Abstract
Objective:
This study explores the efficacy of recombinant factor VIII (rFVIII) low-dose prophylaxis in Chinese pediatric patients with severe hemophilia A from the Retrospective Study in Chinese Pediatric Hemophilia A Patients With rFVIII Contained Regular Prophylaxis (ReCARE) population.
Methods:
This is additional analysis of the multicenter, retrospective ReCARE study, in which the annual bleeding rate (ABR), annual joint bleeding rate (AJBR), and safety of >12-week, low dose (10
Results:
A total of 57 patients (median age: 8.2 [0.4-17.3] years) from the ReCARE study receiving primary (n = 3), secondary (n = 21), and tertiary (n = 33) prophylaxes were included. Low-dose prophylaxis had significant bleeding reduction in all 3 groups compared to the baseline (S = 408.5, P < .001), with median ABR rates of −4.0 (−8.0 to −3.1), −4.0 (−24.0 to 8.0), and −13.9 (−110.6 to 20.6) in the primary, secondary, and tertiary groups, respectively, with a significant difference between the secondary and tertiary groups (P = .008). Median AJBR reduction rates were −2.3 (−6.3 to 8.4) and −14.9 (−61.5 to 19.1) in the secondary and tertiary groups, respectively. But there was no significant difference in AJBRs between the secondary and tertiary groups (P = .061), which was related to damaged joint status. Hence, longer prophylaxis was associated with better prevention of joint bleeding (P = .024).
Conclusion:
Despite significant ABR/AJBR reduction in all 3 groups, the efficacy of the primary prophylaxis was better than the secondary and tertiary prophylaxes.
Keywords
Introduction
The prevalence rate of hemophilia A (HA) in China is remarkably high, with an estimate of 65 000 to 130 000 cases in 2011.
1
As per the Chinese National Hemophilia Registry, the rate of HA in children <14 years of age is as high as 52.3%.
2
Severe HA is manifested by spontaneous or traumatic joint bleeding episodes, especially in the index joints followed by muscle bleeds, due to deficient factor VIII (FVIII: C <1 IU/dL) activity. Lack of appropriate treatment for recurrent bleeding results in intra-articular changes that culminate in progressive joint destruction, irreversible arthropathy, chronic pain, and overall deterioration of disability. Loss of opportunities, school absenteeism, frequent hospital visits, poor quality of life (QoL), stress, and economic burden are other drawbacks associated with the disease.
3
The World Health Organization, World Federation of Hemophilia (WFH), and many national medical/scientific organizations recommend prophylaxis as the first-choice gold standard of treatment.
4
Early prophylaxis is found to be superior to on-demand treatment in reducing the risk of arthropathy and fosters better joint outcomes and improved QoL.
4
–6
The WFH standardized the definitions and standards of prophylaxis and classified it into primary, secondary, and tertiary stages according to the age of onset and index joints status. Prophylaxis initiated before 2 years of age has been reported to provide better outcomes than that initiated at 3 to 5 years or 6 to 9 years of age.
6
–9
Recombinant antihemophilic factor (Kogenate FS, Bayer Healthcare, Whippany, NJ, USA), the first marketed recombinant factor VIII (rFVIII) in China since 2007, is the routinely used prophylactic drug to prevent or reduce the incidence of hemorrhages in adults (25 IU/kg thrice a week) and children (25 IU/kg every alternate day) with HA as well as to reduce the risk of joint damage in children without preexisting joint damage.
10
Recent economic and medical advancements have resulted in increasing the number of children with HA receiving prophylaxis. Furthermore, there is an increase in the incidence of arthropathy with increasing age.
11
Nevertheless, the rate of hemophilia control is limited in China because of limited economy and inadequate treatment availability. Several studies considered very low-dose prophylaxis (10 IU/kg twice a week) as a feasible option
1,12,13
although the outcomes were limited by the number of patients and the focus being on just tertiary prophylaxis.
