Abstract
It is unclear whether initial infection control or anticoagulant therapy exerts a greater effect on early changes in the Sequential Organ Failure Assessment (SOFA) score among patients with sepsis-induced disseminated intravascular coagulation (DIC). This retrospective propensity score cohort study aimed to evaluate whether adequacy of infection control or anticoagulation therapy had a greater effect on early changes in the SOFA scores among 52 patients with sepsis-induced DIC. Inadequate initial infection control was associated with a lower 28-day survival rate among patients with sepsis-induced DIC (odds ratio [OR]: 0.116, 95% confidence interval [CI]: 0.022-0.601; P = .010); however, the adequacy was not associated with an early improvement in the SOFA score. However, despite adjusting for inadequate initial infection control, administration of recombinant human soluble thrombomodulin was associated with an early improvement in the SOFA score (OR: 5.058, 95% CI: 1.047-24.450; P = .044). Therefore, early changes in the SOFA score within 48 hours after the DIC diagnosis were more strongly affected by the administration of recombinant human soluble thrombomodulin than the adequacy of initial infection control.
Introduction
The Sequential/Sepsis Organ Failure Assessment (SOFA) score is important for predicting outcomes among critically ill patients, 1 –4 based on the severity of organ dysfunction and morbidity in 6 physiological systems (the respiratory, coagulation, liver, cardiovascular, renal, and neurological systems). The scores range from 0 (normal) to 4 (most abnormal) for each system, 1 and a high initial SOFA score is associated with a high mortality rate. 2 –4 Changes in the SOFA score are also important in critically ill patients. An increased SOFA score 48 hours after admission to the intensive care unit (ICU) is associated with mortality rates of ≥50%, while mortality rates range between 27% and 35% even for unchanged SOFA scores. 2 Therefore, changes in the SOFA score are used to evaluate treatment response among critically ill patients.
Disseminated intravascular coagulation (DIC) is a common and clinically significant complication of sepsis that causes organ dysfunction. 5 –8 Patients with DIC tend to have a higher SOFA score than those without DIC and typically have a high mortality rate. 9 In addition to DIC being secondary to sepsis, DIC and sepsis adversely affect each other via cross-talk between coagulation and inflammation. 10 Gando et al suggested that DIC, in addition to the underlying causes of DIC, should be promptly treated to avoid organ dysfunction. 11 Thus, several anticoagulants have been investigated for treating sepsis-induced DIC, 12 and recent studies have focused on recombinant human soluble thrombomodulin (rTM) and antithrombin, 13,14 with anticoagulant therapy being linked to improved SOFA scores and a reduced mortality rate among patients with sepsis-induced DIC. 15 –20 However, thrombosis plays an important physiological role in immune defense (immunothrombosis), which involves important immune responses that are induced by the formation of thrombi. 21 Thus, the early administration of anticoagulants may interfere with the treatment of sepsis by reducing the immune response to pathogenic microorganisms. 22
Sepsis-induced DIC develops as sepsis progresses, which highlights the importance of appropriate antibiotic treatment and infection site drainage for sepsis-induced DIC. However, the rate of inappropriate empirical antibiotic therapy can reach 10% to 30% in the ICU. 23,24 Furthermore, additional infection site drainage is often required after the initial drainage, and technical challenges can lead to failed drainage or even unattempted drainage. Therefore, initial infection control is not always adequate for patients with sepsis-induced DIC.
Because DIC and anticoagulant therapy influence the SOFA score, 9,15 –20 physicians should be aware of the factors (eg, adequacy of infection control and/or anticoagulant therapy) that have the greatest influence on early changes in the SOFA score. As no studies have examined this issue, we hypothesized that early changes in the SOFA score (<48 hours after the diagnosis of DIC) would be more strongly affected by the adequacy of initial infection control than anticoagulant therapy among patients with sepsis-induced DIC. This single-center retrospective cohort study aimed to test that hypothesis.
Methods
Study Design, Setting, and Population
This retrospective cohort study evaluated patients who were admitted to our Advanced Emergency and Critical Care Center (Shinshu University Hospital, a tertiary care center) between April 2009 and December 2016. Critically ill patients are either transported directly to this center by emergency services or are referred from the Shinshu University Hospital or other hospitals. Patients were considered eligible if they were ≥18 years old and had sepsis-induced DIC. Patients were excluded if they had (1) sepsis during the treatment of severe trauma, (2) sepsis during the treatment of uncontrolled hemorrhagic disease, (3) sepsis with a terminal malignant disease, (4) a “do-not-attempt resuscitation” order at admission, or (5) died within 48 hours after the DIC diagnosis. Patients were classified according to their change in the SOFA score within 48 hours after the DIC diagnosis, with an improved SOFA score defined as a decrease of ≥1 during the first 48 hours (the SOFA-improved group) and an unimproved SOFA score defined as no change or an increase in the SOFA score (SOFA-unimproved group). The primary outcome was an early improvement in the SOFA score, and the secondary outcomes were DIC resolution, 28-day survival, and in-hospital survival. Early improvement in the SOFA score was defined as a decrease of ≥1 in the SOFA score within 48 hours after diagnosis of sepsis-induced DIC. The 2 groups were compared, and propensity score analysis was performed with inverse probability weighted (IPW) methods to evaluate the association between the adequacy of initial infection control and critical outcomes. Moreover, propensity score analysis, which was adjusted for the adequacy of initial infection control, was performed to evaluate the association between anticoagulant therapy (antithrombin and/or rTM) and early improvement in the SOFA score.
