Abstract
Recombinant human soluble thrombomodulin (thrombomodulin α [TM-α]) has been marketed as a novel anticoagulant for disseminated intravascular coagulation (DIC) in Japan since 2008. Postmarketing surveillance (PMS) has been conducted since its approval. As effectiveness and safety were not previously determined in pediatric patients, this study evaluated PMS data and examined the usefulness of TM-α in treating pediatric DIC. After excluding newborn infants, data for 210 pediatric patients were analyzed and compared to 3786 adult patients. The day after the last TM-α administration, DIC had resolved in 58.5% of the patients. At 28 days after the last TM-α administration, the survival rate was 71.6%. Nineteen episodes of adverse drug reactions were observed in 11 patients but no significant differences were noted for effectiveness and safety. Although this study was limited by its retrospective design, including selection biases and no limitation on concomitant use of other anticoagulants, TM-α appears to be useful for the treatment of DIC in both pediatric and adult patients.
Introduction
Disseminated intravascular coagulation (DIC) is a syndrome characterized by widespread activation of coagulation due to a variety of underlying diseases, such as sepsis, hematological malignancies, and advanced tumors. Thrombotic occlusion in the microvessels of these patients can lead to ischemic multiple organ failure. The subsequent consumption of platelets and coagulation factors that occur during the ongoing coagulation can simultaneously induce bleeding and therefore DIC is referred to as “consumption coagulopathy.” 1 –3 Although recent advances in the management of severe infection and respiratory and circulation failure, which often induce DIC, have helped to reduce the occurrence of DIC, prognosis of patients with DIC remains poor. Unfortunately, prognosis for pediatric patients with DIC is also extremely poor and can even lead to death. 4,5
In order for pediatric patients to be able to recover from DIC, the underlying diseases associated with DIC need to be treated. According to the “The Nelson Textbook of Pediatrics,” 6 anticoagulant therapy should be conducted to prevent ongoing intravascular coagulation in patients where the primary treatment of DIC may be inadequate or incomplete. Although small-scale studies for anticoagulants have already been undertaken, the effectiveness of using these agents has yet to be proven. In particular, there has been no conclusive proof that anticoagulants are useful in pediatric patients with DIC.
Recombinant human soluble thrombomodulin (thrombomodulin α [TM-α]; brand name: Recomodulin Injection 12800 [Asahi Kasei Pharma Corporation, Tokyo, Japan]), which possesses an extracellular domain of membrane-bound thrombomodulin, is produced in Chinese hamster ovary cells. 7 The TM-α binds to the thrombin that is produced during hypercoagulable states. The thrombin–TM-α complex then activates protein C. Subsequently, activated protein C (APC) inhibits coagulation through the inactivation of factors Va and VIIIa. 8 In addition to its anticoagulant activity, it has also been reported that the N-terminal lectin-like domain of TM-α exerts anti-inflammatory activity by decreasing the level of the high mobility group box 1 protein, which enhances the production of interleukin 6 and tumor necrosis factor α in experimental endotoxemia. 9 –13
A phase III randomized double-blind trial was conducted in adult patients with DIC associated with infection or hematologic malignancy for the purpose of comparing the efficacy and safety of TM-α with that of unfractionated heparin (UFH). 7 Results suggested that TM-α was significantly superior to UFH in terms of DIC improvement. Based on the findings of this phase III trial, the Japanese Ministry of Health, Labour, and Welfare granted approval for the use of TM-α for the treatment of DIC in 2008. Since its approval, postmarketing surveillance (PMS) has been conducted to confirm the effectiveness and safety of TM-α administration in general practice settings. 14 Data collected during this PMS included information on pediatric patients with DIC. As the phase III study did not collect any data on pediatric patients with DIC, combined with the fact that only a very small series of patients have been reported since the original approval, 15 a more systematically performed study needs to be undertaken. After evaluating PMS study data from a cohort of 210 pediatric patients, here we report the clinical safety and efficacy of TM-α treatment in pediatric patients with DIC.
Methods
Design and Data Collection
Postmarketing surveillance was conducted in consecutive patients administered TM-α for approximately 2 years between May 2008 and April 2010. 14 The standard dose of TM-α administered was a daily 1 time intravascular drip infusion of 380 U/kg (over a 30-minute period). Since this PMS was conducted in general settings, all patients were treated according to the attending physician’s decisions. As a result there were no limitations placed on the concomitant use of other anticoagulants, blood products, or medication for the treatment of underlying diseases and complications. Of the 4062 patients with DIC enrolled in the PMS, there were 3786 adult patients with DIC who were 15 years and older, 270 pediatric patients with DIC who were less than 15 years of age, and 6 patients with DIC for whom the age was unknown. In addition to the 6 patients of unknown age, 60 newborn patients who were less than 4 weeks old had to be excluded from this analysis, as the background of newborn patients, was much different from that found for the other pediatric patients. The reason for this appears to be related to differences in the physiological status of the coagulation and fibrinolytic systems between newborn infants and pediatric patients older than 4 weeks. Furthermore, the main underlying diseases that induce DIC in newborn infants are generally associated with accidents during the birth process or premature births.