12
–14
To date, sufficient data do not exist regarding the comparison of treatment effect of low-dose prophylaxis among various prophylactic stages (primary, secondary, and tertiary prophylaxes). Considering the high proportion of HA in China, inadequate control with FVIII prophylaxis, and associated economic concerns with the therapy, the authors wanted to explore the effect of low-dose prophylaxis on HA in China. Thus, ReCARE was the first retrospective study conducted across 12 hemophilia treatment centers in China to determine the real-life choice of rFVIII as regular prophylactic treatment (NCT02263066) from November 2007 to May 2013. The study revealed that most patients had been on low-dose prophylactic regimen (10
Materials and Methods
Study Design and Patient Population
This multicenter, retrospective, phase IV, noninterventional study is an additional supplemental analysis of the ReCARE
15
conducted in china to evaluate the efficiency of rFVIII low-dose prophylaxis in reducing bleeding rates of hemophilia and/or to describe the treatment condition of patients during the usage of rFVIII-FS. The trial was approved by independent ethic committees or institutional review boards of all participating sites after the approval of signed informed consents from all patients or their legal guardians, and the study protocol conformed to the declaration of Helsinki and its subsequent revisions. Patients were included in the study if they had severe HA (defined as the presence of FVIII: C<1 IU/dL, except von Willebrand disease and acquired coagulation factor VIII deficiency
16
), received regular rFVIII prophylaxis (administration of ≥2 injections per week, accounting for ≥80% of the prophylactic period
17
) for more than 12 weeks, received low-dose (10
Definitions of the 3 Prophylactic Groups
All patients were divided into primary, secondary, and tertiary prophylactic groups based on the index joints status. The primary prophylaxis was referred to the treatment initiated in the absence of evident joint disease and before the second clinically recognized joint bleed, and before 3 years of age. The secondary prophylaxis referred to the treatment initiated after ≥2 bleeds but before the onset of joint disease as documented by physical examination and/or imaging studies, and/or >3 years of age. Tertiary prophylaxis was the treatment started after ≥2 bleeds or the onset of joint disease at any age. 6 Baseline data were obtained 6 months prior to the initiation of prophylactic therapy.
Study Outcomes
Primary outcomes included the reduction in annual bleeding rate (ABR) and annual joint bleeding rate (AJBR) during the 3 prophylactic periods and their comparison to the baseline. Adverse drug reactions and inhibitor levels were monitored throughout the prophylaxis period and were measured as secondary outcomes.
Statistical Analysis
The SAS-based statistical software JMP Clinical (version 5; SAS Institute Inc, Cary, North Carolina) was used for data analysis. The age, age at first diagnosis, age at first FVIII exposure, duration of diagnosis to starting regular rFVIII prophylactic treatment, ABR, and AJBR were described by median (min-max) for not meeting normal distribution. Factor VIII consumption was described by mean ± standard deviation (SD). Statistical analyses of baseline data (age, age at first diagnosis, age at first FVIII exposure, duration of diagnosis to starting regular rFVIII prophylactic treatment, first regular prophylactic treatment time, and FVIII consumption) among the 3 groups were carried out by Kruskal-Wallis rank sum test. The paired Wilcoxon rank sum test was used to compare the difference between the baseline and the treatment period of ABR and AJBR. The differences between the secondary and tertiary prophylactic groups in ABR and AJBR, considering the unbalance of the baseline in the 2 groups and nonnormality of data, were carried out by rank-based analysis of covariance (CANOVA). Differences were considered as statistically significant when P < .05.
