Abstract
To explore the possible single nucleotide polymorphisms (SNPs) sites in the promoter region of fibrinogen B β (FGB), and construct logistic regression model and haplotype model, so as to reveal the influence of FGB promoter SNPs on susceptibility, hemodynamics and coagulation function of lower extremity deep venous thrombosis (LEDVT) in the genetic background. LEDVT patients (120) and healthy people (120) were taken as case and control objects, respectively. SNPs and their genotypes of FGB promoter were detected by promoter sequencing and PCR-RFLP. The parameters of coagulation system were evaluated. There were 6 SNPs in FGB promoter, which were β-148C/T, β-249C/T, β-455G/A, β-854G/A, β-993C/T and β-1420G/A. The genotype and allele frequency of β-1420 G/A, β-455G/A, β-249c/T and β-148C/T were significantly different between the LEDVT group and the control group, but not β-993C/T and β-854G/A. In addition, we found that the higher the content of Fibrinogen (FG), the higher the risk of LEDVT. The risk of LEDVT increased by 4.579 times for every unit increase of fibrinogen. We also found that FG, PT and APTT in LEDVT group were higher than those in control group, while TT was lower than those in control group; Furthermore, there was no significant difference in all coagulation indexes among 6 SNP genotypes in LEDVT group, while a significant difference was found between the 2 genotypes of β-993C/T in the control group. β-993C/T may indirectly affect the susceptibility of LEDVT by improving the basic level of plasma FG.
Keywords
Introduction
Lower extremity deep venous thrombosis (LEDVT) is known to be caused by abnormal blood coagulation in the deep vein. 1 At present, deep venous thrombosis DVT (DVT) has been widely recognized as the third most common acute cardiovascular disease after stroke and myocardial infarction. 2 As a common peripheral vascular disease, venous valve insufficiency and pulmonary embolism caused by LEDVT are a major risk to the labor force and life safety of patients. 3 It has been reported that LEDVT also can extend to inferior vena cava, even block renal vein and cause renal failure, which is life-threatening. 4,5 Although there have been many methods used in clinical practice to treat LEDVT, it is still difficult to cure and associated with a high recurrence and sequelae rate. 6,7 Therefore, to propose the effective treatment and primary prevention of DVT is of great clinical significance.
It has been reported that the hypercoagulability caused by the increase of coagulation factors is closely related to the formation of DVT. 8,9 Fibrinogen (FG) is the core protein of coagulation system. 10 In the final stage of coagulation process, FG is converted into fibrin monomer by thrombin, and finally forms insoluble fibrin clot to play an important role in blood coagulation. 11,12 Studies on case-control have shown that increased concentration of plasma FG is an independent risk factor for thrombotic and cardiovascular diseases. 13,14 FG was reported that had 3 polypeptide chains which were encoded by 3 independent genes: fibrinogen α (FGA), fibrinogen β (FGB) and fibrinogen γ (FGG). All these genes are located at the end of fourth chromosome long arm l/3 (4q23 ∼ q32). 15 Since the synthesis of FGB chain is the rate limiting step of FG synthesis, 16 any factor that affects FGB gene mutation or transcription will cause the change of plasma FG content. 17 Although, more and more studies have already focused on the role of FGB gene polymorphism in arterial diseases, 18,19 the correlation between FGB promoter polymorphism and clotting function in patients with LEDVT remains unclear.
It has been known that there are more than 10 FGB promoter single nucleotide polymorphism (SNP) that have been found, in which SNPs closely related to gene expression are mainly located in the promoter region of the β chain transcription initiation site. 20 We wonder whether the FGB promoter gene polymorphism affect the susceptibility of DVT via increasing plasma FG level. Since it has been reported that LEDVT is a common and frequently occurring disease in Yunnan Province, and that is located in the plateau area with a high level of plasma FG, 21 we chose the idiopathic LEDVT patients of Han nationality in Yunnan Province to investigate in this study without the interference of regional and ethnic differences on the experimental results. In addition, multiple logistic stepwise regression analysis was used to select the related genotypes and establish haplotype model to explore the influence on idiopathic LEDVT susceptibility, hemodynamics and coagulation function (including plasma FG level).