Prior to the first TM-α administration, the following data were collected for each patient: gender, age, duration of DIC until the first administration of TM-α, treatment of DIC at baseline, underlying disease, severity of underlying disease (as assessed by physicians taking part in this PMS), history of risk factors for central nervous system (CNS) bleeding such as cerebrovascular accidents, CNS surgery, or head trauma, complications (renal impairment, hepatic impairment, and others), bleeding, and organ symptoms at baseline. Based on the underlying diseases that induced the DIC, the patients were divided into the infection group, the hematological malignancy group, and the other group. In cases involving patients with DIC who had 2 or more underlying diseases, each physician judged which of the underlying diseases directly caused the DIC and then placed the patient in the appropriate group. Each group was then evaluated for both effectiveness and safety. The study also collected information on the administration regimens of TM-α (dose and duration), use of other concomitant anticoagulants and/or blood products, and the clinical symptoms related to the bleeding and organ symptoms.
Evaluation of TM-α Effectiveness
Two methods were used to evaluate the effectiveness of TM-α. The first method was the DIC resolution rate (rate of recovery from DIC), which was evaluated on the day after the last TM-α administration. This method was based on the diagnostic criteria of the Japanese Ministry of Health and Welfare (JMHW DIC criteria). 16 The scoring was performed after dividing the patients into groups with or without severe thrombocytopenia due to marked myelosuppression. Patients were excluded from the DIC score analyses if their scores did not indicate DIC at the start of the TM-α administration or if they had not undergone the blood tests that are required for DIC scoring.
The second method evaluated the survival rate on the 28th day after the last TM-α administration. If the physicians could not judge the outcome due to their relocation to another area, patient data were excluded from the survival rate analyses.
Changes in the DIC score, coagulation tests related to coagulation/fibrinolysis in DIC, and the other hematological tests were determined based on values obtained at baseline and on the day after the last TM-α administration.
Evaluation of TM-α Safety
Adverse events (AEs), adverse drug reactions (ADRs), and the level of seriousness were classified in accordance to the International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH) guidelines. All ADRs and serious AEs leading to death were observed until the 28th day after the last TM-α administration. All other AEs were observed until the day after the final TM-α administration. The AEs were coded using the preferred terms from the Medical Dictionary for Regulatory Activities (MedDRA/J) version 13.1.
A multiple analysis was performed in order to identify the background factors responsible for affecting the development of serious bleeding-associated AEs. The items examined included gender, age, medical history related to the risk of bleeding, duration of DIC until the start of TM-α administration, bleeding symptoms at baseline, organ symptoms at baseline, treatment of DIC at baseline, concomitant anticoagulants, coagulation tests (fibrin and fibrinogen degradation products [FDPs], platelet counts, fibrinogen, prothrombin time [PT] ratio, antithrombin [AT], thrombin–antithrombin complex, plasmin-α2-plasmin inhibitor complex, and protein C), and general blood tests (white blood cells, aspartate aminotransferase, alanine aminotransferase, total bilirubin, lactate dehydrogenase, serum creatinine, and C-reactive protein). To investigate the possibility of the production of antibodies against TM-α after the administration of TM-α, serum samples were collected before TM-α administration and on the 28th day after the last TM-α administration. Samples were then measured by enzyme-linked immunosorbent assay for the presence of antibodies.
Data Analysis
Comparisons of the frequency differences between the pediatric and the adult patients for the underlying disease groups were performed using the Kruskal-Wallis and chi-square tests. The Wilcoxon signed-rank test was used to measure the changes in the DIC scores and the coagulation tests that occurred between the start of TM-α administration and the day after the last TM-α administration.
For the multivariate analysis of the risk factors that affect the development of serious bleeding-associated AEs, we performed a chi-square test in which the univariate analysis utilized 22 background factors to identify the candidate factors. A multiple logistic regression analysis was performed using a stepwise method, with the factors found to be significant in the univariate analysis utilized as the explanatory variables.
Results of the analysis were considered significant when the P value was less than .05. The 2-sided statistical testing was performed using JMP software version 8.0 (SAS Institute Co, Ltd, Cary, North Carolina).
