Abstract
Introduction:
Although recent studies have reported the efficacy of antithrombin (AT) supplementation for sepsis-associated disseminated intravascular coagulation (DIC), the factors that influence AT’s effect have not been sufficiently studied. The purpose of this survey was to identify factors that modulate the effects and the adverse effects of AT.
Methods:
We performed a multi-institutional survey. The data from 159 patients with septic DIC with AT ≤70% and who had undergone AT supplementation were analyzed. The patients’ demographic characteristics, including the infection site, baseline sepsis-related organ failure assessment (SOFA) score, baseline DIC score, and baseline AT activity, were analyzed in relation to the 28-day mortality. Bleeding-related adverse events were also examined.
Results:
Overall, 116 patients survived and 43 did not (28-day mortality: 27.0%). A logistic regression analysis revealed that the baseline SOFA score (odds ratio [OR]: 0.816, P = .001), coadministration of recombinant thrombomodulin (rTM; OR: 3.989, P = .006), and respiratory tract infection (OR: 0.129, P = .000) were significantly associated with the survival. Survivors exhibited a higher peak AT activity than nonsurvivors (85.1% vs 65.0%, P = .027). Bleeding events were observed in 4.13% (major bleeding: 1.65%) of the patients, and the coadministration of rTM did not increase the risk of bleeding (with rTM: 4.11% vs without rTM: 4.17%). Heparin was concomitantly used in 22 (18.2%) cases, and its use nonsignificantly increased the bleeding risk (with heparins: 9.09% vs without heparins: 3.03%; P = .224).
Conclusion:
The coadministration of rTM may improve survival without increasing the risk of bleeding in patients with sepsis-associated DIC treated with AT.
Introduction
In the beginning of the 21st century, anticoagulant therapies were expected to be a promising strategy for the treatment of severe sepsis. The efficacy of recombinant activated protein C (APC), 1 high-dose antithrombin (AT), 2 and recombinant tissue factor pathway inhibitor (TFPI) 3 were examined in large-scale randomized controlled trials; however, none of these therapies are currently available. Nevertheless, withdrawing from this field of investigation might not be warranted 4 ; instead, the problems revealed in past studies should be solved in a step-by-step manner.
In previous studies, 5,6 we reported that the effects of AT supplementation for sepsis-associated disseminated intravascular coagulation (DIC) were defined by the initial AT activity and the supplementation dose. We think that it is important for the normal AT level (over 70% approximately) to be restored after treatment. 7 To prove this hypothesis, a baseline AT activity-guided dose selection study, that is, 3000 IU/day for patients with a baseline AT activity <40%, is necessary. 7 However, the Japanese health-care system basically does not approve the use of 3000 IU/day of AT for the treatment of sepsis-associated DIC. Therefore, before proceeding in this direction, we planned to survey the other factors that increase the effect of AT. Thus, the primary purpose of this survey was to identify alternatives for boosting the performance of AT supplementation therapy.
In our second survey, 6 we reported the potential efficacy of a combination therapy consisting of AT and recombinant thrombomodulin (rTM). Thrombomodulin (TM), a cell surface-expressed glycoprotein, is a critical cofactor for the thrombin-mediated activation of protein C. 8,9 Since the anticoagulant mechanisms of the TM-APC system are expressed through a different mechanism from that of AT, additive effects can reasonably be expected. 10 Since 2008, rTM has been clinically available and has been aggressively applied in Japan. 11 However, the concomitant use of AT and rTM has not been widely performed, since additive or synergistic effects have not been confirmed and the safety of concomitant use has not been investigated. We previously examined the effect of AT and rTM combination therapy in preclinical models of sepsis-associated DIC and reported that it provides more favorable effects than the independent use of each agent. 12,13 As the therapeutic strategies are rapidly changing in this field, we planned to examine the additive effect and bleeding-related adverse events of this combination therapy.
