Abstract
Establishing an appropriate prophylaxis regimen for children with hemophilia is a critical challenge in developing countries. Barriers including availability and affordability, catheter-related complications, and inhibitor development risks have led to the introduction of new tailored prophylaxis regimens in different countries. This study emphasizes on the benefits of the Iranian low-dose escalating prophylaxis regimen in a Hemophilia Comprehensive Care Center in Iran. Referred patients with hemophilia less than 15 years of age, who were subject to prophylaxis regimen, are studied retrospectively. A once-weekly prophylaxis regimen of 25 IU/kg was started for the patients primarily. Their prophylaxis regimen was changed to 25 IU/kg twice a week and then 3 times a week when they experienced 3 joint bleedings, 4 soft tissue bleedings, or a 1 life-threatening bleed without a specific trauma history. Overall, 25 patients with severe hemophilia and at least 6-month history of on-demand (OD) treatment were studied. A mean of 1754 IU/kg/yr of coagulation factors, used for OD and prophylaxis purposes, was sufficient to decrease the mean annual bleeding rate (ABR) to 1.86 after prophylaxis. It also reduced the mean hospitalization days and the mean number of target joints to 0.24 and 0.16, respectively. Overall, 19 (76%) patients were continuing their once-weekly regimen at the end of the follow-up. None of the patients needed 3-times-a-week regimen or central venous catheterization and none developed inhibitors in the follow-up. Benefits of the Iranian low-dose escalating prophylaxis regimen prove equal to some of the previous 3-times-a-week prophylaxis regimens in reducing the ABR and hospitalizations.
Keywords
Introduction and Objective
The superiority of prophylaxis over on-demand (OD) or episodic treatment protocols in severe hemophilia has long been proven. 1 Patients with severe hemophilia, with a factor activity level (factor VIII [FVIII]: C) of less than 1%, are susceptible to 20 to 50 bleeding events in a year, which leads to serious disabilities in early adulthood. 2 Prophylaxis regimens were introduced with the rationale of keeping the factor activity level above 1% and therefore converting the severe hemophilia to milder forms with less musculoskeletal disabilities. 3 Initial prophylaxis protocols included 2- or 3-times-a-week infusions, considering the half-life of the coagulation factor concentrates (8-12 hours). 4 However, establishing these prophylaxis regimens nationally required thousands of factor units per kilogram in a year, which makes it quite a challenge for many countries. Moreover, these 3-times-a-week intravenous (IV) infusions brought about limitations, especially in children with limited IV access.
To conquer these challenges in Canada, an escalating prophylaxis regimen was introduced containing 50 IU/kg once weekly with step-up criteria for unexpected bleeding events. Surprisingly, 40% of patients did not need any change in their dosage or frequency of factor infusions after 5 years of follow-up, which could not be explained by the previous rationale of keeping the factor level above 1%. 5 Later, a French prophylaxis regimen was developed containing 50 IU/kg once weekly with milder step-up criteria compared to the Canadian one, which was also successful. 6 Another once-weekly prophylaxis regimen (25 IU/kg once a week) was developed for the first 20 to 50 exposure days to the coagulation factor in previously untreated patients, which reduced inhibitor development rates from 47% to 1% in a follow-up of about 175 exposure days. The primary aim of this regimen was to induce tolerance, and infusion frequency increased back to classic regimen following the tolerating period. 7 Although these regimens were primarily designed as a prophylaxis starting protocol, they interestingly certified as the optimum regimens for many individuals. In a review of 62 hemophilia care centers in the United States, about one-third (20 centers) of them were initiating their hemophilia A prophylaxis regimen with 25 to 50 IU/kg FVIII once weekly contrary to national recommendations to avoid central venous catheterization (CVC) and inhibitor production. 8 Moreover, from 21 centers participated in the European Paediatric Network for Haemophilia Management Registry, 8 centers started their prophylaxis regimen with once- or twice-a-week regimens, with 55% of their patients receiving infusions once in a week. Infusion frequency for these patients was increased based on their bleeding patterns. 9
Altogether, high burden of hemophilia, CVC complications, inhibitor production, and other barriers have pushed different countries toward lower frequency and dosage of prophylaxis regimen along with phenotype-based escalating criteria. These lower doses of prophylaxis are beneficial in reducing the annual bleeding rates (ABRs) and the following musculoskeletal and psychosocial impairments to a great extent, which cannot be explained by the theory of keeping factor trough levels from falling below 1%. 5 Moreover, less frequent infusions and CVC in these prophylaxis regimens are expected to improve the quality of life, especially in children with severe hemophilia. However, these low-dose protocols are still in a trade-off with the risk of unprovoked intracranial hemorrhages.