Results
The ReCARE study included 183 patients who underwent regular prophylactic treatment from 2007 to 2013. Two patients were excluded due to protocol deviation as one of them received regular rFVIII prophylaxis for less than 10 weeks, and another patient was >18 years of age. Finally, 181 patients were included of whom 68 were excluded due to the unavailability of regimen records (n = 14); received non low-dose prophylaxis <10 IU/kg/wk, which cannot be regarded as prophylactic replacement therapy (n = 11); received moderate-dose prophylaxis of 30 to 75 IU/kg/wk (n = 42); and standard prophylaxis of ≥75 IU/kg/wk (n = 1). Overall, there were 113 (72.4%) patients who had received low-dose prophylaxis (10
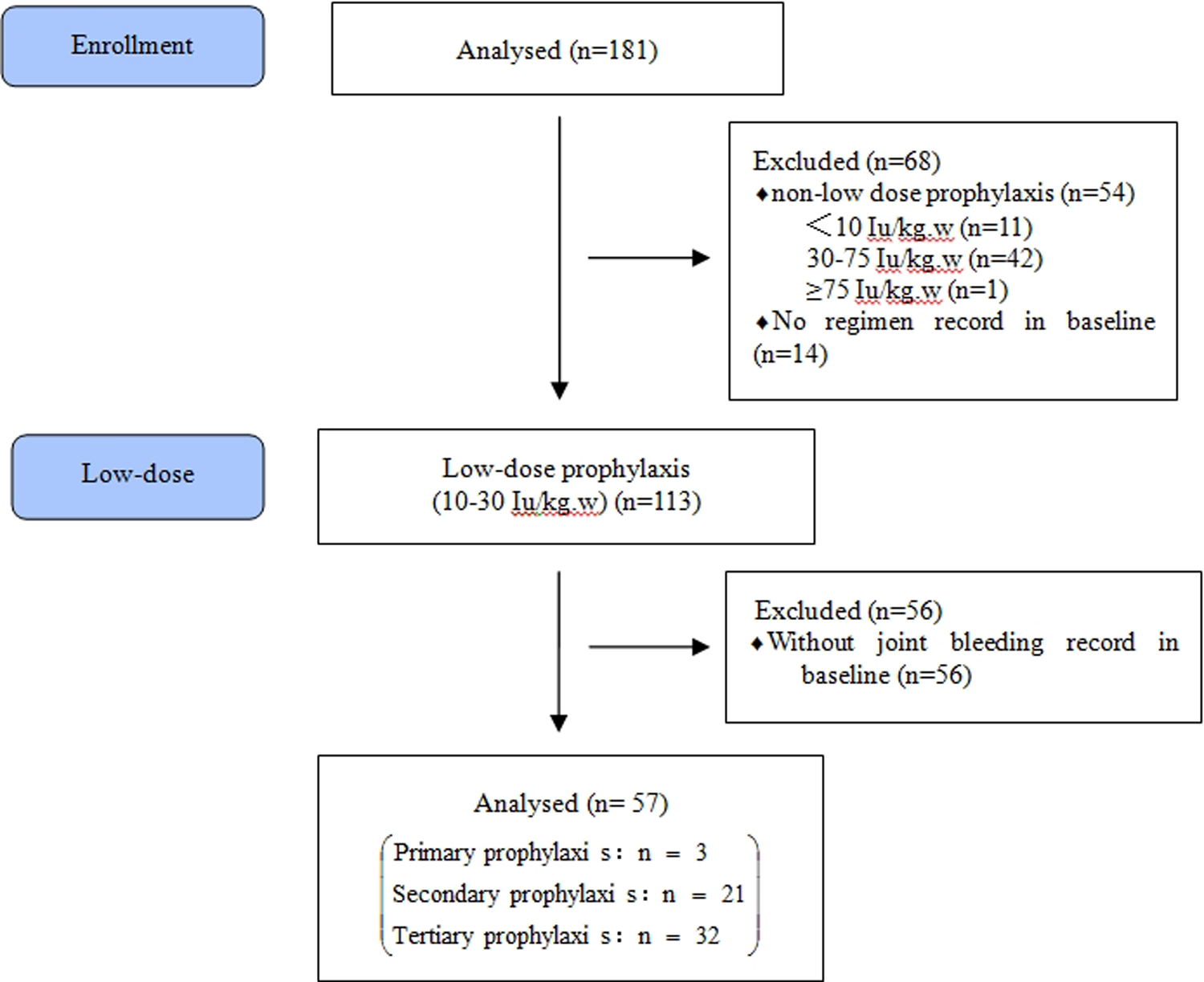
Study flow chart.
Clinical baseline data are reported in Table 1. No statistical significance was observed between the first regular prophylactic treatment time among the 3 groups (P = .322), indicating an equilibrium distribution of treatment duration between the groups; furthermore, there was no statistical significance between the ages of first diagnosis between the 3 groups (P = .133). However, there were statistical significances in the age of first rFVIII exposure (P = .045), duration of diagnosis to start regular rFVIII prophylactic treatment (P = .002), and FVIII consumption (P = .030). All 3 groups started FVIII and regular prophylaxis at different ages—the primary group started treatment at a young age, and a delay in treatment transferred children with hemophilia to tertiary prophylaxis. Furthermore, the rFVIII consumption in the 3 groups was 1157.9 ± 219.7, 1063.5 ± 332.0, and 1303.8 ± 312.1 (IU/kg/y; data not shown), indicating that the factor consumption in low-dose prophylaxis was significantly less than moderate- and high-dose prophylaxis.
