Abstract
The aims of this study were to analyze the clinical features of a large number of cases with disseminated intravascular coagulation (DIC) associated with acute leukemia and to assess the safety and efficacy of thrombomodulin alfa (TM-α) using the French-American-British (FAB) classification of hematological malignancies. We retrospectively examined 644 patients with acute leukemia in postmarketing surveillance for TM-α. M3, M2, M4, M1, and M5 subtypes of acute myeloid leukemia (AML) and L2 and L1 subtypes of acute lymphoblastic leukemia (ALL) have been found more frequently among patients with DIC. Bleeding symptoms at baseline were more frequent in M3 and M7 subtypes. Fibrinogen concentrations were lower, and plasmin-plasmin inhibitor complex values were higher in M3 and Philadelphia-positive (Ph+) ALL. Overall DIC resolution rate was 60.2%, higher in L1 and Ph+ ALL, lower in M1, and generally higher in ALL than in AML. Overall survival rate was generally high, at 79.8%, with higher rates in L3, Ph+ ALL, and M3. Regardless of FAB subgroup, TM-α showed improved bleeding symptoms and DIC scores in clinical practice for DIC patients with acute leukemia.
Keywords
Introduction
Disseminated intravascular coagulation (DIC) associated with hematological malignancy is understood as a type of DIC that exhibits hemorrhagic tendencies with hypercoagulation and hyperfibrinolysis.1,2 However, hematological malignancies include a wide variety of diseases, such as leukemia, malignant lymphoma, and multiple myeloma. In addition, various pathological conditions such as acute, chronic, myeloid, and lymphoid types are included under the concept of leukemia. DIC associated with hematological malignancy should therefore be considered by disease subtype.
The French-American-British (FAB) classification 3 and the World Health Organization (WHO) classification have both been used to classify hematological malignancy. The WHO classification has been widely used in clinical practice, but the FAB classification is also still used. Within the field of DIC research into hematological malignancies, analysis according to the FAB classification has been historically reported.4–6 The FAB classification is still useful, when cytogenetic information of acute myeloid leukemia (AML) has not been fully obtained for the WHO classification.
The frequency of DIC appears higher in AML than in acute lymphoblastic leukemia (ALL). 4 Within AMLs, the frequency of DIC is known to be particularly higher in M3, such as acute promyelocytic leukemia (APL). 4 APL is most frequently associated with hyperfibrinolytic-type DIC and presents with bleeding manifestations, due to the overexpression of annexin II on APL cells. Among AMLs (other than APL), M5, M1, M4, and M2 subtypes have been found more frequently in patients with DIC. 4 AML-FAB subtypes correlated with DIC risk, in particular, DIC was more frequent in M5. 5 Among ALLs, the frequency of DIC was higher in T-cell ALL than in non-T-cell ALL and higher in L2 than in L1. 4 Among children, L2 in ALL and M3, M4, and M5 in AML were found more frequently in patients with DIC. 6 Although some reports have described the incidence and clinical significance of DIC using FAB classifications like these, all such reports have included only limited numbers of DIC cases. Few recent comprehensive epidemiological studies on DIC have thus focused on hematological malignancies.
Thrombomodulin alfa (TM-α) is a recombinant human soluble thrombomodulin. We have previously investigated the efficacy and safety of TM-α in a multicenter, randomized, clinical trial in Japan. 7 We showed that both resolution rate and disappearance rate of bleeding symptoms were significantly better in the TM-α group than in the heparin group among patients with DIC associated with hematological malignancy. 7 TM-α gained approval for the indication of DIC in 2008 in Japan. An all-case registered postmarketing surveillance (PMS) of TM-α has since been conducted. 8 Asakura et al 9 reported the safety and effectiveness of TM-α, with a focus on patients with DIC associated with hematological malignancy according to underlying diseases such as AML, APL, ALL, lymphoma, myelodysplastic syndrome (MDS), and others. This study investigated the clinical features of DIC associated with each acute leukemia group as subdivided by the FAB classification, and the safety and efficacy of TM-α according to detailed classifications based on the FAB classification.
Patients and Methods
Design and Data Collection
We conducted a cohort study using data from 644 patients with AML and ALL from the PMS database for TM-α (from 2008 to 2010). The PMS for TM-α was conducted in accordance with the Japanese Society on Thrombosis and Hemostasis PMS Committee for Recomodulin Injection and the guidelines for Good Postmarketing Study Practice, as required by the Japanese Ministry of Health, Labour, and Welfare. We used existing data without personally identifiable information throughout our study. This study was therefore exempted from local institutional review and formal approval, as well as the requirement for informed consent. The original PMS study was an open-label, multicenter, noninterventional, prospective, observational cohort study of patients with DIC who received TM-α. 8 The study included a total of 4342 patients with heterogeneous underlying diseases treated using TM-α. The subject cohort population in the present analysis was restricted to all patients with DIC associated with hematological malignancy.