Based on previous successful experiences on escalating prophylaxis, especially the Canadian Hemophilia Primary Prophylaxis Study, and individual centers experience in Iran, a new low-dose escalating prophylaxis regimen was proposed to the Ministry of Health and Medical Education. Later on, this new regimen was nationally recommended to hemophilia comprehensive care centers in 2014. This new low-dose prophylaxis regimen was most suitable for Iran with the mean factor use per capita of more than 2 and 50% of its patients with hemophilia (PWH) identified. 10,11 This study is a comparison between the previous OD treatment and the new prophylaxis regimen as the primary report of a 3-year single-center experience, which was the birthplace of the new regimen.
Materials and Methods
All the patients with severe hemophilia (FVIII: C or FIX: C ≤ 1%) with an age of less than 15 years who had at least 6 months of OD treatment to compare with the prophylaxis regimen and yet have not developed inhibitor were eligible for this study. Patients receiving primary prophylaxis (less than 2 soft tissue or joint bleeds and less than 2 years old who have not yet developed any joint damage) were not included since they had no history of OD treatment to compare the results with. The patient recruitment started in March 2013 and ended in January 2015 in the Hemophilia Comprehensive Care Center of Mofid Children Hospital (Tehran, Iran).
The study has been done under the supervision and inspection of Ministry of Health and Medical Education as the sponsor and regular authority of the study, which has strict regulations in factor allocation. Informed consent was obtained from parents before their participation in the prophylaxis program. Detailed data of bleeding events and factor use in the year before participating in the prophylaxis program and follow-up of their prophylaxis experience were documented by the physician and parents through home treatment forms under the supervision of the center. Patients were followed up for 1 to 2 years after the beginning of prophylaxis. Clinical scores were assessed by physicians of the center based on the WFH Physical Exam Score (Gilbert Score). Annual bleeding rates, hospitalization days, target joint numbers, clinical scores, and mean coagulation factor use per kilogram per year for each patient were derived from hospital records as primary outcomes. Descriptive data were shown as mean ± standard deviation and ranges. To compare the outcome measures before and after prophylaxis, data were analyzed with dependent t test. In all the comparisons, a P value of less than .05 is considered significant. All the analyses were done using IBM SPSS Statistics version 20 software.
Iranian Prophylaxis Protocol
The Iranian prophylaxis protocol is designed for patients with severe hemophilia A and B without inhibitors who are under the age of 15. Patients become eligible to receive the initial dose of prophylaxis after they have experienced 2 or more bleeds into soft tissue or joint and yet have not developed an arthropathy or if they reached 2 years of age. All patients are required to have their negative inhibitor levels documented 2 times in the last 3 months. All of the PWH A and B receive 25 IU/kg and 30 to 50 IU/kg once a week, respectively. If they experience 3 joint bleedings, 4 soft tissue bleedings, or 1 major bleeding, their infusion frequency is increased to the next step. Bleedings can occur in different joints and places of the body documented in home treatment forms. After reaching the age 15, the prophylaxis regimen will be discontinued (Table 1). The Iranian prophylaxis protocol exclusively started on plasma-derived FVIII and FIX concentrates. Recombinant FVIII concentrates, Xyntha antihemophilic factor (Pfizer, USA), and Kogenate (Bayer, Germany) were introduced to the Iran pharmaceutical market in the final 6 months of the study and were given to the previously treated patients.