Baseline Characteristics of 57 Patients in the Study.
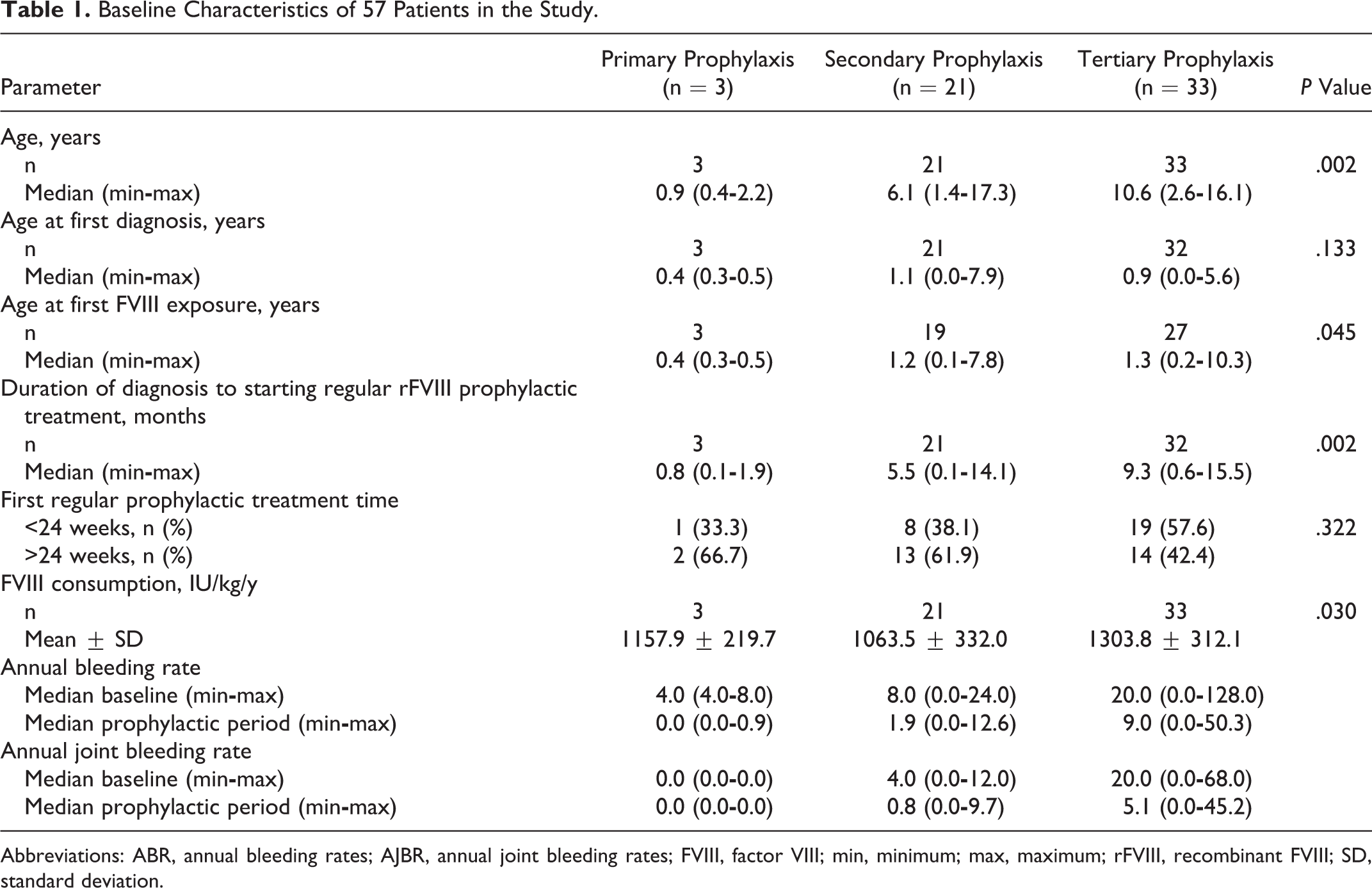
Abbreviations: ABR, annual bleeding rates; AJBR, annual joint bleeding rates; FVIII, factor VIII; min, minimum; max, maximum; rFVIII, recombinant FVIII; SD, standard deviation.