Patients and Methods
Patient Selection
This survey was performed as a multi-institutional, postmarketing survey. A total of 159 patients with sepsis-associated DIC with an AT activity ≤70% who were treated between June 2014 and March 2015 were registered. Patients with a history of allergic shock reaction to AT were excluded. The study was conducted in accordance with the Declaration of Helsinki and Good Vigilance Practice and Good Post-marketing Study Practice. Although the Japanese Ministry of Health, Labour and Welfare judged that the patients’ agreement was not necessary for this survey, the patients’ agreement and consent were obtained when required by the ethics committee of each hospital.
Treatment
When the patients met the Japanese Association for Acute Medicine (JAAM) DIC criteria 14 and had an AT activity level of ≤70%, either 1500 IU/day or 3000 IU/day of AT concentrate (Nihon Pharmaceutical Co. Ltd, Tokyo, Japan) was administered for up to 3 consecutive days unless the patient died or treatment was stopped for any justifiable reason. Antithrombin activity was measured at the time of the diagnosis of DIC (day 1). Although the use of 3000 IU/day is generally permitted only in severe cases, the use of this dosage is not strictly regulated and was dependent on the physicians’ choice. The concomitant use of other anticoagulants was not prohibited. Recombinant TM was administrated intravenously according to the instruction. In all, 380 U/kg of rTM was administered to the patients without renal dysfunctions and 130 U/kg was administered to those whose renal functions were less than creatinine clearance of 10 mL/min. Standard sepsis care was performed, and platelet concentrate and fresh frozen plasma were used as substitution therapy, if necessary. 15
Data Collection
Serial data for the coagulation markers, systemic inflammatory response syndrome (SIRS) score, and AT activity were measured before AT administration (day 1) and on the days after treatment (days 2, 4, and 7). Disseminated intravascular coagulation was judged to have been resolved if the DIC score decreased to less than 4 as of day 7. Survival was recorded until day 28. The bleeding events were recorded throughout the observation period. Major bleeding was defined as bleeding that was fatal, involved a critical organ, or was associated with a decrease in the hemoglobin level of 2.0 g/dL or more or required the infusion of 2 or more units of blood. The platelet count and other coagulation profiles were measured in local laboratories. The following methods were used for the measurements: the electric impedance method was used for the platelet count, the scattered light detection method was used for the prothrombin time, and latex immunoassays were used for the fibrinogen/fibrin degradation products (FDP) and
Statistical Analysis
The numerical values in the text are the median and interquartile range. The univariate association was evaluated using the Fisher exact test, a paired or unpaired Wilcoxon signed-rank test, or a t test. The relationship between the outcomes and the various factors were analyzed using a stepwise logistic regression analysis. The analysis was conducted using the outcome (survived, 1; died, 0) as the criterion variable and respiratory tract infection (yes, 1; no, 0), baseline sepsis-related organ failure assessment (SOFA) score, baseline prothrombin time (PT) ratio, and the coadministration of rTM (yes, 1; no, 0) as additional factors. Results were reported as the ORs, Wald result, P values, and 95% confidence intervals (CIs). Survival rates were calculated using the Kaplan-Meier method and were analyzed using a log-rank test. For all the reported results, P < .05 was considered to denote statistical significance. The above-mentioned analyses were performed using SPSS 22.0 for Windows (IBM SPSS Inc, Chicago, Illinois).
Results
A total of 159 patients were analyzed in this survey. Among them, 116 (73.0%) patients survived and 43 (27.0%) patients died. Twenty-five (15.7%) patients were treated with 3000 IU/day, and the rest were treated with 1500 U/day. Digestive tract infection was the most common underlying disease, followed by respiratory tract infection. Table 1 summarizes the baseline characteristics of the patients. Regarding the background differences, respiratory tract infection was more common among nonsurvivors (P < .01), while no statistical differences were observed for other infectious sites. The baseline SOFA and baseline PT ratio were higher among nonsurvivors (P < .01, .05, respectively), but no difference was seen in the baseline DIC score.
Baseline Patient Demographics.
