Abstract
Background:
Triple antithrombotic therapy is used in patients who require systemic anticoagulation and undergo percutaneous coronary intervention (PCI) requiring dual antiplatelet therapy. Bleeding with this combination is significant; however, few studies have described outcomes with the use of newer oral P2Y12 inhibitors in this setting.
Objectives:
We aimed to compare outcomes among patients prescribed triple therapy with prasugrel or ticagrelor compared to triple therapy with clopidogrel in patients who underwent PCI and required warfarin.
Methods:
We retrospectively evaluated 168 patients who received either prasugrel (n = 32) or ticagrelor (n = 10) and were matched (1:3) to those who received clopidogrel (n = 126) at the time of discharge from the index PCI visit. Matching was performed based on age ±10 years, sex, and indication for PCI. The primary outcome was the incidence of any bleeding during the 12-month follow-up. We also evaluated major adverse cardiovascular and cerebrovascular events (MACCEs).
Results:
Patient baseline characteristics were similar between groups. There was a significant excess of bleeding in patients who received prasugrel or ticagrelor compared to clopidogrel as part of triple therapy (28.6% vs 12.7%; odds ratio, 3.3; 95% confidence interval, 1.38-8.34). No differences were seen between groups in MACCEs.
Conclusions:
The use of prasugrel or ticagrelor as part of triple antithrombotic therapy among patients who underwent PCI and received warfarin was associated with significantly more bleeding compared to patients who received clopidogrel. Therefore, higher potency P2Y12 inhibitors should be used cautiously in these patients.
Introduction
Triple antithrombotic therapy represents a challenging clinical conundrum with an annual bleeding risk of up to 45%. 1 Approximately 10% of the nearly 1 million patients who undergo percutaneous coronary intervention (PCI) in the United States each year have an indication for chronic oral anticoagulation therapy. 2,3 These patients require both dual antiplatelet therapy (DAPT) and systemic anticoagulation to mitigate the risks of stent thrombosis (ST), major adverse cardiovascular event (MACE), and thromboembolism. 1,2 The introduction of newer oral P2Y12 inhibitors, including prasugrel and ticagrelor, reduces the risks of ST and MACE but at the expense of increased bleeding. 4,5 Triple antithrombotic therapy regimens in clinical practice evolved to include these newer P2Y12 inhibitors, despite the lack of rigorous studies evaluating their use. While several studies evaluate clopidogrel as part of triple therapy with aspirin (ASA) and warfarin, data that describe prasugrel or ticagrelor in this setting are limited. 6 Therefore, the purpose of this study was to investigate outcomes among patients prescribed triple therapy with prasugrel or ticagrelor compared to triple therapy with clopidogrel in patients who underwent PCI and required warfarin.
Methods
This retrospective cohort study included patients who underwent PCI at the University of Pittsburgh Medical Center, an integrated delivery system comprised of 22 academic, community, and specialty hospitals. Patients included in this analysis were treated with PCI at 5 different institutions (1 academic and 4 community hospitals) between July 1, 2010, and December 31, 2013, with follow-up through December 31, 2014. Patients were identified through an electronic medical record data repository that contains full-text medical records and integrates information from central transcription, pharmacy, laboratory, finance, administrative, and other departmental databases. 7 To maintain confidentiality, all data were de-identified and extracted through use of an honest broker system (neutral third party acting on behalf of the research team). Criteria for exemption from informed consent were met by the university’s institutional review board.
Patients eligible for study inclusion were consecutive adults (≥18 years of age) identified by International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) codes for PCI for inpatients, whereas Current Procedural Terminology codes for PCI were used for outpatients. Patients with discharge summaries that included documentation of orders for triple antithrombotic therapy, defined as the use of ASA, a P2Y12 inhibitor (clopidogrel, ticagrelor, or prasugrel), and warfarin, were included in the study. For patients with multiple visits during the study period, the first PCI admission was considered the index. Likewise, the first PCI was considered the index procedure for patients who had more than 1 PCI during the study period. Patients discharged on triple therapy with prasugrel or ticagrelor were randomly matched (1:3) to patients treated with clopidogrel based on age ±10 years, sex, and indication for PCI (acute coronary syndrome vs elective procedure).