Annual Bleeding Rate
Wilcoxon matched-pairs signed rank test revealed a significant difference in ABR between the prophylactic period and baseline (S = 408.5, P < .001). Low-dose prophylaxis had a significant effect on bleeding at different prophylactic stages. Annual bleeding rate was lesser in patients who were receiving regular low-dose prophylaxis than the baseline (primary prophylaxis: 0 vs 4.0 [4.0
Rank-Based CANOVA to ABR and AJBR Between Secondary and Tertiary Prophylactic Groups.a,b,c
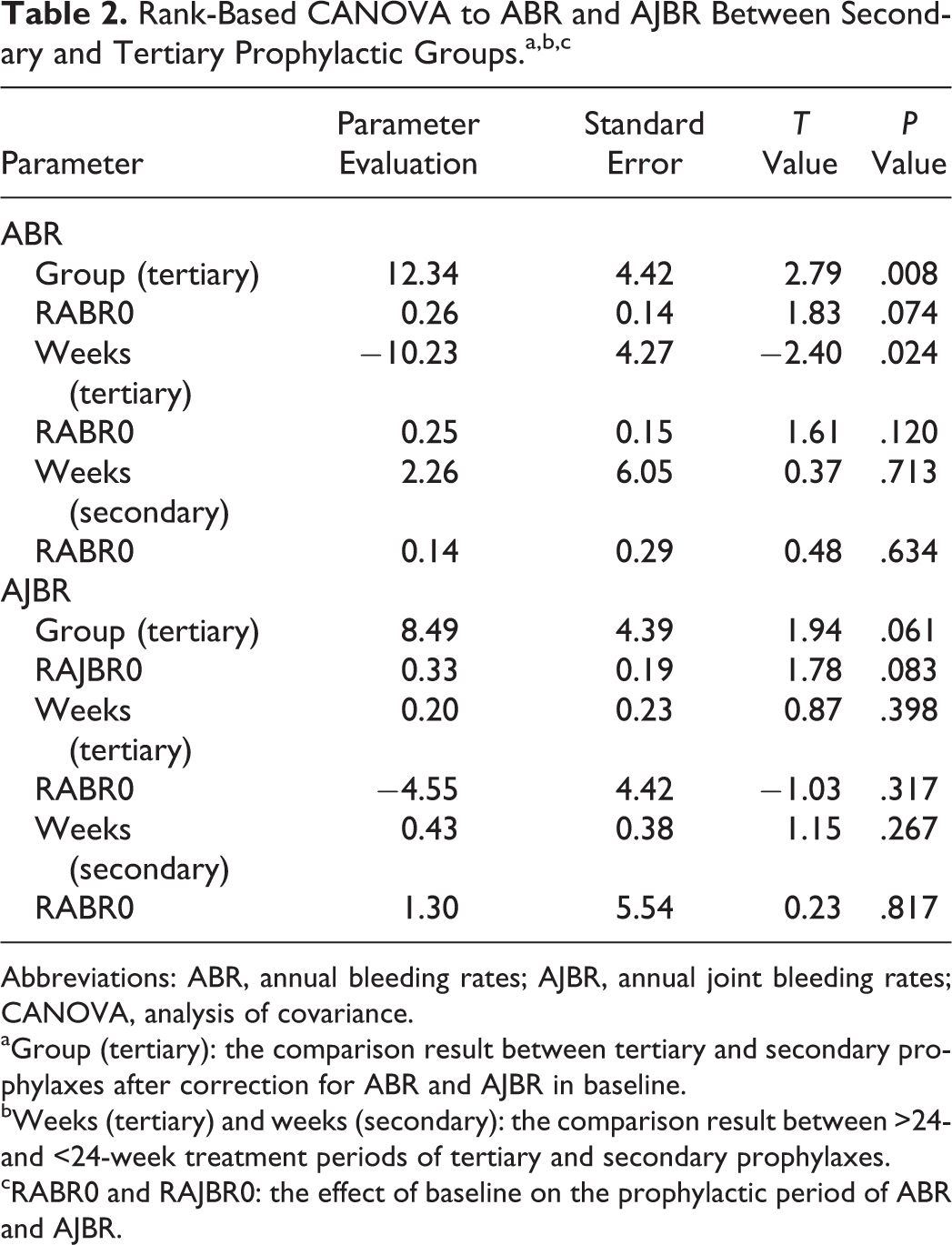
Abbreviations: ABR, annual bleeding rates; AJBR, annual joint bleeding rates; CANOVA, analysis of covariance.
