Abstract
Antihemophilic factor (recombinant) (rAHF; ADVATE®; Baxalta US Inc., a Takeda company, Lexington, MA, USA) is indicated for the treatment and prevention of bleeding in patients with hemophilia A. We aimed to assess the safety and efficacy of standard prophylaxis versus on-demand treatment with rAHF in previously treated Chinese patients with severe/moderately severe hemophilia A. This open-label, sequential, interventional, postapproval study (NCT02170402) conducted in China included patients of any age with hemophilia A with factor VIII (FVIII) level ≤2%. Patients received 6 months’ on-demand rAHF then 6 months’ rAHF prophylaxis (20-40 IU/kg every 48 ± 6 hours). The primary objective was percentage reduction in annualized bleeding rate (ABR) in the per-protocol analysis set (PPAS); secondary objectives included ABR by bleeding subtype, hemostatic efficacy, immunogenicity, and safety. Of 72 patients who received ≥1 rAHF dose, 61 were included in the PPAS. Total ABR was lower during prophylaxis (mean 2.5, 95% CI 1.5-3.7; median 0) versus on-demand treatment (mean 58.3, 95% CI 52.5-64.7; median 53.9), representing a 95.9% risk reduction. Similar findings in favor of prophylaxis were observed for all types of bleeding event by cause and location. rAHF hemostatic efficacy was rated as “excellent”/“good” in 96.1% of treated bleeding events. Transient FVIII inhibitors (0.6-1.7 BU) in 4 patients resolved before study end; no unexpected safety issues were observed. rAHF prophylaxis in this study of previously treated Chinese patients with severe/moderately severe hemophilia A resulted in a clear reduction in bleeding events versus rAHF on-demand treatment, with no change in safety profile.
Keywords
Introduction
Hemophilia A is a rare, recessive, X-linked bleeding disorder caused by deficient activity of coagulation factor VIII (FVIII). 1,2 The tendency to bleed is determined by plasma FVIII levels, with mild, moderate, and severe hemophilia A corresponding to plasma FVIII levels of 5%-<40%, 1%-5%, and <1% of normal, respectively. 2 In China, estimates suggest that 48% of patients with hemophilia A have severe disease and 31% have moderate disease, 3 which is broadly similar to the prevalence in Western countries. 4 –8
Routine prophylactic FVIII replacement therapy with intravenous injections of FVIII concentrates is the standard of care for patients with severe hemophilia A. 9 According to a 2016 survey of adults with hemophilia in China, recombinant FVIII (rFVIII) was the dominant hemophilia therapy, but other treatments such as fresh frozen plasma and cryoprecipitate were used more often than in other countries. 10 Progress has been made in China over the past 2 decades in providing patients with hemophilia A greater access to FVIII products 11,12 and promoting greater usage of prophylaxis 13 to reduce bleeding events and improve joint health.
Antihemophilic factor (recombinant) (rAHF; ADVATE®; Baxalta US Inc., a Takeda company, Lexington, MA, USA) is a third-generation rFVIII replacement therapy that supersedes plasma-derived coagulation factors by eliminating posttransfusion blood-borne infection risk. 14 Structurally similar to naturally occurring FVIII, it is free of human or animal plasma protein additives 14,15 and is considered an effective and well-tolerated replacement therapy based on ≥15 years of prospective study in adult and pediatric patients with hemophilia A, regardless of prior treatment status. 16 –21 In 2012, the China Food and Drug Administration approved rAHF for the control and prophylaxis of bleeding events in patients with hemophilia A. 22
A previous multicenter, prospective, observational clinical study indicated that rAHF was efficacious and well tolerated for on-demand treatment of bleeding events in 58 Chinese patients with mild, moderate, or severe hemophilia A. 23 A postapproval study conducted in the United States and Europe showed that standard prophylaxis significantly reduced bleeding rates versus on-demand treatment. 20 The phase 4 sequential study reported herein assessed the efficacy and safety of standard prophylaxis versus on-demand treatment with rAHF and the pharmacokinetics (PK) of rAHF in previously treated Chinese patients with severe or moderately severe hemophilia A.