Materials and Methods
Study Subjects
From June 2011 to December 2011, 120 patients with idiopathic LEDVT of Han nationality in Yunnan province were included in the present study. All participants were the first time of onset without any treatment (including operation, anticoagulation and thrombolysis, etc.), including 71 males and 49 females, aged 15-80 (53.00 ± 24.75) years old. Routine physical examination was carried out for all patients to exclude heart, brain, liver, kidney, thyroid and adrenal diseases. In addition, patients with malignant tumor, pregnancy, immune and endocrine diseases, bleeding, blood system diseases, trauma and surgical history were excluded, and there was no blood relationship between the above cases. The control group included 120 healthy subjects (59 males and 61 females), aged 8-71 (50 ± 19.75) years old. All of subjects in control group came from physical examination center and outpatient department of First Affiliated Hospital of Kunming Medical University. This study was conducted with Ethics Committee of the First Affiliated Hospital of Kunming Medical University. Written informed consent was obtained from each participant.
Specimen Collection
Fibrinogen and other coagulation functions were detected before treatment on admission. The remaining blood samples were collected and placed in 1.5 ml EP tube and frozen in −80°C for future use (for gene sequencing and PCR-RFLP analysis). Activated partial thromboplastin time (APTT), prothrombin time (Pt), thrombin time (TT), D-dimer and FG level were detected by Sysmex CA-7000 (Japan). D-dimer and FG were determined by immunoturbidimetry.
Gene Sequencing
Peripheral venous blood was collected and stored at −80°C. QiaAmp®DNA Blood Mini Kit (Qiagen, German) was used to extract genomic DNA from peripheral venous blood. All reagents for qRT-PCR were ordered from Takara (Dalian, China). After comprehensive prediction by using multiple promoter prediction software, including Promoter 2.0 Prediction, it is finally determined that the basic range of FGB gene promoter region is -1525- +34, a total of 1560 bp. Primer premier 5.0 was used to design the primers sequence and oligo 6.0 was used to evaluate the primers. The primers needed for the amplification of the FGB gene promoter region were showed in supplementary Table 1.
GeneAmp® PCR System 9600 (Applied Biosystems, USA) was used for the polymerase chain reaction, which was set up to run the following program: 94°C for 3 min, 94°C for 40 s, 58°C for 40 s, and 72°C for 10 min, followed by 32 cycles of 72°C for 10 min, 15°C for 30 min. After the reaction, the PCR products were placed at 4°C for electrophoresis detection or -20°C for long-term preservation. Then, Sanger dideoxy chain termination method was used to complete (1560 bp) sequencing in both positive and negative directions to explore the possible SNP.
Polymerase Chain Reaction-Restriction Fragment Length Polymorphism (PCR-RFLP)
PCR-RFLP was used for the genotype analysis of FGB gene polymorphism. Briefly, the genomic DNA was extracted from blood samples. Primer Premier 5.0 software was used to design the primer of β-1420G/A (rs1800789), β-993C/T (rs2227389), β-854G/A (rs1800791), β-455G/A (rs1800790), β-249C/T (rs1800788) and β-148C/T (rs1800787), which were synthesized by Sangon Biotech Co. Ltd (Shanghai, China), and were shown in supplementary Table 2. The PCR reaction process was: 95°C for 5 min, at 95°C for 30 s, at 68°C for 45 s, at 72°C for 60 s, 20 cycles, then 95°C for 30 s, 58°C for 30 s, 72°C for 40 s, 20 cycles, then extension 6 min at 72°C. β-1420G/A was digested by Bsp143 l, B, β-993C/T was digested by Taal, C, β-854G/A was digested by TaqI, D, β-455G/A was digested by HaeIII, E, β-249C/T was digested by HaeIII, F, β-148C/T was digested by AluI (Figure 1).

Gene polymorphisms of FGB identified by PCR-RFLP assay. A, β-1420G/A was digested by Bsp143 l, B, β-993C/T was digested by Taal, C, β-854G/A was digested by TaqI, D, β-455G/A was digested by HaeIII, E, β-249C/T was digested by HaeIII, F, β-148C/T was digested by AluI.
