Abstract
Background
The aim of this study was to develop and validate a prediction model for predicting the risk of preoperative asymptomatic lower extremity venous thrombosis (LEVT) in patients with knee osteoarthritis (KOA).
Methods
Patients with KOA diagnosed at our hospital between March 2022 and July 2023 were included in this study. Five factors were identified as independent risk factors for the development of preoperative asymptomatic LEVT in patients with KOA by univariate and multivariate logistic regression analysis and a prediction model was constructed by analyzing the results. The performance of the model was evaluated using receiver operating characteristic (ROC) curves. The accuracy of the model and its predictive ability were evaluated using calibration curves. The clinical application value of the prediction model was evaluated using decision curve analysis (DCA).
Results
A total of 354 patients diagnosed with KOA were included in the study. Multivariate logistic regression analysis revealed HDL <1.03 mmol/L (P = 0.004), LDL (P = 0.039), Na+ (P = 0.032), Ca2+ (P = 0.024), and D-dimer >0.55 mg/L (P = 0.049) were the independent risk factors for the occurrence of preoperative asymptomatic LEVT in patients with KOA. The combined-variable ROC curve suggested greater predictability and accuracy (AUC = 0.843, specificity = 90.3%, sensitivity = 71.4%). The calibration curve showed good model agreement and DCA suggested good clinical value of the model.
Conclusion
Low HDL, high LDL, Na+, Ca2+, and D-dimer levels can be used as independent risk factors for the preoperative prediction of asymptomatic LEVT in patients with KOA, and the present study also provides a simple prediction model for clinicians and patients.
Keywords
Introduction
Knee osteoarthritis (KOA) is a common degenerative joint disease that leads to the degeneration, wear and tear of the knee cartilage, which in turn leads to pain, stiffness, and dysfunction. 1 KOA occurs predominantly in older females, with approximately 30% of patients having imaging manifestations of OA, 16% having symptomatic KOA, and 8% having severe KOA.2–5 Treatment modalities for symptomatic KOA include conservative and nonconservative treatments. Conservative treatment includes weight loss, moderate exercise, nonsteroidal anti-inflammatory drugs, injectable steroids, and some adjuvant medications; nonconservative treatment includes arthroscopic debridement, unicondylar knee arthroplasty (UKA), high tibial osteotomy (HTO), and total knee arthroplasty (TKA); and TKA is an effective treatment modality for patients with persistent pain, dysfunction, and advanced imaging manifestations that can significantly reduce pain, restore joint function and improve quality of life.4,6,7
Knee replacement is currently one of the main treatment methods for KOA. With the increasing acceptance of knee replacement and improved quality of life requirements, knee replacement is being chosen by an increasing number of at-risk populations for the treatment of KOA. According to global total joint arthroplasty (TJA) registry data, hundreds of thousands of TKAs are performed annually worldwide, and in the United States, more than 700,000 TKAs are completed annually.5,8,9 In contrast, lower extremity venous thrombosis (LEVT) is a common and serious complication after TJA and is the main cause of mortality and high economic burden after TJA. 10 Symptomatic deep vein thrombosis (DVT) and/or pulmonary embolism (PE) have been reported to occur in approximately 0.45%-5.30% of patients after TKA. 10 One study 11 showed that the incidence of symptomatic DVT associated with postoperative TKA was 1.9% in an Asian population. The incidence of DVT prior to TJA was approximately 17.9%, with a 6.85%-17.9% chance of preoperative DVT in TKA.12,13 In addition, Song et al 14 reported that 66.7% of patients who were diagnosed with venous thrombosis (VTE) prior to TJA could develop VTE at the same site postoperatively.
Preoperative LEVT not only leads to an increased risk of postoperative complications and death but also prolongs patient hospitalization and recovery time. Although several studies have been conducted to investigate the risk factors and prevention strategies for LEVT in patients with KOA, there are insufficient studies on preoperative LEVT in TKA, and most of the studies are still focused on the treatment and prevention of postoperative DVT after TKA. To predict the risk of LEVT, several studies have proposed risk stratification models and scoring systems, such as the Caprini score15–17; however, the correlation and validity of this score with those of the TKA are still controversial, and further research is needed. 18 Therefore, accurately assessing a patient's thrombotic risk and taking appropriate preventive measures are essential to ensure the success of the procedure and the safety of the patient. The aim of this study was to retrospectively analyze the risk factors associated with the preoperative development of asymptomatic LEVT in TKA patients and to construct a nomogram prediction model for use in the clinic to assist clinicians in making more appropriate treatment decisions.
Materials and Methods
Data Source and Cohort Selection
All patients admitted to our hospital with a diagnosis of KOA from March 2022 through July 2023 were collected through the hospital's electronic medical records system. Figure 1 shows the patient enrollment process for this study. We excluded patients who met one or more of the following criteria: had a fracture of the affected limb within one month, had lower extremity varicose veins or symptomatic LEVT, had used of anticoagulant or antiplatelet drugs in the last 3 months, had coagulopathies, had cancer, or had missing or incomplete medical records.
Flowchart for analyzing the development and risk factors for preoperative asymptomatic lower extremity venous thrombosis (LEVT) in knee osteoarthritis (KOA).
Basic patient information included age (years) (≤60, 61–65, 66–70, > 70), gender (male or female), blood type (A, B, AB, O), BMI (kg/m2) (<18.5, 18.5–23.9, 24–27.9, ≥ 28), smoking status, and drinking status. The clinical features included hypertension, diabetes, coronary heart disease, cerebral infarction, a history of other comorbidities, and Kellgren-Lawrence stage (Ⅰ, Ⅱ, Ⅲ, Ⅳ). Preoperative laboratory tests revealed red blood cell (RBC), white blood cell (WBC), hemoglobin (Hb), blood platelet (PLT), albumin (Alb), triglyceride (TG), high density lipoprotein (HDL), low density lipoprotein (LDL), K+, Na+, Cl−, Ca2+, P, Mg2+, plasma fibrinogen (FIB), D-dimer, fibrinogen degradation products (FDP), blood sedimentation (ESR), and lower extremity venous ultrasound findings (no LEVT or LEVT). All of the above tests were completed within 3 days of the patient's admission to the hospital. Among the hematological indices mentioned above, categorical variable analysis can more accurately describe the different effects of different levels of HDL-C on venous thrombosis because of the distribution of HDL-C levels in the study and the nonlinear relationship of HDL-C levels in specific physiopathological processes; therefore, the HDL-C factor was included as a categorical variable in the analyses of this study.
All patients underwent ultrasonography followed by diagnosis by two experienced sonographers. The positive criteria for venous thrombosis in the lower extremities were obstruction or stenosis of the venous trunk, filling defects in the lumen of the vessel, and altered blood flow signal. Both DVT and intermuscular vein thrombosis were included in this study.