Results
Table 1 presents the patient demographics for each of the underlying disease groups. The median age was 3 years, with a range of 4 weeks to 14 years. For the 3 underlying disease groups of DIC, infections were found in 101 patients, hematological malignancy in 53, and other reasons were the cause in 56. Further examination of the infection group showed that the disease in the majority of patients was caused by sepsis/septic shock (n = 40), meningitis (n = 14), pneumonia (n = 8), and encephalitis/encephalopathy (n = 6). For the hematological malignancy group, acute lymphoblastic leukemia (n = 20), acute myeloid leukemia (n = 7), and acute monocytic leukemia (n = 6) were found. The main causes observed in the patients in the other group included hemophagocytic histiocytosis (n = 9) and neuroblastoma (n = 6).
Baseline Characteristics and Treatment of Pediatric Patients.
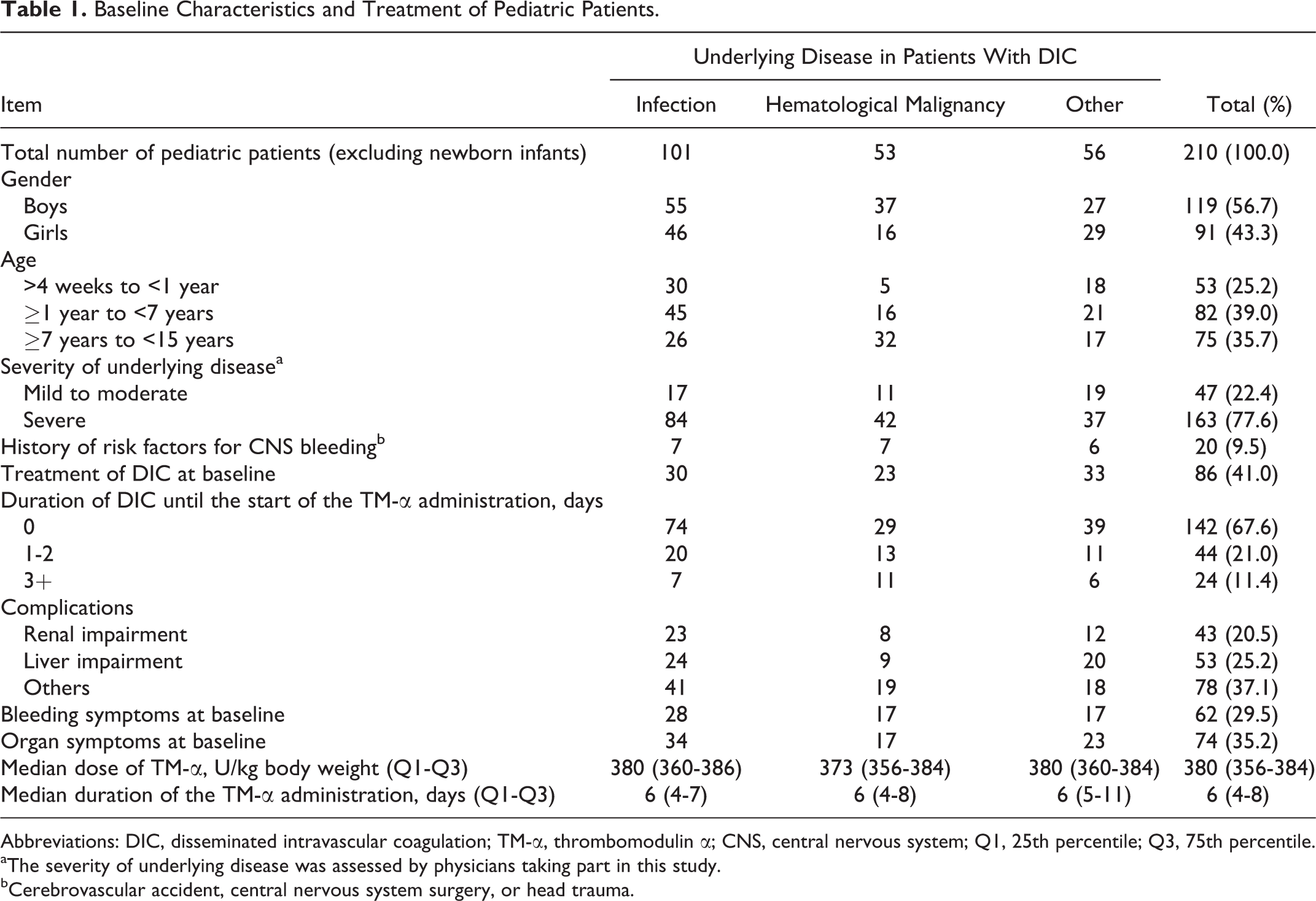
Abbreviations: DIC, disseminated intravascular coagulation; TM-α, thrombomodulin α; CNS, central nervous system; Q1, 25th percentile; Q3, 75th percentile.
aThe severity of underlying disease was assessed by physicians taking part in this study.
bCerebrovascular accident, central nervous system surgery, or head trauma.