Statistical Analysis
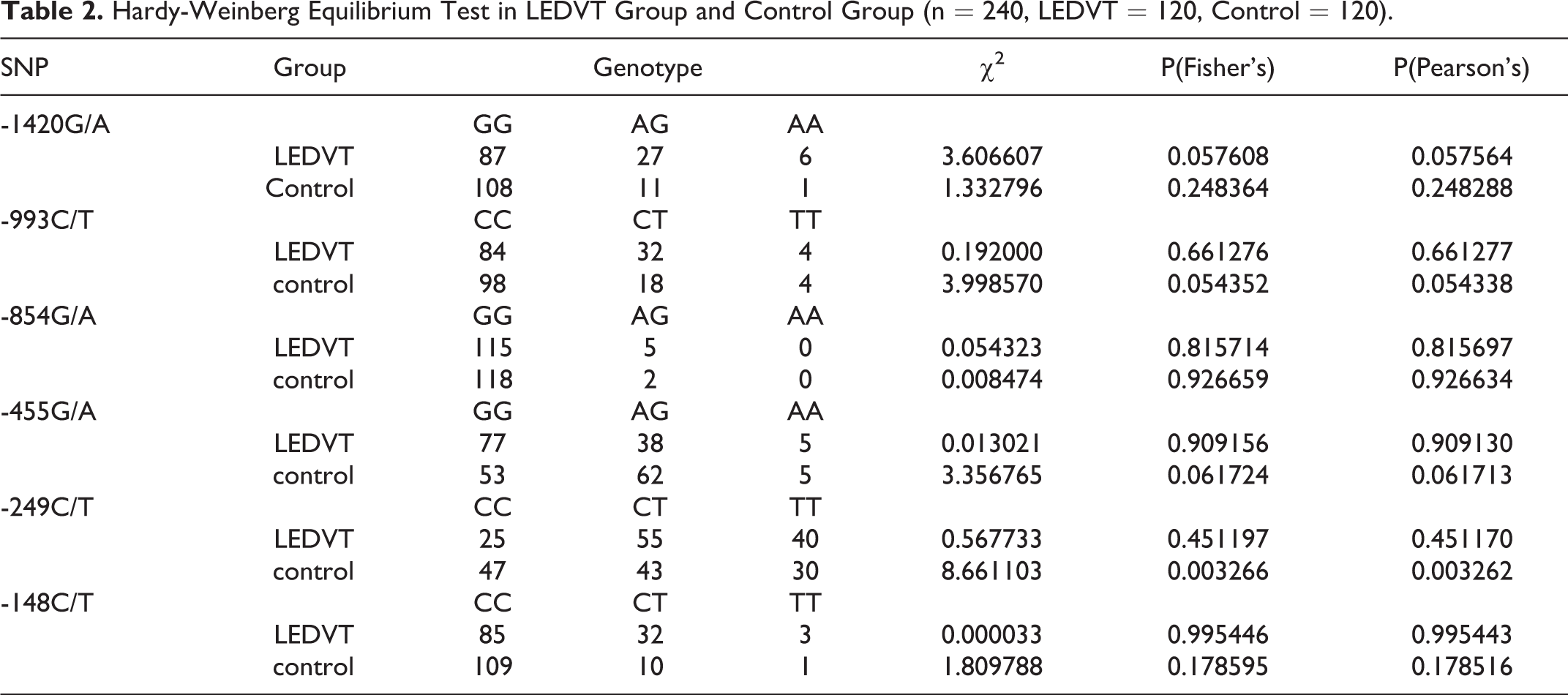
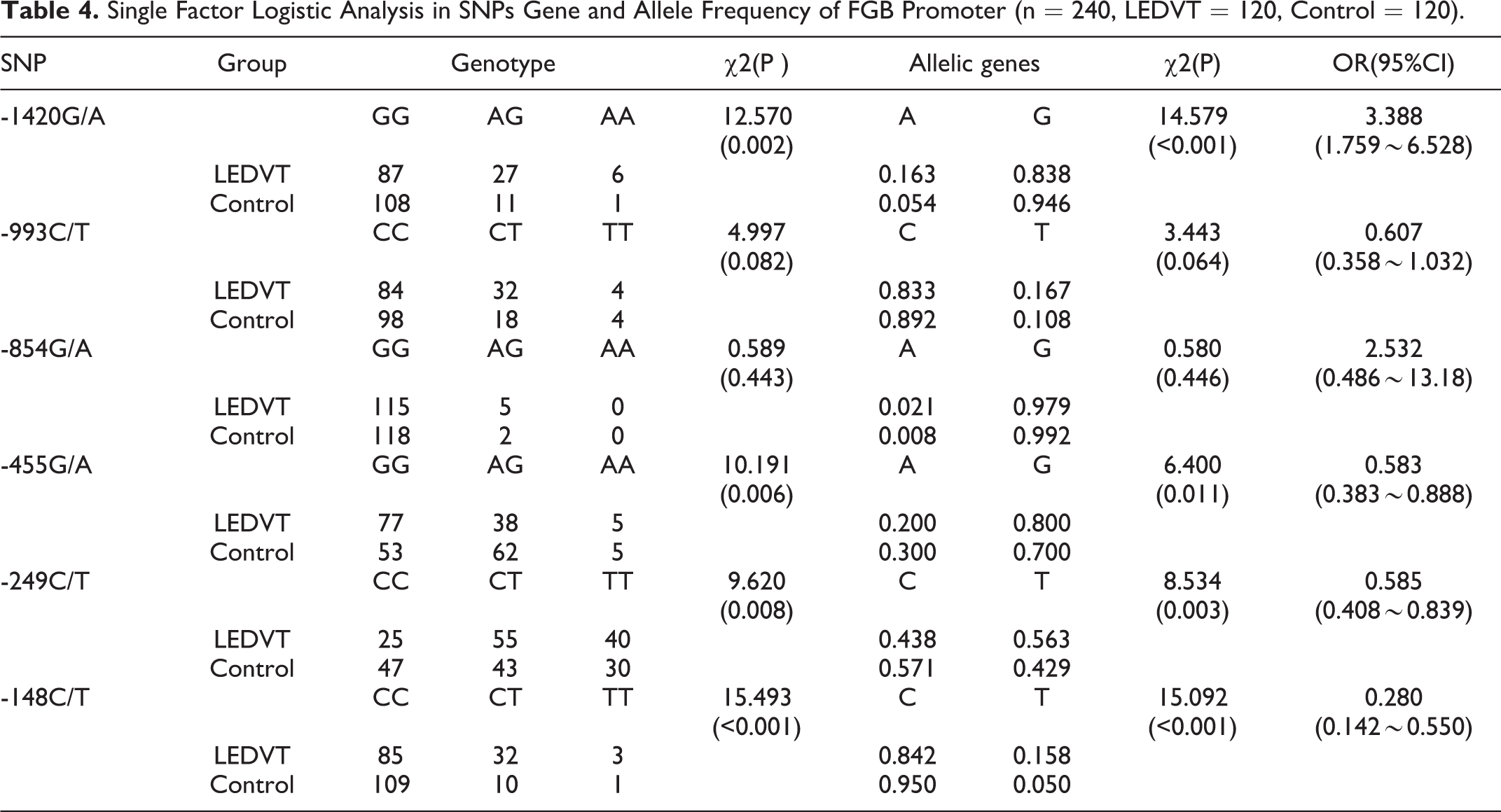
SPSS 17.0 (SPSS Inc., Chicago, IL) was used to analyze the data, which was shown as Mean ± SD or Median ± quartile spacing (M ± Q). The quartile spacing = the upper quartile to the lower quartile (depending on the distribution type of the sample). When compared the 2 groups in this study, Wilcoxon rank sum test or T test was used. Pearson correlation analysis or Fisher exact probability test was used to analyze the correlation. Hardy-Weinberg equilibrium was performed to compare the gene balance between the LEDVT group and Control group (n = 240, LEDVT = 120, Control = 120). Odds ratio (OR) and 95% confidence interval (CI) was used to expresse the differences of genotypes and allele frequency between the case group and the control group. The factors influencing LEDVT was analyzed by Logistic regression analysis. p < 0.05 indicated a statistically significant difference.
Results
The Demographical and Clinical Features of the Participants
As shown in Table 1, no significant difference was found between LEDVT group and control group in the gender and age composition ratio (all p > 0.05), indicating that the 2 groups were comparable in gender and age composition. However, PT, FG and APTT in LEDVT group were higher than that in the control group, while TT was lower than that in the control group.
The Demographical and Clinical Features of the Participants.

Note: mean ± SD.
SNPs of FGB Promoter in LEDVT and Control Groups