Statistical Analysis
SPSS 25.0 (IBM Corporation, Armonk, NY, USA) was used for all the statistical analyses. Quantitative data are expressed as the mean ± standard deviation, and qualitative data are expressed as numbers and percentages. Depending on the type of data, an independent samples t test or the Mann-Whitney test was applied for comparisons between quantitative data, and qualitative variables were used to evaluate differences in demographic and clinical characteristic variables between patients with or without asymptomatic LEVT using Pearson's chi-square test and the rank-sum test. Variables that were statistically significant in the univariate logistic regression analysis were included in the multivariate logistic regression analysis to determine the risk factors for the preoperative development of asymptomatic LEVT in patients with KOA. GraphPad Prism 9.5 software was used to construct the subject-work characteristic (ROC) curve. The ROC curve was used to evaluate the predictive value of the model for the preoperative occurrence of asymptomatic LEVT in patients with the KOA. The nomogram clinical prediction model was developed using multivariate logistic analysis based on the ‘rms’ R package in R 4.3.2 software. The predictive performance of the nomogram was then validated by calibration curve analysis and decision curve analysis (DCA). P values <0.05 were considered to indicate a statistically significant difference.
Results
Patient Selection and Characteristics
The flow chart for patient inclusion and exclusion is shown in Figure 1. The data of 389 patients with KOA from March 2022 to July 2023 were collected, and 35 ineligible patients were excluded according to the exclusion criteria, resulting in the inclusion of 333 patients without preoperative LEVT and 21 patients with preoperative lower-extremity venous thrombosis. According to the presence or absence of preoperative LEVT, the patients were divided into the Non-LEVT group and the LEVT group, with 1 patients having DVT and 20 patients having intermuscular vein thrombosis (IVT) in the LEVT group.
The demographic and clinical characteristics of the included patients are summarized in Table 1. In this study, the participants ranged in age from 47–85 years, with a mean of 65.60 ± 6.18 years and a total of 102 males and 252 females, and the prevalence of preoperative asymptomatic LEVT was significantly greater in females than in males (5.1% of all patients and 85.7% of the incidence population). All patients who developed preoperative asymptomatic LEVT had a BMI outside the normal range. The preoperative comorbidities included hypertension (n = 167), diabetes (n = 51), coronary heart disease (n = 28), cerebral infarction (n = 28), and others (n = 46).
Baseline of the Demographic and Related Clinical Characteristics among Patients.
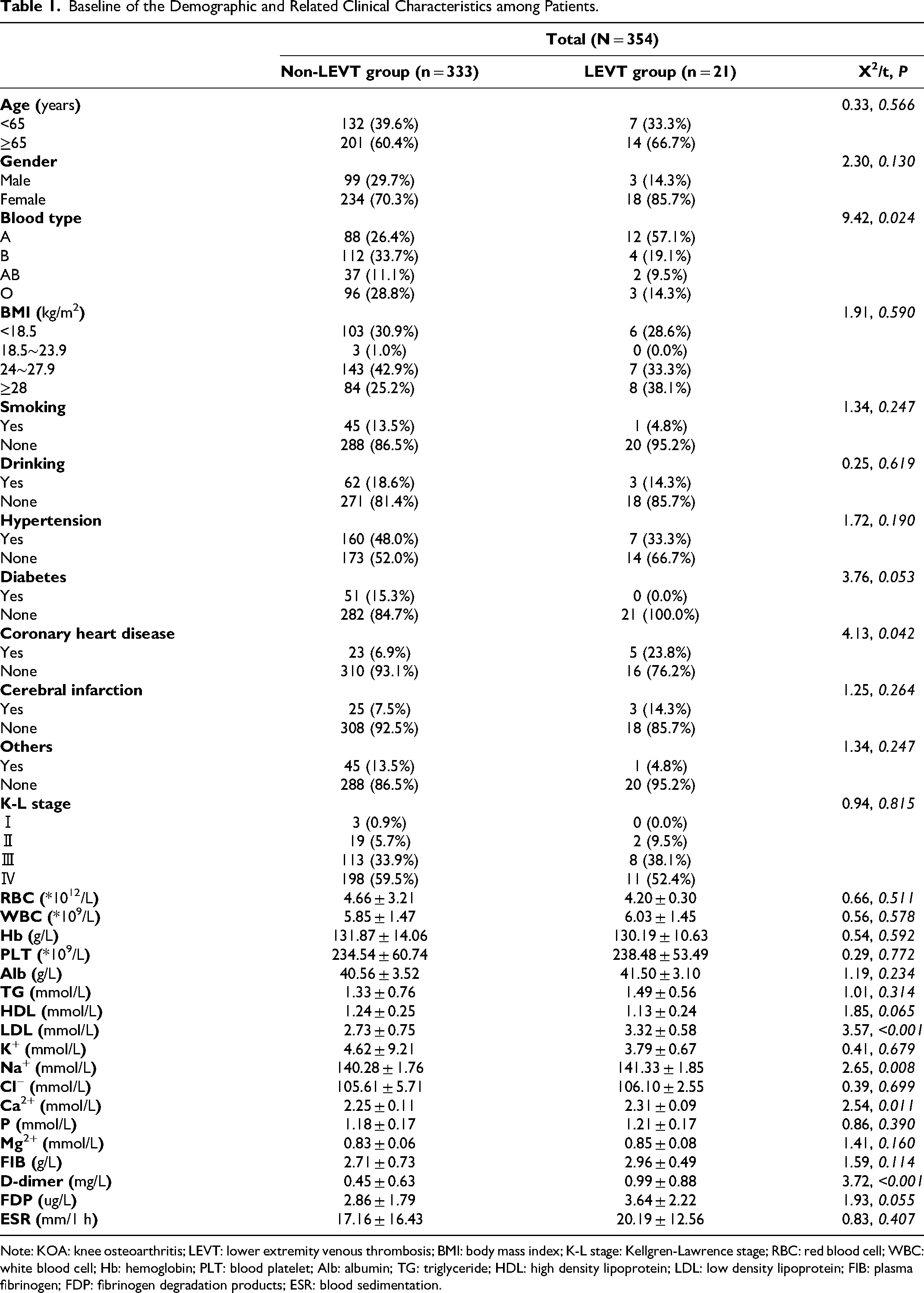
Note: KOA: knee osteoarthritis; LEVT: lower extremity venous thrombosis; BMI: body mass index; K-L stage: Kellgren-Lawrence stage; RBC: red blood cell; WBC: white blood cell; Hb: hemoglobin; PLT: blood platelet; Alb: albumin; TG: triglyceride; HDL: high density lipoprotein; LDL: low density lipoprotein; FIB: plasma fibrinogen; FDP: fibrinogen degradation products; ESR: blood sedimentation.