The median dose of TM-α administered to all patients was 380 U/kg/d (25th percentile [Q1] to 75th percentile [Q3]: 356-384 U/kg/d). The median TM-α doses administered in each of the 3 groups were not significantly different (P = .682, Kruskal-Wallis test). The median duration of the TM-α administration was 6 days (Q1-Q3: 4-8 days). There were no significant differences noted for the duration of the TM-α administrations between any of the 3 groups (P = .165, Kruskal-Wallis test).
Of the 210 pediatric patients, 68 (67.6%) were treated with TM-α alone while the remaining 142 patients were concomitantly administered TM-α with 1 or more of the other anticoagulants. The commonly used anticoagulants were ATIII concentrates (46.2%), UFH (21.9%), and nafamostat mesilate (17.6%). In addition, blood products were also used in 104 (49.5%) of the 210 patients. The most commonly used blood products were platelet concentrate (38.6%), red cell concentrate (35.7%), and fresh frozen plasma (FFP, 32.4%). Of the 210 pediatric patients, 80 (38.1%) were administered both other anticoagulant(s) and blood product(s).
Table 2 lists the efficacy results, which included the DIC resolution rate and the survival rate. Although there was a tendency for a higher survival rate in the pediatric patients compared with adult patients with DIC, the differences noted for the efficacy (DIC resolution and survival) between the pediatric and adult patients were not found to be significant (P = .710, P = .671, P = .053, respectively, chi-square test). The DIC resolution rates for each group of the underlying diseases were 61.5% for the infection group, 65.2% for the hematological malignancy group, and 45.0% for the other group. The rates were not significantly different between the 3 groups (P = .354, chi-square test). Survival rates for each of the underlying disease groups were 65.0% for the infection group, 77.4% for the hematological malignancy group, and 78.2% for the other group. The rates were not significantly different between the 3 groups (P = .124, chi-square test).
Effectiveness and Safety for Pediatric and Adult Patients.a
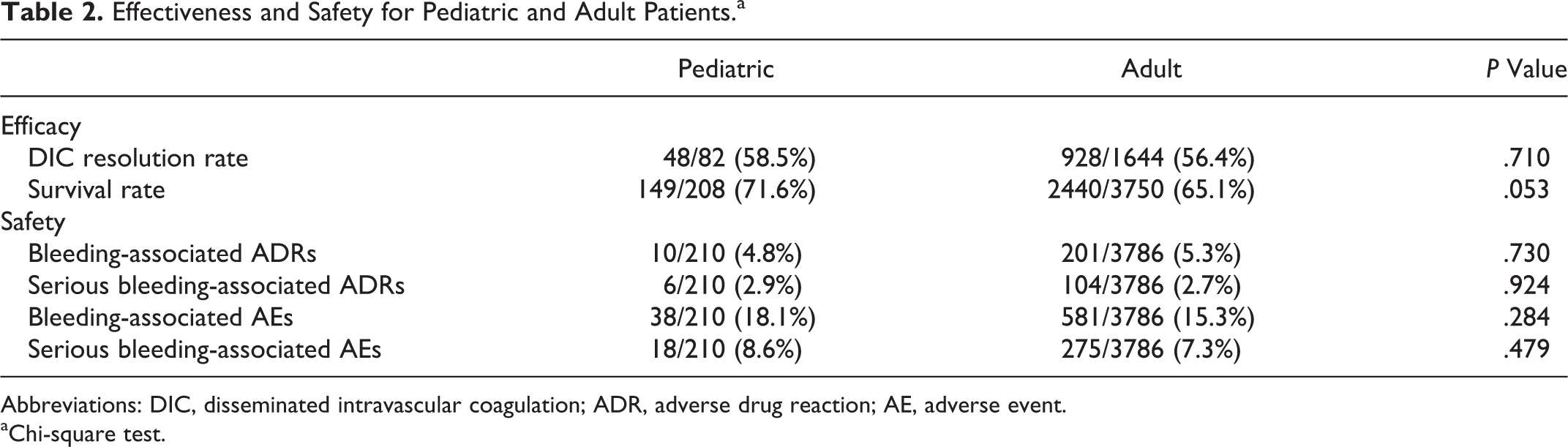
Abbreviations: DIC, disseminated intravascular coagulation; ADR, adverse drug reaction; AE, adverse event.
aChi-square test.