Iranian Prophylaxis Regimen Schedule and Criteria.
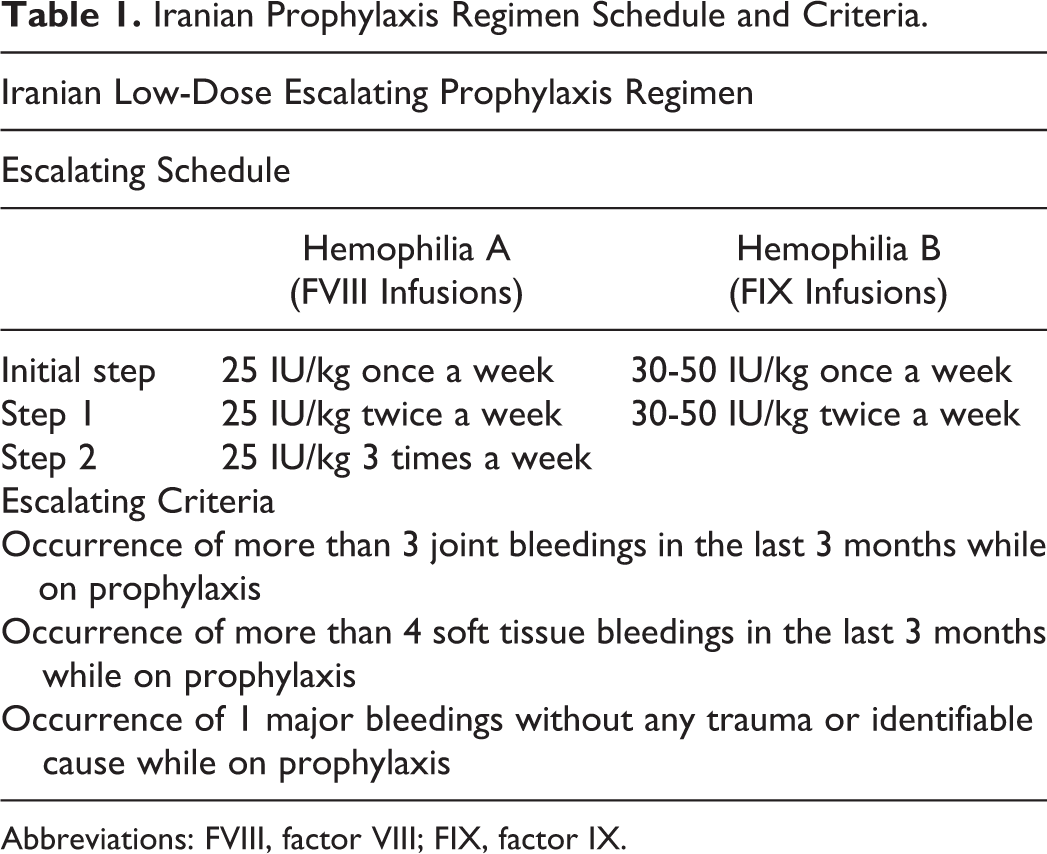
Abbreviations: FVIII, factor VIII; FIX, factor IX.
Results
Overall, 25 patients with severe hemophilia, including 20 PWH A and 5 PWH B, were studied. The mean age at diagnosis of their primary disease was 6.7 ± 5.8 months, and they had received first time factor infusions at an average of 9.6 ± 7.1 months of age. Their age at the start of prophylaxis ranged from 7 months to about 7 years with a mean of 19.8 ± 18.7 months.