The study’s protocol complied with the principles of the Declaration of Helsinki and the Code of Ethics of the World Medical Association for experiments that involve humans. The study protocol was approved by the Ethics Committee of Shinshu University School of Medicine, Matsumoto, Japan (#3665). Information concerning implementation of the research was published on the hospital website to provide patients with an opportunity to refuse participation.
Patient Diagnosis and Treatment Modalities
The diagnosis of sepsis was based on the definition of infection-induced systemic inflammatory response syndrome (SIRS),
25
which was used in our center during the study period. The diagnosis of DIC was based on the Japanese Association for Acute Medicine (JAAM-DIC) scoring system,
11,26,27
which includes the SIRS score, platelet count, prothrombin time, and fibrin/fibrinogen degradation products (FDP), or
Patients were treated according to the Surviving Sepsis Campaign Guidelines and the Japanese guidelines for sepsis management. 28 –30 Although these guidelines have similar recommendations for resuscitation in cases with sepsis, only the Japanese guidelines regard DIC as a target for therapeutic interventions. Most patients received therapeutic interventions using anticoagulant for DIC promptly after the DIC diagnosis, although the attending physician made the final decisions regarding the use of anticoagulants for DIC treatment, the duration of DIC treatment using anticoagulant, and any dose reduction. Therapeutic interventions for DIC are commonly administered at doses of 3000 IU/d for antithrombin and/or 380 IU/kg/d for rTM. Unfractionated heparin, low-molecular-weight heparin, and protease inhibitors were not used for DIC treatment at this hospital. Moreover, patients who required continuous renal replacement therapy (CRRT) or polymyxin B-immobilized fiber column hemoperfusion (PMX-HP) received 20 to 50 mg/h of nafamostat, an anticoagulant with a short half-life for intracircuit anticoagulation via continuous intracircuit infusion. The attending physician determined the initial antibiotic(s), the need for surgical or nonsurgical drainage, and the use of CRRT, PMX-HP, mechanical ventilation, intravenous immunoglobulin (for sepsis), or low-dose hydrocortisone (for septic shock).
Data Collection
The patients’ electronic medical records were reviewed to collect data regarding their demographic characteristics, clinical characteristics, treatments, and outcomes. The data included age, sex, medical history, infected organ(s), bacterial culture results, presence or absence of DIC at admission, presence or absence of septic shock at the DIC diagnosis, laboratory results at the DIC diagnosis, SIRS score, JAAM-DIC score, SOFA score at the DIC diagnosis and 48 hours later, acute physiological and chronic health evaluation (APACHE) II score at the DIC diagnosis, infection, and DIC treatments within 48 hours after the DIC diagnosis, additional infection treatments at >48 hours after the DIC diagnosis, presence or absence of DIC resolution (a JAAM-DIC score that decreased to <4), 28-day survival, and in-hospital survival. The adequacy of initial infection control was also evaluated, with inadequate control defined as (1) pathogenic bacteria that were resistant to the initial antibiotics based on bacterial culture results, (2) additional drainage of the infection site was required at >48 hours after the DIC diagnosis, and (3) drainage of the infection site was deemed necessary but could not be completed or was not attempted. Three reviewers who were blinded to the study hypothesis performed the chart review, categorized the infected organs according to the International Classification of Diseases-10 codes, and judged the adequacy of the initial infection control. The majority opinion was selected in cases with discrepancies regarding the results, or additional personnel were asked to participate when it was necessary to reach a consensus.
Statistical Analysis
The SOFA-improved and SOFA-unimproved groups were compared in terms of their age (continuous variable), sex, medical history, infection status, severity scores (ordinal variables), laboratory results (continuous variables), treatments, and outcomes. Categorical variables were compared using Fisher exact test, while continuous and ordinal variables were compared using the Mann-Whitney U test. All statistical analyses were performed using IBM SPSS software (version 25; SPSS Japan Inc, Tokyo, Japan), and differences were considered statistically significant at P values of <.05.