Patients were diagnosed at the participating institutions. The subtype of hematological malignancy was classified based on the description provided on the case report form. AML and ALL patients were classified based on the FAB classification or Philadelphia chromosome status as determined at an initial stage of diagnosis or treatment as far as possible. Patients were consecutively registered at the initiation of TM-α treatment and were prospectively monitored during the observation period. All patients received TM-α for the treatment of DIC according to the package insert for Recomodulin Injection. The standard dose of TM-α was 380 U/kg/d, the same dose used in the previous phase III trial (0.06 mg/kg/d). 7 No limitation was placed on either the administration period of TM-α or the concomitant use of other anticoagulant drugs, blood preparations, or any drugs for the treatment of complications.
Evaluation
We investigated the clinical features of DIC associated with hematological malignancy and the safety and efficacy of TM-α by detailed classification. In 644 patients associated with acute leukemia in the PMS, DIC resolution rates, DIC improvement rates, 28-day survival rates, underlying disease improvement rates, clinical course of bleeding symptoms, changes in coagulation and fibrinolytic markers, and rates of adverse drug reaction (ADR) and bleeding-related ADR were investigated by detailed classification based on the FAB classification.
The degree of coagulopathy was evaluated by calculating the DIC score according to the diagnostic criteria of the Japanese Ministry of Health and Welfare (JMHW) or that of the International Society of Thrombosis and Hemostasis (ISTH) (Supplementary Table 1).10,11 DIC resolution rates and DIC improvement rates were evaluated at the end of TM-α administration. Resolved DIC status was defined according to JMHW DIC criteria or ISTH DIC criteria. DIC improvement and underlying disease improvement were both evaluated by the attending physician. The 28-day survival rates were evaluated on day 28 from the start of TM-α administration. The clinical course of bleeding symptoms was evaluated at the end of TM-α administration, as previously described.7,9 In brief, the severity of symptoms was evaluated in 4 grades (+++, ++, +, −) based on the evaluation criteria established for each symptom. Changes in the grade of each symptom from the start of TM-α administration to the day after last administration were collectively evaluated on a scale of i to iv: (i) “disappeared”, all symptoms disappeared; (ii) “improved”, total number of improved symptoms was higher than the total number of exacerbated or new symptoms; (iii) “unchanged”, the only confirmed symptom remained unchanged, or total number of improved symptoms was comparable with the total number of exacerbated or new symptoms; or (iv) “exacerbated”, total number of improved symptoms was lower than the total number of exacerbated or new symptoms.
Fibrin and fibrinogen degradation products (FDPs), D-dimer, platelet count, fibrinogen, prothrombin time (PT) ratio, activated partial thromboplastin time (APTT), thrombin-antithrombin complex (TAT), plasmin-α2 plasmin inhibitor complex (PIC), and antithrombin (AT) were measured at each institute. Changes in coagulation and fibrinolytic markers before and after TM-α administration were evaluated.
Rates of ADR and bleeding-related ADR were evaluated from the start of TM-α administration to day 28 after the end of TM-α administration. Safety data were coded using preferred terms from the Japanese version of the Medical Dictionary for Regulatory Activities.
Data Analysis
In the descriptive analysis of baseline characteristics, numerical data are expressed as medians (Q1, Q3; interquartile range). Changes in DIC score from baseline to that of the day after the last administration were examined using the Wilcoxon signed-rank test. Statistical significance was determined with a two-sided P-value < 0.05. P-values were calculated for sample sizes of six or more. All analyses were performed using SAS version 9.4 software (SAS Institute, Cary, NC) by EPS Corporation (Tokyo, Japan) according to the statistical analysis plan. Approval for this study (approval no. 30-019) was obtained from the appropriate ethics committees and institutional review boards.
Results
Patient Characteristics
A total of 644 patients with DIC associated with acute leukemia from the PMS study cohort for TM-α (May 2008 to April 2010) were analyzed (Figure 1). Of these, 509 patients had AML and 135 patients had ALL (Table 1). AML was subdivided into 9 subtypes: M0 (n = 24); M1 (n = 52); M2 (n = 133); M3 (n = 168); M4 (n = 54); M5 (n = 48); M6 (n = 12); M7 (n = 7); and MDS-overt AML (n = 11). ALL was subdivided into 4 subtypes: L1 (n = 37); L2 (n = 72); L3 (n = 9); and Philadelphia-positive (Ph+) ALL (n = 17). The main treatments for AML were based on anthracyclines and cytarabine. The main treatment for APL contained all-trans retinoic acid, arsenic trioxide, tamibarotene, and/or anthracycline. The main treatment for ALL contained vincristine, anthracycline, cyclophosphamide, and/or breakpoint cluster region-abelson (BCR-ABL) tyrosine kinase inhibitors. Baseline characteristics of the 644 patients are shown by underlying disease in Table 1.