Before participating in the prophylaxis program, patients had received an average of 853 IU/kg of plasma-derived factor (VIII, IX) concentrates in a year for OD treatment of their bleeding events. Their mean ABR was 5.60 ± 1.83, which contained joint and nonjoint bleedings. After the beginning of prophylaxis, the mean annual factor use reached 1754 IU/kg/yr for each patient including 181 IU/kg/yr for OD treatment and 1573 IU/kg/yr for prophylaxis (equal to 28 IU/kg in a week for each patient). The new prophylaxis regimen reduced the hospitalization days and also the number of target joints by half and reduced the mean ABR to 1.86 and an annual joint bleeding rate (AJBR) to 0.88. Overall, target joints decreased from 9 to 4 after prophylaxis. Clinical scores also improved from 5.80 to 3.68 by the new regimen (Table 2). At the end of the study from 20 PWH A, 15 (75%) patients were still on the once-weekly regimen (initial step) and 5 (25%) patients were receiving infusions twice a week. From 5 PWH B, 4 (80%) patients received their factor once weekly and 1 patient went through twice-a-week regimen (step 1). Overall, 19 (76%) patients are receiving their factor infusions once a week and 6 (24%) patients have infusions twice a week. None of patients needed a three-times-a-week regimen (step 2) and none needed central lines and catheterizations. None of patients developed inhibitors against the factors (VIII, IX). No major bleedings occurred for patients during the follow-up.
Outcome Data Before and After the Beginning of Prophylaxis Regimen for Each Patient.
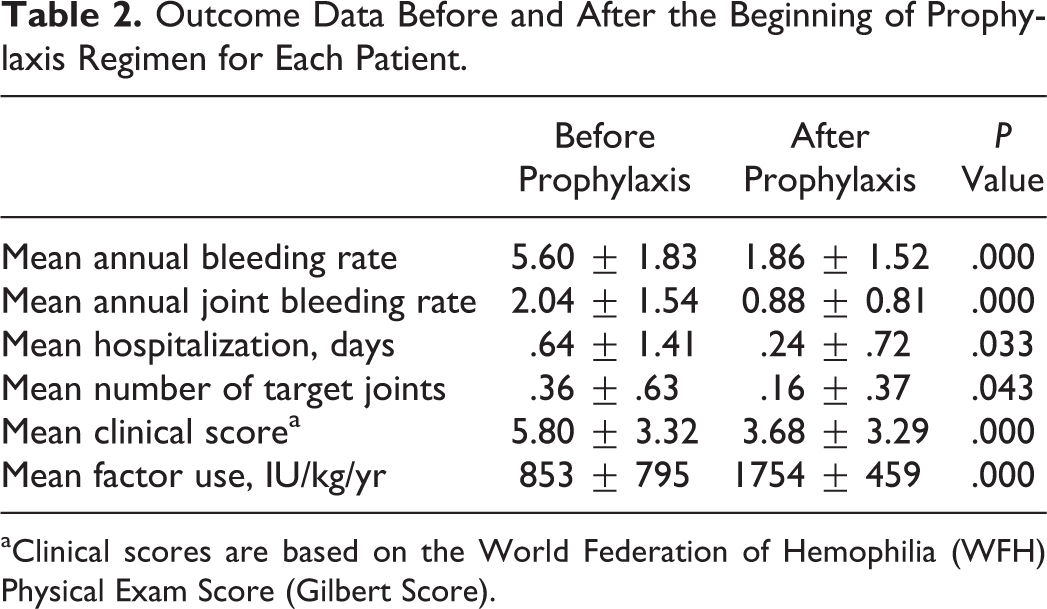
aClinical scores are based on the World Federation of Hemophilia (WFH) Physical Exam Score (Gilbert Score).
For 90 bleeding events, the number of days past from the last infusion was documented. Thirty-two percent of the bleeding events occurred in the first 3 days, which was mostly due to traumatic hemarthroses and hematomas (Figure 1). The distribution of events of hemarthrosis (n = 66) is presented in Figure 2, which mostly includes ankle and knee joints, respectively.