Propensity scores for the adequacy of initial infection control were estimated using a logistic regression model that was based on the following covariates: age, sex, history of diabetes mellitus, chronic organ insufficiency or immunosuppression in the APACHE II score, 31 multiple sites of infection, mixed microorganisms, JAAM-DIC and APACHE II scores at the DIC diagnosis, septic shock, non-β-lactam antibiotics, surgical or nonsurgical drainage, CRRT, PMX-HP, mechanical ventilation, intravenous immunoglobulin, low-dose hydrocortisone, rTM, and antithrombin. The C statistics of the propensity scores was estimated using receiver–operating characteristic curve analysis, and then the propensity scores were used with IPW methods to evaluate the association between inadequate initial infection control and critical outcomes.
Propensity scores were also estimated for the administration of antithrombin and/or rTM. Covariates were considered variables that precluded the administration of each anticoagulant. The C statistics were estimated, and logistic regression analysis was performed to evaluate the association between administration of each anticoagulant and early improvements in the SOFA score. Logistic regression analyses were adjusted for inadequate initial infection control and propensity scores (logit) calculated as: log (propensity scores/[1 − propensity scores]).
Results
Patient Characteristics
During the study period, 7474 patients were admitted to the Advanced Emergency and Critical Care Center, 384 patients were diagnosed with sepsis, and 63 patients had a JAAM-DIC score of ≥4 points. Eleven patients were excluded (5 patients died within 48 hours after DIC diagnosis and 6 patients fulfilled the other exclusion criteria), and 52 patients were included in the final analysis (Figure. 