Figure 1 shows the changes in the DIC score for the infection and hematological malignancy groups after TM-α treatment according to the JMHW DIC criteria. The observed decreases in the DIC score were significant in both groups (P < .001, P < .001, respectively; Wilcoxon signed-rank test).
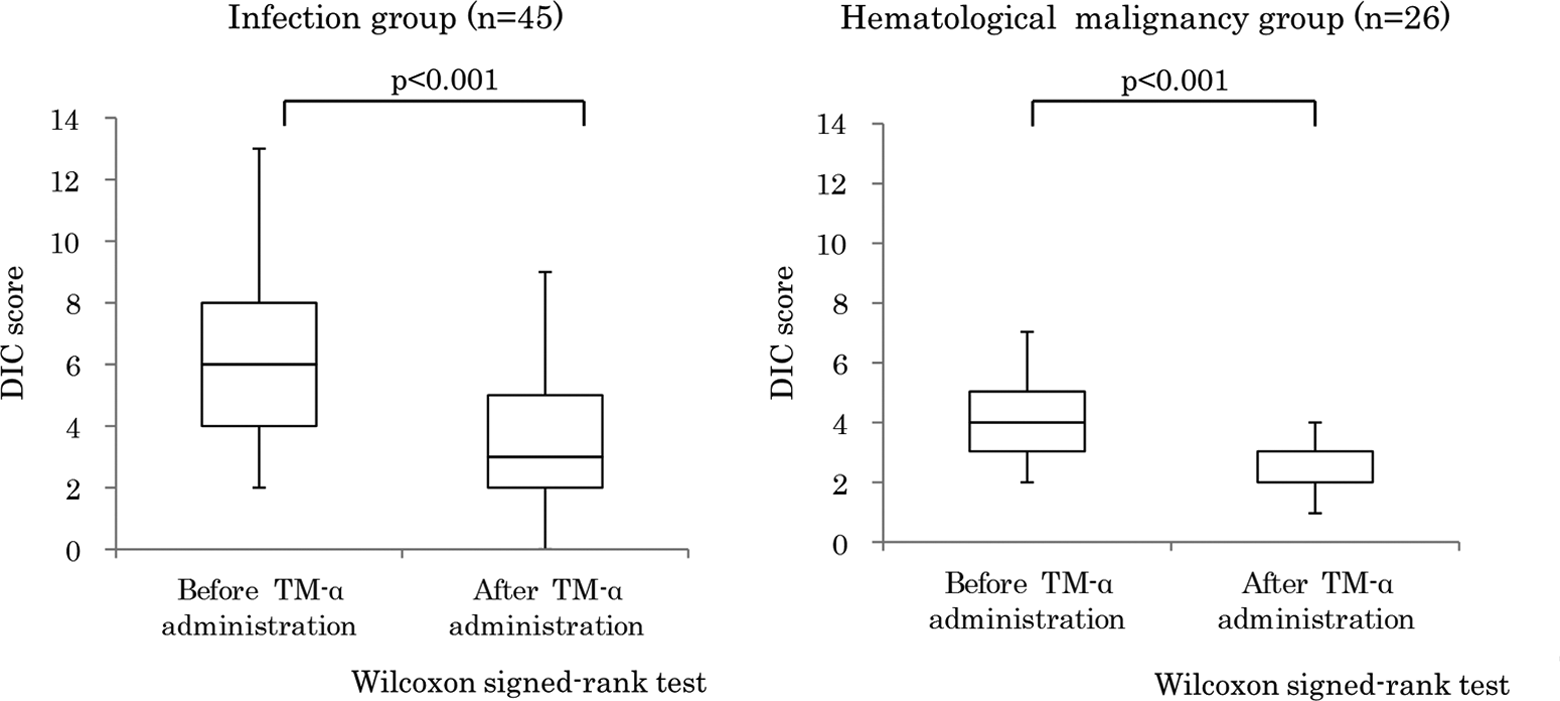
Changes in DIC score after TM-α treatment in the infection and hematological malignancy groups. Values are expressed as means, with error bars indicating the standard deviation. The DIC scores before and after TM-α administration were analyzed statistically. Two-sided P values (Wilcoxon signed-rank test) are shown. DIC indicates disseminated intravascular coagulation; TM-α, thrombomodulin α.
Table 3 shows the changes in the coagulation test and the hematological findings for the infection and hematological malignancy groups during the TM-α treatment. For the patients in the infection group, a significant decrease was seen in the FDP and PT ratio and in the activated partial thromboplastin time on the day after the TM-α administration when compared to the values before the TM-α administration (P < .001, P < .001, P = .039, respectively; Wilcoxon signed-rank test). In addition, significant increases were also seen for the platelet counts, AT, and protein C (P < .001, P < .001, P = .039, respectively; Wilcoxon signed-rank test). For the patients in the hematological malignancy group, there was a significant decrease in the FDP and the PT ratio on the day after the last TM-α administration when compared to the values obtained before the TM-α administration (P < .001, P = .017, respectively; Wilcoxon signed-rank test). Significant increases for AT and protein C were also noted in this group (P = .014, P = .039, respectively; Wilcoxon signed-rank test).