Materials and Methods
Study Design and Conduct
This was a prospective, multicenter, open-label, sequential interventional, postapproval study (NCT02170402) conducted at 11 sites in China between June 2014 and May 2016. The study was approved by the independent ethics committees of all participating sites and conducted in accordance with the standards of Good Clinical Practice and the principles of the Declaration of Helsinki.
Patients
Eligible patients were males (any age) with severe or moderately severe hemophilia A with FVIII ≤2% receiving on-demand FVIII at the time of study enrolment and who had >50 previous exposure days (EDs) to any recombinant or plasma-derived FVIII, a negative inhibitor result at screening (defined as <0.6 Bethesda units/mL [BU/mL]), and no history of inhibitor development. Patients were discontinued from the study if they required a surgical or dental procedure.
Procedures
Patients received intravenous rAHF as on-demand treatment for 6 months and as prophylaxis for the following 6 months. For on-demand treatment of early hemarthrosis, muscle bleeding, oral bleeding, and other bleeding events, 20-100 IU/kg was administered every 6-24 hours, as required by the age of the patient, bleeding severity, and response to treatment, until bleeding resolved or healing was achieved. At the end of the on-demand treatment regimen, standard prophylaxis with rAHF 20-40 IU/kg was administered every other day (48 ± 6 hours).
Twenty-four patients who received rAHF at a dose of 45 ± 10 IU/kg at the start of on-demand treatment (ie, during the first month) were evaluable for the assessment of FVIII PK. Up to 2 patients without a PK assessment at this time were offered a PK assessment at the month 6, 12, or 13 visits.
Information about bleeding events was collected directly from patients via diaries provided at screening and from clinician notes, and included: cause (spontaneous, traumatic, undetermined), location (including information on target joints), and severity (mild [bleeding did not prevent participation in normal activities], moderate [more extensive bleeding did prevent participation in normal activities], severe [life-or limb-threatening bleeding or bleeding threatened an important function]). Patient diaries were used to record treatment and cause of new bleeding events, antihemophilic factor (recombinant) infusion administration details, and response to treatment.
Patients were monitored for the occurrence of any adverse event (AE); those occurring during rAHF therapy were categorized according to Medical Dictionary for Regulatory Activities (MedDRA, version 17.0) dictionary terms and followed up until a conclusion for the AE was determined.
Health-related quality of life (HRQoL) and pain data were collected prospectively using validated patient self-reported questionnaires completed at enrolment, end of on-demand treatment, and end of prophylaxis: 36-Item Short Form Health Survey version 2 (SF-36v2) 24 for patients aged ≥14 years, Canadian Haemophilia Outcomes-Kids Life Assessment Tool (CHO-KLAT) version 2.0 25 for patients aged <14 years, and a visual analogue scale (VAS) for pain (stratified by age <6, 6-12, and >12 years).
Outcomes
The primary outcome measure was percentage reduction in total annualized bleeding rate (ABR) during prophylaxis relative to that during on-demand treatment. Secondary outcome measures were ABRs according to bleeding subtype (joint and nonjoint) and etiology (spontaneous and traumatic); rAHF consumption required to resolve a bleeding event; safety; immunogenicity; and PK. Another secondary outcome measure was overall hemostatic efficacy rated by the patient/caregiver or the investigator at resolution of bleeding using a 4-point scale as follows: “excellent,” for full relief of pain and cessation of bleeding within ∼8 hours of single infusion (no additional infusions required to control bleeding); “good,” for definite pain relief and/or improvements in signs of bleeding within ∼8 hours of single infusion (possibly required additional infusions); “fair,” for probable or slight relief of pain and slight improvement in bleeding within ∼8 hours of single infusion (required additional infusions for complete resolution), and “poor” for no improvement or worsening of condition Tertiary outcome measures were change from baseline in HRQoL and pain, and adherence to prophylaxis (adherence determination included infusions to treat bleeding events during prophylaxis). Adherence was defined with a cutoff of ≥80% compliance to both the dose and frequency of the prophylaxis regimen.
