Abstract
Blood group types are associated with coronary artery disease. However, data are scarce about the impact of blood group types on coronary collateral circulation. In this study, we aimed to investigate the relationship between the blood group types and coronary collateral circulation. Two hundred and twelve patients who underwent coronary angiography in our department and had a stenosis of ≥ 90% in at least one major epicardial vessel were included in our study. Collateral degree was graded according to Rentrop-Cohen classification. After grading, patients were divided into poor coronary collateral circulation (Rentrop grade 0 and 1) and good coronary collateral circulation (Rentrop 2 and 3) groups. The ABO blood type of all participants was determined. The incidence rates of O blood group type were significantly higher in the good coronary collateral group compared to the poor collateral group (37.9% vs 17.1%, P < .001). The O type blood group was an independent predictor of good coronary collateral circulation (odds ratio = 1.83, 95% confidence interval = 1.56-6.18, P = .015). Coronary collateral circulation is associated with blood group types. The O blood group predicts good coronary collateral development among patients with coronary artery disease.
Introduction
Coronary artery disease (CAD) is the leading cause of death worldwide. Many factors play a critical role in clinical outcomes of CAD. Thus, there is great interest in the characteristics of these factors. One such factor is the coronary collateral development. Coronary collateral circulation (CCC) is interarterial coronary anastomoses that occur in the presence of occlusive CAD. 1 It constitutes an alternative source of blood supply to cardiac muscle that may otherwise be lacking oxygen. Good CCC is associated with decreased mortality and smaller infarct size. 2,3 Collateralization varies among patients with similar occlusive CAD. Determinants of good CCC are still under investigation.
Currently, it is acknowledged that blood type groups are associated with CAD. 4,5 Genome-wide association studies have determined a significant association between the blood type group genes and inflammatory marker genes. 6 –9
Blood group types frequency is different in different areas of the world. Race is an important determinant of blood type ratios. 10 In our country it was reported that O blood group type ratio was 32.7%. 11 This ratio is lower than the reported ratio of European countries and United States.
Because inflammatory and genetic markers play a role in CCC development, there might also be a relationship between CCC and blood type groups. In this study, we evaluated the association between blood type groups and coronary collateral development.
Methods
Study Population
From December 2016 to October 2017, a total of 212 patients with stable or unstable angina pectoris and ischemia on noninvasive stress tests underwent coronary angiography at our clinics and had at least one major coronary artery occlusion or stenosis of ≥ 90% with thrombolysis in myocardial infarction (TIMI) grade 1 anterograde flow were enrolled.
Patients with advanced renal disease (estimated glomerular filtration rate ≤ 30 mL/min/1.73 m2), acute myocardial infarction, liver cirrhosis, recent cerebrovascular disease, hematological disease, or severe valvular disease were excluded from the study. Patients receiving antihypertensive therapy and/or having an arterial blood pressure above 140 mm Hg systolic and/or 90 mm Hg diastolic were diagnosed as hypertensive. Patients receiving antidiabetic drugs and or fasting plasma glucose >126 mg/dL were diagnosed as having diabetes mellitus (DM). Patients with total cholesterol >200 mg/dL, low density lipoprotein >130 mg/dL, and triglycerides >150 mg/dL and who were receiving lipid lowering therapy were considered to have hyperlipidemia. Patients who were smoking or who quit smoking within the last year were accepted as smokers. Consistent with the Declaration of Helsinki, the study was approved by the Institutional Ethics Committee, and informed consent was obtained from all study participants.
Laboratory Parameters
Blood samples were drawn from a peripheral vein after a 12-hour fast before the coronary angiography procedure. The ABO blood typing was performed using a hemagglutination test (Erytype S ABO Microplates, Biotest AG, Dreieich, Germany). Erythrocyte count, hemoglobin, hematocrit, and white blood cell count were measured using an automated hematology analyzer XE-2100 (Sysmex Corporation, Kobe, Japan). The biochemical measurements were determined using a molecular analyzer (Roche Diagnostics, Manheim, Germany).
Echocardiography
Baseline 2D and Doppler echocardiographic (GE Vivid 7 Dimension® ultrasound system, General Electric Company, Fairfield, Connecticut) examination was performed before the coronary angiography procedure. Left ventricular ejection fraction, left ventricular diameters, right ventricular function, and valvular pathologies were evaluated.
