Abstract
Elderly individuals are prone to nonvalvular atrial fibrillation (AF) with associated risks of arterial thromboembolic disease. Despite definitive guidelines, oral anticoagulant therapy (OAC) is notoriously underutilized in patients with AF. Physicians cite excessive bleeding risk as one reason they omit OAC for their older patients with AF. Improved understanding of the pathophysiology of age-related bleeding may improve risk–benefit assessments for warfarin and newer antithrombotic agents. We reviewed the literature to identify age-related pathophysiological elements that can exacerbate the likelihood of bleeding. In the context of the Hypertension, Abnormal renal/liver function, Stroke, Bleeding history or predisposition, Labile international normalized ratio, Elderly, Drugs/alcohol concomitantly (HAS-BLED) bleeding risk framework, we highlight age-related physiological dynamics that predispose to hemorrhage. The combination of increased age (>65 years) with the other elements of the risk factor stratification model identifies patients with AF who are especially susceptible to OAC-related bleeding, irrespective of the agent used. Empirically adjusting OAC dose relative to these common bleeding risks may help to achieve an improved risk–benefit therapeutic ratio.
Introduction
Prevalence of atrial fibrillation (AF) soars with advancing age, climbing from 0.1% among those younger than 55 years to 9.0% in patients aged 80 years and older. 1 Studies show that advancing age increases the risk of thromboembolic events and that oral anticoagulant therapy (OAC) with warfarin produces a 68% reduction in the relative risk of stroke versus no therapy. 2 Nonetheless, in a systematic analysis, Pugh et al report that physicians are often reluctant to prescribe warfarin for elderly patients with AF, despite evidence of increased benefit in this population compared with younger patients. 3 Thus, 56% to 85% of older patients with AF without clear contraindications are not anticoagulated. 3
Many have attributed bleeding risks particularly to warfarin therapy, raising expectations that a new generation of antithrombotic agents would offer safer anticoagulation. 4 Yet, although the selective direct factor inhibitors such as dabigatran, apixaban, rivaroxaban, and edoxaban provide advantages in administration, they do not eliminate bleeding risks. In the Randomized Evaluation of Long-Term Anticoagulation Therapy (RE-LY) Trial a direct thrombin inhibitor, dabigatran, was compared with warfarin. 5,6 Both medications were associated with age-related bleeding risks (Figure 1). Nonetheless, a related study indicates that clinicians are more readily prescribing direct thrombin inhibitor therapy to older and relatively more bleeding-prone adults. 7 In that study, over two-thirds of the 7000 patients who received dabigatran, when it was first made available in New Zealand and Australia, were aged over 80 years. By elucidating the pathophysiological underpinnings of bleeding covariates, we aim to enrich the scientific foundation for their application. Such insight also reinforces the rationale to adjust OAC dose relative to each individual’s bleeding risks.
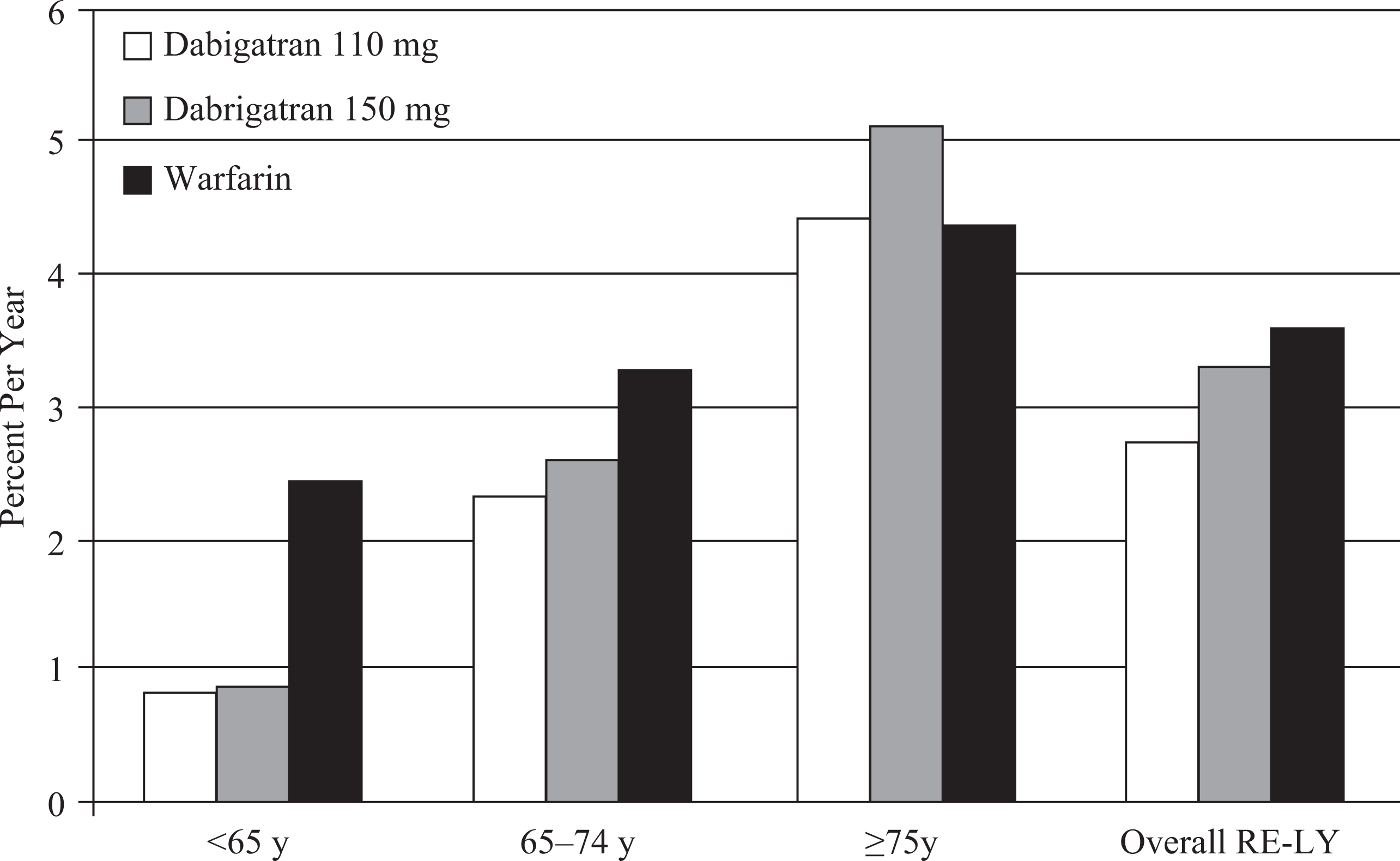
Rates of major bleeding in Randomized Evaluation of Long-Term Anticoagulation Therapy (RE-LY) by age. 5
Bleeding Risk Assessment Tools for Patients With AF