Patient- and treatment-level covariates were collected to determine their impact on patient outcomes. Baseline data collected for each patient included relevant demographic and clinical characteristics. Comorbidities for atrial fibrillation/flutter (AF/AFl) and venous thromboembolism were identified using ICD-9-CM codes, whereas other comorbidities were based on review of the medical record for documentation in the catheterization laboratory procedure summary including diabetes mellitus, history of myocardial infarction (MI), coronary artery bypass grafting (CABG), and/or PCI; cerebrovascular disease; end-stage renal disease on dialysis; valvular heart disease; hypertension; hyperlipidemia; and heart failure. Discharge summary reports were annotated to confirm orders for triple antithrombotic therapy as described above, the indication for PCI, and the dose of ASA prescribed. We also evaluated postdischarge bleeding and major adverse cardiovascular and cerebrovascular events (MACCEs). Sources of data included medication charge codes, clinical reports (emergency department, admission, discharge summary, outpatient visits), laboratory data (hemoglobin [Hb]), and transfusion charges. Smoking status was determined based on annotation of admission reports.
The primary outcome of this study was the incidence of any bleeding during the 12-month period after the index hospitalization. We evaluated for bleeding based on the presence of any nonelective readmission with a primary admission ICD-9-CM code for bleeding. We also evaluated for bleeding based on a decrease in Hb of ≥3 g/dL or the requirement for transfusion of ≥2 units of packed red blood cells. The Hb and transfusion thresholds were adapted from the Efficacy and Safety of Subcutaneous Enoxaparin in Non-Q-Wave Coronary Events criteria for major bleeding. 8 The presence of MACCEs was defined as the cumulative incidence of a composite of cardiac death, nonfatal MI, or nonfatal ischemic stroke within 12 months after the index visit. Nonfatal MI was determined based on the presence of nonelective readmission along with an ICD-9-CM code for MI. Ischemic stroke was defined similarly to MI, based on nonelective readmission with an ICD-9-CM code for stroke.
Statistical Analysis
Parametric and nonparametric comparative tests were used to evaluate differences in baseline characteristics. The χ2 or Fisher exact test were used to compare proportions for nominal data. Student t test was used for continuous data. P values <.05 were considered statistically significant. Univariate logistic regression was used for selection of significant variables to include in the multivariate analysis. A stepwise multivariate analysis was performed to identify clinical factors predictive of bleeding and MACCE. All data were analyzed using IBM SPSS Statistics 22.0 (IBM, Inc, New York, New York).
Results
A flowchart for patient selection is shown in Figure 1. A total of 1051 patient visits were screened for treatment with ASA, a P2Y12 inhibitor, and warfarin at discharge. Of 1051 patient visits, 695 patients were eligible for inclusion (Figure 1). Forty-two patients were discharged on triple therapy with either prasugrel (n = 32) or ticagrelor (n = 10). A total of 653 patients were identified who received clopidogrel and 126 were selected for the 1:3 match. Table 1 summarizes the baseline characteristics of the study patients. Patient demographics and comorbidities were similar between groups, including approximately 50% with a history of AF/AFl. Patients were generally representative of middle-aged Caucasian males. Table 2 provides a description of medication and procedural characteristics of patients. Approximately 60% of patients in each group were treated for acute coronary syndromes and the majority was discharged on low-dose ASA therapy. Patients in the prasugrel/ticagrelor group received a greater proportion of bivalirudin during PCI compared to the clopidogrel group. No other differences in medications were observed, with the exception of a higher proportion of patients in the clopidogrel group who received ASA prior to admission.