1). The median patient age was 71 years (interquartile range [IQR]: 62-77 years), and 46% (24/52) of the patients were males. At DIC diagnosis, the median SIRS, JAAM-DIC, SOFA, and APACHE II scores were 3.0 (IQR: 2.0-3.0), 5.0 (IQR: 4.0-6.8), 8.5 (IQR: 6.0-12.0), and 23.5 (IQR: 17.0-31.8), respectively. Septic shock was experienced by 67% (35/52) of the patients, and sepsis-induced DIC was present at admission for 71% (37/52). The average duration (standard deviation) from admission to DIC diagnosis was 0.4 (0.8) days. All patients who received antithrombin and/or rTM had the first administration within 48 hours of DIC diagnosis. Complete agreement of the infected organ and the adequacy of initial infection control among the 3 reviewers was observed in 79% (41/52) and 90% (47/52) of patients, respectively. Inadequate initial infection control was observed in 33% (17/52) of the patients and the DIC resolved for 77% (40/52) of the patients. The 28-day and in-hospital survival rates after the diagnosis of sepsis-induced DIC were 77% (40/52) and 67% (35/52), respectively. The SOFA score improved within 48 hours after the DIC diagnosis for 60% (31/52) of the patients, and no improvement was observed for 40% (21/52).
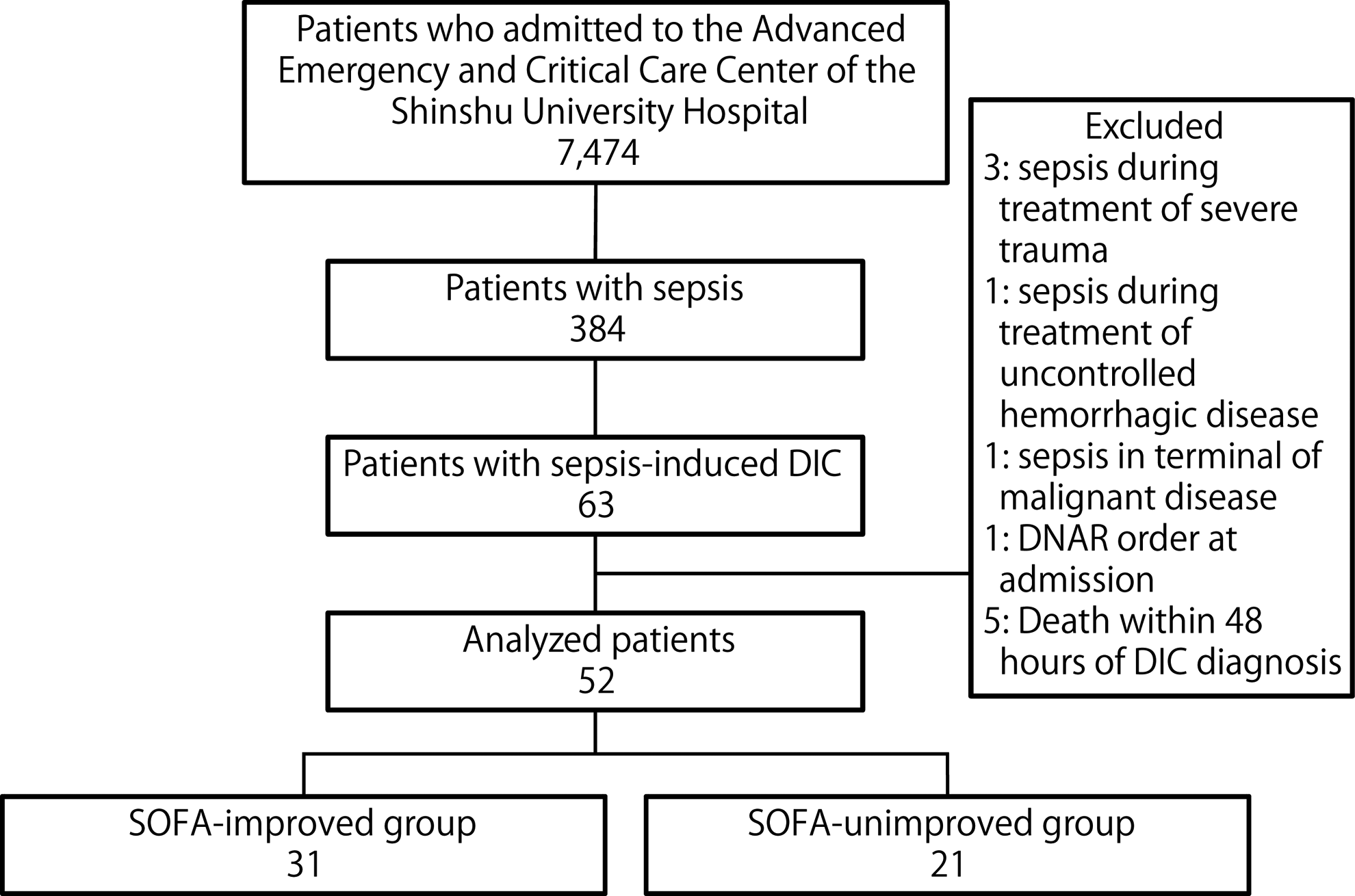
Study flowchart. The numbers of patients are indicated in each box. DIC indicates disseminated intravascular coagulation; DNAR, do-not-attempt resuscitation; SOFA, Sequential Organ Failure Assessment.
Comparing the Groups With Improved and Unimproved SOFA Scores
Table 1 shows the demographic, clinical, and treatment characteristics of the SOFA-improved and SOFA-unimproved groups. No significant differences were observed in terms of demographic characteristics, infected organs, bacterial culture results, severity scores, laboratory results, infection site drainage, life support and adjuvant therapy, and administration of anticoagulants for DIC treatment. All non-β-lactam