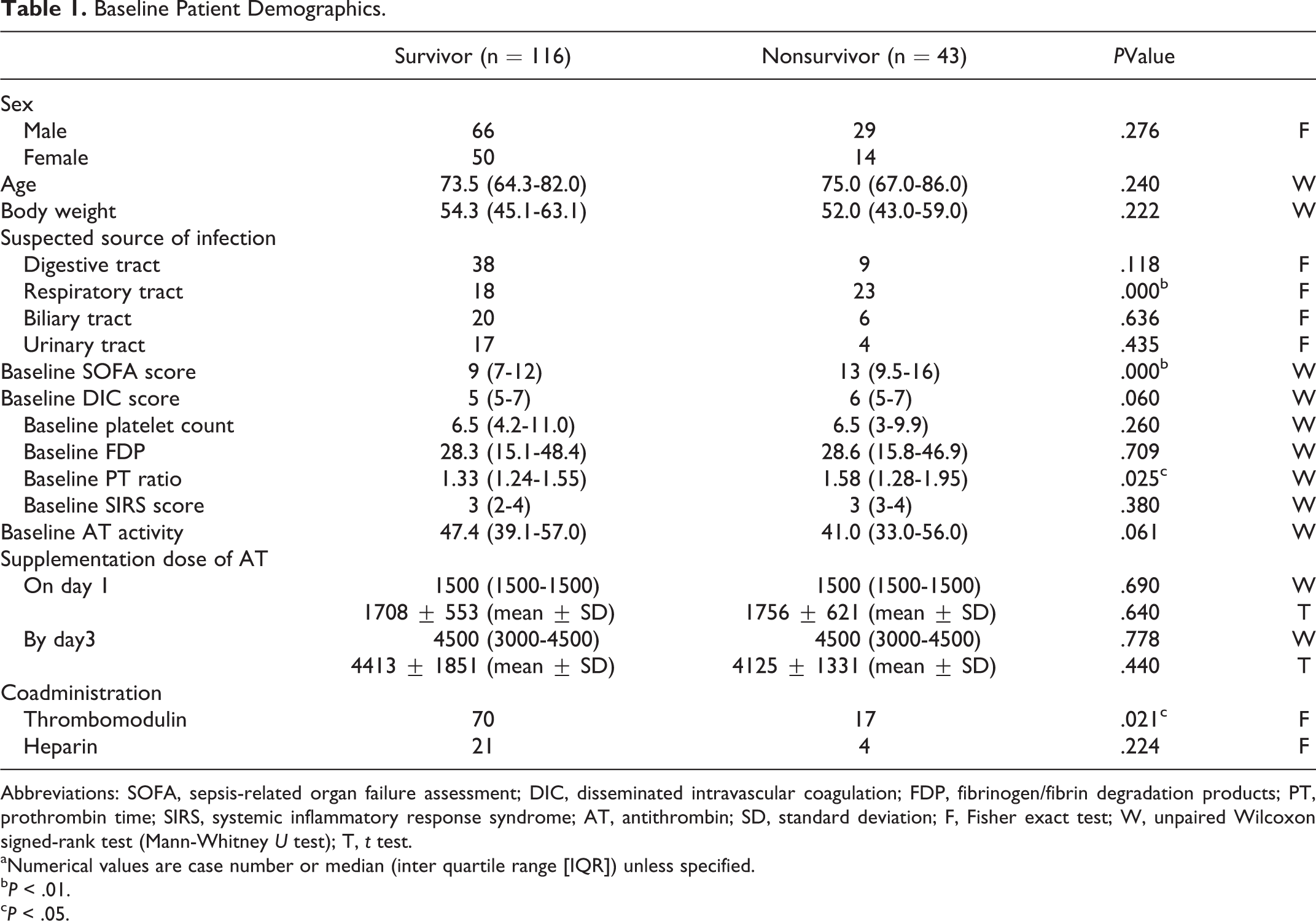
Abbreviations: SOFA, sepsis-related organ failure assessment; DIC, disseminated intravascular coagulation; FDP, fibrinogen/fibrin degradation products; PT, prothrombin time; SIRS, systemic inflammatory response syndrome; AT, antithrombin; SD, standard deviation; F, Fisher exact test; W, unpaired Wilcoxon signed-rank test (Mann-Whitney U test); T, t test.
aNumerical values are case number or median (inter quartile range [IQR]) unless specified.
b P < .01.
c P < .05.