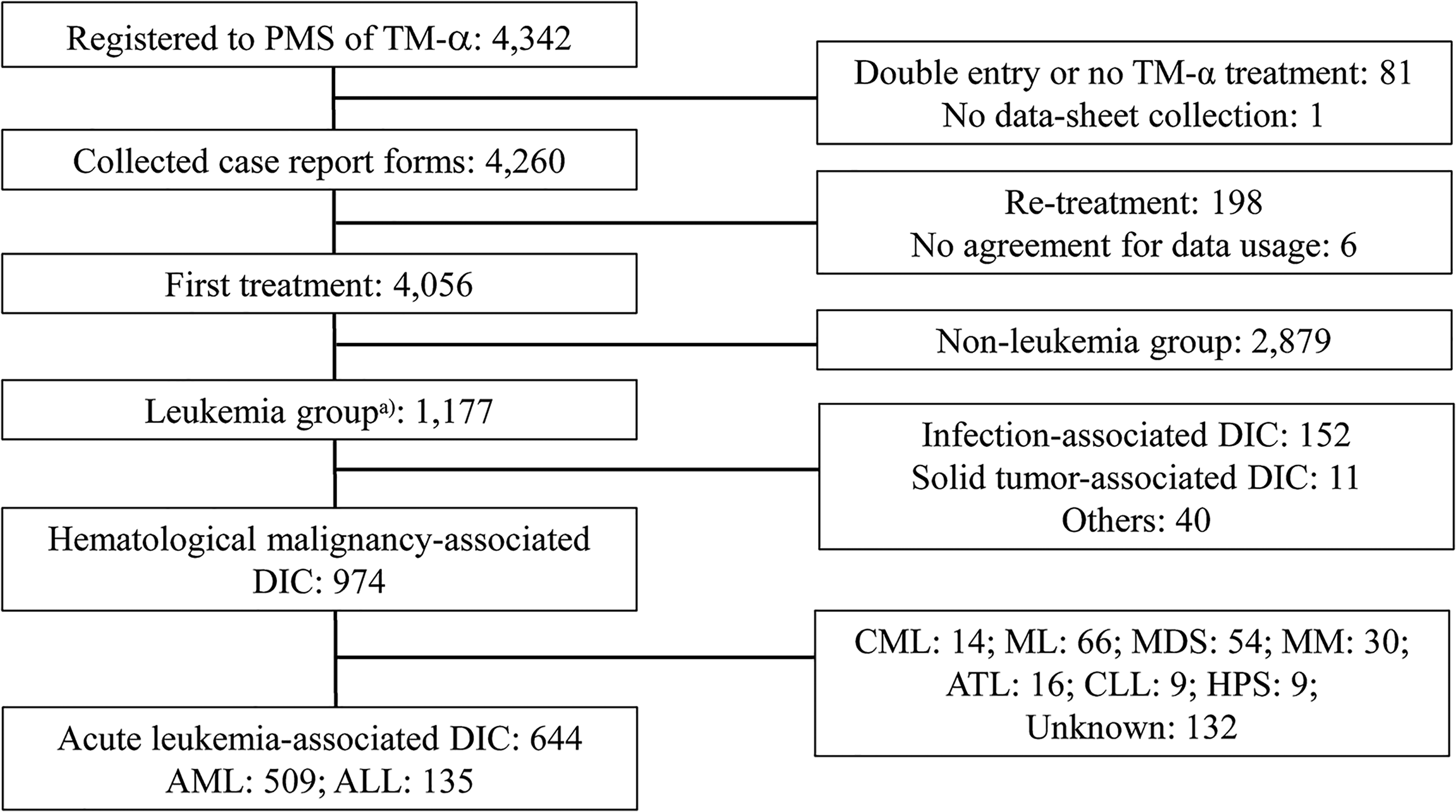
Patient disposition.
Baseline Demographics and TM-α Administration Regimens by Subtype.