Between-Group Comparison of non-LEVT Versus LEVT
According to the data analysis, there were statistically significant differences in blood group (P = 0.024), coronary heart disease (P = 0.042), LDL (P < 0.001), Na+ (P = 0.008), Ca2+ (P = 0.011), and D-dimer (P < 0.001) between the LEVT group and the group without LEVT (Table 1, Figure 2), and there were no differences between the remaining groups.
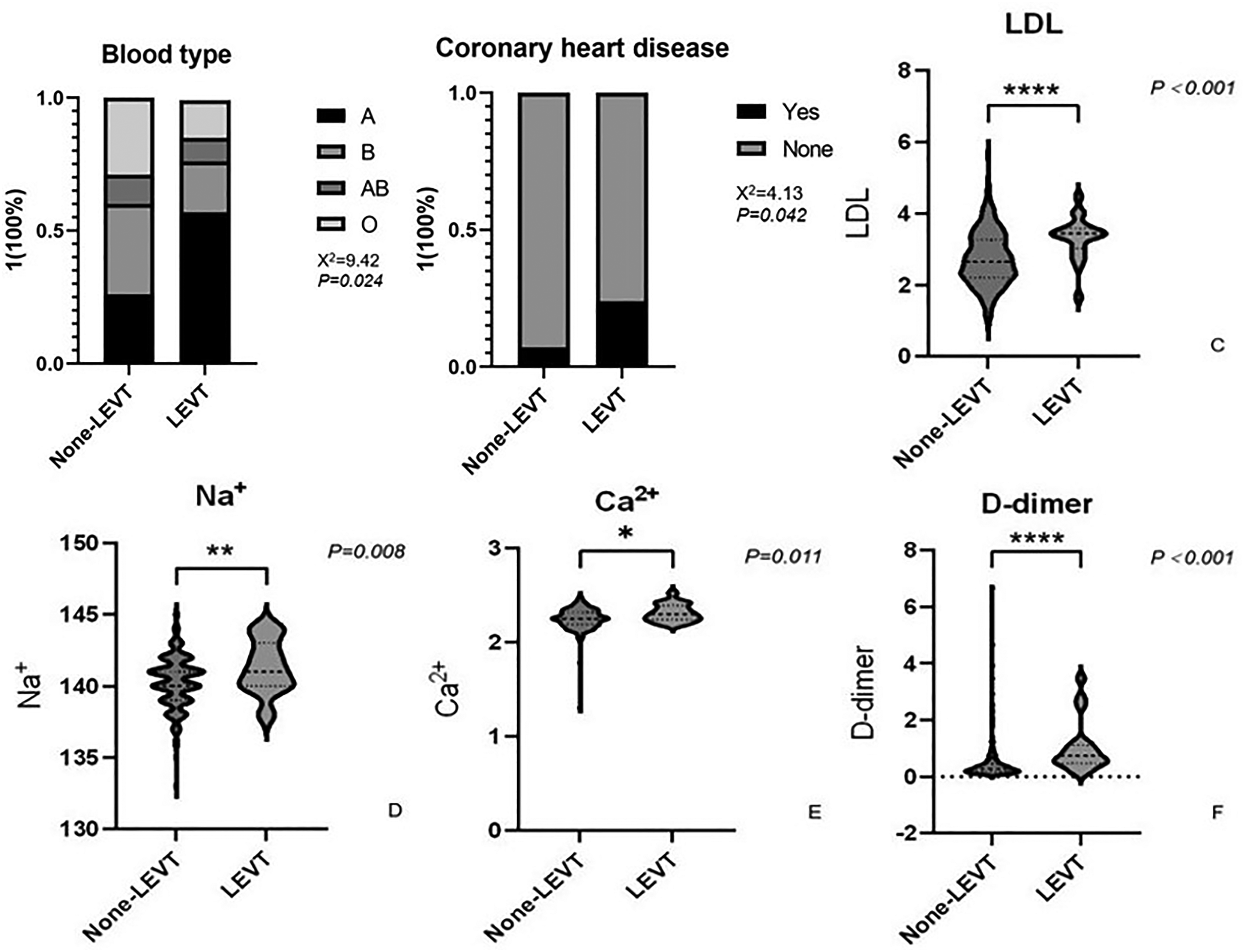
Between-group comparison of none lower extremity venous thrombosis(non-LEVT) versus lower extremity venous thrombosis (LEVT). The chi-square test for blood type(A) and Coronary heart disease(B) between the two groups; The unpaired t-test for low density lipoprotein (LDL) (C), Na + (D),Ca2 + (E) and D-dimer(F) between the two groups. *P<0.05, **P<0.01, ***P<0.005, ****P<0.001.
Analysis of High-Risk Factors for Preoperative Asymptomatic LEVT
Table 2 describes the results of the univariate logistic regression analysis of the occurrence of preoperative asymptomatic LEVT. According to univariate analysis, the risk factors for preoperative asymptomatic DVT in all patients were body mass index (BMI), history of coronary artery disease comorbidity, high-density lipoprotein (HDL) < 1.03 mmol/L, low-density lipoprotein (LDL), Na+, Ca2+, D-dimer (D-dimer) > 0.55 mg/L, FDP >5 ug/mL, ESR >15 mm/1 h (P < 0.05). An interesting finding in our study was that blood groups B and O were protective factors for preoperative LEVT compared with blood group A (P < 0.05).
Univariate Logistic Regression Analysis of Factors Associated with Preoperative Asymptomatic LEVT in Patients with KOA.
