Abstract
This study explored the efficacy and safety of rivaroxaban in elderly patients, at different doses and age of patients, and analyzed risk factors of bleeding. A retrospective analysis was conducted of 299 patients aged 60 years or older who were admitted to the First Hospital of Jilin University between January 2016 and August 2018. It was found that the rate of bleeding events (but not embolism) significantly increased as the dose of oral rivaroxaban increased (P < .001), and with age, especially in patients aged ≥80 years (P = .001, both). The multivariate logistic regression analysis indicated that age (odds ratio [OR]: 2.963, 95% CI: 1.627-5.396) and the daily dose of rivaroxaban (OR: 2.325, 95% CI: 1.483-3.645) were independent risk factors for bleeding. The study determined that rivaroxaban anticoagulant therapy is effective in the elderly patients, but the risk of bleeding increases with age, and is a concern especially in the most old patients. The recommended daily dose of rivaroxaban is effective, but a lower dose is safer for the elderly patients.
Introduction
Thromboembolic diseases may be venous or arterial. Venous thromboembolism is the third most common cause of vascular death after heart attack and stroke, comprising deep vein thrombosis and pulmonary embolism. 1 Arterial thromboembolism is predominantly due to nonvalvular atrial fibrillation. It has been said that patients with atrial fibrillation have a 5-fold higher risk of ischemic stroke compared with healthy individuals, especially in the elderly patients. 2
Warfarin and low-molecular-weight heparin are the traditional medications for treatment of thromboembolic diseases, 3,4 but there are many disadvantages, including the need for regular coagulation monitoring associated with some food-drug interactions, which can aggravate drug compliance. 5 Low-molecular-weight heparin is injected, and patients’ compliance is poor. 6
There are 4 new direct oral anticoagulants (NOACs): rivaroxaban, apixaban, dabigatran, and edoxaban. Rivaroxaban (sold under the trade name Xarelto; Bayer HealthCare) was the first certified for clinical use. 7,8 Rivaroxaban is a direct selective inhibitor of FXa (coagulation factor X), with high oral bioavailability and rapid onset of action, and is relatively easy to take. 9 Rivaroxaban can be used for the treatment of deep vein thrombosis and the prevention of stroke in patients with atrial fibrillation. The effectiveness and safety of rivaroxaban are higher than for some traditional antithrombotic drugs in clinical practice. 10,11 According to a retrospective cohort study of patients with nonvalvular atrial fibrillation, rivaroxaban reduced the incidence of severe stroke and all-cause mortality after the stroke, relative to warfarin. 12
However, the recommended dose of rivaroxaban was determined for patients aged ≥18 years, and there are few studies regarding the effectiveness and safety of its use specifically for the elderly patients. This is especially concerning, because of the greater likelihood of comorbidities and multiple drug interactions in this population. The present retrospective study investigated the effectiveness and safety of rivaroxaban for elderly patients with thromboembolic diseases, and a suitable therapeutic dose.
Methods
The Ethics Committee of the First Hospital of Jilin University (Changchun, China) approved this study. The institutional review board waived a study requirement for informed patient consent.
Patients
This was a retrospective analysis of thromboembolic diseases (deep vein thrombosis or pulmonary embolism) and patients with nonvalvular atrial fibrillation aged 60 years or older who were admitted to First Hospital of Jilin University in northeast China. Patients were included if they had thromboembolic disease or nonvalvular atrial fibrillation ((CHA2DS2-VASc≥2) [Congestive heart failure, hypertension, age 75 years (doubled), diabetes mellitus, stroke (doubled)-vascular disease, 65-74 years and sex category (female)]), required oral rivaroxaban (continuous medication duration ≥3 months), and were aged ≥60 years. Patients with any of the following were excluded: taking other antiplatelet drugs (eg, aspirin, clopidogrel); or a history of severe trauma, major surgery, or gastrointestinal bleeding within the previous 6 months. In addition, patients were excluded if they had severe liver dysfunction (transaminases >3× normal); renal dysfunction (creatinine clearance <30 mL/min); thrombocytopenia or hematological diseases; malignant tumors with a higher risk of bleeding; incomplete clinical data; or switched to other anticoagulant drugs during the medication. Outpatient and telephone follow-ups were conducted to monitor bleeding events and new thromboembolic events within 3 months of continuous medication. All data were collected using EXCEL software.
