 0.11; I 2 = 77%) compared to antiplatelet alone and did not result in a significant difference in myocardial infarction (p = 0.23; I 2 = 5%), death from cardiovascular(p = 0.65; I 2 = 29%) or death from any cause events (p = 0.66; I 2 = 0%) compared to antiplatelet therapy. This meta-analysis showed that oral anticoagulant plus antiplatelet therapy is more effective for reducing risk of acute limb ischemia, stroke, and major amputation than antiplatelet alone. Results of the meta-analysis of the primary outcomes are shown in Figure 2.
Primary and Secondary Outcomes in Clinical Trials.
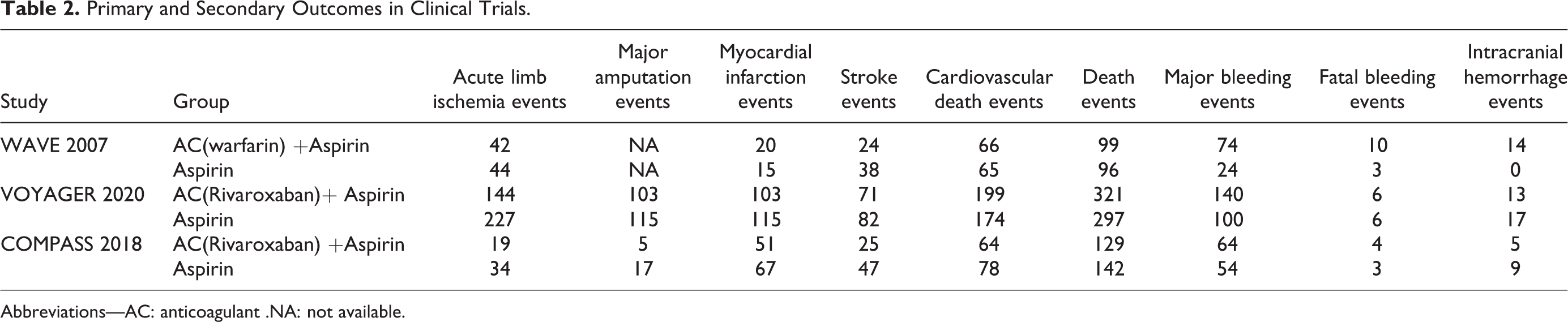
Abbreviations—AC: anticoagulant .NA: not available.

Meta-analysis of primary outcomes of clinical trials.
Secondary Outcomes
The 3 articles 3 –5 contained data about major bleeding events, fatal bleeding, and intracranial hemorrhage events. A statistically significant increase in major bleeding events (p < 0.00001, I 2 = 83%) was reported. We found that heterogeneity of major bleeding events was high (I 2 = 83%), suggesting the need to explore heterogeneity sources. We divided the major bleeding events into 2 types: AC (anticoagulant plus antiplatelet including warfarin): p < 0.00001, I 2 = 83%; and DOAC groups (anticoagulant plus antiplatelet excluding warfarin): p = 0.007, I 2 = 0%). There was no significant difference in fatal bleeding (p = 0.16; I 2 = 0%) or intracranial hemorrhage events (p = 0.43, I 2 = 77%). Because the heterogeneity of intracranial hemorrhage events was high, we will need to explore heterogeneity sources. For this study, we also divided intracranial hemorrhage events into 2 types: AC: p = 0.43, I 2 = 77%; and DOAC: p = 0.23, I 2 = 0%). Figure 3 shows the meta-analysis of bleeding events (major bleeding, fatal bleeding, and intracranial hemorrhage events).