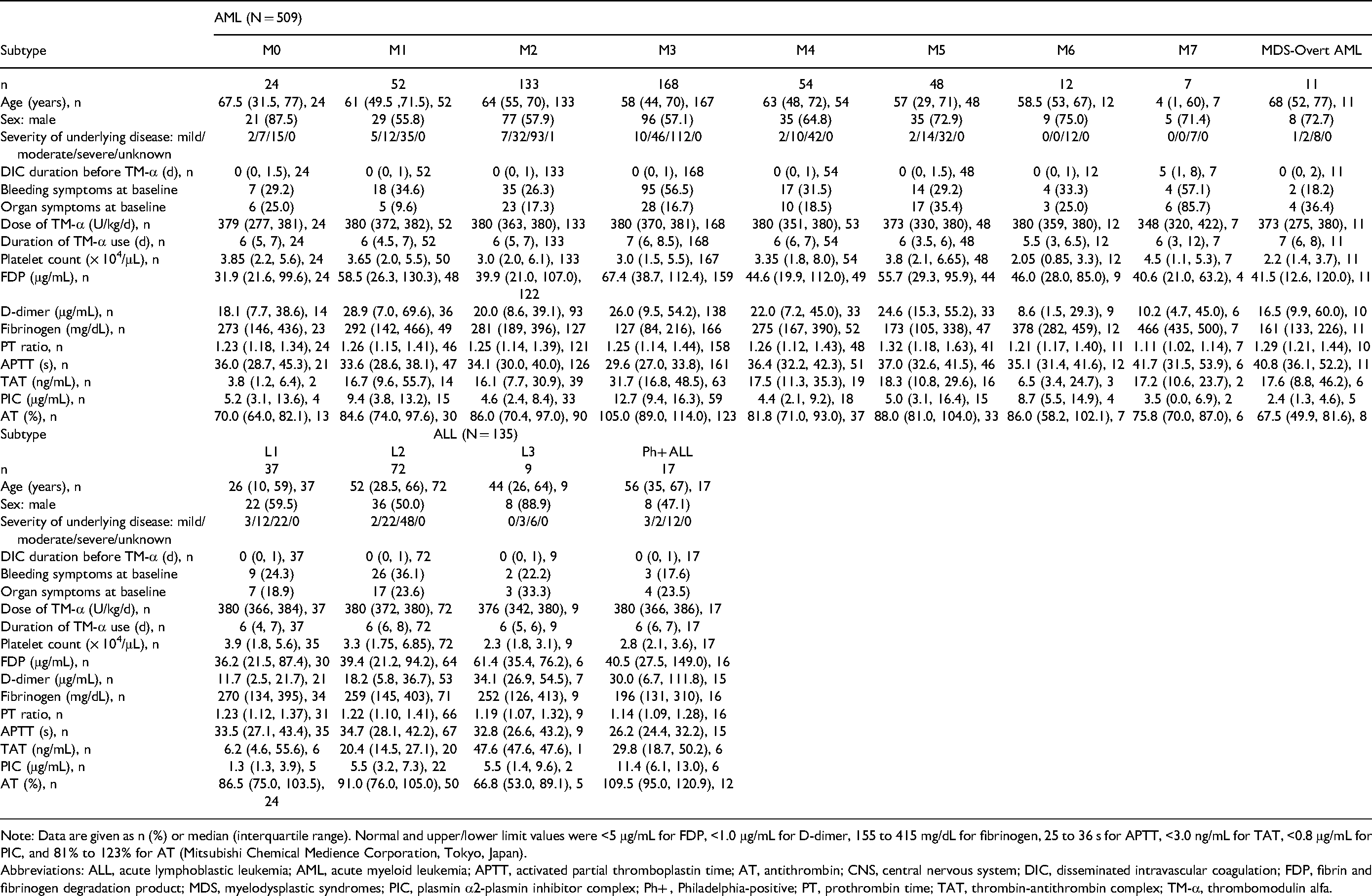
Note: Data are given as n (%) or median (interquartile range). Normal and upper/lower limit values were <5 μg/mL for FDP, <1.0 μg/mL for D-dimer, 155 to 415 mg/dL for fibrinogen, 25 to 36 s for APTT, <3.0 ng/mL for TAT, <0.8 μg/mL for PIC, and 81% to 123% for AT (Mitsubishi Chemical Medience Corporation, Tokyo, Japan).
Abbreviations: ALL, acute lymphoblastic leukemia; AML, acute myeloid leukemia; APTT, activated partial thromboplastin time; AT, antithrombin; CNS, central nervous system; DIC, disseminated intravascular coagulation; FDP, fibrin and fibrinogen degradation product; MDS, myelodysplastic syndromes; PIC, plasmin α2-plasmin inhibitor complex; Ph+ , Philadelphia-positive; PT, prothrombin time; TAT, thrombin-antithrombin complex; TM-α, thrombomodulin alfa.
The proportion of male patients was higher (≥ 70%) as compared to females in M0, M5, M6, M7, MDS-overt AML, L1, and L3. Median age for most of these diseases was around 52 to 68 years, with the exceptions of M7 (4 years), L1 (26 years), and L3 (44 years). Frequency of bleeding symptoms at baseline ranged from about 20% to 35% for many subtypes and was low in Ph+ ALL (17.6%) and MDS-overt AML (18.2%), and high in M7 (57.1%) and M3 (56.5%). Median duration of DIC before TM-α administration was 0 days; that is, TM-α was administered on the same day as DIC diagnosis, except for M7 (5 days). Median duration of TM-α administration was 6 to 7 days, except for M6 (5.5 days). Median dose was near 380 U/kg/d.