Figure 1 shows the changes in AT activities in survivors and nonsurvivors. No difference was seen in the baseline AT activity (47.4% [39.1%-57.0%] for survivors vs 41.0% [33.0%-56.0%] for nonsurvivors). However, the median AT activity on day 4 had increased to within the target range (> 70%) in the survivors, while this level was not reached in the nonsurvivors during the treatment period. A significant difference in AT activity was observed on day 4 (survivors: 85.1% [69.5%-95.5%] vs nonsurvivors: 65.0% [57.5%-92.5%]; P < .05).
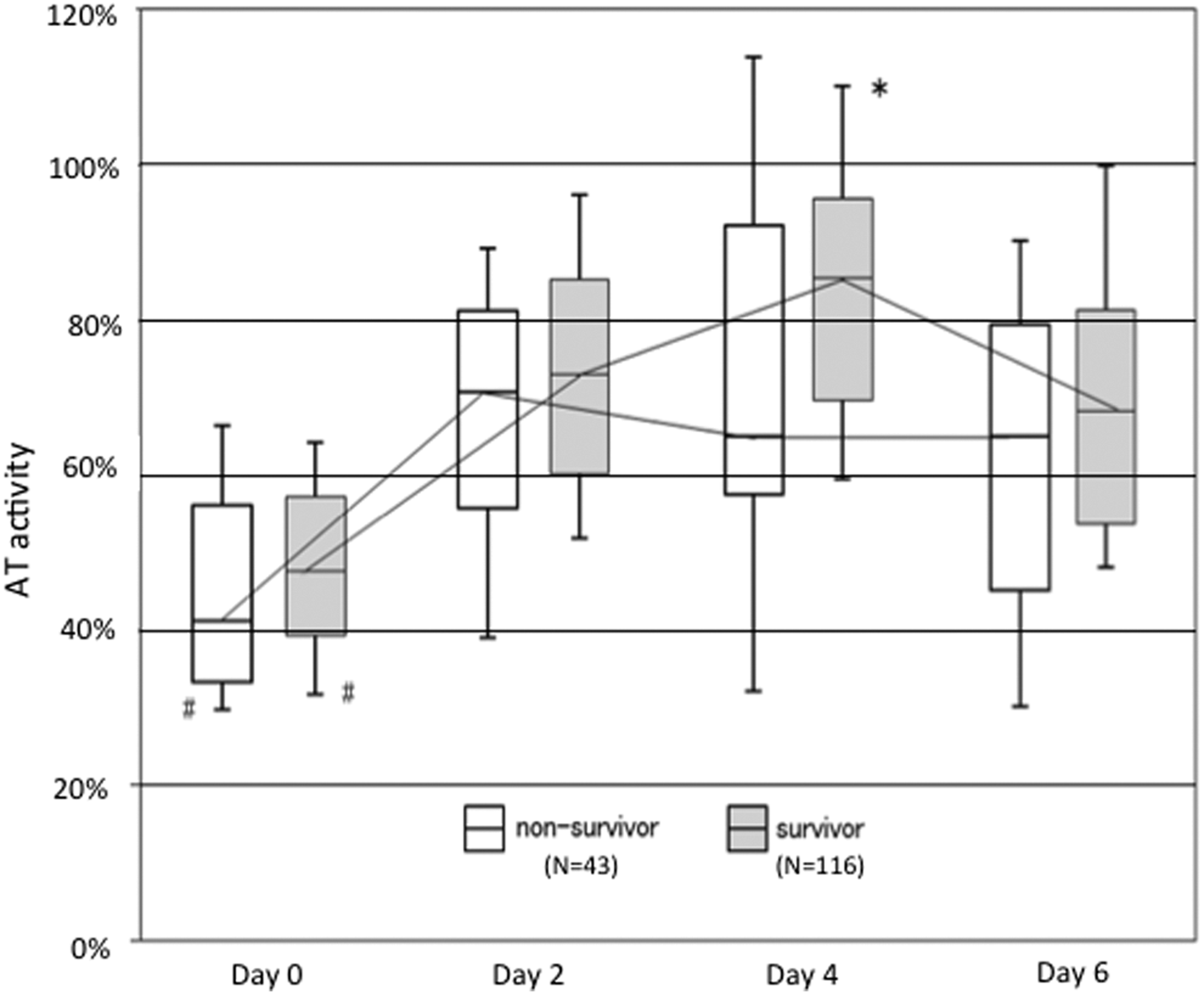
Changes in antithrombin activity. The median baseline antithrombin (AT) activity was less than 50% in both survivor and nonsurvivor groups. The AT activities increased to the normal range 85.1% (IQR: 69.5%-95.5%) in survivors, while this level was not recovered in the nonsurvivors 65.0% (IQR: 57.5%-92.5%) on day 4. The central horizontal bars, columns, and peripheral horizontal bars indicate the median values, the 25th to 75th percentiles, and the 10th to 90th percentiles, respectively. *P < .001 versus AT activity of nonsurvivors on each day. # P < .001 versus AT activity of each group at days 2, 4, or 6.
