Abstract
Background: Information on thrombophilia risk factors for patients with upper extremity deep vein thrombosis (UEDVT) is limited. The genetic, acquired, and coagulation risk factors of an acute episode of lower EDVT (LEDVT) or UEDVT, either isolated or associated with pulmonary embolism (PE), were studied. Materials and Methods: A total of 4503 patients participated in a thrombophilia study. Odds ratio (OR) and 95% confidence intervals (CIs) were calculated. Results: Mean age of the participants was 55 ± 19 years. The risk of LEDVT or UEDVT, isolated or associated with PE, was calculated according to thrombophilia factors. We found association between LEDVT and factor V Leiden ([FVL]; OR: 1.8; 95% CI 1.4-2.4) and resistance to activated protein C ([APC-R]; OR: 1.6; 95% CI 1.1-2.4). The LEDVT + PE presented an association with PTG20210A (OR: 1.5; 95% CI 1.1-2.1). No association was found between the thrombophilic defects studied and UEDVT or UEDVT + PE. Conclusions: Both FVL and APC-R carriers had the risk of developing LEDVT. The PTG20210A carriers had the risk of developing LEDVT + PE. No thrombophilic defects studied presented risk factors for UEDVT or UEDVT + PE.
Keywords
Introduction
Venous thromboembolism (VTE), which includes deep vein thrombosis (DVT) and/or pulmonary embolism (PE), is a multicausal disease.1,2 Its incidence increases sharply with age: it is very rare in young individuals (<1 per 10 000 per year) but increases to ~1% per year in the elderly individuals, which indicates that ageing is one of the strongest and most prevalent risk factors for venous thrombosis. The results obtained from most of the studies undertaken to date indicate that acquired risk factors, malign diseases, the presence of comorbidities, and genetic risk factors such as mutations of the factor V Leiden (FVL) and prothrombin G20210A (PTG20210A) are associated with an increased risk of a lower extremity deep vein thrombosis (LEDVT).2,3 However, the information currently available on risk factors in general and thrombophilia in particular for patients with upper extremity deep vein thrombosis (UEDVT) is limited. 4
The Registro Informatizado de Enfermedad TromboEmbolica (RIETE) initiative is an ongoing, multicenter, international (Spain, France, Italy, Israel, Switzerland, Germany, Argentina, Belgium, Canada, Greece, United States, Portugal, Poland, Republic of Macedonia, Ireland, Egypt, and Venezuela) observational registry designed to gather data on the clinical characteristics, treatment patterns, and outcome in consecutive patients with symptomatic, objectively confirmed, acute VTE.5,6 Data from this registry have been used to study the contribution of genetic and acquired coagulation risk factors to an acute episode of LEDVT or UEDVT, either isolated or associated with PE.
Patients and Methods
Consecutive patients with symptomatic acute DVT, confirmed by objective tests (venography, Doppler ultrasound, impedance plethysmography, or computed tomography scan) for suspected DVT, pulmonary angiography, lung scintigraphy, or helical computed tomography scan for suspected PE, are enrolled in RIETE. Patients were excluded if they were currently participating in a therapeutic clinical trial or if they were not available for the 3-month follow-up. All patients provided oral consent to their participation in the registry, in accordance with the requirements of the ethics committee within each hospital. Patients with central venous catheters (CVCs) were excluded from the study.
Laboratory Methods
Thrombophilia testing was not routinely performed in RIETE, and analyses were performed according to each hospital protocol. The thrombophilia test included homocysteine, factor V Leiden (FVL), and prothrombin G20210A (PTG20210A) mutations, resistance to activated protein C (APC-R), antiphospholipid antibodies (APSs), antithrombin (AT) deficiency, protein C (PC), and protein S (PS) deficiencies, all tested after discontinuation of anticoagulant therapy. The diagnosis of thrombophilia was established as APC-R, PC, PS, or AT deficiency or the presence of FVL, PTG 20210A, or APS. Patients who were homozygous for FVL or PTG20210A and those who were not tested were not considered.
Study Variables and Definitions
The following parameters are recorded in RIETE: patient’s baseline characteristics, clinical status including any coexisting or underlying conditions (chronic lung disease, chronic heart failure, abnormal creatinine levels [1.2 mg/100 mL], or risk factors (postoperative, immobility, cancer, prior VTE, recent travel, estrogenic treatment during the past 2 months, and varicose veins), and location of the VTE.