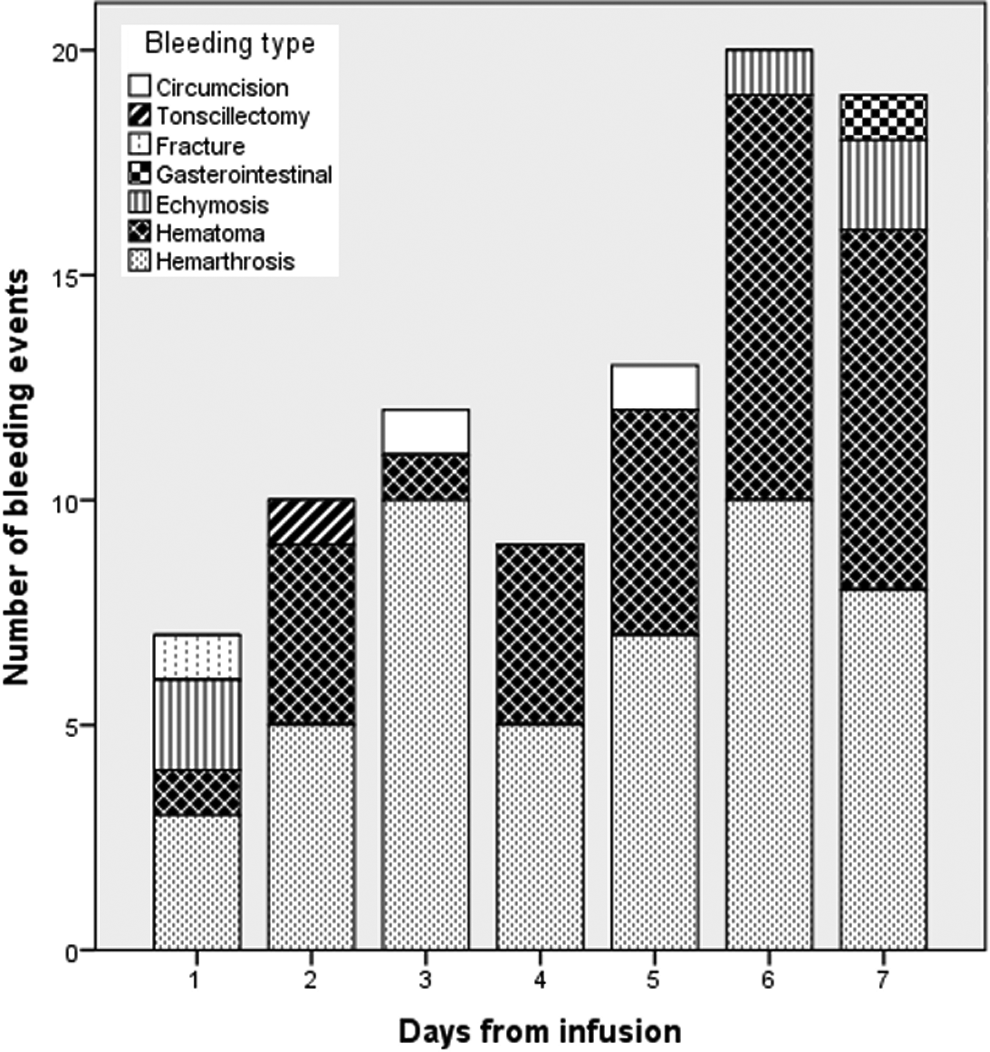
Number of bleeding events and bleeding types based on days from the infusion.
