Abstract
Aim:
To compare the safety and efficacy of direct oral anticoagulants (DOAC) relative to vitamin K antagonists (VKA) for the treatment of left ventricular thrombus (LVT).
Methods:
This retrospective study enrolled patients diagnosed with LVT from 2014-2017. Patient characteristics and outcomes within 12 months of LVT diagnosis were recorded and analyzed. A meta-analysis was also performed by pooling our results with existing data in literature.
Results:
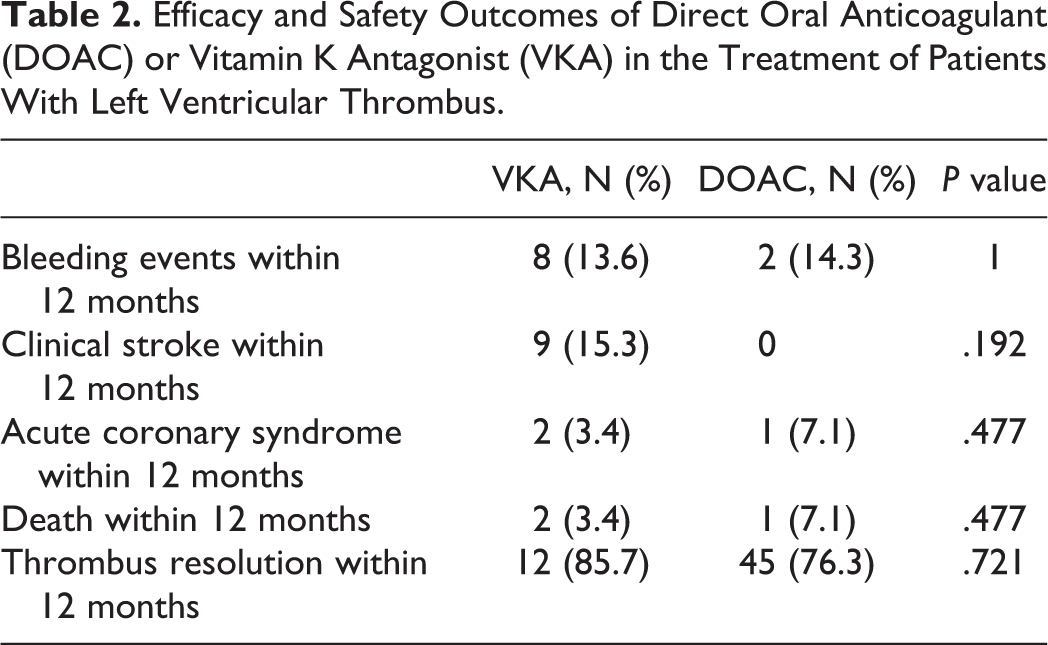
14 DOAC and 59 VKA patients were included. Baseline demographic and clinical characteristics were similar except for age. Although more strokes within 12 months occurred in VKA (15%) than in DOAC (0%) patients, this was not statistically significant (P = 0.189). There were no significant differences in outcomes between patients on DOAC and VKA for acute coronary syndrome (ACS) (7%, vs 3.4%, P = .477), LVT resolution (86% vs 76%, P = .499) or bleeding (14% vs 14%, P = 1) within 12 months. The meta-analysis included 6 studies (n = 408 for DOACs; n = 1207 for VKA). There were no significant differences between DOACs versus VKAs with respect to odds for unresolved thrombus (OR 0.61, 95% CI 0.26,1.41), embolic events (OR 1.24, 95% CI 0.90,1.69), embolic events and death (OR 1.10, 95% CI 0.84,1.45) or bleeding events (OR 1.13, 95% CI 0.74,1.72).
Conclusions:
Our study and meta-analysis suggest similar efficacy and safety of DOACs in the treatment of LVT compared to VKA. These findings underscore the need for a randomized controlled trial.