Groups and Evaluation Criteria
The patients were divided into 4 groups, according to the daily dose of rivaroxaban, as follows: 10 mg every day; 15 mg every day; 10 mg twice a day; or 15 mg twice a day for 21 days and then 20 mg every day. The population was also stratified into 3 groups depending on age: 60 to 69; 70 to 79; and ≥80 years.
The Thrombolysis in Myocardial Infarction standard definitions were applied to assess bleeding as major, moderate, or slight. 13 Specifically, major bleeding was defined as clinically visible (including imaging diagnosis) or important organ and intracranial bleeding with a decrease in hemoglobin ≥5 g/dL. Moderate bleeding was clinically visible (including imaging diagnosis), with decrease in hemoglobin between 3 and 5 g/dL. Slight bleeding was considered blood loss that was insufficient to meet the criteria listed above.
A new thromboembolic event was recorded if, during medication, either of the following occurred: new deep venous thrombosis in a patient with deep venous thromboembolism; or a stroke or vascular occlusion caused by emboli shedding in a patient with atrial fibrillation. A new thrombosis was diagnosed based on angiography or vascular color Doppler ultrasound.
Statistical Analysis
All statistical analyses were performed using SPSS version 22.0. An independent sample t test, χ2 test, or Fisher exact probability method was used. For the R × C contingency table, when the outcome variable was multivariate and orderly, the rank-sum test was used. The logistic regression model was applied to analyze the independent risk factors for bleeding. Differences were considered statistically significant at P < .05. The figure was designed using Prism Software and Adobe Photoshop.
Results
Clinical Characteristics
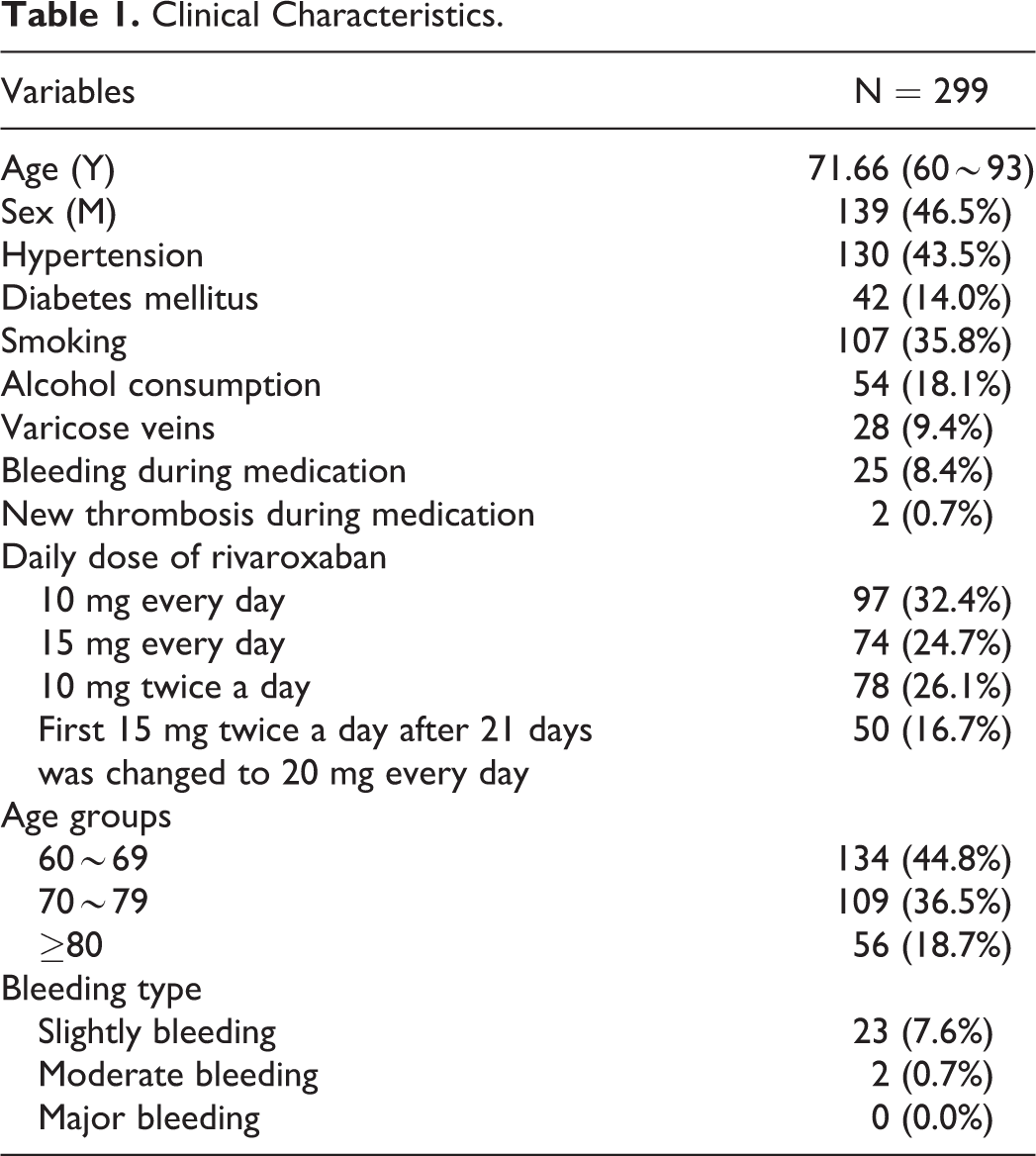
Overall, 299 patients were enrolled, with a median age of 71.66 years (60-93 years; Table 1). During the treatment, 2 (0.7%) and 23 (7.6%) patients experienced moderate and slight bleeding, respectively, with an overall incidence of 8.4%. In addition, 2 patients suffered from new thrombosis; the incidence of new thrombosis was 0.7%.