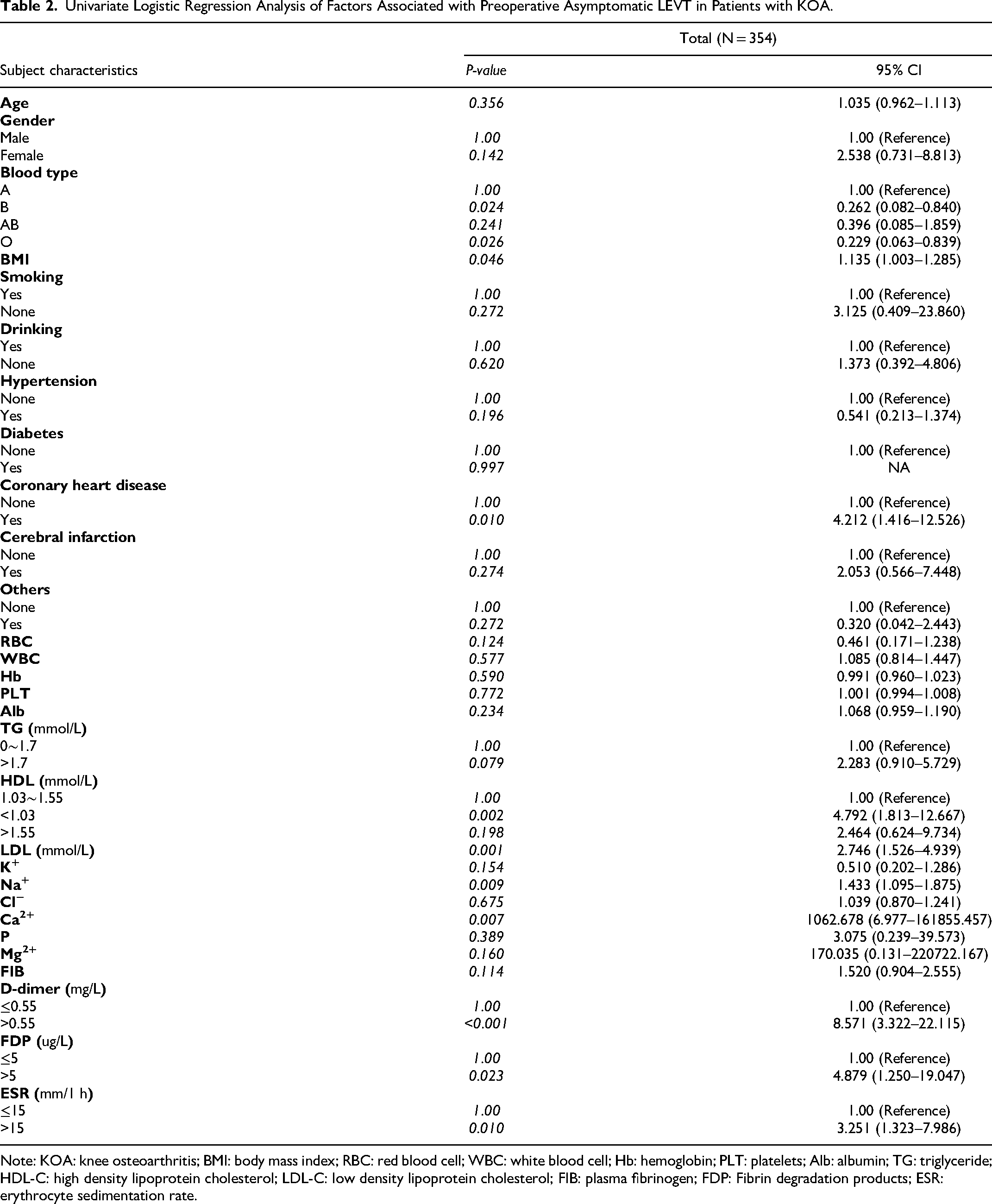
Note: KOA: knee osteoarthritis; BMI: body mass index; RBC: red blood cell; WBC: white blood cell; Hb: hemoglobin; PLT: platelets; Alb: albumin; TG: triglyceride; HDL-C: high density lipoprotein cholesterol; LDL-C: low density lipoprotein cholesterol; FIB: plasma fibrinogen; FDP: Fibrin degradation products; ESR: erythrocyte sedimentation rate.
When the above risk factors were included in the multivariate logistic regression analysis, HDL <1.03 mmol/L (P = 0.004), LDL (P = 0.039), Na+ (P = 0.032), Ca2+ (P = 0.024), and D-dimer>0.55 mg/L (P = 0.049) were found to be independent risk factors for the occurrence of preoperative asymptomatic LEVT in patients with KOA (Table 3). Based on the results of multivariate logistic analysis, we plotted the ROC curves for D-dimer (AUC = 0.7955), HDL (AUC = 0.6509), LDL (AUC = 0.7488), Na+ (AUC = 0.6480), and Ca2+ (AUC = 0.6603) to assess their respective predictive values (Figure 3).
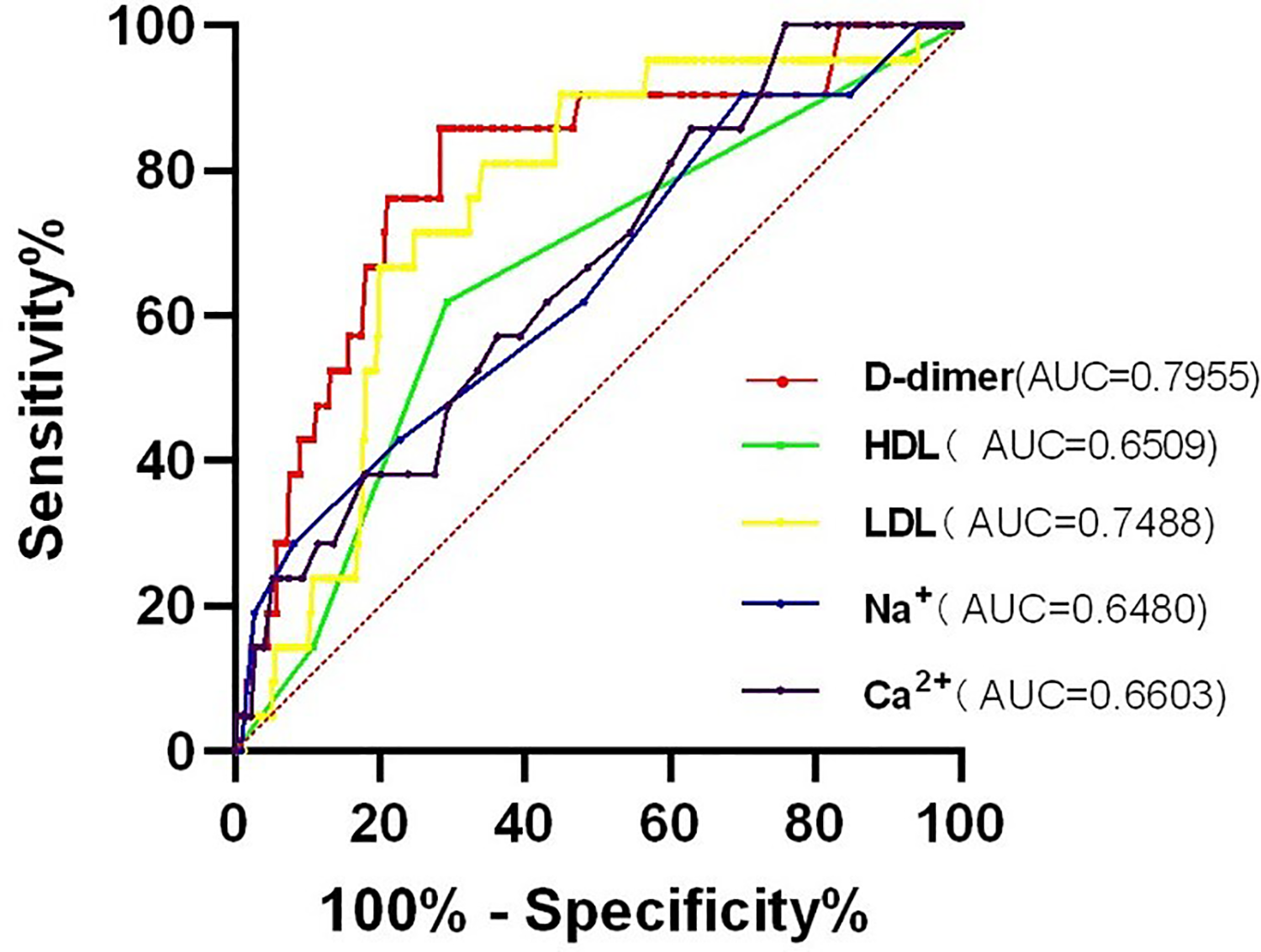
Receiver operating characteristic (ROC) curves plotted based on the results of multifactor logistic regression analysis.