The enter method of logistic regression analysis showed that the absence of a respiratory tract infection and a lower baseline SOFA score were significant factors associated with survival. Regarding treatment factors, the coadministration of rTM was revealed to be associated with an improvement in survival (OR: 3.99, P < .01; Table 2).
Relationship Between the Outcome (28-Day Mortality) and Various Factors.
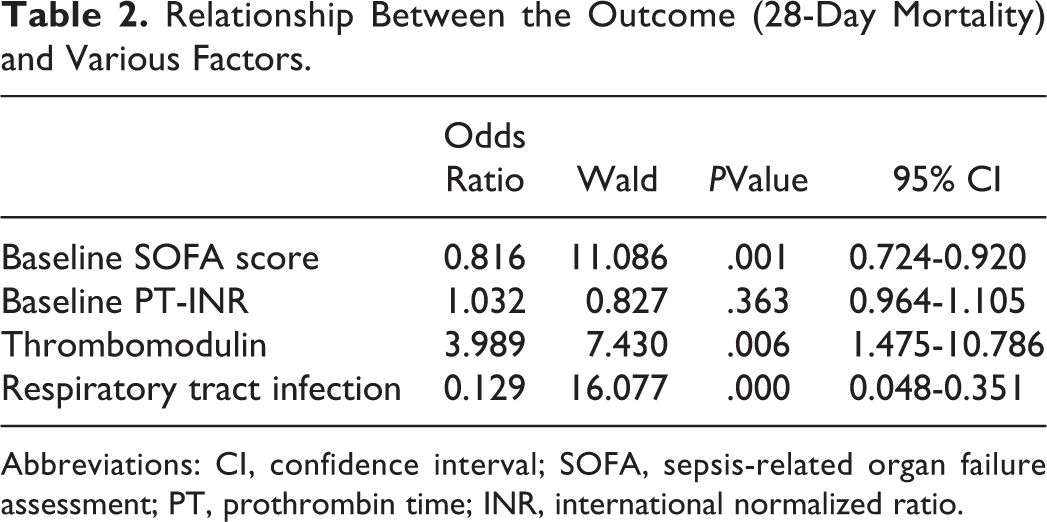
Abbreviations: CI, confidence interval; SOFA, sepsis-related organ failure assessment; PT, prothrombin time; INR, international normalized ratio.
Recombinant thrombomodulin was used in 54.7% of the patients, and the survival rate of patients treated with rTM was 80.5%; meanwhile, the survival rate of patients treated without rTM was 63.9%, which was significantly lower (P = .02). Similar result was seen in the patients with respiratory tract infection-based DIC. The survival of the patients treated with rTM was 68.8% while that of the patients treated without rTM was 28.0% (P = .02). In contrast, the survival was not different in the patients with DIC without respiratory tract infection (84.4% vs 81.0%, P = .79). Kaplan-Meier survival curves for patients treated with or without rTM were calculated using data obtained for a maximum of 28 days after the administration of AT. A significant difference in survival was recognized between the 2 groups (P = .014; Figure 2).