Clinical Characteristics.
Analysis of Effectiveness and Safety of Rivaroxaban in Elderly Patients
The rate of bleeding events was positively and significantly associated with the rivaroxaban dose, and the difference among the groups was statistically significant (χ2 = 12.241, P < .001). The bleeding rate of the group given 15 mg twice a day for 21 days and then 20 mg every day was significantly higher than that of the group given 10 mg every day throughout (P < .008; Supplementary Table 1). The rates of embolism among the dosage groups were not statistically significant (P = .765; Supplementary Table 2). When stratified by age, the rate of bleeding events was positively associated with age, (χ2 = 13.053, P = .001), and bleeding in the ≥80-year group was significantly higher than that of the group aged 60 to 69 years (P < .016; Supplementary Table 3).
Univariate and Multivariable Analysis for Independent Factors of Bleeding
Comparisons of baseline data between the bleeding and nonbleeding groups showed that the following were risk factors for bleeding events (Table 2): age (χ2 = 13.053, P = .001); the daily dose of rivaroxaban (χ2 = 12.241, P < .001); and prothrombin activity (χ2 = 2.008, P = .046). A logistic regression analysis to evaluate the role of these parameters indicated that age (odds ratio [OR]: 2.963, 95% CI: 1.627-5.396) and the daily dose of rivaroxaban (OR: 2.325, 95% CI: 1.483-3.645) were independent risk factors for bleeding (Table 3). An analysis of the association between the daily dose of rivaroxaban and age in the 25 bleeding patients showed that, while the daily dose of rivaroxaban decreased with increasing patient age, patients of advanced age still had a higher risk of bleeding compared with the younger elderly patients (Figure 1).
Analysis of Bleeding Risk Factors.

Abbreviations: ALT, alanine aminotransferase; APTT, activated part thromboplastin time; AST, aspartate aminotransferase; BUN, blood urea nitrogen; Cr, creatinine; D-D; D-Dimer; INR, international standardized ratio; PLT, platelet count; PT, prothrombin time; PTA, prothrombin activity; PTR, prothrombin time ratio; fibrinogen, SD, standard deviation.
a Expressed as frequency and percentage (%).
b Expressed as median (interquartile range).
c Expressed as mean ± SD.
Multiple Logistic Regression Analysis of Influencing Factors of Bleeding Events.

Abbreviation: OR, odds ratio.

A daily dose of rivaroxaban and age distribution in patients with bleeding.
Discussion
New oral anticoagulants prescribed for daily intake can reduce the number of thromboembolic events over the years. The number of users of oral anticoagulants has increased in recent years, especially in the elderly patients who are at the highest risk of stroke and bleeding and show the greatest associated decrease in ischemic stroke without increased bleeding. 14,15 The greatest observed clinical benefit of NOACs is for stroke prevention in atrial fibrillation, in both primary and secondary care. 14 One meta-analysis concerning elderly patients with atrial fibrillation granted that, compared with vitamin-K antagonists, NOACs were associated with a lower risk of systemic embolism, intracranial hemorrhage, hemorrhagic stroke, and fatal bleeding. However, these benefits were accompanied by a higher risk of gastrointestinal bleeding, and there were significant differences in efficacy depending on the individual NOAC agent. 2
Rivaroxaban is a NOAC with a proven lower risk of bleeding. It has been widely used in the treatment and prevention of deep venous thrombosis and nonvalvular atrial fibrillation, as well as for patients with nonvalvular atrial fibrillation with concomitant coronary artery disease, with or without peripheral artery disease. 16 –19 Because of reduced or poor liver and kidney function in the elderly patients, there is higher risk that an anticoagulant will lead to bleeding. However, there is no international recommended dosage for the elderly patients.