Introduction
Left ventricular thrombus (LVT) is a complication of acute myocardial infarction (MI), chronic cardiomyopathy with reduced ejection fraction, and coagulation disorders. 1 -3 The incidence of LVT is estimated to be around 15% in patients who suffer acute anterior ST elevation myocardial infarction (STEMI). 4 If untreated, the risk of thromboembolic events such as embolic strokes or systemic embolism approaches 15%. 5
The current standard of care for LVT is anticoagulation with an oral vitamin K antagonist (VKA), most often warfarin, as endorsed by several major cardiovascular societies. The 2013 American College of Cardiology (ACC)/American Heart Association (AHA) STEMI guidelines recommend VKA for at least 3 months (in addition to indicated antiplatelet therapy) to treat LVT. 6 The 2012 European Society of Cardiology (ESC) STEMI guideline recommended 6 months of VKA therapy for LVT, and advised that repeat echocardiography as early as 3 months after the sentinel event may be used to guide duration of anticoagulant therapy. 7 Subsequent guidelines appear more flexible. The 2017 ESC STEMI guidelines recommend anticoagulation with consideration of bleeding risk and concomitant anti-platelet therapy, while acknowledging the limited data on DOAC in this setting. 8 The 2014 guideline statement from the AHA in conjunction with the American Stroke Association regarding the prevention of stroke in patients with acute MI and LVT advised that non-VKA anticoagulants (including dabigatran, rivaroxaban, or apixaban) could be reasonably used for patients intolerant of VKA. 9
DOACs are an attractive option with fewer drug interactions, no dietary restrictions, reduced need for monitoring, similar efficacy, and lower risk of intracranial bleeding in the treatment of other thromboembolic conditions as compared to VKA. 10 -15 To date, no randomized trials exist to compare the treatment of LVT with DOAC versus VKA. Previously, evidence for the use of DOACs in the setting of LVT have been confined to case reports and case series, and there remain limited comparative studies of DOACs versus VKA. 16 -29
The aim of our study is to add to the current body of literature to assess the safety and efficacy of DOACs compared to VKA for the treatment of LVT in 2 large academic hospitals. We further pooled data of our study with existing studies and performed a meta-analysis to examine the effect of DOAC versus VKA in the treatment of LVT.
Methods and Materials
Consecutive patients with LVT from 2 academic hospitals from January 1, 2014 through December 31, 2017 were included. Patient information was obtained from electronic medical records (EMR). Inclusion criteria: all patients with LVT diagnosed via transthoracic echocardiogram (TTE). All included patients had intravenous contrast used during TTE. Exclusion criteria: patients who were lost to follow-up, LVT involving mechanical circulatory assist devices, pregnant patients, patients who switched between any anticoagulant therapy or who were documented to be non-compliant. Protocol was approved by institutional review boards of each hospital and the affiliated academic institution.
DOAC use was defined as use of apixaban, rivaroxaban, dabigatran or edoxaban as prescribed by physician for treatment of LVT. VKA use was defined as the use of warfarin. The outcomes assessed were ascertained based on medical record documentation and included stroke, acute coronary syndrome (ACS), all-cause death, bleeding (Thrombolysis in Myocardial Infarction (TIMI) bleeding criteria—minimal, minor, and major included), and thrombus resolution. Clinically documented stroke, ACS, and death were assessed within 1, 6, and 12 months after diagnosis of LVT. Bleeding and thrombus resolution were evaluated within 12 months. Thrombus resolution was determined by TTE, when one of the following was present: explicitly stated (i.e., no LVT), when the presence of LVT was no longer noted on the report, or if progress notes explicitly mentioned review of an outside TTE with resolution of LVT.
Patient demographics and clinical characteristics were summarized using median with minimum and maximum values, or frequency with percentage. The outcomes were stratified by VKA and DOAC and compared using the Wilcoxon rank sum, Fisher’s exact, or the Chi-square test. Independent logistic regression was also used to test the association between potential risk factors and outcomes. Factors found to be significant at P < 0.05 were considered for multivariable logistic regression.
For the meta-analysis, a systematic review was conducted using PubMed, EMBASE and Google Scholar searching for studies on DOACs (any of the listed agents as discussed above) in the treatment of LVT. Hits that did not specifically compare DOACs with VKA in LVT were excluded (PRISMA diagram shown in Figure 1). We then pooled our results with results from other studies using Mantel-Haenszel fixed-effect models estimating odds ratios and with 95% confidence interval (CI) for outcomes of thrombus resolution, embolic events, combined embolic events and death, and bleeding events.

PRISMA diagram of study selection for meta-analysis.
Results
Seventy-three patients were included in the analysis (14 in the DOAC group, 59 in the VKA group). Baseline characteristics are presented in Table 1 and were generally similar between the treatment groups but differed in age. Patients in the VKA group were older than patients in the DOAC group (median 62.0 vs 51.5 years, P = .043). The proportion of females was similar (23.7% vs 21.4%, P = 1). Racial distribution in both groups was roughly 5-7% Asian, 35-41% African American, 35% Hispanic, and 20% White. The left ventricular ejection fraction at the time of LVT diagnosis was <40% in 92% of patients in the DOAC group vs 86% of patients in the VKA group (P = .638). The LVT was diagnosed in the setting of acute MI in 43% of DOAC vs 49% in the VKA group (P = .770).