Multivariate Logistic Regression Analysis of Factors Associated with Preoperative Asymptomatic LEVT in Patients with KOA.
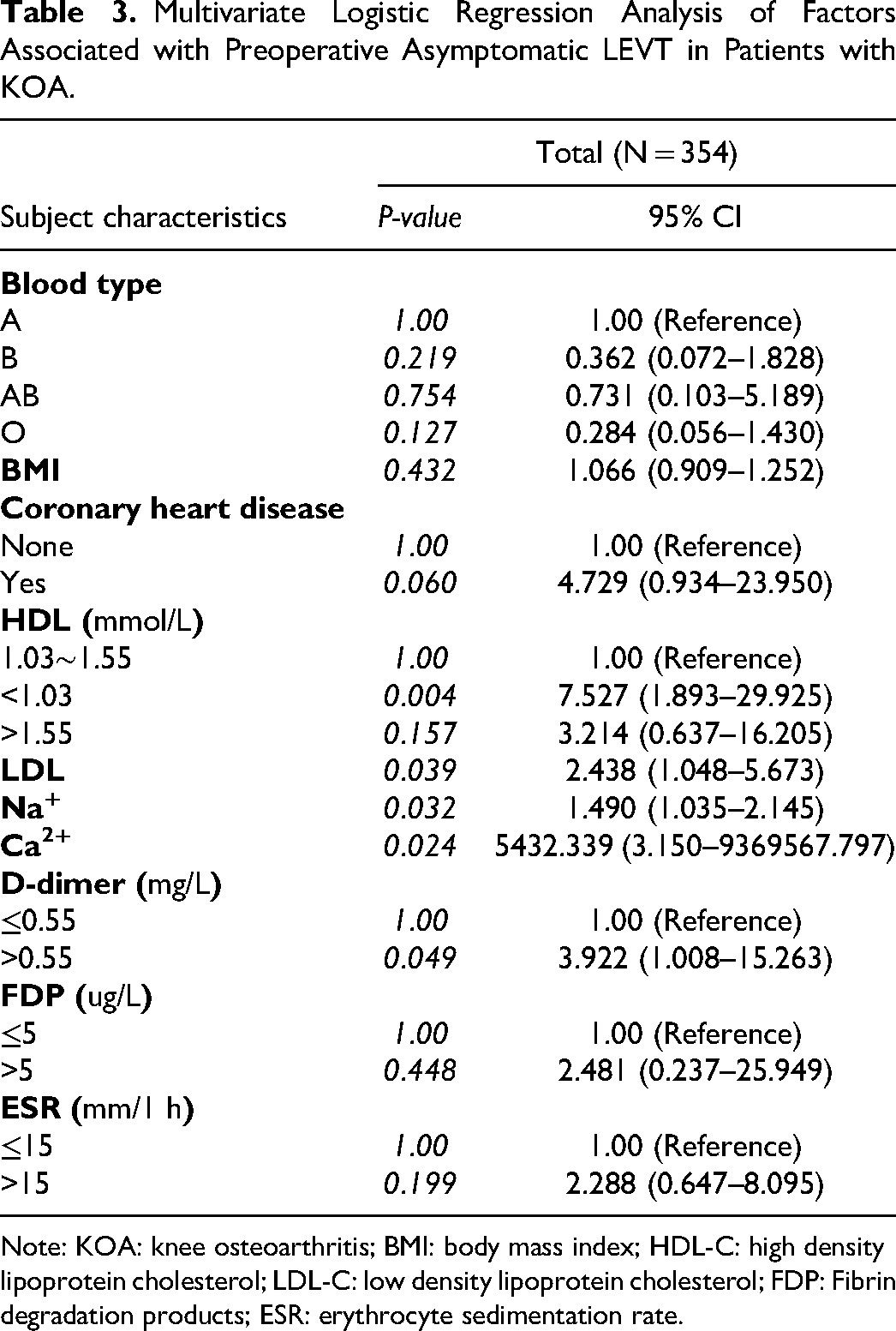
Note: KOA: knee osteoarthritis; BMI: body mass index; HDL-C: high density lipoprotein cholesterol; LDL-C: low density lipoprotein cholesterol; FDP: Fibrin degradation products; ESR: erythrocyte sedimentation rate.
Model Development and Validation
We constructed a nomogram model to predict the occurrence of preoperative asymptomatic LEVT in KOA patients based on the results of logistic regression analysis. The nomogram included five variables: HDL, LDL, Na+, Ca2+, and D-dimer (Figure 4). A ROC curve was used to evaluate the predictive value of each factor, and the joint model was found to be more predictive (area under the curve (AUC) = 0.843, specificity = 90.3%, sensitivity = 71.4%) (Figure 5). The calibration curves showed better agreement between the predictions and the actual risk, with a mean absolute error of 0.018, and the DCA showed that it was more favorable to employ this nomogram model to predict LEVT in all threshold ranges (Figure 6). Taken together, these results suggest that the model has high value for predicting the occurrence of preoperative asymptomatic LEVT in patients with KOA.