initial antibiotics were administered concomitantly with β-lactam, although the SOFA-improved group was significantly less likely to receive non-β-lactam antibiotics (19% vs 48%; P = .038). Although the 2 groups had similar rates of inadequate initial infection control, the SOFA-improved group had significantly higher rates of DIC resolution (87% vs 62%; P = .048), 28-day survival (90% vs 57%; P = .008), and in-hospital survival (84% vs 43%; P = .003; Table 2).
The Demographic and Clinical Characteristics of the Patients in the Groups With Improved and Unimproved SOFA Scores.a
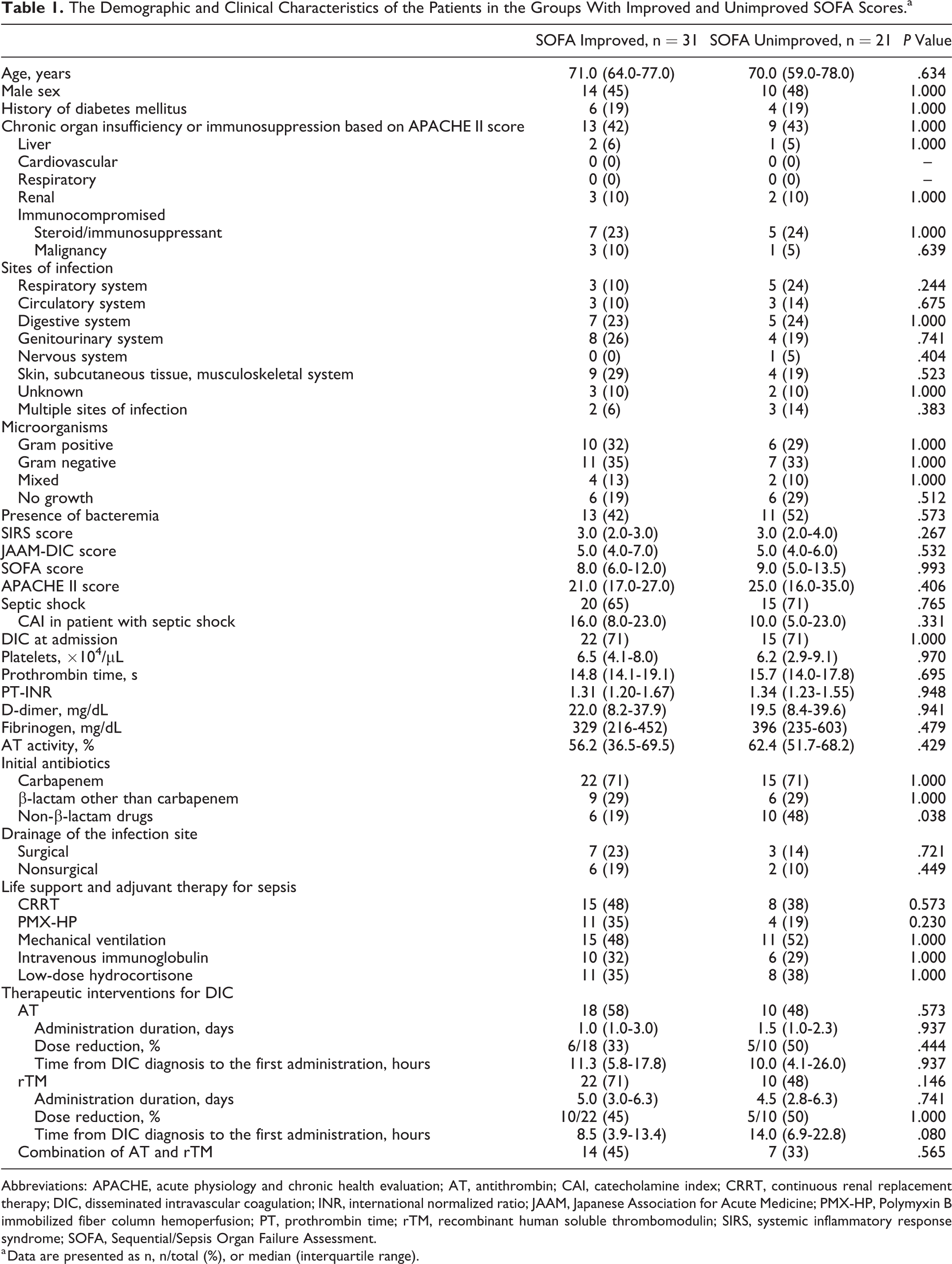
Abbreviations: APACHE, acute physiology and chronic health evaluation; AT, antithrombin; CAI, catecholamine index; CRRT, continuous renal replacement therapy; DIC, disseminated intravascular coagulation; INR, international normalized ratio; JAAM, Japanese Association for Acute Medicine; PMX-HP, Polymyxin B immobilized fiber column hemoperfusion; PT, prothrombin time; rTM, recombinant human soluble thrombomodulin; SIRS, systemic inflammatory response syndrome; SOFA, Sequential/Sepsis Organ Failure Assessment.
a Data are presented as n, n/total (%), or median (interquartile range).
Comparing the Outcomes Among Patients With Improved and Unimproved SOFA Scores.a
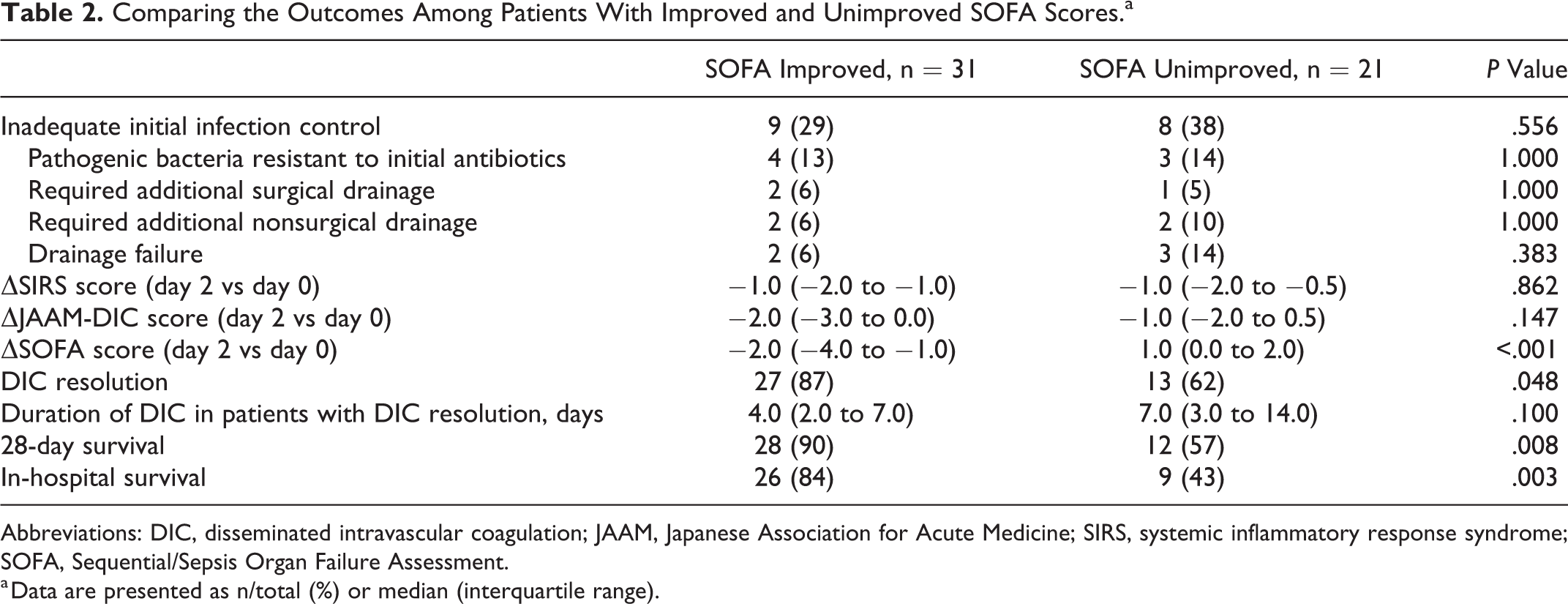
Abbreviations: DIC, disseminated intravascular coagulation; JAAM, Japanese Association for Acute Medicine; SIRS, systemic inflammatory response syndrome; SOFA, Sequential/Sepsis Organ Failure Assessment.
a Data are presented as n/total (%) or median (interquartile range).
Main Effects of Each Treatment Factor on Critical Outcomes