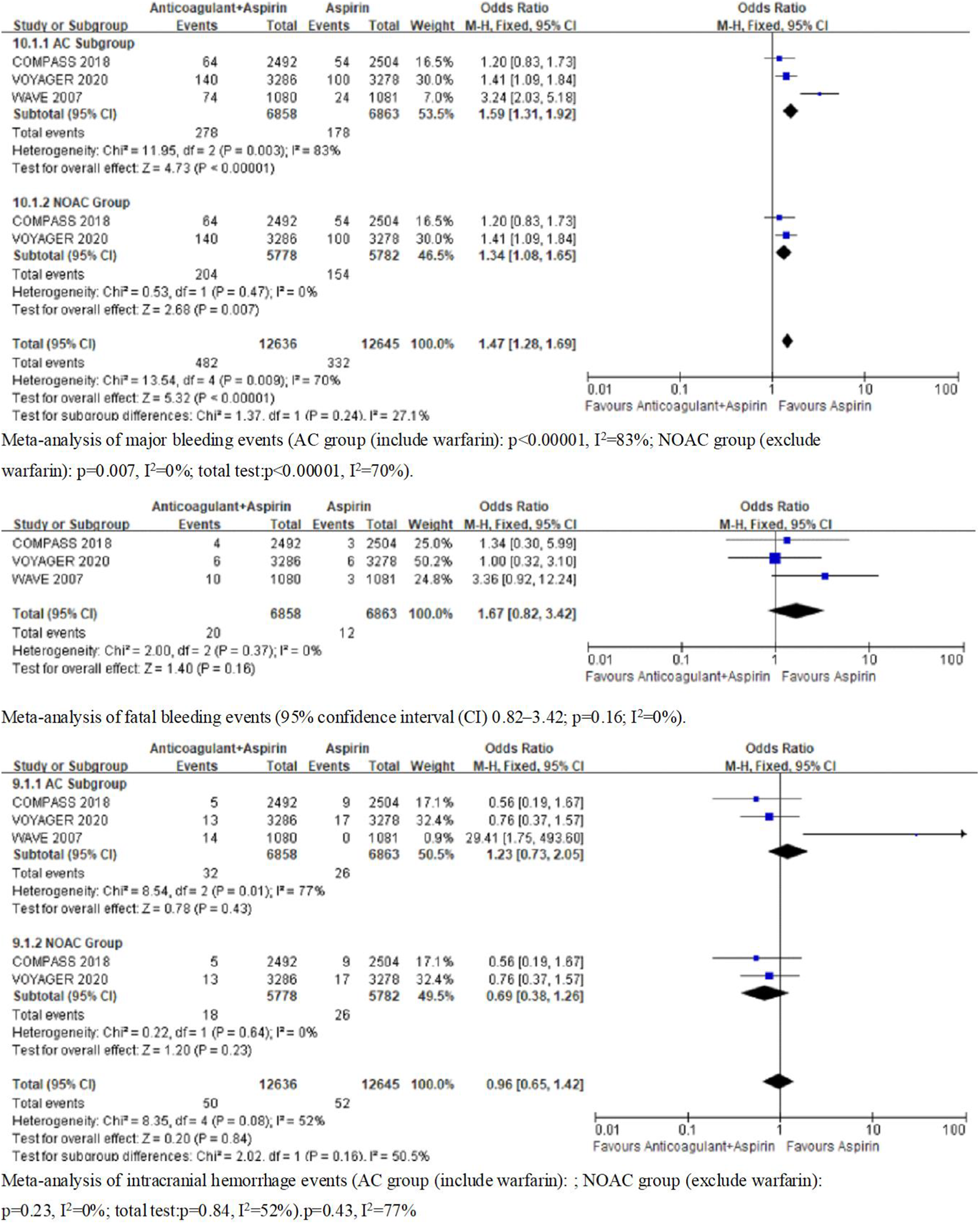
Meta-analysis of bleeding events (secondary outcomes) of clinical trials.
Discussion
PAD is most commonly caused by atherosclerosis and is widespread in the world. 7 Current guidelines recommend antiplatelet monotherapy for prevention of cardiovascular disease with a class IA recommendation. By comparison, dual antiplatelet therapy is regularly prescribed after stent placement in cardiovascular disease. Furthermore, these recommendations contain PAD subgroup analyses in cardiovascular trials. Through underlying antiplatelet therapy, patients with PAD are still at a high risk of atherothrombotic complications. Treatment strategies targeting the decline of cardiovascular morbidity or mortality in PAD have necessarily involved the use of antithrombotic treatment. However, the type, dose, time, and strength of antithrombotic treatment for PAD patients has not yet been determined. As time went on, 3 RCTs 3 –5 provided evidences to antithrombotic treatment for PAD. So we use meta-analysis from 3 RCTs indicating that oral anticoagulant plus antiplatelet therapy for PAD may improve acute limb ischemia, major amputation, or stroke risk compared with antiplatelet therapy alone.