The complete sequencing confirmed that there were 6 kinds of SNPs in the FGB promoter region. As shown in Figure 1, they were: β-249c / T, β-148C / T, β-455G / A, β-993c / T, β-854 g / A, and β-1420 g / A. In order to compare the gene balance between the 2 groups from the perspective of genetics, we carried out Hardy-Weinberg balance test in the experimental control group. As shown in Table 2, except for β-249C / T in the control group (χ2 = 8.661103, p < 0.05), all 6 SNPs in the LEDVT group and the other 5 SNPs in the control group were corresponding with Hardy-Weinberg equilibrium (p > 0.05). Moreover, as shown in Table 3, we found that the linkage disequilibrium between β-1420G/A and β-993C/T, β-1420G/A and β-455G/A, β-993C/T and β-455G/A, β-993C/T and β-249C/T, β-1420G/A and β-148C/T, β-993C/T and β-148C/T, β-455G/A and β-148C/T, β-455G/A and β-249C/T, β-249C/T and β-148C/T has statistical significance (p < 0.05). To investigate whether 6 kinds of SNPs in the FGB promoter associated with the risk of LEDVT. As shown in Table 4, the genotype frequency and allele frequency of β-1420G / A, β-455G / A, β-249C / T, β-148C / T were significantly different between LEDVT group and control group (all p < 0.01). The single factor Logistic regression analysis of SNPs showed that the OR values of β-1420A, β-455A, β-249C and β-148C were 3.388, 0.583, 0.585 and 0.280 times of β-1420G, β-455G, β-249 T and β-148 T, respectively. In addition, we further evaluated that association between FG and the risk of LEDVT. As shown in Table 5, the higher the FG content, the higher the risk of LEDVT. The risk of IDVT increased by 4.579 times for every increase of FG.