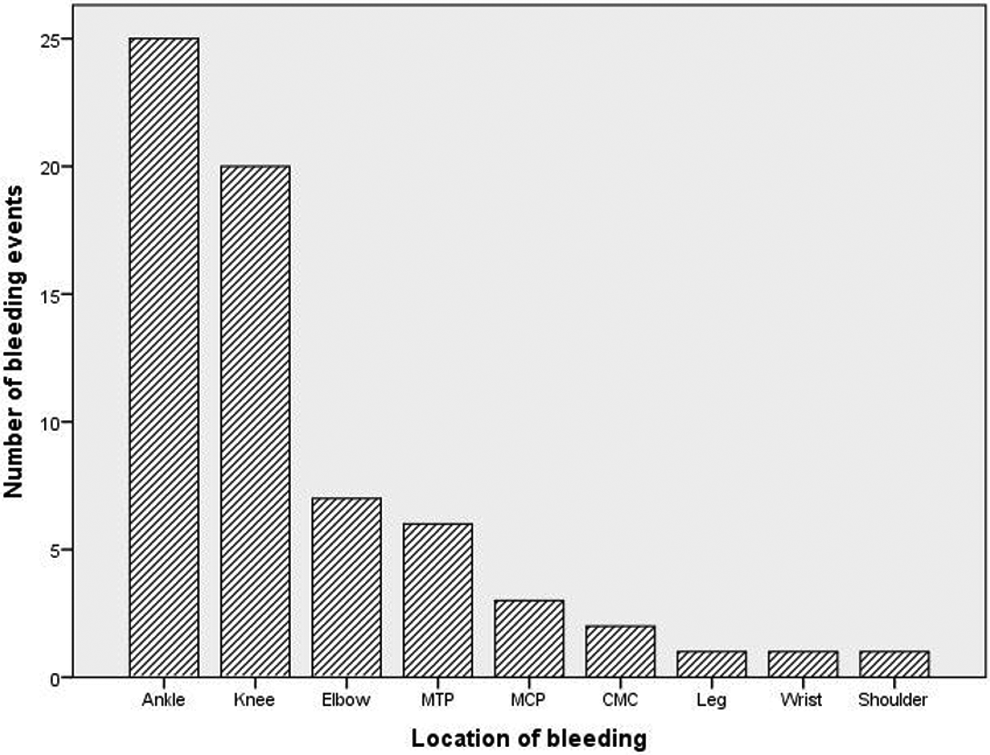
Number of joint bleeding events in the prophylaxis period based on their location. MTP indicates metatarsophalangeal joint; MCP, metacarpophalangeal joint, CMC, carpometacarpal joint.
Discussion
Many experts have recognized the disproportionate improvement in clinical situation of their patients with hemophilia after establishing a minimum low-dose escalating prophylaxis regimen in their individual practice experience. This study is an attempt to document this observation as a single-center experience.
Literature Review
About 2 to 3 decades ago, it was suggested that to prevent hemarthrosis effectively, a regimen of 3000 IU/kg/yr should be used for each child with severe hemophilia with a prophylaxis regimen divided into 2 or 3 times a week. 12 Later, the classic 3-times-a-week Swedish prophylaxis regimen (HA: 25-40 IU/kg 3 times a week) was developed using 4000 IU/kg/yr for each patient to reduce the AJBR approximately to zero. 3,13 Although ideal, the high factor use made it less feasible for many countries to follow the new recommendations. The next intermediate-dose Dutch prophylaxis protocol (HA: 15-25 IU/kg 2-3 times a week) used an average of 2100 IU/kg/yr for each patient to result in an AJBR of 1.3. 13 However, with this new regimen, venous access still remained a major barrier, especially in young children. Later, escalating protocols were introduced to benefit from individual differences in the phenotype of the disease. To lower factor use and also to avoid central lines, they used a low-frequency regimen for starting prophylaxis and step-up regimens with higher frequencies whenever bleeding patterns required. The Canadian-tailored prophylaxis regimen (HA: starting with 50 IU/kg once a week) used an average of 3656 IU/kg/yr to reduce the mean of AJBR to 1.2, with 40% of patients needing no step-ups. 5
Eventually, countries with 1 to 3 units per capita FVIII use were recommended to start prophylaxis models of their own (ex-10 IU/kg twice a week for a country with 1 IU/capita), since lower dose of prophylaxis is still considerably effective. 14 Some examples of these low-dose models were implemented in China, Algeria, Tunisia, and other countries. A fixed low-dose secondary prophylaxis regimen in China (HA: 10 IU/kg twice a week over a period of 12 weeks) by using equal to 1000 IU/kg/yr factor reduced joint bleeds by 78%. 15 Another multicenter trial in China proved the results again by using about 80 to 103 IU/kg per month and reducing joint bleedings to 0.7 per month (79% reduction). 16 Tunisia with a median factor use of 30 IU/kg per week resulted in an ABR of 0.93 (42 bleeds in 45 children registered), which was compatible with a mean 0.99 factor unit per capita. 17 Algeria also introduced a regimen with yearly consumption of 1751 IU/kg, which considerably reduced the rates of bleeding. 18
Current Situation in Iran