Although many clinicians base their decisions on whether to use OAC on the Congestive heart failure, Hypertension, Age ≥75 years, Diabetes mellitus, and prior Stroke or transient ischemic attack (CHADS2) score, this score gauges only thrombosis risks and provides no clarity regarding bleeding vulnerability. 8 Alternatively, several risk stratification tools have been developed to predict the risk of bleeding among candidates for OAC, including Hypertension, Abnormal renal/liver function, Stroke, Bleeding history or predisposition, Labile international normalized ratio, Elderly, Drugs/alcohol concomitantly (HAS-BLED), Anticoagulation and Risk factors In Atrial fibrillation (ATRIA), Hepatic or renal disease, Ethanol abuse, Malignancy, Older (age >75 years), Reduced platelet count or function, Hypertension (uncontrolled), Anemia, Genetic factors, Excessive fall risk, and Stroke (HEMORR2HAGES), modified Outpatient Bleeding Risk Index (mOBRI), and National Registry of Atrial Fibrillation (NRAF) (see Table 1). 9–13 Data for these risk prediction models were developed by systematic reviews and statistical analyses of 1 or more trials of OAC in patients with AF. Although each tool is slightly different, they all assess an individual’s risk of bleeding based on a group of clinical variables. The sum of the individual elements (often weighted) is used to classify patients into low, intermediate, and high-risk strata, with annual risks of major bleeding approximately ≤1%, 2% to 4%, and ≥5%, respectively. 14
Abbreviations: INR, international normalized ratio; HAS-BLED, Hypertension, Abnormal renal/liver function, Stroke, Bleeding history or predisposition, Labile international normalized ratio, Elderly, Drugs/alcohol concomitantly; ATRIA; Anticoagulation and Risk factors In Atrial fibrillation; HEMORR2HAGES, Hepatic or renal disease, Ethanol abuse, Malignancy, Older (age >75 years), Reduced platelet count or function, Hypertension (uncontrolled), Anemia, Genetic factors, Excessive fall risk, and Stroke mOBRI, modified Outpatient Bleeding Risk Index; NRAF: National Registry of Atrial Fibrillation; CYP, cytochrome.
a Specifically CYP2C9 polymorphism.
In this review, we particularly focus on the HAS-BLED score.
12
It has been endorsed by multiple professional societies and allows a straightforward categorization and discussion of the multiple factors that interact with age and impact the risk of bleeding in patients with AF.
15–17
The HAS-BLED was derived by analysis of the medical records of 3978 ambulatory and hospitalized patients with AF from the Euro Heart Survey, only 10.2% of whom had not received any antithrombotic therapy.
12
Antithrombotic strategies included OAC in 64.8% (n = 2242) of the patients, 12.8% (n = 286) of whom were also receiving antiplatelet therapy with either aspirin or clopidogrel and 24.0% (n = 828) who received only antiplatelet therapy. The mean age of the population was 66.8 years and 41% were women. Univariate analyses were used to identify clinical factors associated with major bleeding events. The latter was defined as any bleeding requiring hospitalization and/or associated with a >2 g/dL decrease in hemoglobin and/or requiring transfusion that was not categorized as a hemorrhagic stroke. After multivariate analyses and stepwise removal of less strongly associated risk factors (P > .10), the remaining factors from the derivation cohort were combined with more historical risk factors (OAC therapy, hypertension, alcohol use) to create the model. The HAS-BLED incorporates 9 clinical factors:
The HAS-BLED Model
The relative risk attributed to age varies among the various bleeding risk factor stratification models, with the highest weight attribution in mOBRI and ATRIA (Figure 2). The threshold for age as a risk factor varies between ≥65 years in mOBRI and ≥75 years in ATRIA. 14 In HAS-BLED, patients who are >65 years of age are assigned 1 point and, in the absence of any other risk factor, are considered to be at low risk of bleeding.