Changes in Coagulation and Hematological Test Findings After TM-α Treatment.a
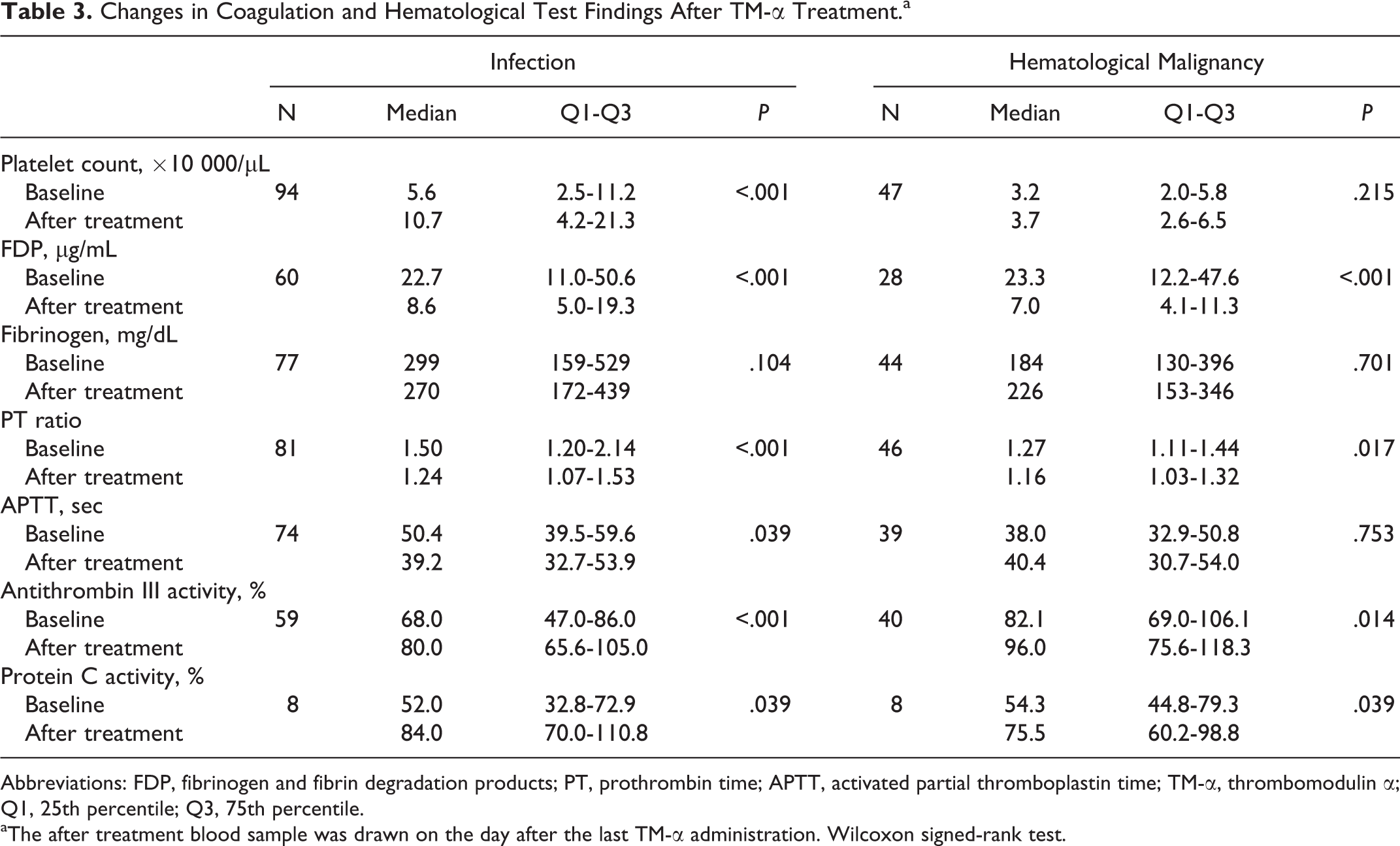
Abbreviations: FDP, fibrinogen and fibrin degradation products; PT, prothrombin time; APTT, activated partial thromboplastin time; TM-α, thrombomodulin α; Q1, 25th percentile; Q3, 75th percentile.
aThe after treatment blood sample was drawn on the day after the last TM-α administration. Wilcoxon signed-rank test.