Hardy-Weinberg Equilibrium Test in LEDVT Group and Control Group (n = 240, LEDVT = 120, Control = 120).
Linkage Disequilibrium Tests in LEDVT Group and Control Group (n = 240, LEDVT = 120, Control = 120).

Single Factor Logistic Analysis in SNPs Gene and Allele Frequency of FGB Promoter (n = 240, LEDVT = 120, Control = 120).
Multivariate Logistic Regression Analysis of LEDVT.

Effect of SNPs in FGB Promoter on Clotting Function
In order to investigate the effect of SNPs in FGB promoter on clotting function, the coagulation function parameters of SNPs were evaluated. We found that no significant difference was found in all coagulation function parameters (PT, FG, TT and APTT) among all SNPs genotypes (β-854 g / A, β-148C / T, β-249c / T, β-993c / T, β-455G / A and β-1420 g / A) in LEDVT group (data not shown, all p > 0.05). Although there was also no significant difference in all coagulation function parameters (PT, FG, TT and APTT) among SNPs genotypes (β-854 g / A, β-148C / T, β-249c / T, β-993c / T, β-455G / A and β-1420 g / A) (data not shown, all p > 0.05), as shown in Table 6, there were significant differences in FG between the β-993C / T 2 alleles in control group (p < 0.05). It is suggested that β-993C / T 2 alleles can affect the basic level of FG in the blood of health people, that is to say, in the basic state of health body, the FG of genotype carrying mutation site T is slightly higher than that of wild homozygote genotype Zygote genotype.
Wilcoxon Rank Sum Test of Coagulation Indexes of β-993C / T Genotypes.

Discussion
DVT is one of most common acute cardiovascular disease after coronary heart disease and stroke. 2 The hospital mortality rate is high, which is the most common cause of sudden death, accounting for about 10% of the total hospital mortality. 3 This brings about a high medical security risk. At present, in addition to the acquired clinical high-risk factors, the risk factors of congenital heredity have been paid more and more attention. 21,22 Therefore, systematic and in-depth study of patients with LEDVT is conducive to avoid the influence of confounding factors, so as to reveal the internal genetic rule of LEDVT occurrence and development, and provide reference for disease prevention and treatment, especially for screening and intervention of high-risk LEDVT population. In the present study, the clinical features of the participants suggesting that the clotting function was different in LEDVT patients compared with healthy people. We confirmed that there were 6 kinds of SNPs in the FGB promoter region by complete sequencing and PCR-RFLP. Among them, the genotype and allele frequency of β-1420 G / A, β-455G / A, β-249c / T and β-148C / T were significantly different between the LEDVT group and the control group, but not β-993C/T and β-854G/A. Furthermore, there was no significant difference in all coagulation indexes among 6 SNP genotypes in LEDVT group, while a significant difference in FG was found between the 2 genotypes of β-993C/T in the control group.