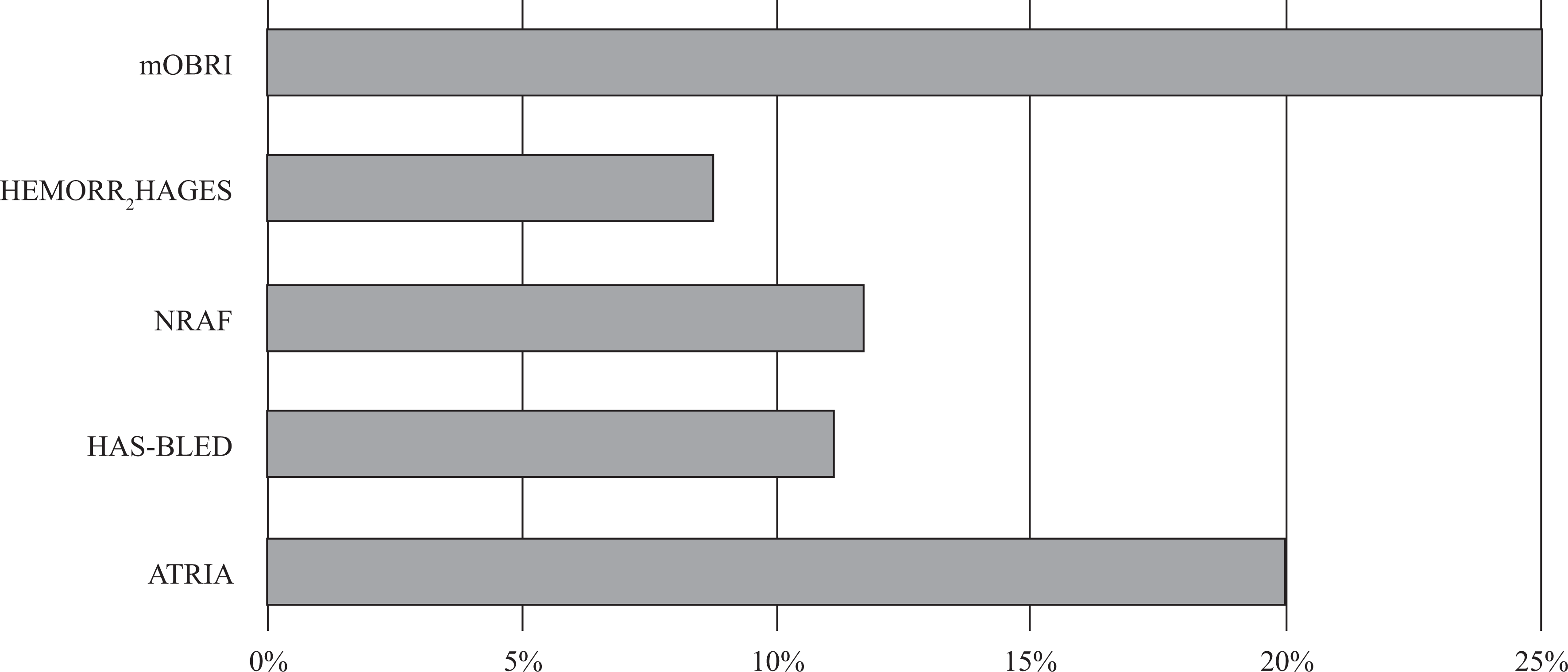
Weighting of age versus other risk factors in Hypertension, Abnormal renal/liver function, Stroke, Bleeding history or predisposition, Labile international normalized ratio, Elderly, Drugs/alcohol concomitantly (HAS-BLED) and 4 other contemporary bleeding risk models. 14
Age >65 Years Increases OAC Bleeding Risk
Although we will discuss pathophysiologic features incorporated within HAS-BLED by the sequential order of representative letters as they appear within the acronym, we will begin out of sequence with “E” for elderly individuals, given the focus of this article. In HAS-BLED, age >65 years contributes 11% of the overall total score. 12 Even in the absence of comorbidities, aging of the cardiovascular system is associated with phenotypic changes that increase the susceptibility to bleeding. 18 Although age >65 years in some respects corresponds to other bleeding-associated covariates that accumulate with age, there are unique aspects of aging that directly increase bleeding risk. Among a list of age-related vulnerabilities, (1) changes in vascular biology; (2) increased vulnerability to falls, and (3) changes in pharmacokinetics (PK) and pharmacodynamics (PD) are key factors that increase OAC-related bleeding.