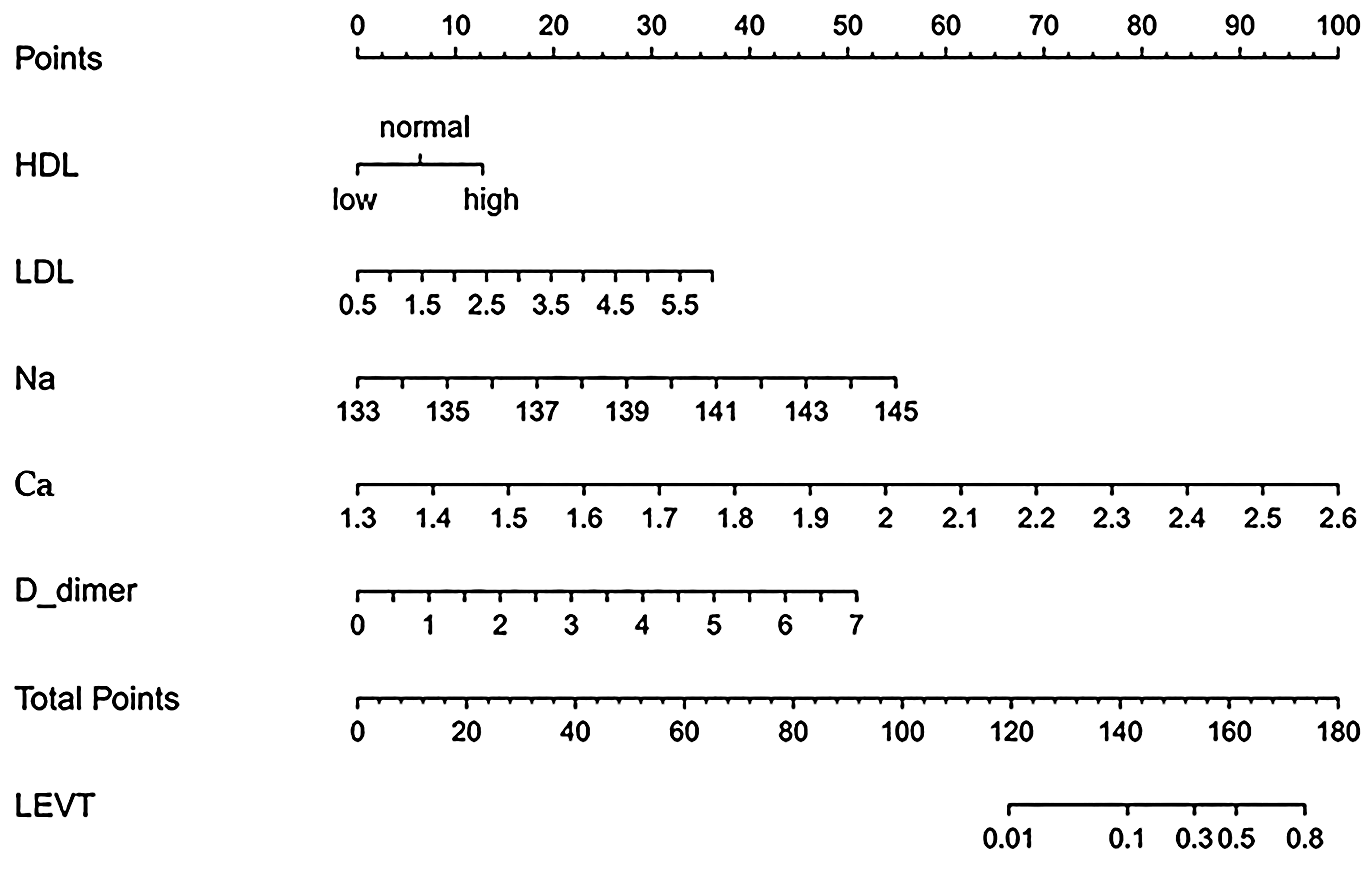
Nomogram to predict the occurrence of preoperative asymptomatic LEVT in patients with KOA. To calculate the incidence of preoperative asymptomatic LEVT in a patient, points for each parameter can be identified from the corresponding risk factor on the point axis and the sum of the points plotted on the total point axis. The incidence of preoperative asymptomatic LEVT in a patient is the probability value of being on a perpendicular line to the corresponding total number of points. LEVT = lower extremity venous thrombosis.
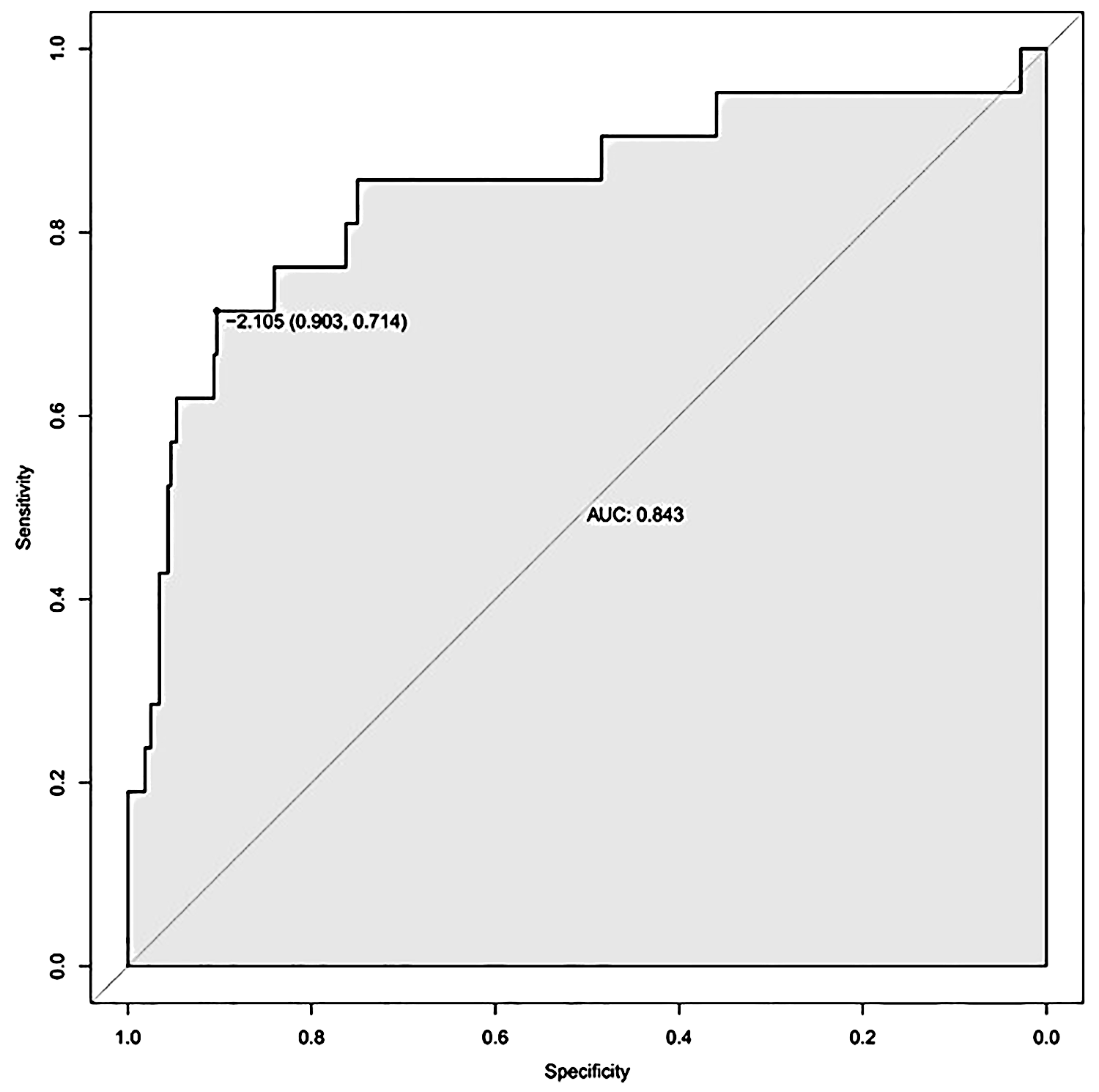
Multifactorial joint model shows better ROC results.