aGroup (tertiary): the comparison result between tertiary and secondary prophylaxes after correction for ABR and AJBR in baseline.
bWeeks (tertiary) and weeks (secondary): the comparison result between >24- and <24-week treatment periods of tertiary and secondary prophylaxes.
cRABR0 and RAJBR0: the effect of baseline on the prophylactic period of ABR and AJBR.
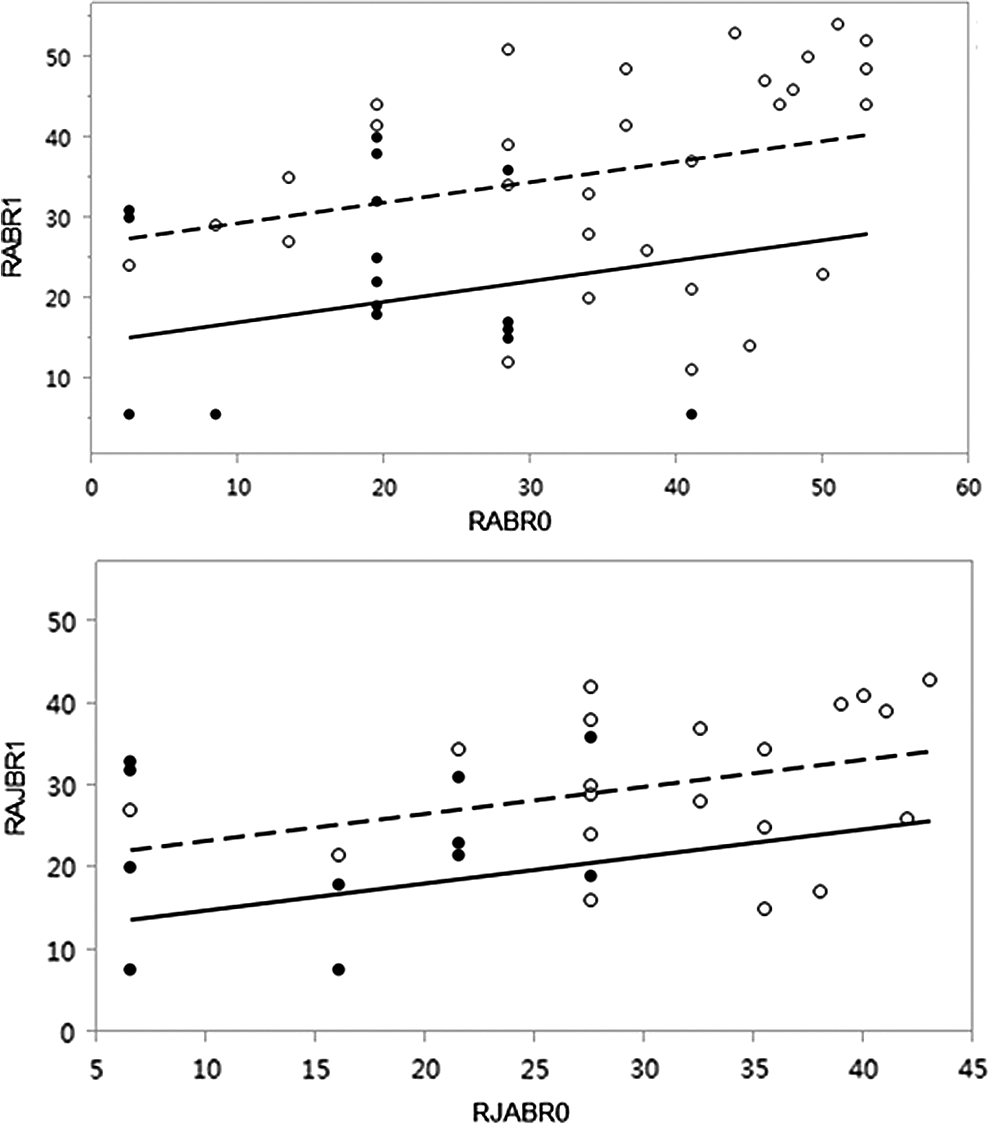
Rank-based CANOVA analyzing ABR and AJBR between the secondary and tertiary prophylactic groups. The difference between the secondary and tertiary prophylactic groups in ABR and AJBR were carried out by rank-based CANOVA for the unbalance of the baseline and nonnormality distribution, which means the data in the baseline and during prophylactic period were ranked first and then CANOVA was performed for the ranked values. RABR0 and RAJBR0 mean the rank of ABR and AJBR in the baseline; RABR1 and RAJBR1 mean the rank of ABR and AJBR during the prophylactic period. The closed symbols and broken line indicate the tertiary group, whereas the open symbols and straight line indicate the secondary group. ABR indicates annual bleeding rates; AJBR, annual joint bleeding rates; CANOVA, analysis of covariance.