The OAC Bleeding Risks in Relation to Age: Omnipresent Vascular Changes
Age as a bleeding risk relates to fundamental changes in vascular biology that predispose to hemorrhagic events. With age, capillaries within the skin demonstrate increasing hemorrhagic vulnerability that extends throughout the vascular tree. Senile purpuras exemplify such predisposition. These purple-red lesions on the extensor surfaces of the forearms and hands occur in 10% to 20% of geriatric patients. 19 Systemic or topical steroids and platelet dysfunction often produce similar changes. Presumed mechanisms include age-related loss of soft tissue supporting cutaneous blood vessels compounded by higher capillary pressures that are also common with age. 20
Although primarily of historical interest, the Rumpel-Leede test illustrates the relevance of connective tissue changes and small-vessel fragility. The test was first described in 1909 as a simple tool to evaluate capillary fragility. 21 Although normally capillaries can withstand pressures of 90 mm Hg for 3 minutes, elderly adults and those with Cushing disease, scurvy, thrombocytopenia, vasculitis, diabetes, and some viral exanthems may show petechiae distal to the cuff inflated to 10 mm Hg above the diastolic blood pressure. Biopsies of the site show congestion of venules and extravasation of erythrocytes into the subpapillary dermis. Small-vessel fragility increases with age 22 and is associated with an increased risk of hemorrhagic events. An epidemiologic study of cerebrovascular disease in 4186 Japanese patients, 40 to 79 years of age, demonstrated that the relative risk of cerebral hemorrhage versus stroke was significantly less for those with normal capillary fragility as measured by the Rumpel-Leede test. 23
The OAC Bleeding Risks in Relation to Age: Increased Risks of Falls
Age also constitutes a bleeding risk due to the age-related predisposition to falls. Falling predilections among elderly individuals are often multifactorial. Age-associated changes in gait, proprioception, neuroautonomic function, vision, and posture, 24 as well as the use of sedatives (including alcohol) and other medications that compromise alertness, acuity, or balance, all increase susceptibility. Age is also associated with increased frailty, a broad concept that refers to generalized weakening and cognitive slowing, and which thereby compounds risks of falling. 25 Ironically, although many clinicians tend to avoid OAC for older patients with AF due to concerns about falls, detrimental consequences of thromboembolism may be the greater hazard. As one decision analysis model demonstrates, a patient would need to fall 295 times per year before the risk of OAC-associated traumatic intracranial hemorrhage (ICH) outweighs that of ischemic stroke. 26 Thus, although falls are a legitimate concern, strategies by which to factor falling risks into management decisions remain imprecise.
The OAC Bleeding Risks in Relation to Age: Age-Related Changes in Pharmacology
Progressive loss of organ function with aging begins in early adulthood and can significantly affect both the PK and the PD of oral anticoagulant drugs in the elderly individuals. 27,28 Changes in these pharmacologic properties with advancing age may be associated with increased risk of OAC-related bleeding. For example, stenosis of the mesenteric arteries is present in 17.4% of free-living patients 70 years of age and older. 29 Experimentally, chronic celiac axis and superior mesenteric artery-associated ischemia can decrease drug absorption and inhibit P-glycoprotein (P-gp) messenger RNA production and enzyme function and have been shown in one study to be independently and significantly associated with annualized weight loss (P = .028). 29,30 The P-gp serves as a transporter for all of the new and investigational direct factor inhibitors. 31 Thus, patients taking a drug that is a P-gp substrate may have decreased clearance of the anticoagulant and increased risk of bleeding. In healthy elderly patients, there is a significant negative correlation between plasma albumin levels and age with both linked to an increased risk of frailty. 32,33 Changes in protein binding can also significantly alter available drug and distribution of medications such as warfarin between the vascular space and other extracellular fluid compartments. Furthermore, advancing age is associated with increased risk of polypharmacy; consequently, elderly patients with AF receiving antithrombotic therapy may be taking drugs that alter the absorption, distribution, and metabolism of the traditional oral anticoagulants or of the direct factor inhibitors and increase the risk of bleeding. 34
The OAC-Related Bleeding Risks: Anemia in the Elderly Individuals
Anemia is the most common hematologic problem of the elderly individuals and increases in prevalence with age. 16 In the nursing home population, 48% to 63% of residents are anemic, reflecting poor health status of institutionalized older adults. 35 In 97% of the cases of anemia in the elderly individuals, the anemia is mild with a hemoglobin of ≥11. 35 It may be a primary manifestation of silent bleeding or secondary to an age-related comorbidity that also increases the risk of bleeding such as abnormal renal/hepatic function or drugs such as excessive alcohol consumption. In a series of 278 consecutive elderly patients (mean age 69 years) with AF and an indication for OAC who underwent percutaneous stenting, anemia was present at baseline in 41%. 36 In this population, patients with anemia having AF had a higher rate of gastrointestinal (GI) tract major bleeding (20.0% vs 10.7%; P = .035) and a lower event-free survival period (77% vs 92%; P = .006). Anemia also contributes directly to increased risk of bleeding. In patients with a normal red blood cell count, the flow and number of erythrocytes force platelets centrifugally toward the endothelial lining. As a result, platelets are in close apposition to the vessel wall. When vascular integrity is disrupted, this relationship facilitates platelet adhesion and aggregation. In patients who are anemic, this interaction is encumbered and platelet adhesion is decreased. 37 Anemia also decreases the amount of adenosine diphosphate (ADP) that is available to contribute to collagen-induced platelet aggregation at the site of injury. 38,39