Statistical Analyses
All efficacy-related analyses were conducted on the per-protocol analysis set (PPAS; defined as patients who completed 6 months ± 2 weeks of on-demand treatment and 6 months ± 2 weeks of prophylaxis, with ≥80% compliance to both the dose and frequency of the prophylaxis regimen). The safety analysis set comprised patients who received ≥1 infusion of rAHF.
Descriptive statistics of study endpoints were generated for each treatment regimen (on-demand, prophylaxis) and age category (<6 years, 6-12 years, and >12 years of age). ABR was analyzed using the generalized estimating equations (GEE) framework with a negative binomial option. The 2 treatment regimens (on-demand and prophylaxis) were compared in each patient in terms of mean and median ABR within the GEE model framework (with a logarithmic link function, the default for the negative binomial distribution option), accounting for the fixed treatment effect and the follow-up time (in years) as an offset and an unstructured working correlation matrix. Percentage change in ABR between treatments was based on mean ABR. Ratios between treatment means (95% confidence interval [CI]) were estimated in this model using the GENMOD procedure of SAS.
Analysis of inhibitor incidence was computed for 2 risk categories, based on previous FVIII treatment: patients with 51-150 EDs (higher risk) and those with >150 EDs (lower risk). Changes in HRQoL and VAS pain scores between baseline and the end of on-demand treatment and between baseline and the end of prophylaxis were tested for statistical significance using Wilcoxon signed-rank tests for paired samples. The association between adherence (continuous) and ABR was evaluated using a nonparametric approach via Spearman’s correlation analysis. FVIII PK parameters were derived using the preinfusion-adjusted activities of FVIII by noncompartmental analysis methods with Phoenix WinNonlin Professional version 6.4 (Pharsight Corp., St Louis, MO, USA).
All data were analyzed with SAS version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Patients
Of 79 patients enrolled, 72 received rAHF and were included in the safety analysis set (Figure 1 and Table 1). The study (on-demand and prophylaxis periods) was completed by 67 patients. Of 5 patients who discontinued from the study, 3 withdrew consent after ∼13 months of treatment, 1 refused to attend a clinic visit, and 1 was withdrawn because prophylaxis was administered at the beginning of the study. Sixty-one patients comprised the PPAS.

Patient disposition. FAS indicates full analysis set; PPAS per-protocol analysis set; rAHF, antihemophilic factor (recombinant); SAS, safety analysis set.
Baseline Characteristics of the Chinese Males (Safety Analysis Set)a.

Abbreviation: FVIII, factor VIII.
b This patient was withdrawn from the study because he had not been receiving on-demand treatment at the time of enrolment.
Bleeding Events
Among 61 patients in the PPAS, 1745 bleeding events occurred during on-demand treatment and 72 during prophylaxis. During on-demand treatment, 1066 (61.1%) bleeding events in 55 patients were spontaneous, 400 (22.9%) events in 50 patients were traumatic, and 278 (15.9%) events in 37 patients were of undetermined cause. There were 1389 mild, 349 moderate, and 7 severe bleeding events during the on-demand treatment period. During prophylaxis, 15 (20.8%) bleeding events in 9 patients were spontaneous, 38 (52.8%) events in 19 patients were traumatic, and 19 (26.4%) events in 11 patients were of undetermined cause: there were 51 mild, 21 moderate, and zero severe bleeding events in the prophylaxis period.