Annual Joint Bleeding Rates
In the primary prophylactic group, there were no joint bleeding events during the baseline and prophylactic treatment. On comparing the baseline and prophylactic period results of ABR using Wilcoxon matched-pairs signed rank test (S = 237.0, P < .001), the results were similar to ABR results. During the regular low-dose prophylaxis, AJBR was less than the baseline throughout the primary, secondary (4.0 [0.0
Treatment Periods (>24 Weeks vs <24 Weeks)
The primary, secondary, and tertiary prophylaxes were analyzed between the treatment periods of >24 and <24 weeks. There was only significant difference in ABR between the secondary and tertiary prophylaxes (P = .024). None of the patients reported inhibitor levels or adverse events during the entire prophylactic period.
Discussion
Early prophylaxis has better outcomes in preventing life-threatening and spontaneous bleeding and maintaining joint status with improvement in the QoL compared to episodic treatment.
6,18
Children receiving FVIII therapy as a routine preventative treatment demonstrated 8 times fewer joint bleeds in a year than those receiving enhanced on-demand treatment.
19,20
In addition, initiating early prophylaxis may help prevent inhibitor formation.
21
Based on the administration guidelines of standard FVIII prophylaxis, FVIII must be infused at 25 to 40 U/kg every 48 hours to maintain FVIII trough levels at more than 1 IU/dL; however, hemophilia medical professionals have been attempting new treatment regimens and individualized prophylaxis such as moderate-dose prophylaxis (10
The present study was a further analysis of the ReCARE trial. 15 Data from the ReCARE trial revealed that low-dose prophylaxis accounted for a major proportion of patients (72.4%; n = 113). The number of patients receiving median- and standard-dose prophylaxes was lower than that receiving low-dose prophylactic treatment, which represents the current status of FVIII prophylactic therapy in China. Furthermore, patients in the ReCARE study received regular rFVIII prophylaxis for more than 12 weeks. Therefore, analyzing the data from the ReCARE trial will help in understanding the effect of using low-dose rFVIII as prophylaxis (primary, secondary, and tertiary prophylaxes) in Chinese children with severe HA across geographically well-distributed centers in China.
Because high cost of the primary prophylaxis is a major limitation in developing countries, most patients from China and other developing countries prefer to receive low-dose prophylaxis. Multiple studies have evaluated the effect of low-dose prophylaxis on joint function and the QoL compared to on-demand treatment in children with hemophilia. 1,13,21 –27 Few studies in China have shown better effect of low-dose prophylaxis on joint bleeding compared to on-demand treatment. 1,13 A control study of low-dose secondary prophylaxis (currently defined as tertiary prophylaxis) and on-demand treatment by Wu et al revealed that both long- and short-term low-dose tertiary prophylaxes can significantly improve joint bleeds and functions compared to on-demand treatment. 11 Another study revealed a 6-fold (85%) reduction in the risk of joint damage in children with hemophilia receiving prophylaxis (25 IU/Kg every other day) than those receiving intensive on-demand treatment. 28 In their study, Nilsson et al demonstrated that patients receiving a prophylactic regimen of 10 to 20 IU/Kg every 3 to 5 days initiated at an age of 3 to 13 years had only 1.6 to 16 joint bleeds/year with a radiologic score of 0 to 41. 29 Prophylaxis has shown minimal/no bleeds at all if initiated at an age of less than 3 years. 1,7,30 In a recent study by Verma et al, it was found out that very low-dose FVIII prophylaxis (10 IU/kg twice a week) was effective in the joint and overall bleeding preventions, in addition to being cost-effective. 27 In the current study, there was an age-dependent trend of prophylaxis regimen in which the youngest patients had the primary prophylaxis followed by the secondary and the oldest children had the tertiary prophylaxis, which means children diagnosed early with hemophilia were on primary prophylaxis and those who had delayed diagnosis due to varied reasons had ultimately turned to tertiary prophylaxis after the onset of joint status.