Hypertension Increases the Risk of OAC-Associated Intracerebral Bleeding
Hypertension is the “H” in HAS-BLED. 12 More than 90% of the individuals who are normotensive at age 55 years will eventually become hypertensive 40 ; by age 60 years, more than 65% of the men and women have systolic hypertension. 41 Thus, hypertension comigrates with age as a bleeding risk factor. In HAS-BLED, hypertension is weighted at 11% of the maximum score. 10 The HAS-BLED is the only model that provides a specific threshold for hypertension and confirmed increased risk of bleeding at systolic blood pressure >160 mm Hg. 14 Hypertensive vasculopathy is the most common pathophysiology in those with hypertension. Histologic examination of the sites of hypertensive vasculopathy-associated bleeding demonstrates hyperplasia and hyalinosis of the small vessels and microaneurysms. 42 Subclinical leaks may be common. 43 The most feared major bleeding complication of OAC is ICH. The risk of ICH from hypertensive vasculopathy is estimated to be increased 2- to 6-fold over that of normotensive individuals. 44,45 Magnetic resonance imaging changes of microhemorrhages were identified in 60% of the patients with nontraumatic ICH versus only 5% of the healthy adults. 46 The ICH is associated with an increased risk of death. Among Medicare beneficiaries, the in-hospital and 30-day mortality rates from ICH in Joint Commission-certified primary stroke center hospitals were approximately 28% and 40%, respectively. 47 Cerebral microbleeds are also more common in patients treated with OAC or antiplatelet therapy than others and may contribute to the risks of clinically significant ICH in patients with hypertension with AF who are treated with antithrombotic therapy. 48
Abnormal Renal/Hepatic Function Increases the Risk of OAC-Associated Bleeding
Chronic kidney disease (CKD) relates to age, anemia, as well as hypertension and other CV risk factors, but it also independently compounds bleeding risks. 49 Abnormal renal and/or hepatic function are the “A” in HAS-BLED and have a weight of 1 point each. 12 Abnormal renal function (serum creatinine ≥2.26 mg/dL; 200 µmol/L), chronic dialysis, and a history of renal transplantation are also considered to be surrogates for this variable. 12 Beginning in the third decade of life, a patient’s glomerular filtration rate (GFR) decreases by ∼10% per decade. 27 Thus, even in the absence of CKD, a 70-year-old’s GFR and ability to excrete drug metabolites may be reduced by as much as 50%. Therefore, even in the absence of contributory factors such as diabetes and hypertension, by age 85 years the GFR of a patient can be anticipated to be ∼60 mL/min/1.73 m2. Of the approximately 26 million people with CKD in the United States, ∼70% are ≥60 years of age, 50 most with stage 3 CKD, that is, GFR 30 to 59 mL/min/1.73 m2.
Uremic manifestations begin to accumulate as GFR approaches 60 mL/min. 51 Uremic toxins that may be contributory to the defect include cyclic guanosine monophosphate (cGMP), cyclic adenosine monophosphate (cAMP), phenolic acids, methylguanidine, and others. Patients with chronic renal failure and not on hemodialysis are particularly susceptible to GI bleeding as well as increased genitourinary bleeding. 52,53 Patients with CKD also often have prolonged bleeding times. It has been postulated that increased levels of proinflammatory cytokines in those patients upregulate nitric oxide synthase (iNOS). 54 The resultant nitric oxide activates guanylate cyclase and increases levels of cGMP. This can lead to reduced levels of the endogenous platelet aggregators thromboxane A2 and ADP. 55–57 Levels of prostacyclin (PGI2) also increase, with increased vasodilation and decreased platelet aggregation. 58,59 Finally, patients with CKD demonstrate decreased levels of factor VIII function. It has been proposed that this may be due to increased binding of factor VIII by von Willebrand factor or increased amounts of the multimeric protein. 60,61
In HAS-BLED, abnormal liver function is defined as chronic hepatic disease or biochemical evidence of it and weighted as 1 point, independent of the presence of abnormal kidney function. Liver disease is also a risk factor for OAC-associated bleeding in HEMORR2HAGES. Since both blood flow and size of the liver decrease by about 25% to 35% with advancing age, the ability of the organ to clear drugs declines with time. 62 There is a consistent negative association between age and total cytochrome (CYP) P450 content, NADPH cytochrome c reductase activity, and levels of CYP2E1 and CYP3A proteins. Furthermore, the liver is the site of synthesis for all major coagulation proteins except for factor VIII. Varices in patients with cirrhosis are a significant risk factor for bleeding.