Efficacy
In the PPAS, the mean total ABR for on-demand rAHF treatment was 58.3 (95% CI 52.5, 64.7; median 53.90; range 16.7-121.7), versus 2.5 (95% CI 1.5, 3.7; median 0; range 0.0-22.1) during prophylaxis, representing a mean ABR reduction ± standard deviation of 95.9 ± 7.1%. The ratio of mean total ABR for prophylaxis/on-demand was 0.04 (95% CI 0.03, 0.06; P < .001). A consistent improvement in ABR, favoring prophylaxis, was observed for all types of bleeding events (joint and nonjoint) and etiologies (spontaneous and traumatic; Figure 2).
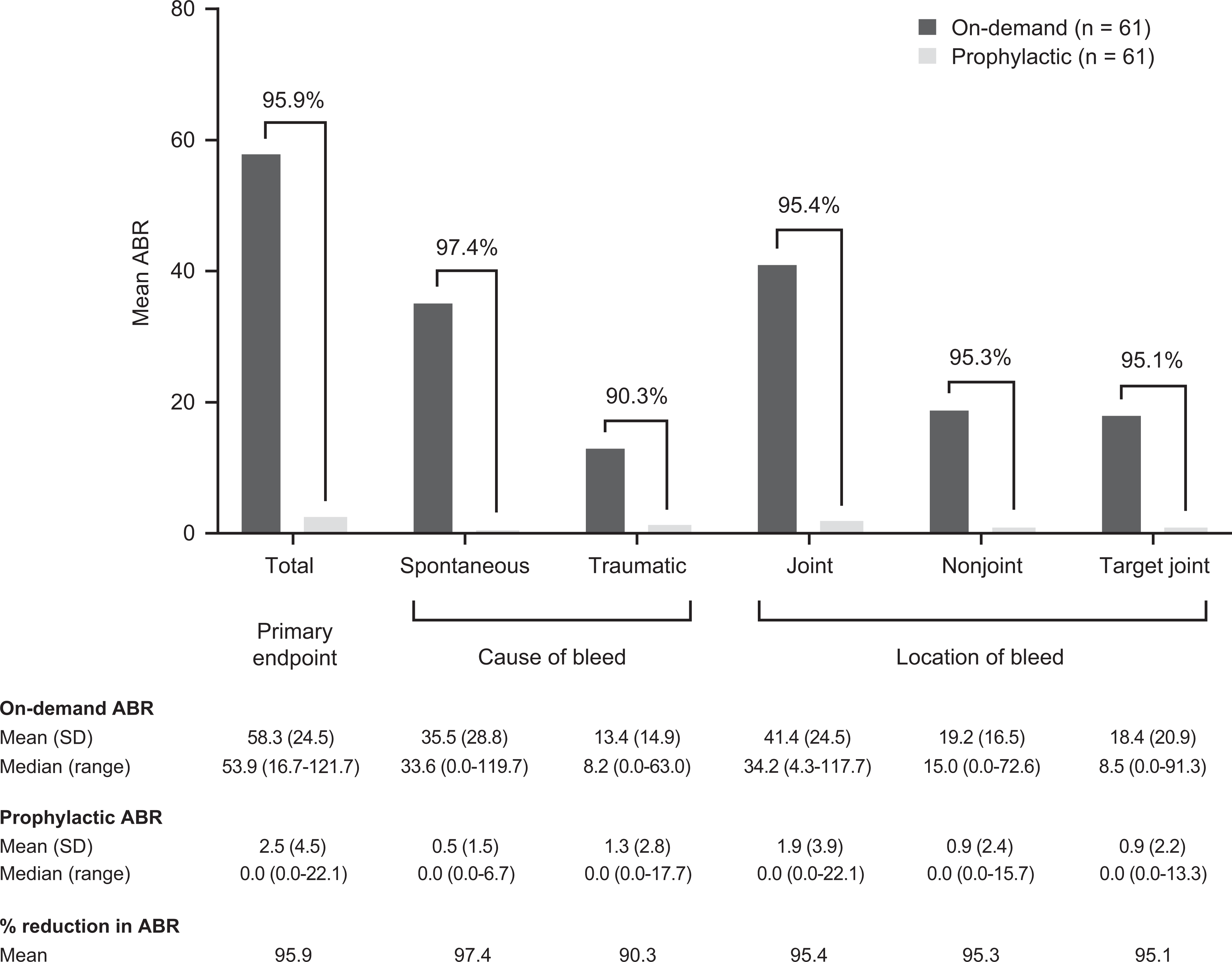
Intra-patient comparison of mean ABRs in the rAHF on-demand treatment and prophylaxis periods by bleeding subtype (PPAS). ABR indicates annualized bleeding rate; PPAS, per-protocol analysis set; rAHF, antihemophilic factor (recombinant); SD, standard deviation.