In 11 of the 210 pediatric patients with DIC, there were 19 episodes of ADRs reported in which a causal relationship with the TM-α administration could not be ruled out. The ADRs observed included 3 episodes of epistaxis, 2 episodes of pulmonary bleeding, and 1 episode each of hemorrhagic cerebral infarction, pulmonary alveolar bleeding, respiratory tract bleeding, hemothorax, oral bleeding, gastrointestinal bleeding, small intestinal bleeding, melena, hematochezia, urinary bladder bleeding, hematuria, puncture site bleeding, catheter site bleeding, and liver dysfunction. Unexpected ADRs were not observed in the pediatric patients. Of the 6 reported patients with serious bleeding-associated ADRs, there were 3 with gastrointestinal bleeding, 2 with respiratory bleeding, and 1 with a cerebral infarction caused by bleeding. However, none of the patients with serious bleeding-associated ADRs died as a result of the bleeding.
Table 2 presents the rate of patients with bleeding-associated ADRs, serious bleeding-associated ADRs, bleeding-associated AEs, and serious bleeding-associated AEs for the pediatric and adult patients with DIC. There were no significant differences in the safety between the pediatric and adult patients with DIC. The rates of patients with serious bleeding-associated AEs for each of the underlying diseases were 5.0% for infection, 15.1% for hematological malignancy, and 8.9% for the patients in the other group, with no significant differences noted for any of these 3 groups (P = .101, chi-square test).
Table 4 shows the results of the univariate analysis, which was performed in order to identify the background factors that could be affecting the development of serious bleeding-associated AEs. Results showed that the use of concomitant anticoagulants had no influence on the development of serious bleeding-associated AEs. Our multivariate analysis determined that there were 2 background factors that were risk factors, higher age and organ symptoms at baseline. The odds ratio and the 95% confidence interval for age (increases for each year) and organ symptoms at baseline (absent vs present) were 1.12 (1.10-1.24, P = .029) and 3.43 (1.27-9.89, P = .015), respectively.
Results of Logistic Regression Models Used to Evaluate the Effects of Various Factors.
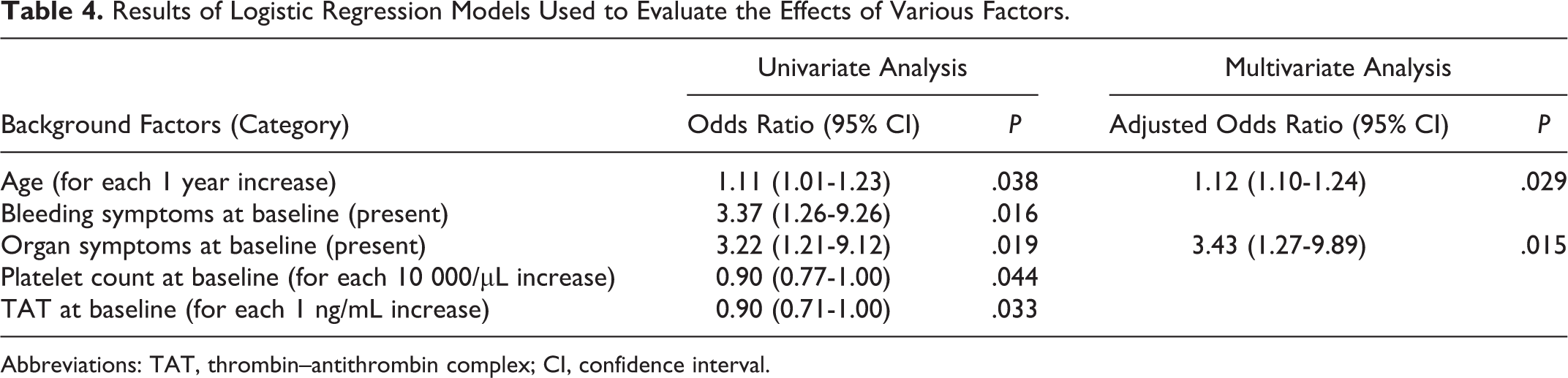
Abbreviations: TAT, thrombin–antithrombin complex; CI, confidence interval.
To investigate the possibility of the production of antibodies induced by the administration of TM-α, 118 serum samples were measured. None of these patients exhibited any positive antibody titers against TM-α.
Discussion
The current PMS study indicated that TM-α appears to be useful for the treatment of DIC in both pediatric and adult patients. Our findings also suggested that there appears to be a tendency for a higher survival rate in the pediatric patients compared with the adult patients. In addition, this study also showed there was a significant improvement in the DIC score and in many of the coagulation tests after the administration of TM-α. On the other hand, this study found that there were no significant differences for the safety between the pediatric and the adult patients, indicating that TM-α administration would be useful for treating DIC in both pediatric and adult patients.