History of Stroke Increases the Risks of OAC-Associated Bleeding
The “S” in HAS-BLED is a mnemonic for stroke. 12 A history of a stroke contributes 1 point to the overall score. Factors that increase stroke risk such as age often overlap with those associated with an increase the risk of bleeding. Furthermore, patients with AF who have had a stroke may receive more aggressive thromboprophylaxis and thus increased risk of bleeding. In addition, cerebral amyloid angiopathy (CAA) is strongly linked to advanced age and increased risk of intracranial bleeding. Based upon a report of 784 autopsies, the prevalence of moderate to severe CAA was estimated to increase from 2.3% between the ages of 65 and 74 years to 12.1% for ages ≥85 years. 63 The disorder is characterized by deposition of Congo red-positive material in the walls of cerebral and meningeal small- to mid-size blood vessels. Accumulations of amyloid within intracranial blood vessels can weaken their walls and predispose to hemorrhage. Patients with CAA may have microhemorrhages and transient neurologic findings. 64 The most common location of CAA-related spontaneous ICH is within the cortex and subcortical white matter of the posterior lobes of the brain and cerebellum, rather than deep within the hemispheres as with hypertensive vasculopathy.
History of Prior Bleeding Increases OAC Bleeding Risks
The “B” in HAS-BLED represents history of or predisposition to bleeding and contributes 1 point to the overall score. 12 Historical criteria for pertinent bleeding histories vary among different clinical standards and range from remote bleed, recent bleed, previous bleed, GI bleed in previous 2 weeks, and risk of rebleeding. With the exception of the mOBRI bleeding risk score, none of the bleeding models specify the site for risk of bleeding. 14 In the elderly individuals, the GI tract is the most common site of bleeding. The baseline risk of major GI bleeding in patients aged 65 years and older is 1.17% per year. 65 Both upper and lower GI bleeding increases with age. 26,66,67 In a wide range of patients (11-91 years; mean age 51.5 years), 75% of the upper GI bleeds were gastroduodenal and due to peptic ulcer disease. 68 Other upper GI bleeding sites identified by endoscopy include Mallory-Weiss tears (∼10%), esophageal varices (∼5%), mucosal inflammatory disorders (∼5%), and gastric cancer (∼1%). Patients with a recently resolved upper GI bleed unrelated to nonsteroidal anti-inflammatory drugs (NSAIDs) do not appear to be at increased risk of bleeding if they have been evaluated and treated as indicated for Helicobacter pylori. 69,70 The incidence of acute major lower GI tract bleeding is approximately 20% to 30% that of major upper tract bleeding. 66 Common causes vary among reports, but etiology and midrange incidences are diverticulosis (24%), ischemia (12%), benign anorectal disease (12%), tumors (7%), colitis not otherwise specified (14%), angiodysplasia (2%), inflammatory bowel disease (3%), and other (26%). Caution should be exercised in bleeding risk assessment in patients in whom the bleeding site is obscure; in this instance, there is a significant increased risk of rebleeding, particularly within the first 6 months. 71
Labile INR (Due to Changes in PD) Increases the Risk of OAC-Associated Bleeding
Age-related alterations in PD may be even more important than PK changes in patients treated with warfarin. A labile INR is the “L” in HAS-BLED and contributes 1 point to a patient’s HAS-BLED score and is not a feature of the other bleeding risk models. 12 Advancing age is associated with inhibition of the synthesis of vitamin K-dependent clotting factors independent of warfarin therapy. In parallel studies conducted in both humans and rodents, the elderly patients had greater anticoagulant response despite administration of a lower weight-related dose. 72 Despite these PD changes, there were no appreciable differences in half-life, volume of distribution, plasma clearance, or the rate of degradation of the vitamin K-dependent coagulation factors (ie, factors II, VII, IX, and X) in either group. In a study of the INR levels of patients followed in an anticoagulation clinic, 57% of whom were aged 65 years or older, INRs >6.0 occurred during the induction phase in 11% of the patients and were significantly more common in those 65 years of age and older (P < .05). 73 Despite lower doses of warfarin during the maintenance phase, INRs were significantly and positively correlated with advancing age. In a study of tolerability of warfarin in the elderly individuals, age ≥80 years and INR ≥4.0 were associated with increased risk of bleeding during the first 90 days of treatment. 74 In addition, elderly patients are slower to normalize their INR and may require more aggressive management of excessive anticoagulation. 75 These PD alterations may also be important considerations in patients treated with the direct factor inhibitors since both factors II and X are vitamin K dependent, and thus their age-related decline may be exaggerated when these new agents inhibit the activity of their targeted protein.