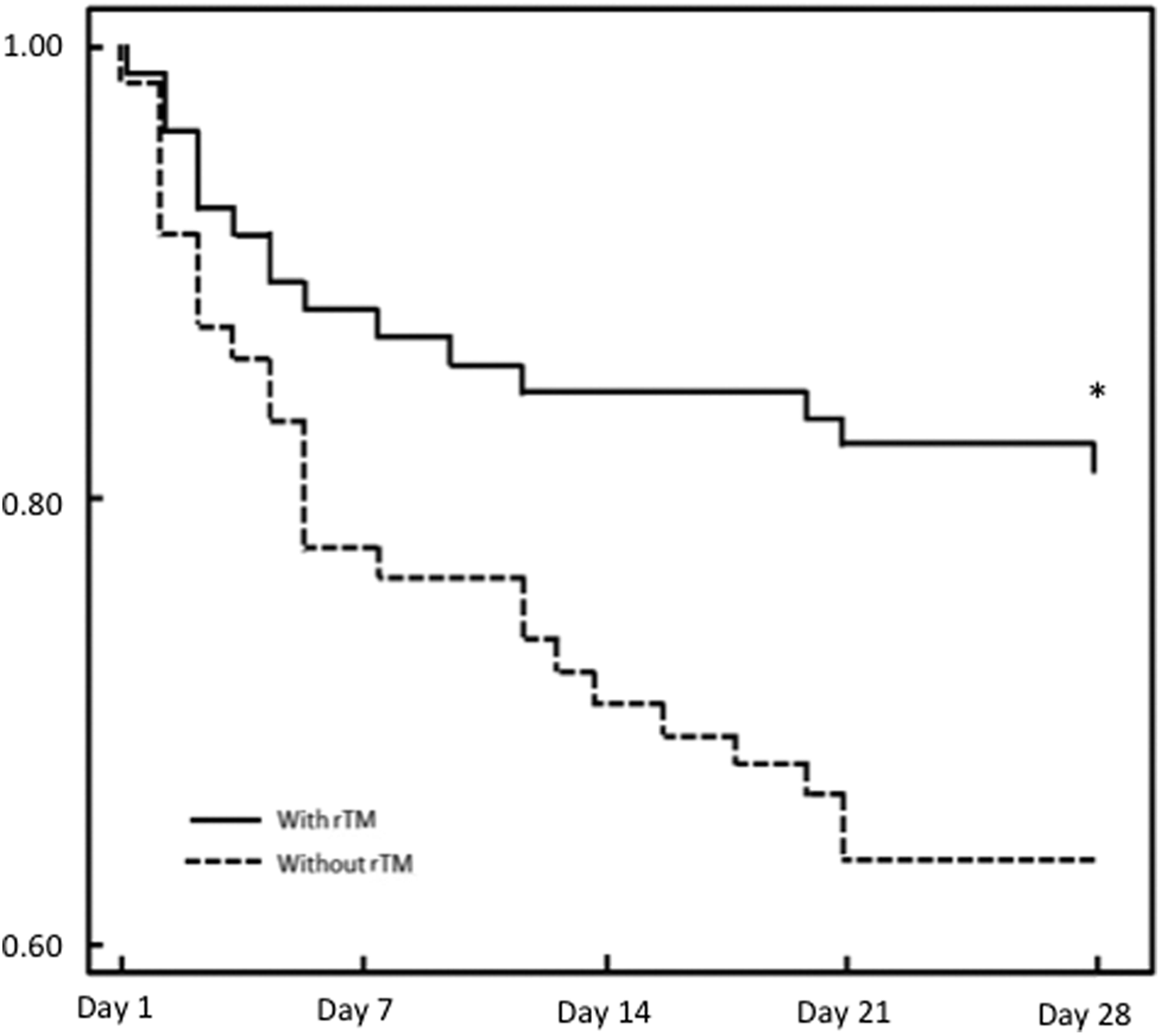
Comparison of survival. A Kaplan-Meier curve was calculated to compare survival between patients treated with antithrombin (AT) with recombinant thrombomodulin (rTM) and those treated with AT without rTM. A significant difference was recognized on day 28 (P = .014, log-rank test).