El-Nawawy et al carried out a prospective study in order to evaluate the outcome of early DIC management among high-risk patients who ranged in age from 1 month to 5 years and who were admitted to a pediatric intensive care unit. They reported the mortality of patients with overt DIC and pre-DIC (positive
Since TM-α inactivates coagulation via the activation of protein C, it is thought that the effect of TM-α on DIC can be influenced by the plasma level of protein C. Mohri have reported that TM-α exhibits an adequate inhibition effect on prothrombinase activity when the plasma concentration of TM-α is higher than approximately 1 μg/mL, even if the plasma level of protein C is 10%. 17,18 In the current study, measurement of the plasma level of protein C was performed in 64 patients, and the results showed that there were no patients who had a plasma level of protein C less than 10%.
When TM-α is administered to a pediatric patient with DIC, special attention to serious bleeding-associated ADRs as well as the effectiveness of DIC needs to be carefully considered. Data from the current PMS study showed that serious bleeding-associated ADRs occurred in 6 (2.9%) patients, which was equal to the incidence observed in adult patients with DIC (2.7%). In the placebo-controlled clinical trial (RESOLVE) that was undertaken in order to confirm the usefulness of recombinant APC against pediatric patients with severe sepsis, numerically there were more CNS bleeding events noted in the APC group during the 28-day study period (APC vs placebo: 11 events [4.6%] vs 5 events [2.1%], P = .13). 19 In the current PMS study, there were 2 events (1.0%) with serious CNS bleeding-associated AEs reported (one from the infection group and one from the hematological malignancy group). Unfortunately, it was impossible to compare the large-scale clinical trial with the current PMS study due to differences in the inclusion criteria, patient’s background, and the type of treatment performed in each of the patients. However, it is assumed that the risk of TM-α to provoke and aggravate bleeding will be quite low, as it does not show any anticoagulant action unless thrombin is present.
Since bleeding can be easily induced by consumption of platelets and coagulation factors in patients with DIC, it was difficult to judge whether the bleeding-associated AEs had a causal relationship with the administered anticoagulants in this study. In order to clarify the causal relationship, we attempted to examine the background of patients with serious bleeding-associated AEs. Our investigation determined that there were 18 serious bleeding-associated AEs, with the bleeding most frequently observed in gastrointestinal and respiratory sites, which correspond to the general sites of bleeding. The results of our multivariate analysis determined that higher age was one of the background factors responsible for affecting the development of serious bleeding-associated AEs. Of the 9 patients who had gastrointestinal bleeding, 6 were older than 7 years of age and all of them exhibited complicated thrombotic microangiopathy (TMA) or graft versus host disease (GVHD) after stem cell transplantation. However, in these patients, we believe the underlying disease had a greater influence than their age. Since these patients with DIC having TMA and/or GVHD are prone to bleeding after stem cell transplantation, 20 special caution needs to be taken to ensure that gastrointestinal bleeding will not occur when TM-α is administered. Additionally, as our multivariate analysis also determined organ symptoms at baseline to be a background factor, it is quite likely that further bleeding could occur in severely compromised patients. Therefore, the use of anticoagulants should be carefully evaluated prior to any administration.
It should be noted that there were several limitations for the current PMS study. First, data were collected from patients who were not subjected to any inclusion criteria, as patients with DIC were consecutively registered at the initiation of the TM-α administration. Second, the evaluation time after the start of the TM-α administration differed from patient to patient, as there was no limitation on the duration of the TM-α administration. Third, patients undergoing concomitant use of other anticoagulants or other treatments of DIC were not excluded from the study. Furthermore, there was no pediatric control group in this study. Therefore, it cannot be definitively concluded from the current PMS study data that TM-α is a safe and effective drug in pediatric patients with DIC. Nevertheless, even when these background differences are considered, the results of the current PMS study indicated that TM-α appears to be similarly useful for the treatment of DIC in both pediatric and adult patients. While the results collected from the current PMS study are promising, additional studies that examine the concomitant method of FFP, which contains protein C, in patients who have a low level of protein C activity will need to be undertaken in the future.
Footnotes
Acknowledgments
We thank all participating physicians and registered patients who took part in this surveillance. We would also like to thank Katsuhito Nihashi and Yuka Fujiki for their assistance with the statistical analysis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Naoya Kitamura and Goichi Honda are employees of Asahi Kasei Pharma Corporation. The other authors have no conflicts of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study used the TM-α post-marketing surveillance database. This surveillance was supported in part by the Japanese Society on Thrombosis and Hemostasis.