Drugs and/or Alcohol Increase the Risk of OAC-Associated Bleeding
Weighted as 1 point (11%), the use of NSAIDs, antiplatelet drugs, or concomitant alcohol is the “D” in HAS-BLED and the final element of the score. 12 The NRAF, on the other hand, weights the contribution to the overall risk of bleeding from antiplatelet agents and alcohol/drug abuse to 8% and 17%, respectively. 13 Conventional NSAID use (without cytoprotection) and significant alcohol consumption are relative contraindications to OAC. 76 The use of NSAIDs increases with age and is particularly significant in the elderly individuals who may be taking 1 or more NSAIDs for chronic cardiovascular or rheumatologic disorders. 77,78 The primary risk of NSAID therapy is dose-related GI toxicity. The GI toxicity from NSAIDs is a function of the systemic effects of the drugs on upper GI mucosal cyclooxygenase activity. 79 Chronic NSAID use can also produce a significant number of macroscopic small-bowel lesions (P < .01). The literature suggests that aspirin or warfarin monotherapy increases the risk of major GI bleeding by 1.2- to 2.4-fold. 80 In the elderly individuals, combination of NSAID plus warfarin increases the risk of major GI bleeding by ∼9-fold. Cytoprotection with either misoprostol or proton pump inhibitor therapy decreases the risk of NSAID-associated major GI bleeding by ∼50%. Results of the Rotterdam Scan Study indicate that inhibitors of platelet function such as aspirin increase the risks of cerebral microbleeds in the elderly individuals, independent of hypertension or the use of antithrombotic drugs. 81 The baseline risk of subdural hematoma and of intracerebral hemorrhage in the elderly patients are 0.04% and 0.10%, respectively. 80 Aspirin doubles the risk of both subdural and intracerebral hemorrhage in the elderly patients, while warfarin increases the risks by 3.25- and 7.5-fold, respectively. 80 Acetaminophen has minimal effects upon peripheral cyclooxygenase activity. However, the medication produces a dose-related increase in INR >6.0 that becomes significant at doses ≥2.3 g/week. 82
Ethanol abuse is associated with an increased risk of anticoagulant-related bleeding and contributes 9% and 17% to bleeding risk in HEMORR2HAGES (ethanol abuse) and NRAF (alcohol/drug abuse), respectively, but is grouped with drugs in HAS-BLED. 17 Surrogates of alcoholism such as increased erythrocyte mean corpuscular volume and elevated levels of γ-glutamyl transpeptidase are more common in patients with spontaneous ICH than in those without. 83 The risk associated with alcohol abuse seems to be multifactorial. Excess alcohol intake can cause hypertension, and it is possible that increased blood pressure following a binge may predispose to an ICH. Alcohol also has a direct toxic effect upon megakaryocytes that can cause thrombocytopenia, generally mild, in 3% to 43% of well-nourished, nonacutely ill alcoholics. 84 It can also cause a thrombocytopathy that impairs platelet aggregation and function. Other factors that may contribute to the risks arising from excess ethanol consumption include trauma from falling or accidents while the patient is intoxicated, alcoholic gastropathy, and metabolic effects on the liver.
The Emerging Class of Selective, Direct Factor Inhibitors—How to Better Manage Patients With AF
Even with increasing awareness of various bleeding risk assessment tools, there is no generally accepted tool to integrate risks of bleeding and individualize dosing. As already noted, underthromboprophylaxis in patients with AF is notorious. New and investigational direct inhibitors of thrombin (dabigatran) and factor Xa (rivaroxaban, apixaban, edoxaban) are touted for their potential to improve AF care. All have been evaluated in large-scale randomized trials of patients with AF. As we have examined the various elements that increase the risk of OAC-associated bleeding in elderly patients with AF, it is reasonable to consider whether the various biologic elements could provide clues to simplify identification of the optimal antithrombotic dosing regimen.
Reassessing the RE-LY Trial for Clues to Age, Risk of Bleeding, and Dosing
In the fourth quarter of 2010, the US Food and Drug Administration (FDA) approved dabigatran for the reduction in the risk of stroke and systemic embolism in patients with nonvalvular AF. The decision to license the drug was based upon analysis of the results of the RE-LY trial of dabigatran in 18 113 patients with nonvalvular AF and at least 1 risk factor for stroke. 6 Patients were randomized to receive fixed doses of dabigatran 110 mg (n = 6015) or 150 mg (n = 6076) twice daily or dose-adjusted warfarin (INR 2.0 to 3.0, n = 6022). The annual rates of stroke or systemic embolism with the vitamin K antagonist and the lower and higher doses of the direct thrombin inhibitor were 1.71%, 1.54%, and 1.11%, respectively. Although both the dabigatran doses were noninferior to warfarin (P < .001), since the higher dose was also superior to the vitamin K antagonist (P < .001), the agency only licensed the 150-mg dose. 85 This decision was the result of a risk–benefit assessment. The FDA based this decision on 2 principles: (1) the adverse impact of stroke and systemic embolism in patients with AF are greater than nonfatal bleeding; and (2) nonfatal bleeding is much more common than life-threatening bleeding in patients with AF treated with OACs.
In its assessment, the agency relied on 3 bleeding risk factors to assess the net clinical benefit of the 2 dosing strategies, Age/elderly, Bleeding history, and Chronic kidney disease /impaired renal function, also known as the ABC approach. 85 Bleeding risks evaluated by these ABCs are assessed relative to the risks of stroke. The FDA was unable to identify any population for whom the 110-mg dose would improve dabigatran’s risk–benefit profile and concluded that most, if not all, patients should receive the 150-mg dose. The FDA investigators commented that they were concerned that approval of the lower dose might appear to endorse a “play-it-safe” approach that would encourage the use of a less effective regimen. Based upon PK/PD modeling, however, a 50% reduction in the approved dose (ie, 75 mg) was approved in patients with severe renal impairment.