Heparins were used in 15.7% of the patients, and the survival of these patients was higher than that of patients who were treated without heparins, but the difference was not statistically significant (84.0% vs 70.9%, P = .22).
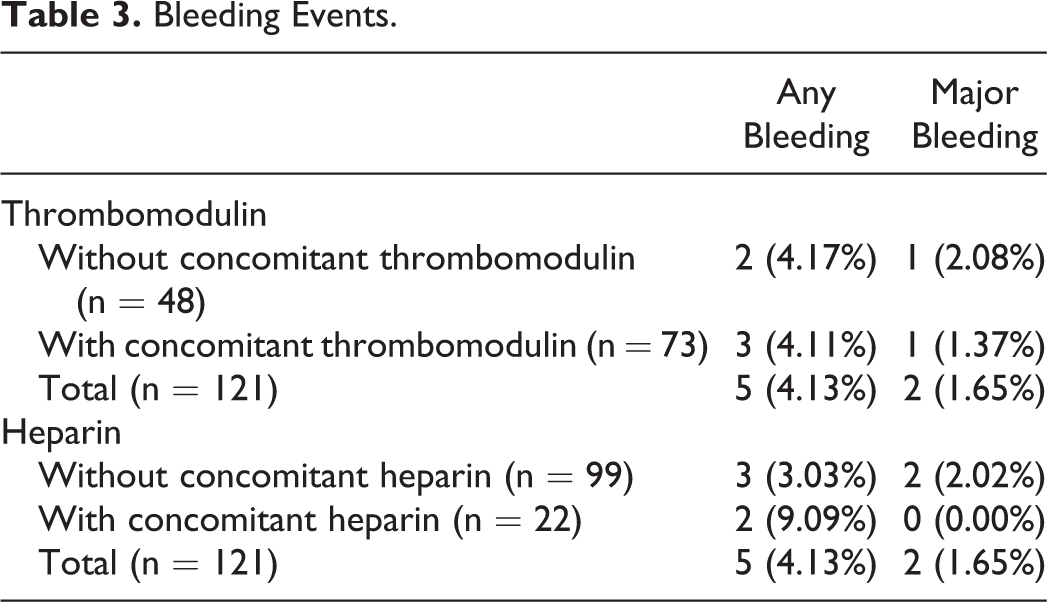
Overall, bleeding events were observed in 5 (4.13%) patients, and 2 (1.65%) of these events were classified as major. The 2 patients with major bleeding had been treated with 1500 IU/day of AT. As for the concomitant use of rTM, bleeding was recognized in 4.11% (major: 1.37%) of the patients, and the incidence was lower than that for patients treated without rTM (4.17%; major: 2.08%). Regarding the use of heparin, the incidence was 9.09% (major: 0.00%) for patients treated with heparins versus 3.03% (major: 2.02%) for patients treated without heparins (Table 3).
Bleeding Events.
Discussion
In this past decade, a certain advance has been made in the treatment of severe sepsis. 16 –18 However, to achieve further advances, we need to step toward a new area that has been left for development. 19 Reductions in the levels of endogenous anticoagulants including AT, 20,21 APC, 21 and TFPI 22 are commonly observed during sepsis. Because these endogenous anticoagulants have been reported to exert anti-inflammatory activities as well as the ability to inhibit thrombin generation, 9,10 supplementation with these anticoagulants may be an option to improve the outcomes of patients with sepsis-associated DIC. However, the fact is that “not a single endogenous anticoagulant has been confirmed as a therapeutic agent for septic DIC.” 23,24(pp1-3) Former studies have pointed out some possible reasons, 25 –28 and each of these problems should be fixed one by one.
Our first survey examining the superiority of 3000 IU/day of AT was performed between 2006 and 2009. In this survey, 3000 IU/day was used for 10.8% of the total patients. 5 In the present survey, the rate increased to 15.7% but continued to be less than expected. Thus, the purpose of this survey was to find an alternative way of boosting the effects of AT supplementation.