The present study was a retrospective analysis of clinical data of 299 elderly patients taking rivaroxaban. It was found that the total bleeding rate was 8.4%, which was either slight or moderate bleeding, and of the skin or mucosa. The signs of gastrointestinal bleeding are dark stool or slightly bloody stool (data not shown). There was no major bleeding. In addition, the incidence of embolism among the groups was insignificant, which indicates that rivaroxaban is effective in anticoagulation therapy.
Interestingly, compared with the bleeding rate reported in the Phase III clinical trial of rivaroxaban, 20,21 the bleeding rate of the current patients overall was lower. However, specifically for our patients aged 80 years or older, the bleeding rate was significantly higher than that shown by the RECORD III clinical trial. With further analysis, we found that even when the daily dose of rivaroxaban was reduced in patients 80 years or older, there was still a higher risk of bleeding. This suggests that elderly people should receive a lower dose of rivaroxaban; for best effectiveness, small doses are safer. Studies have found that a rivaroxaban dose of 10 mg once daily is better tolerated and is more suitable for the elderly patients. 22,23 In the present research, 10-to-15 mg per day was both effective and safe for older people.
The present analysis also determined that prothrombin activity contributes to bleeding. Prothrombin activity is a sensitive indicator of the severity and prognosis of hepatocyte necrosis. Most coagulation factors are synthesized in the liver. When liver parenchyma is damaged and a large number of hepatocytes are necrotic, the synthesis of coagulation factors is limited and the risk of bleeding increases. Studies have found that thrombin allele knockout mice have a higher risk of bleeding, which included important organs such as the heart and brain. 24 However, whether prothrombin activity can be used as an evaluation index for bleeding risk of rivaroxaban, further research is needed.
This analysis of the associations among rivaroxaban-related bleeding events, the daily dose of rivaroxaban, and age, provided some guidance for anticoagulation therapy with rivaroxaban in the elderly patients. However, this study is a retrospective analysis of clinical data. Patient data were collected through both telephone follow-up and the hospital’s medical records. Many factors that may affect the study results were excluded. The study sample was small, which may also affect the results. The reliability of the conclusion requires more scientifically rigorous, multicenter, and large-scale experiments to confirm. Further studies should be conducted that include the elderly patients with different thromboembolic diseases, and whether the dose of rivaroxaban can be reduced from the recommended dose while maintaining effectiveness and safety.
Conclusion
By evaluating the effectiveness and safety rivaroxaban in elderly patients, the anticoagulant effect of rivaroxaban was confirmed. However, for elderly patients aged 80 years or older, selection of the dose of anticoagulant drugs still requires care. While effectiveness is the primary goal, taking less (10-15 mg/d) than the recommended dose may be safer for elderly patients. Risk factors for bleeding in the elderly should be reassessed.
Supplemental Material
Supplementary_Table_1 - Analysis of Effectiveness, Safety, and Bleeding Related to Rivaroxaban in Elderly Patients
Supplementary_Table_1 for Analysis of Effectiveness, Safety, and Bleeding Related to Rivaroxaban in Elderly Patients by Huimin Hou, Ang Li, Liping Zhang, Xiujiao Qin, Yanfang Jiang and Huiying Zhao in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
Supplementary_Table_2 - Analysis of Effectiveness, Safety, and Bleeding Related to Rivaroxaban in Elderly Patients
Supplementary_Table_2 for Analysis of Effectiveness, Safety, and Bleeding Related to Rivaroxaban in Elderly Patients by Huimin Hou, Ang Li, Liping Zhang, Xiujiao Qin, Yanfang Jiang and Huiying Zhao in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
Supplementary_Table_3 - Analysis of Effectiveness, Safety, and Bleeding Related to Rivaroxaban in Elderly Patients
Supplementary_Table_3 for Analysis of Effectiveness, Safety, and Bleeding Related to Rivaroxaban in Elderly Patients by Huimin Hou, Ang Li, Liping Zhang, Xiujiao Qin, Yanfang Jiang and Huiying Zhao in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Authors’ Note
All authors had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. The need for informed consent from the patients for publication of this information was waived by the institutional review board of the hospital for this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