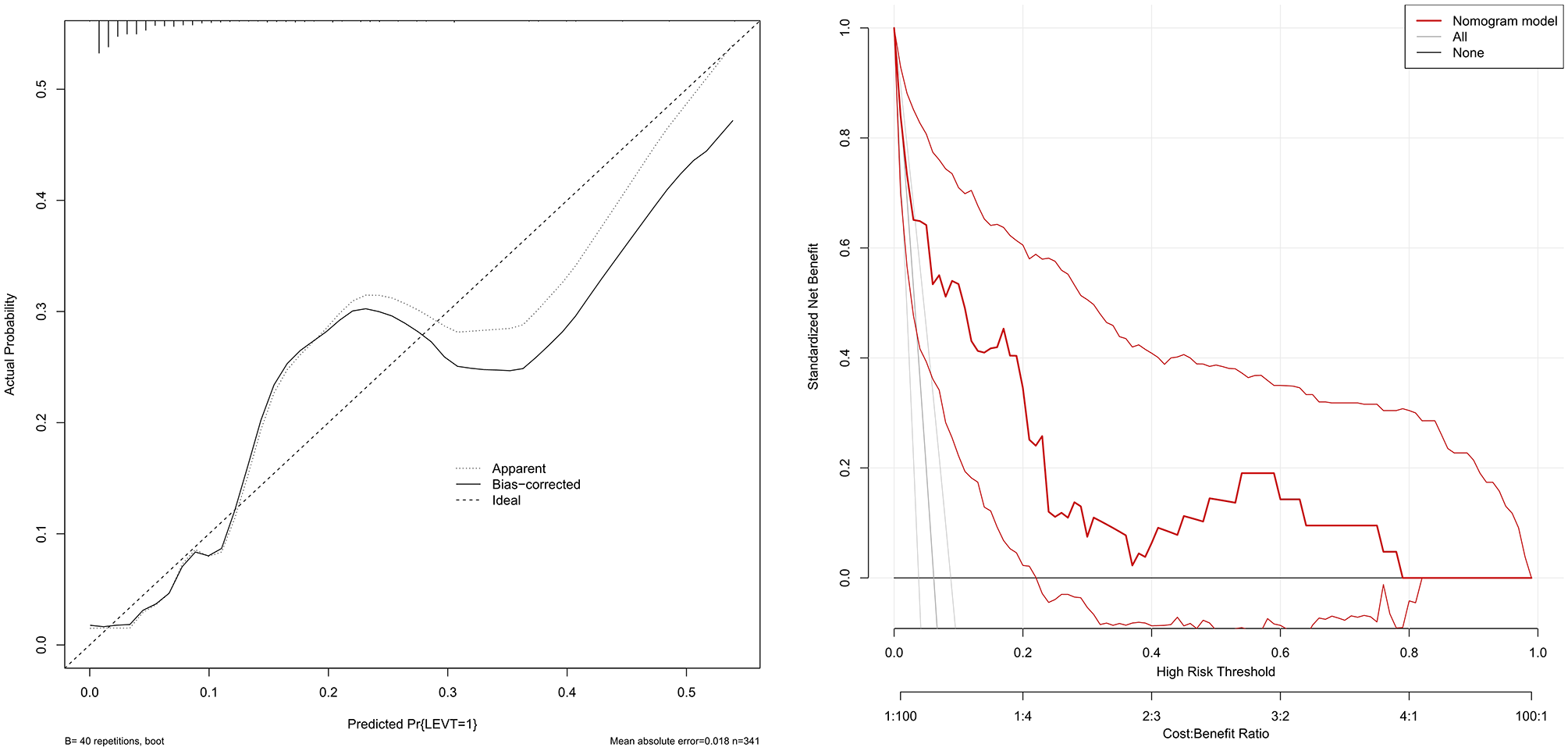
Calibration curve and decision curve analysis (DCA) of predictive models. (A) Calibration curves for preoperative asymptomatic LEVT incidence in knee osteoarthritis (KOA) show best agreement with nomogram predictions. (B) DCA for assessing the value of predictive models for clinical applications. The Y-axis represents the net benefit, the first X-axis represents the threshold probability, and the second X-axis represents the cost-benefit ratio. The red line represents the nomogram for predicting preoperative asymptomatic LEVT, the grey line represents the assumption that asymptomatic LEVT occurs in all patients, and the black line represents the assumption that asymptomatic LEVT does not occur in all patients. The range of threshold probabilities of positive net benefit for the model based on the values on the x-axis corresponding to the points of intersection of the red line with the grey and black lines.
Discussion
Studies have shown that the preoperative incidence of LEVT in TJA patients ranges from 6.85% to 29.4%.12–14 Xiong et al 19 reported that in patients with DVT before TJA, approximately 74% of thrombi occurred in the distal vessels. In our study, approximately 95% of patients with preoperative LEVT were IVT, which demonstrated a high incidence of distal vessel thrombosis as in previous studies. Smith et al 20 reported out that small thrombosis may also progress to PE, suggesting that we should pay attention to medium- and small-size distal vessel thrombosis in patients with total hip arthroplasty (THA) and that IVT may develop and dislodge in this group of patients who are undergoing surgical and other treatments, resulting in PE and even death. In addition, among THA patients diagnosed with preoperative lower limb venous thrombosis, the probability of a regenerating thrombus at the same site after undergoing THA is approximately 66.7%. 14
From the point of view of patient financial burden and medical insurance, patients with VTE may experience postoperative recurrence or postthrombotic syndrome (PTS), with the most common symptoms including persistent or intermittent limb pain, swelling, itchy skin, or cramping. This will extend the patient's hospital stay and add additional financial burden to the patient.21,22 A literature review suggested that who underwent major orthopedic surgery for DVT in the United States have higher hospitalization costs than patients without DVT (approximately $12,000-$17,000 higher). 23 This all adds an additional financial burden to the patient's family and to national health care coverage; therefore, it is important to identify risk factors for preoperative DVT in patients with TJA.