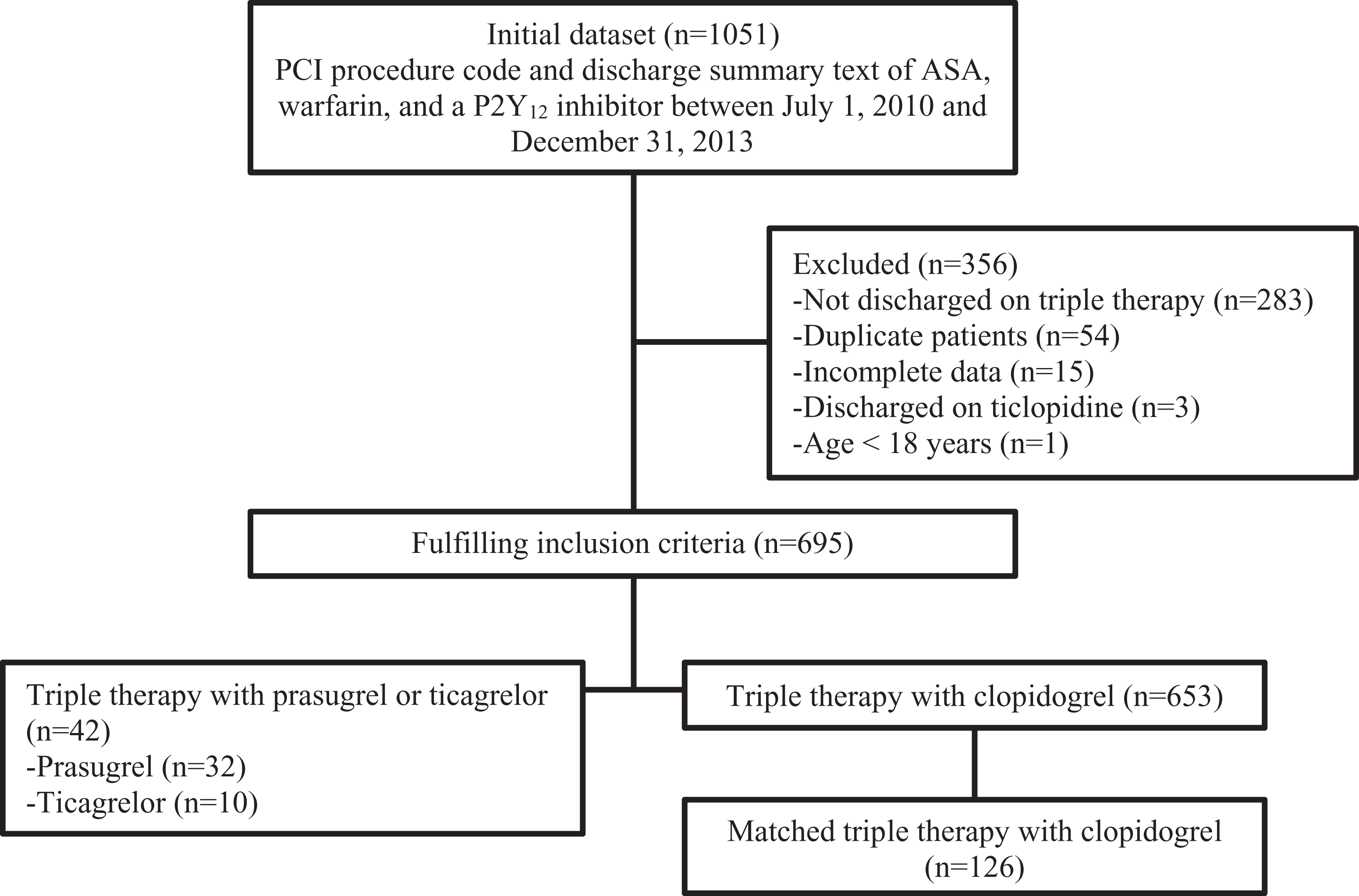
Flowchart.
Baseline Characteristics of the Study Population According to P2Y12 Inhibitor Group.a

Abbreviations: CABG, coronary artery bypass grafting; MI, myocardial infarction; PCI, percutaneous coronary intervention; VTE, venous thromboembolism.
aData are shown as mean ± standard deviation (SD) or number (percentage).
bMatched on age and sex for selection of cohort.
cConfirmation of current smoking status could only be determined for 131 (78%) of 168 of the study cohort.
dFisher exact test.
Treatment and Procedural Characteristics of the Study Population According to P2Y12 Inhibitor Group.a

Abbreviations: ACE-I, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; NSTEMI, non-ST-segment elevation myocardial infarction; STEMI, ST-segment elevation myocardial infarction; UA, unstable angina.
aData are shown as number (percentage).
bConfirmation of medication prior to admission could only be determined for 131 (78%) of 168 of the study cohort.
cMatched on PCI indication for selection of cohort.
dOne patient in the clopidogrel group was discharged on aspirin 162 mg/d; comparisons of all aspirin dose categories by Fisher exact test.
Clinical outcomes are presented in Table 3. There was a significantly greater proportion of bleeding among patients who received ticagrelor or prasugrel compared to clopidogrel as part of triple therapy. Several clinical variables were evaluated in univariate analyses for effects on bleeding: history of chronic kidney disease (P = .11), dialysis (P = .021), stroke (P = .67), home anticoagulation use (P = .21), body weight (P = .68), and clopidogrel use (P = .043). Only dialysis and P2Y12 inhibitor use were included in the multivariate regression model for bleeding. Bleeding events were significant only for the P2Y12 inhibitor prescribed and for history of dialysis by multivariate regression. There was a significant increase in bleeding among patients who received ticagrelor or prasugrel compared to clopidogrel (odds ratio [OR], 3.3; 95% confidence interval [CI], 1.38-8.34). The proportion of patients who required dialysis was low, but this variable also influenced the bleeding outcome (OR, 12.5; 95% CI, 1.9-81.6).