Propensity scores were calculated for the adequacy of initial infection control, which revealed a C statistic of 0.884, a Hosmer-Lemeshow χ2 value of 6.541 (df = 8), and a P value of .587. Logistic regression analyses were then performed using the IPW method, which revealed that inadequate initial infection control was not significantly associated with an early improvement in the SOFA score, although inadequate initial infection control was associated with significantly reduced rates of DIC resolution (odds ratio [OR]: 0.160, 95% confidence interval [CI]: 0.029-0.882; P = .035), 28-day survival (OR: 0.116, 95% CI: 0.022-0.601; P = .01), and in-hospital survival (OR: 0.215, 95% CI: 0.047-0.987; P = .048; Table 3).
Main Effects of Each Treatment Factor on Critical Outcomes.
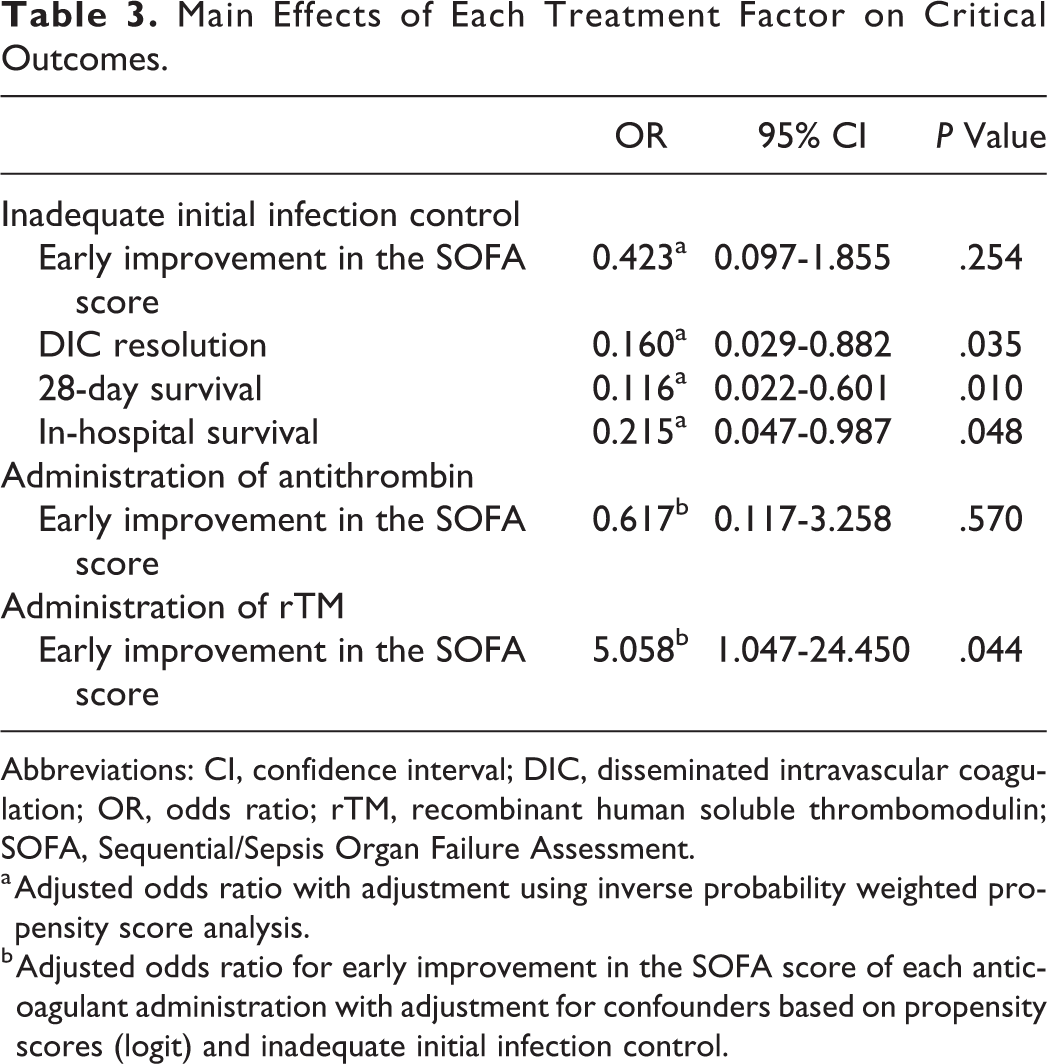
Abbreviations: CI, confidence interval; DIC, disseminated intravascular coagulation; OR, odds ratio; rTM, recombinant human soluble thrombomodulin; SOFA, Sequential/Sepsis Organ Failure Assessment.
a Adjusted odds ratio with adjustment using inverse probability weighted propensity score analysis.
b Adjusted odds ratio for early improvement in the SOFA score of each anticoagulant administration with adjustment for confounders based on propensity scores (logit) and inadequate initial infection control.
Propensity scores were also calculated for the administration of antithrombin, which revealed a C statistic of 0.948, a Hosmer-Lemeshow χ2 value of 4.712 (df = 8), and a P value of .788. Logistic regression analysis, which was adjusted for propensity score (logit) and inadequate initial infection control, revealed that the administration of antithrombin was not significantly associated with an early improvement in the SOFA score (Table 3).
Propensity scores were calculated for the administration of rTM, which revealed a C statistic of 0.856, a Hosmer-Lemeshow χ2 value of 4.535 (df = 8), and a P value of .806. Logistic regression analysis, which was adjusted for propensity score (logit) and inadequate initial infection control, revealed that the administration of rTM was significantly associated with an early improvement in the SOFA score (OR: 5.058, 95% CI: 1.047-24.450; P = .044; Table 3).