The median number of infusions required to resolve a bleeding event was 1.0 (range 0.0-5.0) during on-demand treatment and 1.0 (range 0.0-6.0) during prophylaxis. The median number of units/kg body weight to resolve a bleeding event was 31.3 (range 8.6-200.0) during the on-demand treatment period and 38.4 (range 20.8-160.0) during the prophylaxis period.
Overall, most treated bleeding events (96.1% [1746/1817]) were rated by patients/caregivers or investigators as having an “excellent” (66.9% [1215/1817]) or “good” (29.2% [531/1819]) hemostatic efficacy response, and ratings were similar between treatment regimens (Figure 3).

Hemostatic efficacy rating for the treatment of bleeding events in the PPAS. PPAS indicates per-protocol analysis set.
Exposure to rAHF
In the safety analysis set (n = 72), the median duration of treatment was similar in the on-demand period (5.7 months; range 1.0-7.8) and prophylaxis period (5.9 months; range 3.0-6.3). During these timeframes, the rAHF median cumulative dose (999.1 vs 2341.1 IU/kg), dose intensity (173.8 vs 400.3 IU/kg/month), and number of infusions received (36.0 vs 90.0) was less for on-demand treatment than for prophylaxis.
Safety
A total of 101 AEs occurred in 38 of 72 (52.8%) patients during or after infusion with rAHF in the on-demand and prophylaxis treatment periods (Table 2). During the on-demand treatment period, 4 serious AEs (1 mild and 3 severe) occurred in 4 patients. These were 4 instances of transient FVIII inhibitor development (range 0.6-1.7 BU/mL; considered related to study treatment) in 3 children and 1 adult. All FVIII inhibitor levels returned to 0.0 BU/mL at the next study visit. FVIII inhibitor development was only reported in patients with 51-150 prior EDs to FVIII. No serious AEs occurred in the prophylaxis period and there were no deaths during the study. Only 2 other nonserious AEs (fatigue and chest discomfort) in 2 patients were considered to be related to rAHF.
Incidence of AEs (Safety Analysis Set).