Coronary Angiography Procedure
Selective left and right coronary angiography was performed using the Judkins technique via the femoral or radial artery. All recordings were evaluated by 2 experienced interventional cardiologists who blinded the clinical parameters of the patients. Collateral grading was carried out on the major coronary vessel with ≥90% stenoses. The Rentrop-Cohen grading scale was used to quantify the extent of collateral filling. The most opacified projection was used for grading. The following values were assigned according to the scale: 0 = no visible filling of any collateral vessel or collateral channels, 1 = filling of side branches of the artery to be perfused by collateral vessels without visualization of the epicardial segment, 2 = partial filling of the epicardial artery by collateral vessels, or 3 = complete filling of the epicardial artery by collateral vessels. If there were more than one coronary collateral vessel, the highest Rentrop-Cohen grade was used for analysis. Severe multivessel CAD was defined as presence of at least 2 narrowed (≥90% stenosis) major coronary arteries. After the grading, Rentrop 0 and 1 collateral flow were classified as the poor CCC group, and Rentrop 2 and 3 collateral flow were classified as the good CCC group.
Statistical Analysis
Statistical analysis was performed using IBM SPSS Statistics 22.0 statistical software. For univariate analyses, 2-sample t tests were used to compare continuous variables, whereas the χ2 test or Fisher exact test was used to compare categorical variables between the 2 groups. For the quantitative parameters, the Mann-Whitney test was used to compare differences between 2 independent groups. The continuous variables were described as the means ± standard deviation, whereas the discrete variables were reported as frequencies and percentages. We analyzed the effects of different variables on the occurrence of coronary collateral development in univariate analysis and determined the variables whose unadjusted P value was <.10 as potential risk markers and these were included in the full model. We composed the model by using forward elimination at multivariate regression analysis, and we eliminated potential risk markers by using likelihood ratio tests. A P value of <.05 was accepted as statistically significant.
Results
Of the 212 patients who were included in the study, 124 were in the good CCC group and 88 were in the poor CCC group. The clinical characteristics and laboratory assessment of the patients are summarized in Table 1.
The Clinical Characteristics and Laboratory Parameters of Study Groups.a

Abbreviations: ACE, angiotensin converting enzyme; AT II, angiotensin II; BMI, body mass index; CAD, coronary artery disease; CCB, calcium channel blocker; HDL, high density lipoprotein; LDL, low density lipoprotein; LVEF, left ventricular ejection fraction; MPV, mean platelet volume; NLR, neutrophil to lymphocyte ratio; PDW, platelet distribution width; RDW, red blood distribution width; SD, standard deviation.
a Data were given as mean ± SD.
The mean age of the study groups was 65.4 ± 10.8 years in the good CCC group and 64.6 ± 11.4 in the poor CCC group (P = .135). The percentage of male patients was significantly higher in the patients with good CCC group compared with the poor CCC group (75% vs 42%, P < .012). The ratio of patients with hyperlipidemia and diabetes was higher in the poor CCC group (43.1% vs 30.6%, P = .028; 43.1% vs 19.3%, P = .030, respectively).
There was no statistically significant difference among the groups forage, body mass index (BMI), smoking status, creatinine, and hematological parameters such as hemogram, hematocrit, red blood cell distribution width (RDW), mean platelet volume (MPV), and platelet distribution width (PDW). However, the good CCC group had significantly higher ejection fraction (EF) values compared to the poor CCC group (56.4% vs 48.3%, P = .039) and higher neutrophil to lymphocyte ratio (NLR; 2.26 ± 0.9 vs 4.28 ± 1.8, P = .007), respectively. There was no statistically significant difference between the groups with respect to the medications taken by the patients, including angiotensin converting enzyme inhibitors, angiotensin receptor blockers, acetyl salicylic acid, beta blockers, statins, fibrates, oral nitrate, and calcium channel blockers. The percentage of the O blood group was statistically higher in the good CCC group (37.9% vs 17.1% P < .01). The percentage of O blood type was highest in the Rentrop 3 group and lowest in the Rentrop 0 group (86.9% vs 43.5% P < .001; Figure 1A). The percentage of non-O blood type was highest in the Rentrop 0 group and lowest in the Rentrop 3 group (56.5% vs 13.1%, P < .001; Figure 1B). There was no statistically significant difference between the groups with respect to the duration of ischemic symptoms. The mean number of coronary arteries with severe stenosis or occlusion was higher in the good collateral group (1.48 ± 0.4 vs 1.08 ± 0.8, P = .016; Table 2). Severe multivessel coronary stenosis frequency were higher in the good collateral group (37.9% vs 15.9%, P = .002). However, we did not determine any significant difference between the groups for particular diseased vessels (left anterior descending, circumflex and right coronary arteries). To determine independent predictors of good and poor collateral development, univariate and multivariate logistic regression analyses were performed (Tables 3 and 4). In multivariate regression analysis, O blood group (odds ratio [OR]: 1.83 [95% confidence interval, CI: 1.56-6.18], P = .015), male gender (OR: 0.83 [95% CI: 0.24-1.36], P = .034) and presence of total occlusion (OR: 4.73 [95% CI: 1.64-18.66], P = .038) remained independent predictors of good CCC. Determinants of poor CCC were NLR (OR: 1.33 [95% CI: 0.88-3.2], P < .010), non O blood type (OR: 1.83 [95% CI: 1.56-6.18], P = .015), and DM (OR: 3.11 [95% CI: 1.94-9.76], P = .008).