In contrast to the FDA, the European Medicines Agency (EMA) approved both the 110- and 150-mg doses of dabigatran. Moreover, the European Society of Cardiology applied the HAS-BLED score (0-2 vs ≥3) to identify patients who would be candidates for the 150-mg and the 110-mg doses, respectively. 86 Age >65 years, bleeding (history of, predisposition to, or anemia), and abnormal kidney function correspond to a HAS-BLED score of 3 and are used to identify candidates for whom the 110-mg dose provides an improved risk–benefit profile.
Similarly, the Japanese dabigatran package insert recommends that the 110-mg dose should be considered for patients who are 70 years of age and older or have a history of GI bleeding or CKD. 87 Japanese prescribing guidelines also suggest that the PK effects of concomitant therapy with an oral medication (“Drug”) that inhibits P-gp is another reason to consider substituting the 110-mg for the 150-mg formulation. Thus, the elements of the Japanese dosing guidelines can be referred to as the ABC/Ds.
Applying the ABC/D Dose Adjustment Model to Other Antithrombotic Agents in Patients With AF
Although the premise of using ABC/D factors for risk assessment and dosing has only been applied to dabigatran, the same logic generalizes to the other selective direct factor inhibitors. In general, (1) patients with AF are generally older 88 ; (2) with advancing age, there is an increased prevalence of comorbidities that increase their risks of bleeding 89,90 ; (3) the prevalence of CKD increases rapidly with age 91 ; (4) depending upon the agent, renal excretion is responsible for elimination of 25% to 80% of the direct factor inhibitors 4,92 ; and (5) older patients and those with AF may be taking drugs that interact with the P-gp efflux pump (eg, both verapamil and quinidine are strong P-gp inhibitors) and can alter OAC activity.
Edoxaban is an investigational selective, direct factor Xa inhibitor with some novel properties that may provide theoretical advantages for older patients with AF. In the phase 2 trial of the drug, the risk of bleeding was a function of the Cmin rather than Cmax. 93 Although the explanation for this observation is not clear, it may be mediated by restoration of normal hemostasis between doses as a function of either the properties of the drug or the dosing schedule. Clinical trial simulations were used to select once-daily 30-mg and 60-mg doses for clinical trials. 94 Edoxaban is currently being compared with warfarin (INR 2.0-3.0) in the phase 3, multinational Effective aNticoaGulation with factor xA next GEneration in Atrial Fibrillation–Thrombolysis In Myocardial Infarction study 48 (ENGAGE AF-TIMI 48) trial 95 of approximately 20 500 patients with AF and CHADS2 score of ≥2. 95 Elements of this trial have the potential to provide additional insight into the role of the various ABC/D elements of antithrombotic therapy and bleeding risk in the elderly patients with AF. The trial design explores low- and high-dose exposure strategies to further define its clinical properties. Patients are initially randomized in a 1:1:1 schedule to warfarin or edoxaban 60 or 30 mg. If an edoxaban recipient has clinical factors that may increase drug exposure and thus the risk of bleeding, the patient can receive a 50% dose adjustment either at randomization or during participation in the trial. Dose reduction is recommended if patients have any of the following: (1) calculated creatinine clearance of 30 to 50 mL/min (Cockcroft-Gault formula); (2) weight ≤60 kg; or (3) treatment with a strong P-gp inhibitor. Depending upon the results of the trial, patients may be candidates for 60-mg, 30-mg, or 15-mg doses.
Conclusions
Atrial fibrillation is common and highly detrimental to older adults. Although novel OAC agents are becoming available, bleeding risks persist. In this review, we focus on the elements of bleeding risk that are inherent in aging and in age-related physiology/medical management. The 9-variable HAS-BLED bleeding risk score for patients with AF who are candidates for OAC was used in this analysis for the following reasons: (1) it broadly covers pathophysiologic risk factors for bleeding in this population; (2) it is easily implemented in the clinic; and (3) it has been endorsed by a number of professional societies. 15–17 In practice, choices of OAC may be enhanced by such perspective regarding patients themselves, tailoring medication types and doses to each patient’s bleeding risk profile. Both the European and the Japanese models use the ABC/D approach to tailor dabigatran therapy, and this HAS-BLED-based analysis demonstrates that it may provide a practical guideline for dose selection for recently introduced and other novel oral antithrombin agents in development.
Footnotes
Acknowledgments
The authors would like to thank Daiichi Sankyo for providing unrestricted funding for editorial support in the preparation of this manuscript. Lawrence Hargett provided the editorial support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DF has no conflicts of interest. RG has served as a consultant to Bayer Schering Pharma, Boehringer Ingelheim, Bristol-Myers Squib, Daiichi Sankyo, and Sanofi.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Editorial work was funded by Daiichi Sankyo.