Immobilized patients are defined as patients who had been immobilized for ≥4 days in the 2 months prior to VTE. Postoperative patients are defined as those who underwent an operation in the 2 months prior to VTE. The etiology of VTE was defined as secondary (especially in those with recent surgery, immobility, cancer, estrogen use, leg varicosities, or who traveled recently) or idiopathic. 7 Body mass index (BMI) was calculated in kilogram per meter squared.
Fatal PE was defined as any death occurring shortly (<10 days) after PE diagnosis (either the initial episode or recurrent PE), in the absence of an alternative cause of death. Major bleeding was defined as a bleed that required a transfusion of 2 or more units of blood, was retroperitoneal, spinal or intracranial, or was fatal.
Active cancer was defined as newly diagnosed cancer or when receiving antineoplasic treatment of any type (surgery, chemotherapy, radiotherapy, hormonal therapy, support therapy, or combined treatments). Recent travel was defined as any travel for >6 hours occurring less than 3 weeks before VTE diagnosis. Varicose veins were defined as subcutaneous dilated veins equal to or more than 3 mm in diameter measured in the upright position. 8 Type, dose, and duration of treatment received on VTE diagnosis and clinical results after anticoagulant therapy were recorded.
Data Collection and Monitoring
Physicians ensure that eligible patients are consecutively enrolled. Data are recorded onto a computer-based case report form at each participating hospital and submitted to a centralized coordinating center through a secure Web site. Data encoding is used to enhance confidentiality and security. Data quality is regularly monitored and documented electronically to detect inconsistencies or errors, which are resolved by the local coordinators. Data quality is also monitored by periodic visits to participating hospitals or by contracted research organizations that compare the medical records with the data on the Web. A data audit is performed at periodic intervals. Patients' identities remain confidential as they are identified only by a number assigned by the study coordinating center, which is responsible for all data management.
Statistical Analysis
Odds ratios (ORs) and 95% confidence intervals (CIs) were calculated using unconditional logistic regression. Crude models and models adjusted for fibrinogen, chronic lung disease, chronic heart failure, abnormal creatinine levels, postoperative, immobility, cancer, prior VTE, travel >6 hours in the last 2 weeks, BMI, estrogenic treatment during the past 2 months, varicose veins in the lower extremities, and idiopathic VTE were employed. All tests of statistical significance were 2 sided. All these statistics were calculated using the SPSS v 17.
Results
Between January 2001 and December 2008, 22 847 patients with symptomatic, acute VTE had been enrolled in RIETE. Of these, 4503 (19.7%) patients were tested for thrombophilia: 41% presented positive results, of whom 8.4% had FVL, 6.8% had PTG20210A, and 3.0% had APC-R. Mean age of the population was 55 ± 19 years. Patients with UEDVT were 12 years younger than those with LEDVT (42.7 ± 17.8 and 54.8 ± 18.7, respectively). Their clinical characteristics and outcomes are shown in Table 1 .
Clinical Characteristics, Risk Factors, and 3-Month Outcomes in Patients With Test of Thrombophilia of the RIETE Registry

Abbreviations: LEDVT, lower extremity deep vein thrombosis; PE, pulmonary embolism; UEDVT, upper extremity deep vein thrombosis; UFH, unfractioned heparin; LMWH, low-molecular-weight heparin; AVK, anti-vitamin K; SD, standard deviation.
The risk of LEDVT or UEDVT, either isolated or associated with PE, was calculated according to thrombophilia factors. The OR calculated expresses the association between the different characteristics of the VTE and the different types of thrombophilic abnormalities. In the unadjusted analysis, an association was found between LEDVT and FVL (OR: 1.8; 95% CI 1.5-2.3), and APC-R (OR: 1.7; 95% CI 1.2-2.5), and PS deficiency (OR: 1.5; 95% CI 1.0-2.1). No association was found between any of the thrombophilic defects studied and UEDVT or UEDVT + PE (Table 2 ).