Baseline Characteristics of Included Patients With Left Ventricular Thrombus Treated With a Direct Oral Anticoagulant (DOAC) or Vitamin K Antagonist (VKA).

There were no significant differences in outcomes between patients on DOAC and VKA in stroke within 1 month (annotated as DOAC vs VKA: 0% vs 8.5% P = 0.576), 6 months (0% vs 6%, P = 0.337), or 12 months (0% vs 15%, P = 0.192). There was no difference in ACS or mortality between patients on DOAC and VKA within 1 month (0% vs 0%, P = 1), 6 months (0% vs 1.7%, P = 1), or 12 months (7% vs 3.4%, P = .477). Bleeding rates within 12 months were similar as well between DOAC and VKA groups (14% vs 14%, P = 1). The rate of LVT resolution within 12 months for DOAC was 86% and for VKA, 76% (P = .721). Outcomes are presented in Table 2. There were no patient demographics or baseline comorbidities that were associated with mortality, bleeding, or thrombus resolution. Prior history of stroke was associated with stroke within 12 months (OR 9.44, 95% CI 1.90,46.79) and ACS within 12 months (OR 18.00, 95% CI 1.44,224.67).
Efficacy and Safety Outcomes of Direct Oral Anticoagulant (DOAC) or Vitamin K Antagonist (VKA) in the Treatment of Patients With Left Ventricular Thrombus.
Meta-Analysis
After systematic review, a total of 6 studies (2 fully published papers and 3 published abstracts), in addition to our current study were included (n = 408 for DOACs; n = 1207 for VKA). 30 -34 Overall, there was no significant differences between patients treated with DOACs versus VKAs for LVT with respect to odds for unresolved thrombus (OR 0.61, 95% CI 0.26,1.41), embolic events (OR 1.24, 95% CI 0.90,1.69), or bleeding events (OR 1.13, 95% CI 0.74,1.72) (Figure 2). No significant heterogeneity was noted. Because there were a significant number of cross-over (n = 64) between DOACs and VKA in the study by Robinson et al, we also performed a sensitivity analysis for embolic events using available data from their intention-to-treat analysis and found no significant change in our results (OR 1.05, 95% CI 0.77,1.42) Table 3.
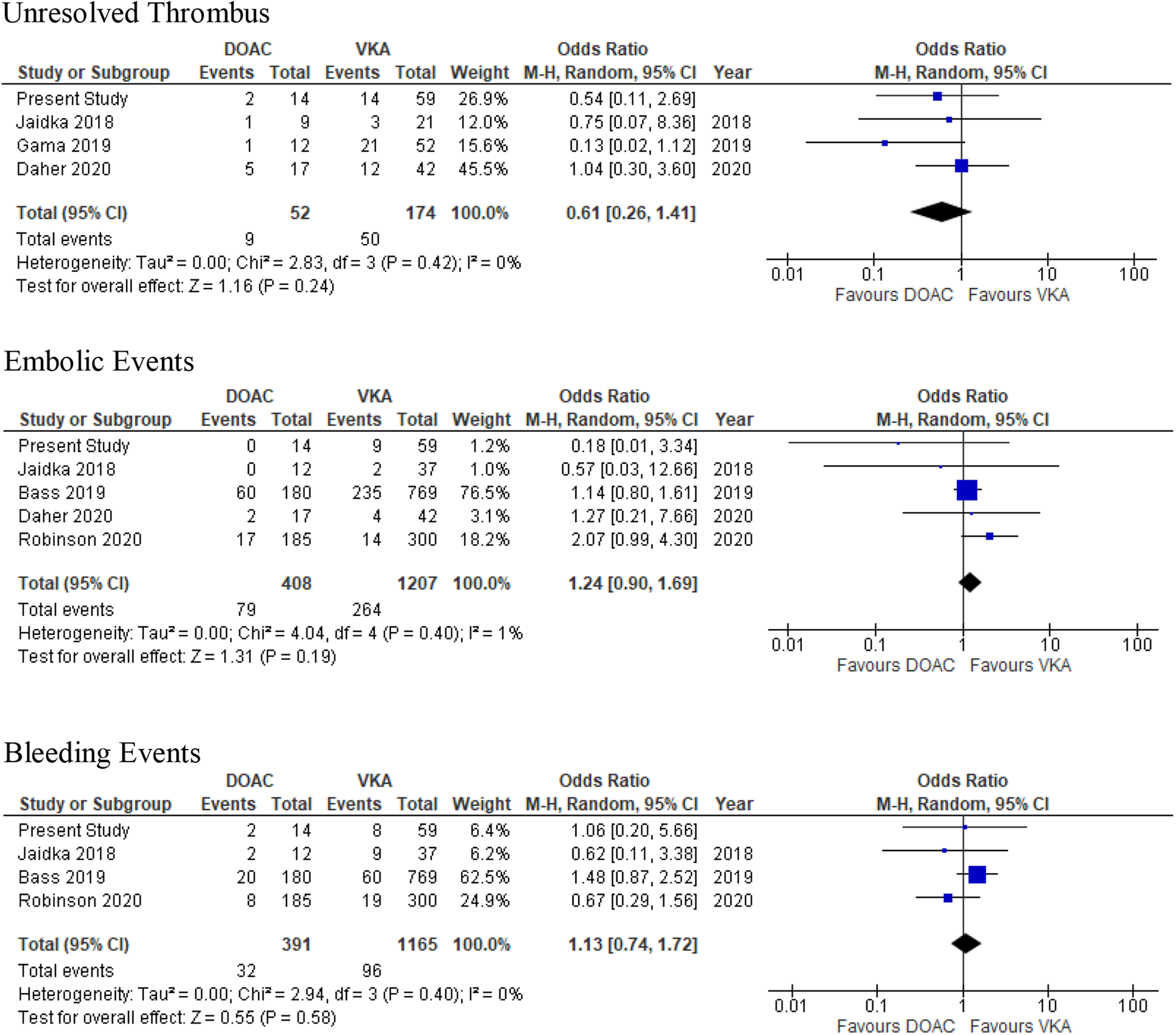
Meta-analysis of direct oral anticoagulant versus vitamin K antagonist in left ventricular thrombus comparing efficacy and safety outcomes.
Characteristics of Included Studies in the Meta-Analysis.

Discussion
LVT is a significant complication of acute myocardial infarction and heart failure requiring anticoagulation to prevent stroke, systemic embolism, and mortality secondary to thromboembolic events. Recent data suggest presence of LVT was associated with high risk for major adverse cardiovascular events and mortality while total LVT resolution while on anticoagulation therapy was associated with reduced mortality. 35 Limited data exists comparing the use of DOAC vs VKA for the treatment of LVT. In this retrospective study of patients with LVT, clinical efficacy (as assessed by thrombus resolution, stroke, all-cause mortality, ACS) and safety (as assessed by bleeding rates) did not differ significantly between the 2 anticoagulation groups. We also did not observe significant differences in efficacy and safety outcomes between the 2 treatments in a meta-analysis pooling our results with existing data.