Clinical Features at Baseline
Clinical features at baseline are shown in Table 1. Markedly abnormal values of each hemostatic marker were as follows. Median concentration of FDP at baseline was at least 40 μg/mL (defined as 3 points in the JMHW DIC scoring system) for M3 (67.4 μg/mL), L3 (61.4 μg/mL), M1 (58.5 μg/mL), M5 (55.7 μg/mL), M6 (46.0 μg/mL), M4 (44.6 μg/mL), M7 (40.6 μg/mL), and Ph+ ALL (40.5 μg/mL). Median concentration of fibrinogen (reference range is usually 150-350 mg/dL) at baseline was less than 150 mg/dL for M3 (127 mg/dL) and more than 400 mg/dL for M7 (466 mg/dL). Median PT ratio at baseline was at least 1.25 (defined as 1 point in the JMHW DIC scoring system) for M5 (1.32), MDS-overt AML (1.29), M4 (1.26), and M1 (1.26). Median APTT at baseline was more than 40 s (reference range, 26-38 s) for M7 (41.7 s) and MDS-overt AML (40.8 s). Median AT at baseline was 70% or less (lower limit of normal) for L3 (66.8%) and MDS-overt AML (67.5%). Although the number of cases was small, median TAT at baseline was high for L3 (47.6 ng/mL) and M3 (31.7 ng/mL), and median PIC at baseline was high for M3 (12.7 μg/mL) and Ph+ ALL (11.4 μg/mL).
Safety
Safety outcomes are shown in Table 2. The ADR rate was high in M6 (16.7%), L3 (11.1%), and L1 (10.8%). The serious ADR rate was high in L3 (11.1%). The bleeding-related ADR rate was >5% for L3 (11.1%), L1 (8.1%), Ph+ ALL (5.9%), M1 (5.8%), M4 (5.6%), and M2 (5.3%). The serious bleeding-related ADR rate was >5% for L3 (11.1%) and L1 (8.1%).
Safety Outcomes of TM-α in DIC Patients Associated With Hematological Malignancy by Subtype.

Abbreviations: ADR, adverse drug reaction; ALL, acute lymphoblastic leukemia; AML, acute myeloid leukemia; DIC, disseminated intravascular coagulation; MDS, myelodysplastic syndromes; Ph+ , Philadelphia-positive; TM-α, thrombomodulin alfa.
Efficacy
Efficacy outcomes are shown in Table 3. JMHW DIC resolution rates ranged from 45.5% to 94.7%. The DIC resolution rate was over 70% for L1 (94.4%) and Ph+ ALL (88.9%) and under 50% for M1 (45.5%). The 28-day survival rate was over 80% for L3 (100%), Ph+ ALL (88.2%), M3 (86.9%), L2 (86.1%), L1 (83.3%), and M1 (82.7%) and under 60% for MDS-overt AML (54.5%) and M7 (57.1%). The improvement rate of the underlying disease was over 60% for L3 (77.8%), M6 (75%), M3 (74.9%), L2 (68.1%), M1 (67.3%), M4 (66.7%), and L1 (62.2%) and under 40% for MDS-overt AML (18.2%). In M3 and M7, in which the frequency of pre-existing bleeding was high (at 56.5% and 57.1%, respectively, Table 1), the ratios of disappeared, improved, or unchanged symptoms in the clinical course of bleeding symptoms after administration of TM-α were 94.7% and 100%, respectively. The ratio of disappeared, improved, or unchanged symptoms in the clinical course of bleeding symptoms after administration of TM-α was over 70% for all subtypes. The ratio of exacerbated symptoms was over 20% for M5 (28.6%) and M4 (21.4%) (Table 3).
Efficacy Outcomes of TM-α in DIC Patients Associated With Hematological Malignancy by Subtypes.

Abbreviations: ALL, acute lymphoblastic leukemia; AML, acute myeloid leukemia; MDS, myelodysplastic syndromes; DIC, disseminated intravascular coagulation; JMHW, Japanese Ministry of Health and Welfare; Ph+ , Philadelphia-positive; TM-α, thrombomodulin alfa.
Changes in Coagulation and Fibrinolysis Markers
Changes in DIC score during TM-α administration are shown in Figure 2. Although median JMHW DIC score at baseline was higher for M1, M3, and M5, the postadministration DIC scores for each subtype were significantly improved compared with those preadministration, except in M6, M7, and MDS-overt AML. A comparison of changes in coagulation and fibrinolysis markers that could be measured both before and after TM-α treatment by subtype is presented in Supplementary Table 2. Maximum median values of FDP before TM-α administration were 67.2 μg/mL, 62.9 μg/mL, and 61.4 μg/mL for M3, M5, and L3, respectively, decreasing to 8.6 μg/mL, 10.6 μg/mL, and 8.5 μg/mL, respectively, after TM-α administration. High median values of D-dimer before TM-α administration were 34.1 μg/mL, 32.2 μg/mL, 30.0 μg/mL, 26.9 μg/mL, and 37.9 μg/mL for L3, Ph+ ALL, M1, M3, and M5, respectively, decreasing to 11.9 μg/mL, 4.7 μg/mL, 5.2 μg/mL, 4.0 μg/mL, and 7.2 μg/mL after TM-α administration. Abnormal fibrinogen before TM-α administration was 125 mg/dL for M3, but normalized to 243 mg/dL after TM-α administration. High median value of PT ratio before TM-α administration was 1.32 for M5, respectively, decreasing to 1.11 after TM-α administration. Minimum median value of AT before TM-α administration was 66.8% for L3, increasing to 90.4% after TM-α administration. High median values of TAT before TM-α administration were 37.6 ng/mL and 31.4 ng/mL for M1 and M3, respectively, but decreased to 8.5 ng/mL and 4.8 ng/mL after TM-α administration. High median values of PIC before TM-α administration were 16.4 μg/mL, 13.2 μg/mL, and 12.5 μg/mL for L1, M1, and M3, respectively, decreasing to 0.6 μg/mL, 2.6 μg/mL, and 2.1 μg/mL after TM-α administration.