Iran with identified PWH of about 5000 (HA: 4230) has invested much effort in improving factor per capita use and factors proper allocation. Considering the overall population of 80 million and current FVIII per capita index of 2.7 unit, 51 063 units of FVIII is available per year for PWH A 11 . If an average weight is taken to be 25 kg, then 2042 IU/kg FVIII in a year is available for every patient. The newly introduced Iranian low-dose escalating prophylaxis regimen requires a mean of 1754 IU/kg/yr factor (FVIII/IX) for both prophylaxis and OD use, to result in an ABR of less than 2 (1.86) and an AJBR of 0.88. The overall factor use is equal to regular OD approaches and therefore does not impose any additional costs to societies.
Goal setting for a prophylaxis regimen should be specific to every society with its specific priorities. With the current status of the factor availability in Iran, the most fitting prophylaxis approach would promise functional joints toward adolescence and early adulthood and postponing functional dependence and disability to the third and fourth decades of life, until a typical patient with hemophilia has gone through a suboptimal normal growth and has developed an adjusted lifestyle to their disease. 19
Prophylaxis Program for Hemophilia in Iran
The Iranian prophylaxis protocol insists on the primary prophylaxis, based on WFH recommendations, to be optional for children and their parents; therefore, consent should be obtained from patients’ families for the start of the program. 4 It also recommends not starting the prophylaxis too early and postponing the procedures such as circumcision to a later time in life. On the other hand, the threshold for starting the prophylaxis is lower than classic prophylaxis including soft tissue bleedings to reduce danger signals while exposing to the factor. 7 Eventually, to induce toleration to the plasma-derived factor, the initial starting dose is of low dose and frequency. The protocol consists of starting with low-dose infusions (25 IU/kg once weekly) combined with escalating criteria of the Canadian regimen. 5 Based on the phenotype of the disease, patients are transferred to the next step, which is an addition in frequency of the infusions of the same dose.
Single-Center Experience
The classic prophylaxis regimen was shown to result in an AJBR of less than 1. 19 However, our escalating prophylaxis protocol, combining low-dose and low-frequency together, competes with both high-dose and intermediate-dose regimens with an ABR of 1.86 and an AJBR of 0.88. These bleeding rates are achieved while 76% of patients are still on their primary 25 IU/kg once-weekly regimen. The regimen with overall factor use of less than 2000 IU/kg/yr, rare IV line complications and inhibitor development, and low bleeding rates has overcome many barriers to the previous prophylaxis regimens. On the other hand, low frequency of infusions by reducing the discomfort of infusions along with less attending to clinics seems to have brought improvements to the quality of life in patients with hemophilia and their adherence to their prophylaxis regimen. However, documenting the exact improvement in quality-of-life score should be the subject of further studies.