Univariate Analysis of the Risk of LEDVT, LEDVT + PE, PE, UEDVT, and UEDVT + PE Associated With Thrombophiliaa
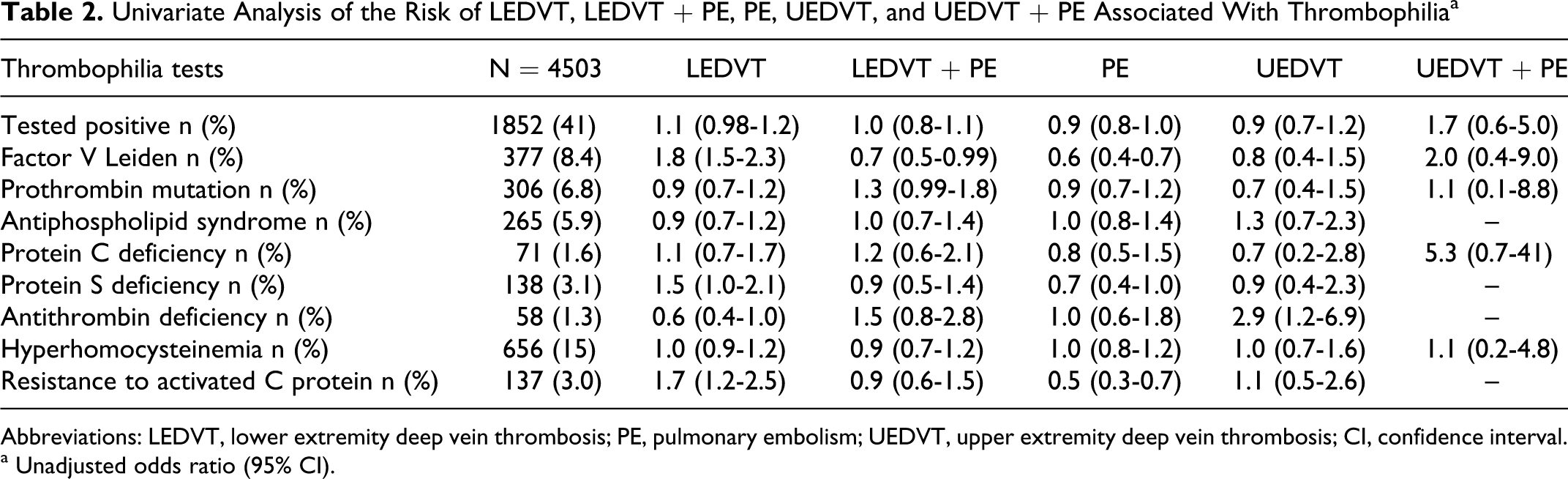
Abbreviations: LEDVT, lower extremity deep vein thrombosis; PE, pulmonary embolism; UEDVT, upper extremity deep vein thrombosis; CI, confidence interval.
a Unadjusted odds ratio (95% CI).
In order to study the possible effect of the factors associated with these illnesses, a new analysis was undertaken adjusted for the following variables: recent journeys, fibrinogen, creatinine, surgery, chronic lung disease, chronic heart failure, immobilization, cancer, BMI, use of estrogens, prior VTE, and varicose veins. The association found in the crude analysis was of approximately the same magnitude for LEDVT and FVL (OR: 1.8; 95% CI 1.4-2.4) and APC-R (OR: 1.6; 95% CI 1.1-2.4). However, the statistical significance was lost with PS deficiency, and LEDVT + PE presented a positive association with PTG 20210A (OR: 1.5; 95% CI 1.1-2.1). No association was found between any of the thrombophilic defects studied and UEDVT or UEDVT + PE (Table 3 ).