In the WAVE (Warfarin Antiplatelet Vascular Evaluation) trial, 5 the study compared the effectiveness and safety of combination antithrombotic therapy with oral anticoagulant (warfarin) plus antiplatelet therapy with antiplatelet therapy alone for PAD. The results showed that the combination of oral anticoagulant (warfarin) plus antiplatelet therapy was no more effective than antiplatelet therapy alone in preventing primary cardiovascular complications. In contrast, anticoagulant (warfarin) plus antiplatelet therapy was associated with a substantial excess of moderate or life-threatening bleeding events. The WAVE trail’s results emphasize the need to appraise alternatives to warfarin in patients with PAD. A low dosage Factor Xa inhibitor (rivaroxaban) plus antiplatelet therapy has been shown to lower cardiovascular ischemia risk. The COMPASS (Cardiovascular Outcomes for People Using Anticoagulation Strategies) study 4 demonstrated that patients with PAD who were enrolled in the COMPASS trial and received the combination of rivaroxaban 2.5 mg twice a day plus 100 mg of aspirin a day had a 28% reduction in major adverse cardiovascular events, a 46% reduction in major adverse limb events, and a 31% reduction in the composite of major adverse cardiovascular or limb events, compared with the aspirin alone group. Although this combination was associated with an increased risk of major bleeding, there was no excess in fatal or critical organ bleeds. The European Society for Vascular Surgery (ESVS) 2020 guidelines 8 have added recommendations in acceptance of the COMPASS study for PAD patients with chronic limb threatening ischemia.
Acute limb ischemia (ALI) is a common complication in PAD and lower extremity vascular reconstruction is an effective measure for preventing limb loss. 9 –11 However, new evidence suggests that patients who had lower limb recanalization had a quadruple increased risk of ALI and higher risk of cardiovascular events, including an almost 30% increased risk of myocardial infarction. 12–13 Acute limb ischemia is a complication that is combined with long hospital stays and high risk of amputation, disability, and death. 14 –17 On the basis of COMPASS study, the Vascular Outcomes study of ASA along with rivaroxaban in endovascular or surgical limb revascularisation for peripheral artery disease (VOYAGER PAD) 3 was designed to assess the effectiveness and safety of 2.5 mg twice daily rivaroxaban plus aspirin in high risk PAD patients at high risk for lower extremity vascular reconstruction compared with aspirin alone. A low dose of rivaroxaban with added aspirin was associated with a significantly lower morbidity of ALI, major amputation for vascular causes, myocardial infarction, ischemic stroke, or death from cardiovascular causes compared to aspirin alone. The incidence of TIMI (Thrombolysis in Myocardial Infarction) major bleeding did not significantly differ between the 2 groups. The incidence of ISTH (International Society on Thrombosis and Haemostasis) major bleeding was significantly higher in the rivaroxaban plus aspirin group.
Previous studies for antithrombotic therapy in endovascular and surgical revascularization were not definitive, as they were premised on subgroups of patients with coronary artery disease or extensive atherosclerosis patients. 18 –20 The VOYAGER PAD study provides new, high-quality evidence that the benefits for long-term use of this novel dual pathway regimen reduces the risk of acute cardiovascular events and ALI events after lower limb revascularization. The data from the VOYAGER PAD study complements the COMPASS study, which revealed a significant reduction in cardiovascular events and ALI events in PAD patients. Our meta-analysis has highlighted a new antithrombotic therapy for patients with PAD. Platelet inhibition plus low-dose anticoagulant would depend on status to balance ischemic and bleeding risks when selecting type, dose, and intensity of antithrombotic treatment for PAD. 21 –23 Our study has some limitations. In this meta-analysis, the number of RCT trials was low and major bleeding events are a severe problem for antithrombotic therapy. Because the studies used aspirin and rivaroxaban for antiplatelet/anticoagulant therapy, it is still unclear whether similar curative effect would be verified by other categories antiplatelet agents (P2Y12 inhibitors, phosphodiesterase inhibitors, glycoprotein IIb/IIIa receptor inhibitors) and DOACs (direct thrombin inhibitors). The efficacy/safety of rivaroxaban plus aspirin in patients with PAD compared with dual antiplatelet treatment is unknown. We hope that the questions we have raised will be addressed soon by additional trails.
Conclusions
The results of our meta-analysis indicate that oral anticoagulant plus antiplatelet therapy for PAD may improve acute limb ischemia, major amputation, or stroke risk compared with antiplatelet therapy alone. The risk of myocardial infarction, death, fatal bleeding or intracranial hemorrhage remains debatable. However, it was evident that substantially more major bleeding events occurred in the oral anticoagulant plus antiplatelet group.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