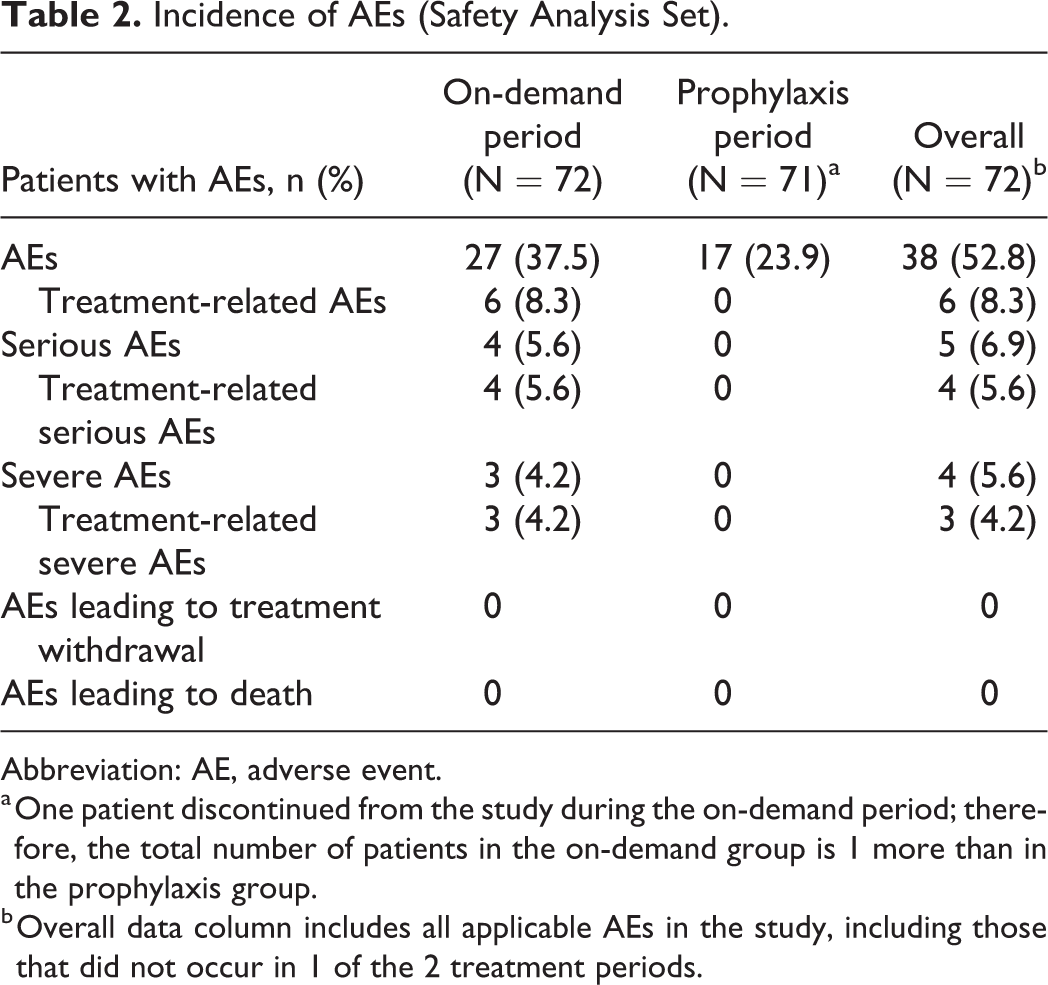
Abbreviation: AE, adverse event.
a One patient discontinued from the study during the on-demand period; therefore, the total number of patients in the on-demand group is 1 more than in the prophylaxis group.
b Overall data column includes all applicable AEs in the study, including those that did not occur in 1 of the 2 treatment periods.
Patient-Reported Outcomes
In evaluable patients, statistically significant improvements (numerical increase) over baseline in the SF-36v2 domains of physical functioning, role-physical, bodily pain, general health, social functioning, and physical component score were detected at the end of on-demand treatment and prophylaxis (Table 3). Statistically significant improvements (numerical increase) over baseline for vitality and role-emotional were detected at the end of prophylaxis only. CHO-KLAT 2.0 summary scores and VAS pain scores also improved (numerical decreases) over baseline at the end of the on-demand and prophylaxis periods. Of all patient-reported outcomes, only VAS pain scores had improved at the end of prophylaxis compared with at the end of on-demand treatment.
Scores for PRO Questionnaires Completed at Baseline, End of On-Demand Treatment, and/or End of Prophylaxis.