A total of 354 patients were enrolled in this study. LEVT was confirmed after bilateral lower extremity venous ultrasonography, and the preoperative incidence of asymptomatic LEVT was 5.93%. In patients with severe KOA, demographic characteristics, laboratory findings, and clinical features may all be associated with preoperative LEVT. According to the results of the multivariate logistic regression analysis, we confirmed that high D-dimer, high LDL, high Ca2+, low HDL and high Na+ levels were the high risk factors for preoperative asymptomatic LEVT in patients with knee osteoarthritis, and we evaluated the model with good accuracy and discriminability through internal verification. According to the DCA, our model provides a framework for comprehensively considering the cost benefit risk relationship and has good clinical decision-making guidance significance.
The study by Xiong et al 24 indicated that age over 69.5 years was an independent risk factor for preoperative DVT in patients undergoing total knee arthroplasty (TJA). However, age was not an independent risk factor in our study, and the study by Whiting et al 25 supports our view. Considering that our study may be due to the large age of the KOA patient population and small sample size, we need to expand the sample size and age range for further research. Slow blood flow, hypercoagulability and vascular endothelial injury are the three factors associated with venous thrombosis. 26 All the factors that contribute to the above factors will increase the probability of LEVT. Smoking is harmful to the vascular endothelium, and previous studies have shown that smoking is associated with a higher incidence of DVT.27,28 However, more research is needed to confirm whether smoking is an independent risk factor for LEVT formation. 25
In our study, we found that HDL < 1.03 mmol/L and high LDL were high risk factors for asymptomatic LEVT before TKA. Although in our study, the t-test showed that HDL-C was not statistically different from Non-LEVT patients in the population of LEVT patients, we still confirmed that it was an independent risk factor for the occurrence of preoperative LEVT by logistic regression analysis. This is where the use of logistic regression analysis compensated for the shortcomings of traditional difference analysis. Aviram et al 25 found that decreased HDL was associated with increased platelet activity, which greatly increased the probability of thrombosis. Moreover, a previous study has shown that a decrease in HDL cholesterol and an increase in inflammatory cells are associated with the occurrence of DVT. 29 In chronic inflammation, KOA may cause changes in blood lipid levels through the lipid metabolism pathway, thus increasing the incidence of LEVT. From our study, we also found that all patients with LEVT had abnormal BMIs. But whether abnormally high or abnormally low BMI is strongly associated with the development of LEVT has also not been confirmed in some studies.19,24 In this study, the association between high and low BMI and preoperative LEVT was not clear (P > 0.05), which may be due to insufficient sample size of LEVT, and we will increase the sample size in future studies to confirm the association between the two. On the other hand, abnormal lipid levels are strongly associated with cardiovascular disease, and high LDL levels and low HDL levels are considered risk factors for cardiovascular disease. 30 In our study, we found that the incidence of coronary heart disease in the LEVT group was significantly different from that in the non-LEVT group, which may also indicate that dyslipidemia has an impact on the occurrence of asymptomatic LEVT before TKA. More studied are needed to further explore whether lipid metabolism can directly affect venous thrombosis and the underlying mechanism involved. In addition, our study revealed that when Na and Ca ions were elevated, the risk of preoperative asymptomatic LEVT in patients with KOA increased, and these two parameters were found to be independent risk factors. Melge et al 31 found that Na+ plays a crucial role in the coagulation process and that a high Na concentration is capable of placing the blood in a rapid, procoagulant state, which may increase the risk of thrombosis. The blood coagulation reaction plays an important role in a complex of vitamin K-dependent serine proteases and nonenzymatic cofactor proteins assembled on membrane surfaces in a calcium-dependent manner, whose activity is regulated by calcium ions and promotes coagulation in the presence of high calcium.32,33 Therefore, more attention should be given to the relationship between ion metabolism and thrombosis in clinical practice, and more studies are needed to elucidate the mechanism and relationship between ion metabolism and LEVT before TKA. In clinical practice, D-dimer is widely used as an indicator of high sensitivity to thrombosis, an increase in D-dimer is significantly associated with the formation of lower limb venous thrombosis, and the D-dimer level is greater in patients with proximal vascular thrombosis was higher than that in patients with IVT.34,35
Previously, several scores, such as the Caprini score, have been proposed to help clinicians and patients assess the risk of LEVT formation. 15 However, whether the current commonly used scoring systems are suitable for the assessment of LEVT in TKA remains to be investigated. This study may be the first to use a nomogram to assess preoperative asymptomatic LEVT in patients with KOA. Nomograms are graphical computational tools commonly used in cancer research to predict patient prognosis and are tools that facilitate clinical decision-making and improve predictive accuracy.36,37 We now analyze our common clinical features to identify independent predictors of preoperative asymptomatic LEVT and construct a nomogram prediction model, which will make our assessment and screening of KOA patients for preoperative asymptomatic LEVT more relevant. The results of this study will provide better accuracy and efficiency in predicting preoperative asymptomatic LEVT in patients with KOA, and aggressive imaging screening and prophylactic anticoagulation for high-risk patients will be more conducive to patient disease management and reduce the economic burden.
There are several limitations to our study. First, our study is a retrospective study, which may introduce selection bias. Second, in our study, only five independent predictors were used, and as we did not find a suitable public dataset for external validation, our results require additional data and multi studies for further validation. Finally, we focused only on the patient's most recent bilateral lower extremity venous ultrasound findings from the time of consultation and did not expand the time frame of the study's examinations to focus on the progression of thrombosis. Further studies will be needed in follow-up to verify that the model can be of real benefit in the clinic.
Conclusion
In summary, we confirmed that high D-dimer, high LDL, high Ca2+, low HDL and high Na+ are independent risk factors for the preoperative occurrence of asymptomatic LEVT in patients with KOA. To the best of our knowledge, this study is the first to develop a nomogram-based clinical prediction model for the preoperative occurrence of asymptomatic LEVT in patients with KOA, which will help clinicians to identify those at high risk for the preoperative occurrence of asymptomatic LEVT, and will contribute to proactive screening and prophylactic measures.
Footnotes
Author Contributions
(Ⅰ) Conception and design: J Duan and Y Du; (Ⅱ) Administrative support: D Wu and Y Du; (Ⅲ) Provision of study materials or patients: J Duan and W Qin; (Ⅳ) Collection and assembly of data: J Duan, W Qin, and T Xing; (V) Data analysis and interpretation: J Duan and W Qin; (VI) Manuscript writing: J Duan; (VII) Manuscript revision: T Xiao, Z Wang and Yuanliang Du; (Ⅷ) Final approval of manuscript: All authors.
Consent to Participate
As this was a retrospective study, and data were analyzed anonymously, informed consent was therefore waived by the committee.
Data Sharing Statement
The data used during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Considerations
This study was approved by the Ethical Review Committee of Chengde Medical College Hospital. This study was conducted in accordance with the guiding principles of the 1964 Declaration of Helsinki and its subsequent amendments.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the the Cultivating Programme Plan for Clinical Excellent Talent of Hebei in 2024, (grant number ZF2024240).