Outcomes According to P2Y12 Inhibitor Group.a

Abbreviation: MACCE, major adverse cardiovascular and cerebrovascular events.
aData are shown as number (percentage). Proportions compared between groups using χ2 or Fisher exact test.
Event rates for composite MACCE and individual end points did not differ between groups (Table 3). Clinical variables tested in univariate analyses for effects on MACCE included current nonsmoker (P = .02), history of CAD (combined from history of MI, CABG, and PCI; P = .96), history of diabetes mellitus (P = .29), dialysis (P = .23), and clopidogrel use (P = .91). The only significant variable was smoking status, so there was no need to proceed with multivariate analysis. The risk of MACCE was lower among patients who were not current smokers (OR, 0.4; 95% CI, 0.17-0.91). The P2Y12 inhibitor group did not affect the MACCE outcome.
Discussion
Our findings demonstrated a significantly greater risk of bleeding when prasugrel or ticagrelor was prescribed as part of triple antithrombotic therapy compared to those who received clopidogrel. We observed no between-group differences in ischemic outcomes, including the MACCE composite and individual end points. Our population was generally representative of a real-world cohort of higher risk patients who required DAPT and also had an indication for oral anticoagulation. Patients who require triple antithrombotic therapy present a challenging clinical dilemma due to the significant risks of major bleeding with clopidogrel-based regimens. 1 However, our experience provides additional insight into outcomes in the context of newer oral P2Y12 inhibitors.
Current guidelines recommend triple antithrombotic therapy with ASA, clopidogrel, and warfarin in patients requiring oral anticoagulation after PCI, even though such regimens have not been adequately studied in prospective trials. Strategies to mitigate bleed risk include using a lower international normalized ratio (INR) target for warfarin (2.0-2.5), lowering the ASA dose, and using the triple therapy combination for as short a time as possible depending on stent type and bleed risk. 9,10 Recent prospective studies have provided insights into the triple therapy dilemma post-PCI. The What is the Optimal antiplatelet and anticoagulant therapy in patients with oral anticoagulation and coronary StenTing (WOEST) study evaluated dual therapy (clopidogrel and vitamin K antagonist [VKA]) versus triple therapy (ASA, clopidogrel, and VKA) in an open-label fashion. The authors reported significantly less bleeding in the dual therapy versus triple therapy group without an excess of thrombotic events. 11 The triple therapy in patients on oral anticoagulation after drug-eluting stent implantation (ISAR-TRIPLE) study demonstrated no difference in net clinical outcome, including major bleeding, between 6-week and 6-month durations of clopidogrel in patients who also received ASA and an oral anticoagulant. 12 The PIONEER AF-PCI study (an open-label, randomized, controlled, multicenter study exploring 2 treatment strategies of rivaroxaban and a dose-adjusted oral vitamin K antagonist treatment strategy in subjects with atrial fibrillation who undergo percutaneous coronary intervention) found significantly lower rates of clinically significant bleeding among patients randomized to low-dose or very-low-dose rivaroxaban plus a P2Y12 inhibitor or DAPT (1-12 months) compared to a VKA plus DAPT (1-12 months). 13 However, the use of prasugrel and ticagrelor was limited in this trial and was excluded from both the WOEST and ISAR-TRIPLE studies. 11 –13
Observational studies have provided some insights into the clinical experience with using newer oral P2Y12 inhibitors as part of triple antithrombotic therapy. Sarafoff et al reported results from a study of 377 patients who underwent drug-eluting stent placement and received triple antithrombotic therapy. 6 Of the patients studied, 21 (5.6%) received prasugrel whereas 356 (94.4%) received clopidogrel. Overall, the rate of bleeding according to the Thrombolysis in Myocardial Infarction (TIMI) major and minor definitions was 28.6% in the prasugrel group compared to 6.7% in the clopidogrel group (hazard ratio [HR], 4.6; 95% CI, 1.9-11.4; P < .001). 6 No differences were found in MACCE. Prasugrel treatment was independently associated with bleeding at 6 months in their multivariable model (adjusted HR, 3.2; 95% CI, 1.1-9.1; P = .03). 6
Registry data from the Acute Coronary Treatment and Intervention Outcomes Network Registry-Get With the Guidelines reported results for 1370 patients who were discharged on triple therapy for acute MI and AF. 14 The majority of these patients (n = 1346) were discharged on clopidogrel, whereas only 19 patients received prasugrel. The primary safety outcome was bleeding readmission within 2 years of the index PCI, and this was significantly higher in the triple therapy compared to the dual therapy group (adjusted HR, 1.61; 95% CI, 1.31-1.97). Readmissions that involved intracranial hemorrhage were also higher in the triple therapy group (adjusted HR, 2.04; 95% CI, 1.25-3.34). 14 Direct comparisons between clopidogrel and prasugrel were not reported.