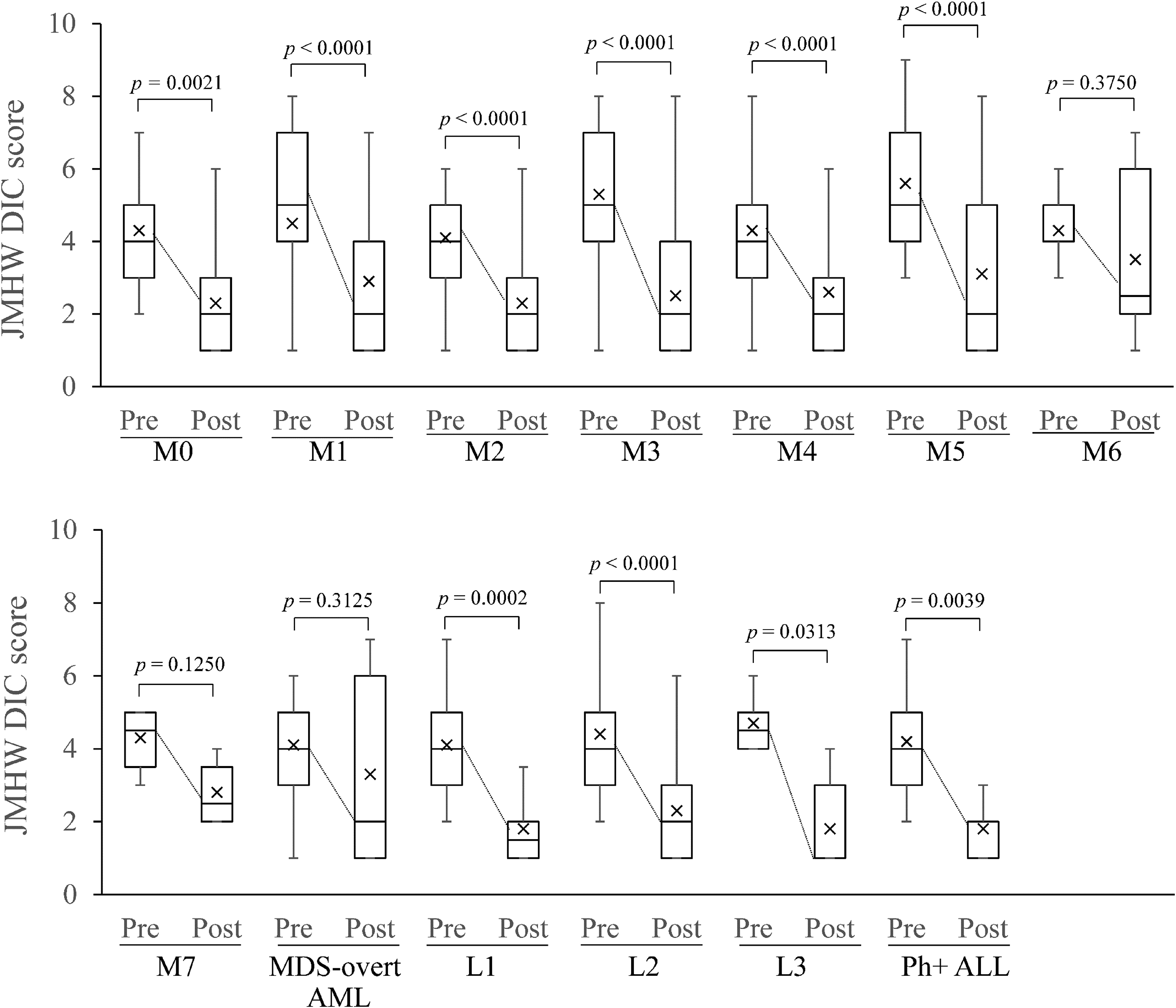
Changes in JMHW DIC scores during TM-α treatment by subtype. Data are shown as box plots with lower extreme, lower quartile, median, upper quartile, and upper extreme values. Rectangles represent lower and upper limits of the interquartile range, and median values are demarcated inside rectangles. Vertical lines (whiskers) represent the spread of data. Upper line represents the upper, or third quartile, plus 1.5 × (interquartile range), and lower line represents the lower, or first quartile, minus 1.5 × (interquartile range). Outliers are not indicated. P-values from the Wilcoxon signed-rank test are indicated. Cross symbols represent mean values.