Multivariate Analysis of the Risk of LEDVT, LEDVT + PE, PE, UEDVT, and UEDVT + PE Associated With Thrombophiliaa
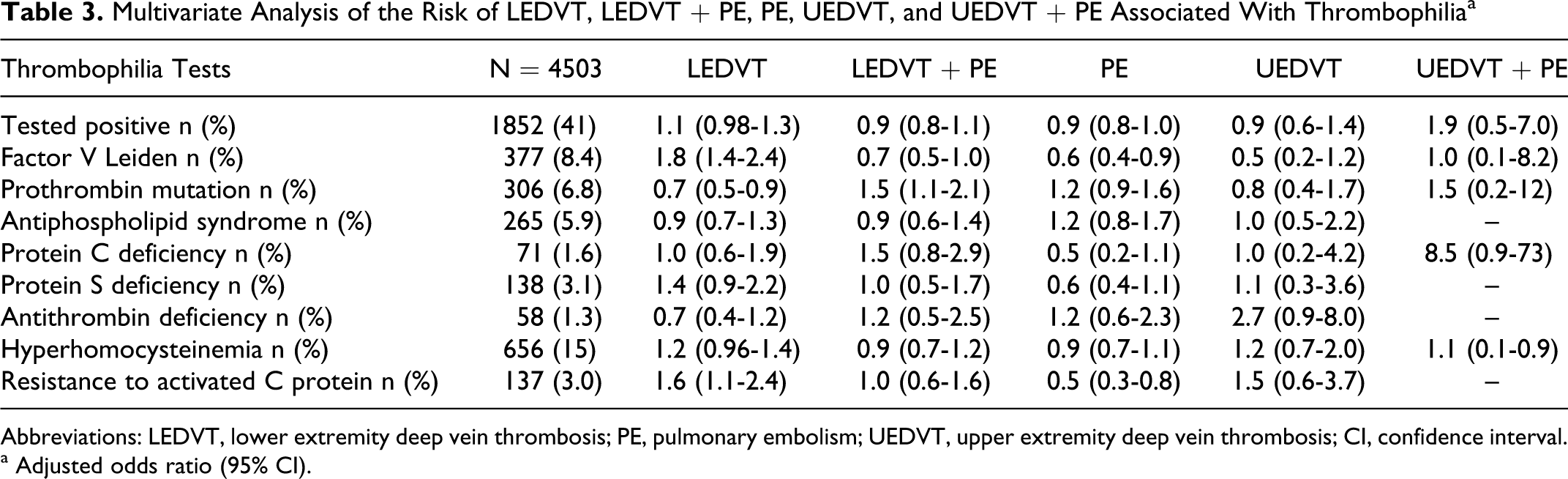
Abbreviations: LEDVT, lower extremity deep vein thrombosis; PE, pulmonary embolism; UEDVT, upper extremity deep vein thrombosis; CI, confidence interval.
a Adjusted odds ratio (95% CI).
Discussion
From the 4503 patients objectively diagnosed with VTE on whom a thrombophilia study was carried out, 41% presented positive results. This figure is very similar to that published by other authors,9,10 where thrombophilic defects were observed in at least 30% to 40% of patients with a first episode of VTE, FVL and PTG20210A being the most common.
Our results show that both FVL and APC-R are low risk factors (OR: 1.8 and 1.6, respectively), only for LEDVT. Likewise, PTG20210A has also been shown to be a low risk factor only for the LEDVT + PE. These findings remain valid even after adjusting for confounding factors and are in line with those of other studies in which the FVL and the PTG20210A, are not very relevant as risk factors for VTE.1,11–13 Studies that have analyzed thrombophilia in relation to age have observed that between 15 and 30 years of age the FVL and PTG20210A are high risk factors, which then become medium risk factors between the ages of 31 to 45 years. However, it is from 46 years of age when they decrease as risk factors for VTE.1,11–13 In part, this could explain our results, as our population has a high mean age. These results suggest that although conventional risk factors, malign diseases, and the presence of comorbidities and genetic factors are associated with an increased risk of thrombosis, the incidence of the disease increases as the age of the patient increases subsequently. The effect of a factor on the risk of thrombosis may be different in young and middle-aged populations compared with the elderly individuals.2,3 Age, therefore, becomes the strongest and the most prevalent determinant of the risk of a first event of thrombosis.
In our study, none of the thrombophilic defects studied were shown to be risk factors for patients with UEDVT, even despite being more than 12 years younger than patients with LEDVT, at manifestation. This observation is in agreement with those of other investigators14,15 who reported that advancing age and overweight, 2 of the most common risk factors for LEDVT, do not predispose to UEDVT. A review of other data indicates that a role for familial thrombophilia and in particular FVL and PTG20210A in UEDVT has not been demonstrated conclusively.16,17 All these suggest that genetic risk factors would play an even less prominent role in the pathogenesis of the UEDVT than in that of the LEDVT.