Comparing our data to prior studies of LVT evaluating VKA anticoagulation, the rates of embolization were similarly low. 36,37 Evidence supporting the efficacy of DOACs in treatment of LVT for apixaban, rivaroxaban, and dabigatran had previously been limited to case reports and single-armed case series. 16 -29 The largest systematic review evaluating the clinical question of DOAC for LVT was performed by Kajy et al, and found high efficacy of thrombus resolution with low bleeding rates in data spanning 30 articles and 41 patients. 38 However, recently, a few studies including our own have examined the efficacy and safety of DOAC in the off-label treatment of LVT as compared to the current standard of care, VKA. Most of these studies and our meta-analysis suggest similar efficacy and safety between the 2 treatments. On the other hand, a large and comprehensive study performed by Robinson et al (included in the meta-analysis) found significantly higher rates of embolic events to be associated with DOAC use when compared to warfarin. 28 Overall rates of clot resolution were about 65% in this study, which is much lower than that percentage seen in previous data and results from our study. Interestingly, some patients developed embolic events even after documented thrombus resolution. Because the comprehensive nature and large size of the Robinson et al study, clinicians should continue to exercise caution in the off-label use of DOACs in the treatment of LVT. A randomized control trial is warranted to more definitively assess the efficacy and safety of DOAC for this purpose.
There are several limitations to our study including small sample size and retrospective design. With respect to meta-analysis, 3 of the studies included were published abstracts without full manuscripts which limited our ability to compare baseline characteristics. Further limitations include residual confounding and publication bias.
Conclusion
Since their implementation for venous thromboembolism and atrial fibrillation, DOACs have provided an attractive alternative to VKA for patients and clinicians alike, with reduced need for monitoring, no dietary restrictions, and a lower risk of intracranial bleeding. However, there remains a lack of sufficient data regarding efficacy and safety of DOACs in the treatment of LVT. Our study suggests that DOACs have similar efficacy and safety compared to VKA for this purpose. However, the results must be viewed with caution in the context of available evidence, demonstrating urgent need for a randomized control trial.
Footnotes
Authors’ Note
John M. Cochran and Xiaoming Jia contributed equally.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by Baylor College of Medicine.