Toleration to Inhibitor Development
Ninety-five percent of inhibitor productions occur in the primary 50 exposure days to factors. There is evidence that starting the prophylaxis regimen with a low threshold such as a simple soft tissue bleed at a low dose and frequency reduces the risk of developing inhibitors. This happens by avoiding danger signals (ex-major bleeding, infection, surgery) while gradually exposing to mild doses of the coagulation factor. 7 Although danger theory has been described mostly for previously untreated patients and primary prophylaxis, it has been beneficial in decreasing the inhibitor production rate to zero in this study.
Pharmacokinetics
The distribution of coagulation FVIII is mostly intravascular with a noncovalent bond to von Willebrand factor. 20 Therefore, with the short half-life of FVIII, obtaining high factor levels for a lifetime is difficult in patients with severe hemophilia. In times when the factor trough level is below 1%, joints are theoretically thought to be susceptible to bleeding events, since bleeding events occur rarely in patients with moderate hemophilia. 3,21 Therefore, factor infusions are divided into 2 or 3 times a week to keep the trough level from falling. However, published and unpublished data support the fact that a considerable percentage of patients (76% in our study) do not experience clinically determined bleeding with once-weekly regimens. The heterogeneity in half-life of the factor (6-25 hours) can at best explain a 3-fold difference in infusion intervals needed for patients. 20 However, still a successful low-dose once-weekly prophylaxis experience points at an unknown pathophysiology or local storage of the factor as suggested in the Canadian study.
Limitations
Regular OD approaches usually lead to an average of 1200 to 1700 IU/kg/yr factor use per PWH. 22,23 The overall use of 853 IU/kg/yr before starting the prophylaxis in this study could be due to the age distribution among the patients with a mean of 19.8 ± 3.7 months and a median of 12 months at the start of prophylaxis. At this age range, patients are less likely to develop severe and chronic arthropathies, and therefore, their overall factor use is lower compared to other studies.
A further limitation could be the lack of control group of the same age. Since the same study population are compared before and after prophylaxis, the impact of aging can cover a part of improvements in ABR, clinical score, and target joints. The distribution of activity among patients is not equal and most of the patients are moving through a more active life, thereby experiencing more bleeds in joints and soft tissue.
Future Aspects
Lack of efficient national registries has led to unmergeable data from different hemophilia care centers and problems with data transfer among them. To more accurately introduce the Iranian low-dose escalating prophylaxis regimen and its benefits, prophylaxis protocols should be unified among the 54 hemophilia care centers and protocol violations should be minimized. A comprehensive list of necessary entries should be defined and completed accurately by all centers and changes be updated in time. Long-term follow-up of patients in different age groups including those under OD approach is required to analyze data in subgroups. Based on the primary data, accurate quality-of-life studies and cost–benefit analysis studies could be done to further establish the new regimen.
To understand why low-dose low-frequency prophylaxis regimens are efficacious, the pharmacokinetics of FVIII and the actual predictors of the clinical disease as well as the pathophysiology of the disease should be scrutinized again. Whether the infused factor is stored in the reticuloendothelial system should also be reconsidered.
Conclusion
The Iranian prophylaxis regimen, established since 2014, is a combination of a low-dose low-frequency protocol with escalating criteria for increasing the frequency according to patient phenotype. This protocol is ideal for countries with the factor use of 2.5 to 3 units per capita, resulting in an AJBR of less than 1. The annual factor consumption in this protocol is comparable to an ordinary OD approach with no additional burden to societies. Considering all the benefits, utilizing this protocol seems to be a necessity for all the countries of the region.
Footnotes
Authors’ Note
The research has not been presented orally.
Acknowledgments
The authors express their gratitude to Iran Food and Drug Administration, especially the chief advisor Dr Abdul Majid Cheraghali for their assistance through the establishment of the prophylaxis program. The authors are also indebted to Department of Transplantation and Special Diseases affiliated with the Ministry of Health and Medical Education, especially Dr Katayoun Najafizadeh and Dr Mohammad Aghighi for their kind cooperation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