In the current study, after at least 12 weeks of observation, we found that low-dose prophylaxis could significantly reduce bleeding times for each of the groups, with the highest reduction being observed in the primary prophylactic group, indicating that primary prophylaxis also prevented bleeding from further aggravation of the disease. The median ABR of the primary prophylactic group was 0 times (range: 0
It is noticeable that ABR and AJBR were positively correlated in the baseline and prophylactic period with low-dose prophylactic treatment. Severe lesions will have poor prognosis, and therefore, bleeding and joint disease cannot be fully improved. This study strengthens the findings from previous studies that concluded that early diagnosis and initiation of prophylaxis, especially the primary and secondary prophylaxes, can be more effective in the prevention of bleeding in children with hemophilia and will delay further progress of the joint disease. Furthermore, this study also supports a multinational study involving 477 patients, in which prophylaxis was associated with significant reduction of arthropathy compared to on-demand treatment, which was directly associated with decreased direct and indirect health-related costs and significant reductions in the number of hospital admissions and school absenteeism with a favorable psychological development in hemophilia children. 31 Furthermore, we inferred that only longer treatment period resulted in lesser bleeding in the tertiary prophylactic group, representing that longer prophylactic duration will have better prognosis in already established target joint status. Hence, as standard therapy, prophylaxis should be given regularly as early as possible before the onset of joint disease. Furthermore, we also observed that FVIII consumption was significantly less in low-dose prophylaxis regimen compared to moderate-dose and high-dose prophylaxis regimens, which meant that low-dose prophylaxis could have an economic as well as an acceptable short-term outcome.
Therefore, low-dose prophylaxis will have a direct impact on the cost factor and will make treatment an affordable option in developing countries, thus yielding better control rates of HA. Although the total cost of high-dose factor prophylaxis is 66% higher, the outcomes are only slightly better than intermediate-dose prophylaxis. 27 We suggest for low-dose prophylaxis, patients with severe HA in developing countries could receive 10 to 15 IU/KG, 1 or 2 times per week to avoid frequent bleeding to keep their normal QoL as much as possible. Our study has certain limitations. First, the retrospective design of the study demands future confirmation of the findings by prospective, randomized, large-group studies. Second, data collection from different centers was inconsistent and there was a large amount of missing data. Third, data of only 57 patients were analyzed. Evaluation for joint structures using ultrasound, X-ray, and MRI test for index joints and the evaluation of function using tools such as Hemophilia Joint Health Score (HJHS) were not available because of the retrospective nature of the study. One of the major limitations was that data of only 3 patients in the primary prophylactic group were analyzed due to the lack of awareness of Chinese families about hemophilia resulting in late initiation of prophylaxis, thereby resulting in increased proportion of the secondary and tertiary prophylaxes, which suggested that we need to strengthen public awareness and education as one of the aims in the future. Furthermore, the prophylactic period in the present study was short and further validation is needed.
Conclusion
Low-dose prophylaxis significantly reduced the frequency of bleeding and joint bleeding. Initiating early prophylaxis for a longer duration will result in better prevention of joint damage. However, further randomized prospective research is warranted to support this conclusion, including assessment of the joint structures and functions by assessment tools in a longer prophylactic duration study cohort.
Footnotes
Acknowledgments
The authors thank Dr Amit Bhat and Miss Navya Reddy from Indegene Pvt Ltd, Bangalore, India for providing medical writing assistance to this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