As the core protein in coagulation system, FG plays important role in blood coagulation and / or hemostasis. 23 Thrombotic test confirmed that there was a strong correlation between pulmonary embolism and plasma fibrinogen level. 24 Lowering the level of plasma FG was also confirmed that it could help to the treatment of cardiovascular diseases such as coronary heart disease and deep vein thrombosis of the lower extremities. 25,26 In molecular structure, 3 polypeptide chains—α, β and γ of FG are encoded by 3 independent genes FGA, FGB and FGG respectively . In the direction of transcription, the synthesis of β-polypeptide chain is just opposite to that of α chain and γ chain, so the synthesis of fibrinogen β-polypeptide chain is the rate limiting step of the whole FG molecule synthesis . Multivariate logistic regression analysis indicate that FG is a risk factor of LEDVT. As a kind of acute phase reactive protein, FG can be synthesized and released in large quantities under stress or inflammatory reaction. Meanwhile, FG can promote the adhesion of neutrophils to endothelial cells through intercellular adhesion factor-1, thus causing vascular intima damage. 27,28 FG can also act as a matrix to promote the aggregation and aggregation of neutrophils and platelets. 29 FG is the substrate of thrombin and directly participates in the coagulation process. In addition, FG promotes platelet aggregation, increases blood viscosity, and increases erythrocyte adhesion, all of which promote thrombosis formation. 30
At present, it has been found that there are more than 10 SNPs of FGB gene, while most of these studies are from Caucasians in the West. 31 Genetic risk factors are often different among different races and regions. 32 The genetic risk of DVT in FGB promoter region polymorphism has not been reported in Chinese Han population. Our study will contribute to the understanding and treatment of Chinese Han LEDTV patients. In our study, the basic range of FGB gene promoter from Han nationality in Yunnan Province was determined to be −1525 (upstream) ∼ + 34 (downstream), a total of 1560 bp. The complete sequencing and PCR-RLFP confirmed that there were 6 SNPs in the FGB promoter. The combination of the 2 methods provides a more convenient method for DVT related gene chip manufacturing in the future, that is, only need to quickly detect the key SNP sites of mutation, and do not need to obtain all the sequence information. The results showed there was a statistical difference in FG content between −993C and −993 T genotypes in the health examination group, but no statistical difference in PT, TT, and APTT. However, in the LEDVT group, no statistical difference was found in PT, TT, FG and APTT among all SNPs genotypes. These results indicate that the FG level of −993 T allele (CT + TT) is slightly higher than that of −993CC wild homozygote, that is −993 T may affect the susceptibility of LEDVT by improving the basic level of plasma FG. It should be noted that although there was no statistical difference in coagulation indexes among the genotypes of all SNPs in the LEDVT group, the effect of SNPs in the FGB promoter on the coagulation function of LEDVT cannot be negated. As mentioned in a previous experiments, LEDVT is actually a dynamic development pathophysiological process. 33 When the body’s coagulant anticoagulant fibrinolytic network is activated one after another, the clotting state is reversed with the remission and recurrence of thrombus, which is not unchangeable. 34 Therefore, in the natural course of LEDVT, hypercoagulation or hypocoagulation may occur alternately and present dynamic balance, even difficult to define. The apparent result of coagulation test seems contradictory, but it is reasonable. Since LEDVT is the common result of many genes involved in human body, 35,36 the role of a single gene is not enough to have a decisive impact on its occurrence and development. A previous study has been suggest that the environment may have a great influence on the FG level of these populations, even exceeding the genetic effect. 37
Like most disease-related SNPs study, LEDVT research is difficult to grasp the dynamic changes of course related parameters, and it is difficult to obtain hemodynamic and coagulation parameters in the first time, which makes the results qualitative but not quantitative. There is a limitation in this study. Smoking, 38 hypertension, 39 obesity (body mass index) 40 and other factors may also be related to the level of FG, but we did not take these factors into account because of the lack of partial information of patients in the multi factor analysis. Although we have not found the there was significant difference in coagulation function (PT, FG, TT and APTT) among all 6 SNPs in FGB in LEDVT group, with the large-scale joint exploration of multiple genes in the future, it is gradually possible for us to predict the risk of LEDVT in advance in the genetic level. Early intervention, early detection, removal, anticoagulation, and even surgery (including without vena cava filter) intervention will have a positive effect on the prevention and treatment of DVT in DVT high-risk population, so as to reduce incidence rate, disability and mortality.
Supplemental Material
Supplemental Material, sj-pdf-1-cat-10.1177_1076029620967108 - The Correlation Between FGB Promoter Polymorphism and Clotting Function in Patients With Idiopathic Lower Extremity Deep Venous Thrombosis
Supplemental Material, sj-pdf-1-cat-10.1177_1076029620967108 for The Correlation Between FGB Promoter Polymorphism and Clotting Function in Patients With Idiopathic Lower Extremity Deep Venous Thrombosis by Shengbin Han, Bin Yang, Yaoyu Feng, Lingfeng Zhao, Qun Feng, Hongxi Guan, Donghui Song, Fang Yin and Li Zhuang in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present study was supported by the Foundation of Applied Basic Research Program of Yunnan Province (Kunming, China; grant nos. 2019FE001(-216)).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