Abbreviations: CHO-KLAT 2.0, Canadian Haemophilia Outcomes-Kids Life Assessment Tool version 2.0; FAS, full analysis set; HRQoL, health-related quality of life; PPAS, per-protocol analysis set; PRO, patient-reported outcome; SF-36v2, 36-Item Short Form Health Survey version 2; VAS, visual analogue scale.
a In patients ≥14 years of age.
b The PPAS for HRQoL analysis was defined as the subset of the FAS HRQoL set who had completed questionnaires at the screening visit, at the end of on-demand treatment, and at the end of prophylaxis.
c Significant increase (P < .05) from baseline (ie, improvement).
d The FAS for HRQoL analysis was defined as patients who had completed questionnaires at the screening visit or at the end of on-demand treatment or at the end of prophylaxis.
e In patients <14 years of age.
f Significant decrease (P < .05) from baseline (ie, improvement).
g P < .001 versus the end of on-demand treatment.
Pharmacokinetics
Slightly higher clearance and a shorter half-life of FVIII were observed among patients aged 6-12 years versus patients aged >12 years (Table 4). Incremental recovery increased with age-group stratum.
Summary of FVIII Pharmacokinetic Parameters After Intravenous Administration of rAHF as Prophylaxis in Chinese Males With Severe or Moderately Severe Hemophilia A.

Abbreviations: AUC, area under activity-time curve; CL, systemic body clearance of drug from plasma; Cmax, maximum activity; CV, coefficient of variation; FPKAS, full pharmacokinetic analysis set; FVIII, factor VIII; IR, incremental recovery; max, maximum; min, minimum; MRT, mean residence time; PPPKAS, per-protocol pharmacokinetic analysis set; rAHF, antihemophilic factor (recombinant); t
a PK parameters measured by one-stage clotting assay.
Treatment Adherence
Patient adherence to prophylaxis was excellent, with 100% dose compliance and 98% frequency compliance. Only 1 of 72 patients was <80% adherent to prophylaxis. No significant relationship was observed between ABR and prophylaxis dose (P = 0.699) or frequency adherence (P = 0.193).
Discussion
In this sequential phase 4 study of rAHF treatment in previously treated patients with severe or moderately severe hemophilia A, total ABR was statistically significantly lower during the 6-month prophylaxis period than during the 6-month on-demand treatment period. Compared with on-demand treatment, prophylaxis significantly reduced spontaneous and traumatic bleeding events, as well as hemarthroses and bleeding into non-joint anatomical sites. Further, 7 severe bleeding events occurred during the 6 months of on-demand treatment compared with zero severe bleeding events during the 6 months of prophylaxis.
The efficacy and safety profile of rAHF observed in this phase 4 study aligns with that observed in other previously treated cohorts of patients with moderately severe to severe hemophilia A. 17 –20 The pronounced impact of rAHF prophylaxis on total ABR, relative to on-demand treatment (median 0.0 vs 53.9), is consistent with observations from a global prospective randomized phase 4 clinical study of rAHF in 66 previously treated patients with moderate or severe hemophilia A (median ABR 1.0 for prophylaxis vs 43.9 for on-demand treatment). 20 In a smaller and shorter sequential-treatment study in 30 previously treated Chinese children with severe hemophilia A, 12 weeks of prophylaxis with a sucrose-formulated rFVIII reduced the ABR relative to a prior 12-week period of on-demand treatment (median 0.0 vs 57.5). 26
The trend toward higher clearance and lower half-life observed in patients aged 6-12 versus >12 years is consistent with PK profiles observed in other FVIII studies. 18 The hemostatic efficacy ratings for bleeding events treated with rAHF during the on-demand and prophylactic treatment periods were similar, consistent with previous rFVIII data. 26 The number of infusions and units/kg body weight required to resolve a bleeding event were similar for both regimens and, as expected, rAHF consumption was higher in the prophylaxis period than in the on-demand treatment period.