In the second survey, we demonstrated a superior effect of 3000 IU/day in severer cases with a baseline AT activity level of less than 40%. 6 In the current survey, though the difference was not significant, the survival of patients with an initial AT activity <40% who were given 3000 IU/day of AT was 66.7%, while that of patients given 1500 IU/day was 60.0%. Other than the points mentioned earlier, the first and second surveys also suggested the importance of target activity-oriented supplementation. 7 A prospective intervention study is required to prove this hypothesis, but the observation in this study that the survival of patients who fulfilled the target activity (approximately 70%) was 86.1%, whereas the rate was only 64.5% among patients who did not meet this target (P = .02) does provide supportive data.
Although it was revealed that the coadministration of rTM was significantly associated with the improvement of survival (OR: 3.85, P = .04) in the second survey, the number of patients who received rTM was 5.9% (18 out of 307). 7 In the present survey, more than half of the patients were treated with rTM, and the survival of the patients treated with both agents was over 80%, which was significantly higher than that of the patients treated with AT alone. Interestingly, Tagami et al 29 reported that they could not find the additional effect of AT on rTM. We think this was because the treatment was different between our survey and theirs. We speculate that AT and rTM were administered sequentially in many of the cases in Tagami’s study because they collected the data from 2010 to 2013. Recombinant thrombomodulin was released in 2008, and most of the physicians did not use rTM synchronously with AT to avoid the bleeding complications in the first several years. On the other hand, we only used the data from 2014 to 2015 to evaluate the effect of coadministration of AT and rTM. To the best of our knowledge, this is the first clinical report to describe the usefulness of combination therapy. Recently, combination therapy has become popular in Japan, and the rate of its use is rapidly growing. One possible reason for the quick spread may be the recognition of the safety of combination therapy. Indeed, the bleeding rate was somewhat lower among the patients who received both agents in the current study. However, since the number of cases was small, a larger scale survey is currently underway to confirm the safety of combination therapy.
The logical background for combination therapy remains uncertain, but the fundamental theory is based on the following: First, both AT and TM are significantly reduced either from the bloodstream or from the endothelial surface 16,17 ; second, both anticoagulants target different coagulation factors 30 ; and third, they do not interfere with each other. As most anticoagulant systems are disrupted during severe sepsis, 31 treatment using multiple agents, rather than a single agent, may be necessary. 12,13
The concomitant use of heparins was recognized in approximately one-fourth of the patients in our first survey, 5 and this rate decreased to 15.7% in the present survey. Our former survey revealed that the concomitant use of heparins did not increase the bleeding risk associated with AT supplementation, but the incidence tended to increase with heparin use in the present survey. Aside from adverse bleeding events, one concern is that heparins may abolish the antiinflammatory effects of AT by inhibiting binding to glycosaminoglycan on the endothelial surface, 2,8,32 and the rate of heparin use has gradually been decreasing. However, the pros and cons of heparin use have not yet been concluded decisively.
Finally, the present study had some limitations. The number of patients was relatively small, making the benefit and risk difficult to discuss. In addition, though the effect of concomitant use of rTM had a strong influence on survival in patients with DIC having respiratory tract infection, such effect was not recognized among the patients without respiratory tract infection. As the pathophysiology and mortality rate of DIC are likely to be different based on the infection sites, 33 individual analysis according to the presence or absence of respiratory tract infection should be performed in a larger scale survey.
Conclusion
For the treatment of sepsis-associated DIC, the coadministration of AT and rTM could be a useful alternative treatment. The concomitant use of AT and rTM may increase the effect of AT without increasing the risk of bleeding. These observations need to be confirmed in future studies.
Footnotes
Authors’ Note
This work was performed as a postmarketing surveillance by Nihon Pharmaceutical Co. Ltd.
Acknowledgments
The authors thank all the institutes that cooperated with this survey. The authors also thank M. Arakawa and K. Kinoshita for their support.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TI and SG are members of the ISTH/DIC subcommittee.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