The Cardiovascular Percutaneous Intervention Trial registry included a comparison of 27 and 125 ticagrelor and clopidogrel-treated patients, respectively, who also received background therapy with ASA and a VKA after PCI. 15 Bleeding events were based on TIMI and Bleeding Academic Research Consortium definitions; however, the presence of non-CABG-related major bleeding comprised the net adverse clinical event (NACE) composite. 15 Overall, no significant differences were seen between groups for NACE or the bleeding outcomes. The authors cited baseline differences between groups as a potential reason for the lack of difference in outcomes.
The primary strength of our study was that it focused on the use of newer oral P2Y12 inhibitors in the setting of patients who received triple therapy after PCI. Our primary finding was a significant excess of bleeding in the prasugrel or ticagrelor group compared with clopidogrel-treated patients. Using multivariate regression, we found an approximate 3-fold excess risk of bleeding within the prasugrel or ticagrelor arm of the study. This relative risk increase in bleeding was concordant with limited observational studies of prasugrel, which demonstrated an approximate 2.4- to 3.2-fold risk compared to clopidogrel-based triple therapy. 6,14 The implications of our findings with ticagrelor are more limited due to the smaller sample size, but nonetheless a signal for increased bleeding appears to exist. Interestingly, our ticagrelor results differed from a study of registry data. 15 The discordant results may relate to selection bias and baseline differences in patient groups.
Limitations
Several limitations are acknowledged in our analysis and are inherent to the retrospective design. First, the decision to prescribe a particular P2Y12 inhibitor was at the discretion of the treating physician and could have resulted in selection bias. We did, however, control for some of the factors that impact bleeding and thrombotic risk through matching, such as age, sex, and indication for PCI. Based on the relatively small sample size, we decided to aggregate the prasugrel and ticagrelor patients into a single group and compared them to clopidogrel-treated patients. Therefore, we were unable to delineate any possible differences in bleeding or thrombotic risk between the 2 newer P2Y12 inhibitors. We collected a number of other important comorbidities and concurrent medications that could have also explained differences in reasons for antiplatelet selection and outcome. The groups were well-balanced overall; however, patients in the prasugrel/ticagrelor group were more likely to have received bivalirudin, whereas more patients in the clopidogrel group received ASA prior to admission. The impact of these differences could not be discerned; however, the decision to use bivalirudin could have been based on an effort to mitigate bleed risk in the patients who received newer, and higher potency, P2Y12 inhibitors. Certain other medications were also unknown such as proton pump inhibitors and selective serotonin reuptake inhibitors.
Approximately half of our study population had a history of AF/AFl, although all indications for anticoagulation could not be determined. With regard to warfarin, we were also unable to obtain INR values and time in therapeutic range during follow-up. Other variables were also not collected, including specific duration of and adherence to DAPT and certain procedural characteristics such as arterial access and type of stent. Finally, we had limited Hb and transfusion requirements that were seen in follow-up. Therefore, our bleeding definition relied to a greater extent on readmissions with primary ICD-9-CM code documentation of bleeding. This proved to be an efficient means of capturing bleeding based on the ability to leverage a comprehensive electronic medical record data repository from a large integrated delivery system. However, we were only able to capture clinical outcomes in patients who received care within our health system.
Conclusions
The use of prasugrel or ticagrelor as part of triple antithrombotic therapy among patients who underwent PCI and received ASA and warfarin was associated with significantly more bleeding compared to patients who received clopidogrel. Therefore, the newer P2Y12 inhibitors should be used cautiously in these patients and avoided to the extent possible.
Footnotes
Authors’ Note
This material in this article has not been previously presented elsewhere.
Acknowledgments
The authors acknowledge Melissa Saul for her expertise and contributions to data management.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