Discussion
DIC associated with hematological malignancy is understood to be a type of DIC that exhibits a hemorrhagic tendency with hypercoagulation and hyperfibrinolysis,1,2 but no reports appear to have investigated the characteristics of DIC by detailed classification regarding disease type. We investigated the clinical features of DIC associated with hematological malignancy, especially acute leukemias, and the safety and efficacy of TM-α by detailed classification based on the FAB classification. 3
Clinical Features of DIC Among FAB Subgroups
Among AML, M3 was the most common in this cohort, and TM-α was used for many cases of DIC with bleeding. Next, TM-α was used for DIC associated with M2, M4, M1, and M5; DIC can therefore be seen to be widely associated with AML subtype. Among ALL, L2 was the most common in this cohort, followed by L1. These results were consistent with reports that DIC is frequently associated with M3, M5, M1, M4, M2 of AML, and L2 of ALL.4,5 Furthermore, this study revealed that many cases of DIC are seen in other L1.
Among coagulation and fibrinolysis markers, FDP or D-dimer was high in almost all DIC associated with hematological malignancy, indicating that DIC associated with hematological malignancy is characterized by hypercoagulation and hyperfibrinolysis. Ribeiro and Pui 5 showed that DIC was associated with a decrease in the remission rate of acute leukemia, reducing outcomes in children, and Fenaux 12 reported that lethal bleeding due to DIC associated with APL was a major cause of remission failure. Uchiumi et al 13 reported that the survival period tended to be longer in the non-DIC group than in the DIC group among cases of AML (except for APL), although the difference was not significant. Dixit et al 14 considered that careful monitoring for DIC was necessary in all cases, because DIC associated with leukemia showed bleeding symptoms in 40.3% and leukemia patients developed DIC associated with bleeding or infection after day 7 from start of chemotherapy. These are important data that DIC or coagulopathy associated with hematological malignancy and during chemotherapy are directly linked to treatment results and the prognosis of patients.
M3 and Ph+ ALL showed typical hyperfibrinolytic DIC characteristics with high PIC and low fibrinogen level. In most DIC pathologies, FDP and D-dimer increase in parallel. However, in hyperfibrinolytic DIC, not only fibrin degradation but also fibrinogen degradation progresses and a dissociation between FDP and D-dimer values (that is, a significant FDP increase and slight D-dimer increase) are observed. Avvisati et al 15 reported that M3 patients with hyperfibrinolysis showed sufficiently increased plasmin activity to develop acquired α2-antiplasmin deficiency. Also in this cohort, in M3, a dissociation between FDP and D-dimer values was observed. On the other hand, no such dissociation between FDP and D-dimer values was observed in Ph+ ALL, because annexin II expression on these cells appeared to be not as high as in M3. In M7, APTT was prominently extended, and bleeding symptoms at baseline were frequent.
In this cohort, the frequency of bleeding symptoms at baseline ranged from about 30% to 40% for acute leukemia and from about 10% to 30% for nonleukemia. Frequency of bleeding symptoms at baseline was >50% for M7 (57.1%) and M3 (56.5%). The most common pathological classification causing fatal cerebral hemorrhage was M3 (43.9%), which requires treatment and hemostasis management.16,17 Fibrinogen <100 mg/dL was a risk of bleeding in studies on adult ALL. 18 Bleeding was often severe in adults with L2 and L1. 19 Serious bleeding during induction of M3 was significantly associated with low fibrinogen and high white blood cell counts of ≥20,000/μL in addition to PT prolongation.20,21 The findings of the present study are consistent with these reports.
Safety and Efficacy of TM-α Among FAB Subgroups
Asakura et al 9 reported the safety and effectiveness of TM-α focusing on patients with DIC associated with hematological malignancy according to underlying diseases such as AML, APL, ALL, lymphoma, MDS, and “other” in the same cohort as this study. In that report, the overall JMHW DIC resolution rate was 55.9%, the overall 28-day survival rate was 70.7%, the overall ADR rate was 6.3%, and the bleeding-related ADR rate was 4.6%. 9
The DIC resolution rate ranged from 45.5% to 94.4%, and the 28-day survival rate ranged from 54.5% to 100% among subtypes. The DIC resolution rate increased in the order of L1 and Ph+ ALL, all of which exceeded 70%, indicating that the DIC resolution rate was generally high for lymphoid tumors and generally low for M6, M7, and MDS-overt AML. The 28-day survival rate increased in the order of L3, Ph+ ALL, M3, L2, L1, and M1, indicating that the survival rate was generally high for lymphoid tumors and myeloid tumor such as M3 and M1, and generally low for M7 and MDS-overt AML. The DIC resolution rate and the 28-day survival rate approximate the percentage improvements in the underlying disease. Thus, supportive therapy such as DIC treatment becomes important with the appropriate chemotherapy in the early days of AML and ALL.