Various clinical studies4,18 have shown that malign diseases are the important risk factors for UEDVT, something that has not been proved true in our case. One possible explanation could be the exclusion of patients with CVC in our study. Population-based data revealed that more than 60% of all patients with UEDVT had a history of recent CVC placement at the corresponding upper extremity. 19 Large-scale registers have also provided contemporary profiles of patients with UEDVT underlining the paramount role of CVC placement and cancer for both development and outcome of UEDVT. 20
The proportion of patients with UEDVT + PE was 0.3%. The data available on the frequency of PE among patients with UEDVT vary between the 0.75% of the large registers of patients with UEDVT not associated with CVC 20 and the 30% of patients with UEDVT of whichever etiology. 21 Patients with UEDVT differ from those with LEDVT with regard to the incidence of clinical and laboratory risk factors, including age and carriage of thrombophilic defects. Patients with UEDVT have a lower degree of in vivo hemostatic system activation and in vitro thrombin generation capacity and are at a much lower risk of concomitant PE. 15
We should, however, not overlook the possible objections that this type of register may provoke. Nevertheless, we are aware that the RIETE registry also provides data on the management of patients with VTE in a real-world situation with a unselected patient population. It can help to identify practices for providing treatment to patients and factors associated with better or worse patient outcomes as it helps when designing new randomized clinical studies.5,6,22 One limitation is that testing was performed according to each hospital protocol, and some centers probably did not test for all known thrombophilias. This has led to some data not being available for either APC-R or the factor V Leiden tests and, therefore, having results for only 1 test, when there should ideally be the same number for both factors. Certainly, this could lead to an underestimation of some thrombophilic disorders. However, the proportion of patients testing positive in our series was similar to that reported in several prospective studies.3,22–24 This is important because, unlike the careful patient selection that characterizes clinical trials, our patient population reflects routine, unmonitored medical practice involving a broad spectrum of patients with VTE. Selection bias was avoided by including consecutive patients with objectively confirmed, symptomatic, VTE who were referred to study centers.
In short and with regard to the risk found in this study in relation to thrombophilia, only FVL and APC-R carriers were at risk, even though this risk of developing an LEDVT is low. For patients with LEDVT+PE the mutation PTGA2021A was the only risk factor. In contrast, none of the thrombophilic defects studied appeared to be a risk factor for UEDVT or UEDVT + PE.
With regard to antiphospholipid syndrome, the prevalence in patients with a first episode of VTE ranges from 5% to 8% in different studies,25,26 which corresponds with the results of our study. However, most VTE cases are not linked to specific effects of coagulation or to underlying medical conditions, 27 which could be due to the fact that VTE is a multifactorial disease that depends on the individual’s genotype, on other genetic defects, and on the influence of environmental risk factors, such as oral contraceptive pills, cancer, or APS itself, which may interact with each other. All these mean that some individuals do not develop VTE, whereas others develop severe and recurrent episodes at early ages. 24
Bearing in mind all the above, we think that the information contributed by our results should make us reflect upon the usefulness of indiscriminately carrying out thrombophilia tests in clinical practice as these would only be indicated in those patients with a suspected risk of recurrent thrombosis, defined by a clinical, personal, or family history of VTE, of family thrombophilia, under 50 years of age, or to continue with high
In conclusion, for all these reasons and in accordance with the recommendations made by other authors and international bodies,30,31 the undertaking of thrombophilia tests on patients with a first episode of VTE is not advisable.
Footnotes
Appendix A
Acknowledgments
We express our gratitude to Sanofi-Aventis Spain for supporting this Registry with an unrestricted educational grant. We also express our gratitude to Bayer Schering Pharma for supporting this Registry. Bayer Schering Pharma’s support was limited to the international part of RIETE (excluding patients from Spain), which accounts for 11.13% of the total patients included in the RIETE Registry. We also thank the Registry Coordinating Center, S & H Medical Science Service, for their quality control, logistic, and administrative support and Prof Salvador Ortiz, Universidad Autónoma de Madrid and Statistical Advisor S&H Medical Science Service for the statistical analysis of the data presented in this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Manuel Monreal (Spain) is a coordinator of the RIETE Registry. Dr Hervè Decousus (France) Dr Paolo Prandoni (Italy), and Dr Benjamin Brenner (Israel) are the RIETE Steering Committee Members. Dr Raquel Barba (Spain), Dr Pierpaolo Di Micco (Italy), and Dr Karine Rivron-Guillot (France) are the RIETE National Coordinators. S&H Medical Science Service is the center of RIETE Registry Coordinating Center
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