Discussion
The present study demonstrated that inadequate initial infection control among patients with sepsis-induced DIC was associated with unfavorable outcomes, such as DIC nonresolution, 28-day mortality, and in-hospital mortality. However, a significant negative effect of inadequate initial infection control on the early improvement in the SOFA score (within 48 hours after the diagnosis of sepsis-induced DIC) was not revealed. In contrast, even after adjusting for inadequate initial infection control, the administration of rTM was significantly associated with an early improvement in the SOFA score.
Ferreira et al have reported that a high initial SOFA score was associated with a high mortality rate and that trends in the SOFA score during the first 48 hours of ICU admission were important for predicting survival outcomes among critically ill patients. 2 For example, they found that the mortality rates among medicosurgical ICU patients were ≥50% when the SOFA score increased, 27% to 35% when it did not change, and <27% when it decreased. 2 Thus, the SOFA score is widely used to evaluate treatment responses among patients who are admitted to the ICU. However, not all patients who develop sepsis-induced DIC have the DIC at the time of their ICU admission. Therefore, we evaluated changes in the SOFA score during the first 48 hours after the DIC diagnosis. Despite the fact that DIC can modify the SOFA score, 9 our findings indicate that the SOFA-improved group had significantly higher rates of DIC resolution, 28-day survival, and in-hospital survival, which suggests that early changes in the SOFA score after the DIC diagnosis might be important for evaluating treatment responses among patients with sepsis-induced DIC.