In M3 and M7, in which the frequency of pre-existing bleeding was high, the total ratios of disappeared, improved, or unchanged symptoms in the clinical course of bleeding symptoms after TM-α administration were 94.7% and 100%, respectively, indicating that TM-α, which has both anticoagulant and antifibrinolytic effects, may represent a good treatment for those types of DIC that exhibit hemorrhagic tendencies with hypercoagulation and hyperfibrinolysis. Except in M6, M7, and MDS-overt AML, for which there were relatively few cases, DIC scores were significantly decreased for most of the treatments in all subgroups.
For a recent historical cohort, gabexate mesilate (FOY) and nafamostat mesilate (FUT) were compared for DIC with hematological malignancies. 22 DIC resolution rates on day 7 of FOY and FUT were approximately equal at the end of TM-α administration of the study, at 40.3% and 45.5%, respectively. Our data for DIC overall resolution rate at the end of TM-α administration showed a rate of 55.9%, superior to the DIC resolution with protease inhibitors. In particular, many disease types showed a DIC resolution rate of at least 80%.
Limitations
Some limitations need to be considered when interpreting the results obtained from the present investigation. The PMS data used in this study lacked a control arm. As all patients in this study were treated with TM-α, investigation of the incidence of DIC and the current status of treatment intervention in Japan were not completely possible. In addition, detailed analyses of the onset of DIC in leukemia, such as whether DIC was due to leukemic disease including kinds of cytogenetic abnormalities or instead due to treatments such as chemotherapy, could not be examined. However, this investigation was conducted by the continuous registration format and under conditions of actual clinical usage. The FAB classification does not include cytogenetic judgments but may be applicable in most clinical cases in the DIC treatment of leukemia, which provides many opportunities for determination at an initial stage of diagnosis or treatment. This investigation also provides new useful information about the efficacy and safety profile of TM-α with regard to types of acute leukemia that could not be sufficiently obtained from previous studies.
Epidemiological studies of DIC with leukemia remain limited to old reports. In the future, we must investigate and clarify the incidence, severity, and causes of DIC in true clinical practice and clarify the characteristics of DIC patients who should be treated using anticoagulant therapy.
Conclusions
This study showed the clinical features of DIC associated with acute leukemia according to FAB classifications and also provided safety and efficacy profiles of TM-α by detailed classifications based on the FAB classification in clinical practice. M3, M2, M4, M1, and M5 subtypes of AML and L2 and L1 subtypes of ALL have been found more frequently among patients with DIC who were treated with TM-α. Regardless of the FAB subgroup, TM-α was associated with improved bleeding symptoms and DIC scores in clinical practice for DIC patients with acute leukemia.
Supplemental Material
sj-doc-1-cath-10.1177_10760296211054094 - Supplemental material for Clinical Features of Disseminated Intravascular Coagulation According to the French-American-British Classification in Patients With Acute Leukemia and Thrombomodulin Alfa Treatment—A Cohort Study Using a Postmarketing Surveillance Database
Supplemental material, sj-doc-1-cath-10.1177_10760296211054094 for Clinical Features of Disseminated Intravascular Coagulation According to the French-American-British Classification in Patients With Acute Leukemia and Thrombomodulin Alfa Treatment—A Cohort Study Using a Postmarketing Surveillance Database by Seki Yoshinobu, Goichi Honda and Noriaki Kawano, Toshimasa Uchiyama, Kazuo Kawasugi, Seiji Madoiwa, Naoki Takezako, Ikezoe Takayuki, Hideo Wada in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
We would like to thank all investigators who participated in this postmarketing surveillance study. We express our sincere appreciation to Ms. Hideyo Ohshige and Ms. Fumiyo Komatsu for their helpful suggestions.
Authors’ Note
Approval for this study (No. 30-019) was obtained from the appropriate ethics committees and institutional review boards of Uonuma Institute of Community Medicine. The data that support the findings of this study are available from Asahi Kasei Pharma Corporation, but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available.
Author Contributions
Seki planned the study design, interpreted the data, and wrote the article. Seki provided oversight and supervised the current study. Kawano, Uchiyama, Kawasugi, Madoiwa, Takezako, Takayuki, and Wada advised on study design and interpretation of the data and reviewed the article. Honda planned the study design.
Declaration of Conflicting Interests
Yoshinobu, Toshimasa, Madoiwa, and Takezako received personal fees from Asahi Kasei Pharma Corporation outside the present work. Kawasugi and Takayuki received grants and personal fees from Asahi Kasei Pharma Corporation outside the present work. Wada received grants and personal fees from Asahi Kasei Pharma Corporation and Japan Blood Products Organization outside the present work. Honda is an employee of Asahi Kasei Pharma Corporation. Kawano has not received any grants or personal fees.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Asahi Kasei Pharma Corporation, which funded the analysis and English editing of the article.
Ethical Approval
Ethical approval to report this case was obtained from Ethics Committee of Uonumakikan Hospital (approval number/30-019).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