Data pertaining to the 4 cases of transient FVIII inhibitor development during the on-demand treatment phase should be interpreted with due consideration to the heterogeneous population, who had a range of prestudy exposures to FVIII. Generally, children and adults require FVIII EDs of >50 and >150, respectively, to be regarded as previously treated (and at less risk of inhibitor development). 27,28 Some adults and adolescents in this Chinese cohort had ≤150 EDs and did not fully meet the criteria for PTP (including the 1 adult who developed a transient high-titer inhibitor) and therefore were regarded as high risk. Nevertheless, the duration of this study was sufficient to evaluate efficacy and safety beyond the initial 50 EDs, the period of highest risk for inhibitor development in children. 27 rAHF was well tolerated across all age groups and no new safety signals emerged when compared with the 2015 study of rAHF by Shapiro et al. 18
After only 6 months of on-demand treatment and 6 months of prophylaxis with rAHF, patients had statistically and clinically significant improvements in several HRQoL domains during each treatment period. Despite a reduction in ABR after the switch from on-demand treatment to prophylaxis, there was no accompanying improvement in scores from the HRQoL instruments (SF-36v2 and CHO-KLAT), although there was a significant improvement in VAS pain scores at the end of prophylaxis.
Conclusions
In the past decade, health insurance and drug regulatory agencies in China have been promoting the use of prophylaxis for the treatment of hemophilia, 13 as prophylaxis use in China was shown to be lower than several other countries based on world-wide survey results reported in 2016. 10 Although updated, real-world data are warranted to obtain the current treatment patterns utilized in China, the clinical study results reported herein provide evidence that support the use of rAHF prophylaxis in Chinese patients.
The efficacy and safety of rAHF in previously treated Chinese patients with severe or moderately severe hemophilia A appear to be similar to those described in other rAHF studies. The reduction in bleeding (total ABR) provided by prophylaxis with rAHF indicates that this management approach is a more effective alternative to on-demand treatment in this population. This reduction in ABR with prophylaxis vs on-demand rAHF treatment was dramatic, although, there are some patients who still had high ABRs despite receiving standard prophylaxis. Further personalization of the prophylactic treatment regimen may help improve bleed outcomes in these patients.
Footnotes
Authors’ Note
All authors contributed to the study concept and design. YZ, YH, JJ, XZ, XW, RW, DW, RY, FY, and QH were clinical trial investigators and recruited and treated patients. All authors participated in reviewing and interpreting the data and reviewing the manuscript. All authors read and approved the final version of this manuscript. The datasets, including redacted study protocol, redacted statistical analysis plan, and individual participants’ data supporting the results reported in this article will be made available within 3 months from initial request, to researchers who provide a methodologically sound proposal. The data will be provided after its de-identification, in compliance with applicable privacy laws, data protection, and requirements for consent and anonymization. Data requests should follow the process outlined in the Data Sharing section on: ![]() . The study protocol, informed consent form, and all amendments were reviewed and approved by the relevant ethics committees prior to implementation. All patients (or their legally authorized representatives) provided written informed consent before enrollment.
. The study protocol, informed consent form, and all amendments were reviewed and approved by the relevant ethics committees prior to implementation. All patients (or their legally authorized representatives) provided written informed consent before enrollment.
Acknowledgments
The authors would like to thank all patients and their caregivers who took part in the study. Medical writing and editorial support was provided by Malcolm Darkes, PhD, MPS, and Daniella Babu, PhD, of Excel Medical Affairs (Fairfield, CT, USA) and was funded by Baxalta US Inc., a Takeda company, Lexington, MA, USA.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: YZ has received speaker and consultancy fees from Bayer, Novo Nordisk, Pfizer, and Takeda (Shire). RY has received speaker/consultancy fees from Bayer, Novo Nordisk, Pfizer, Roche, and Takeda (Shire). HF is an employee of Shire BioScience (Shanghai) Co., Ltd., a Takeda company, Shanghai, China. WE is an employee of Baxalta Innovations GmbH, a Takeda company, Vienna, Austria and an owner of Takeda stock.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the sponsors, Baxalta US Inc., a Takeda company, Lexington, MA, USA, and Baxalta Innovations GmbH, a Takeda company, Vienna, Austria.