Several studies have indicated that improvements in the mortality rate were not observed among all patients with sepsis but only among patients with DIC. 12,32,33 Thus, as anticoagulant treatment might be reserved for patients with sepsis-induced DIC, the present study only included patients with sepsis-induced DIC based on the JAAM-DIC score, which can predict multiple organ dysfunction syndrome, mortality risk, and treatment response among patients with sepsis-induced DIC. 11 Several investigators have also reported that rTM administration may improve the SOFA score and reduce the mortality rate among patients with sepsis-induced DIC, 15 –20 with Yatabe et al suggesting that early rTM administration is better than delayed administration after the diagnosis of DIC. 34 These findings support the early administration of rTM, which is supported by our observation of an association of rTM administration with an early improvement in the SOFA score, independent of the adequacy of initial infection control. In this context, the change in SOFA score is a useful measure for evaluating the effects of interventions on survival outcomes among critically ill patients. 35 Therefore, although the present study was not designed to investigate the potential survival benefit of rTM administration, an early improvement in the SOFA score after the DIC diagnosis may help identify patients with sepsis-induced DIC who would benefit from receiving this drug.
Appropriate treatment of infection using effective antibiotics and infection site drainage are the most important therapies for sepsis-induced DIC, with Zahar et al suggesting that early appropriate antibiotic therapy has the greatest influence on survival outcomes among patients with sepsis. 36 Unfortunately, the rate of inappropriate empirical antibiotic therapy can reach 10% to 30% in the ICU, 23,24 which agrees with our observed rate of 33%, despite the fact that there may have been differences in the definition of inadequate infection control. In this context, no studies have strictly evaluated the rate of inadequate infection site drainage, although the current guidelines recommend rapid intervention to achieve source control after the diagnosis of sepsis. 37 However, there is no established definition for inadequate infection control, although our data indicate that antibiotic resistance, and inadequate drainage had the greatest effects on this factor. Moreover, we found that inadequate initial infection control was significantly associated with critical outcomes, such as DIC nonresolution, 28-day mortality, and in-hospital mortality among patients with sepsis-induced DIC. Therefore, adequacy of initial infection control within 48 hours after the DIC diagnosis is an important factor that should be evaluated while treating patients with sepsis-induced DIC.
Interestingly, despite the adequacy of initial infection control being significantly associated with critical outcomes, the present study revealed that rTM administration had a greater effect on early improvement in the SOFA score within 48 hours after the DIC diagnosis. Thus, it appears that early rTM administration may help improve the SOFA score in this setting, even in cases with inadequate initial infection control. Therefore, when treating DIC using rTM, the adequacy of initial infection control must be judged using non-SOFA indicators.
This study has several limitations. First, the single-center retrospective observational design is associated with a risk of unmeasured or unknown biases, despite the fact that we used the IPW method to reduce the influence of confounders. 38 Moreover, because the study hospital is a tertiary center, the included patients may have been more severely ill than patients who are treated at other centers. Second, the sample size was relatively small, which limits the generalizability of the findings and the ability to detect relatively minor differences. Moreover, the patients who died within 48 hours after DIC diagnosis, who accounted for nearly 10% of the cohort, were excluded because their state could not be evaluated 48 hours after DIC diagnosis. These selection criteria might introduce the risk of bias. Therefore, we confirmed that the significant effects of the initial infection control and anticoagulants on critical outcomes did not change in an additional analysis that included those patients. Third, the present study did not control for provider-related factors, and it is possible that unobserved treatments may have the affected outcomes. Fourth, sepsis was diagnosed based on definitions that were proposed in 1991 (for infection-induced SIRS), 25 although all of the included patients also fulfilled the new criteria for sepsis diagnosis, which changed during early 2016 (an elevated SOFA score of ≥2). 39 Fifth, the study evaluated data from a 7-year period and it is possible that evolutions in the treatments for DIC or sepsis might have influenced our findings, although our center had a fairly consistent protocol for resuscitation in cases of sepsis during the study period. 28 –30 Sixth, the diagnosis of DIC was based on the JAAM-DIC score, which is predominantly used in Japan, and our findings may not be observed in hospitals that use the International Society of Thrombosis and Hemostasis (ISTH) criteria. 40 However, we found that the JAAM-DIC score could diagnose most cases of overt DIC, similar to the ISTH criteria, 11,26,27,41 and the JAAM-DIC score provides the added benefit of being able to identify patients with DIC who might benefit from anticoagulant treatment. 33 Seventh, the presence of DIC at admission was observed in 71% of the patients, which obscures the timing of DIC onset, although we also commonly encountered patients with sepsis who developed DIC during their ICU admission. Further prospective multicenter studies are needed to validate our findings, which may help improve the treatment of sepsis-induced DIC.
In conclusion, inadequate initial infection control was associated with unfavorable outcomes among patients with sepsis-induced DIC. However, rTM administration exerted a stronger effect than adequacy of infection control on early changes in the SOFA score within 48 hours after the DIC diagnosis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Mochizuki has received personal fees from Asahi Kasei Pharma Co. and from Toray Medical Co., which were unrelated to the present study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