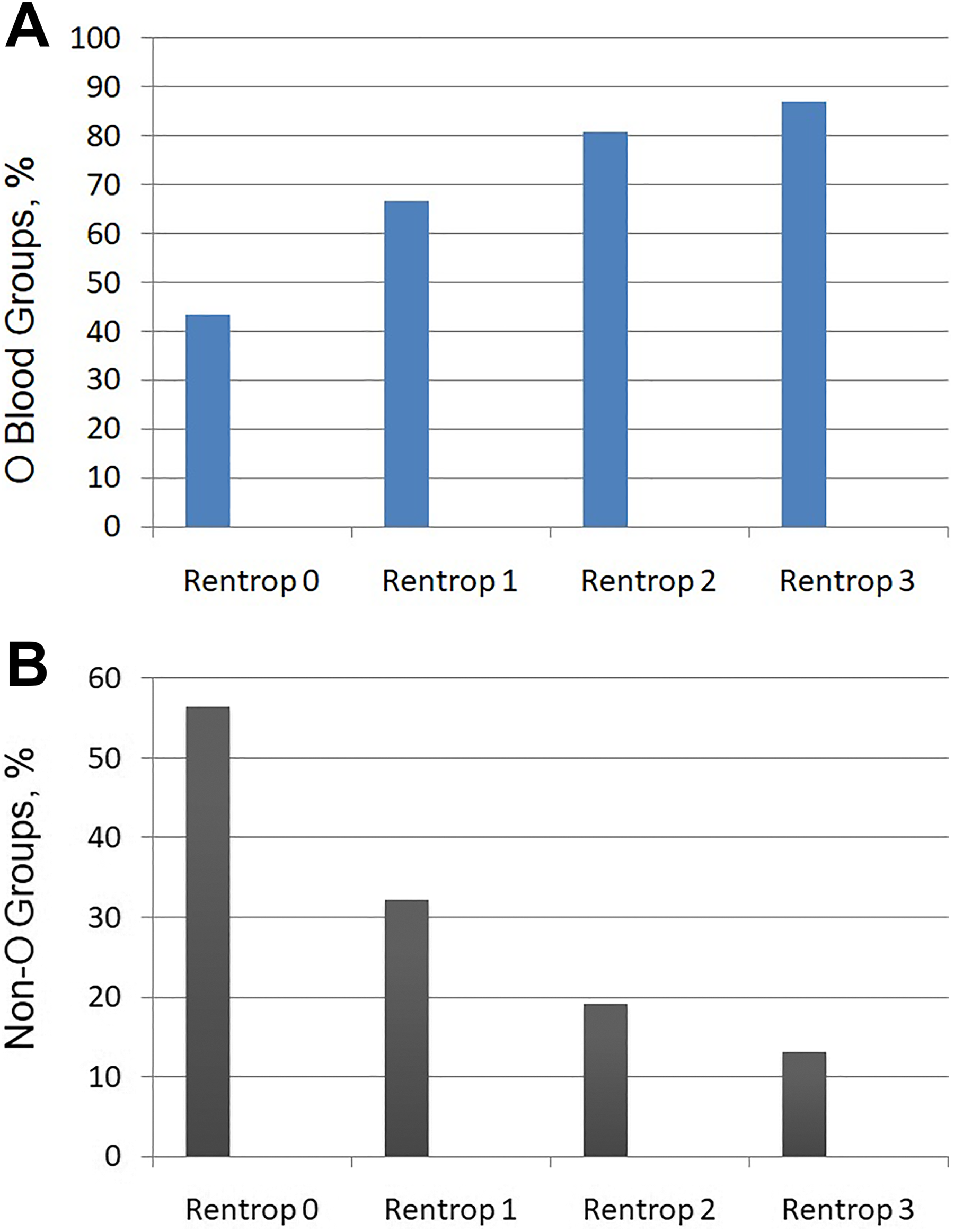
Percentage of patients with blood type among the Rentrop classes. A, Percentage of patients with blood type O. B, Percentage of patients with blood type non O.
Coronary Angiographic Data of the Study Groups.a
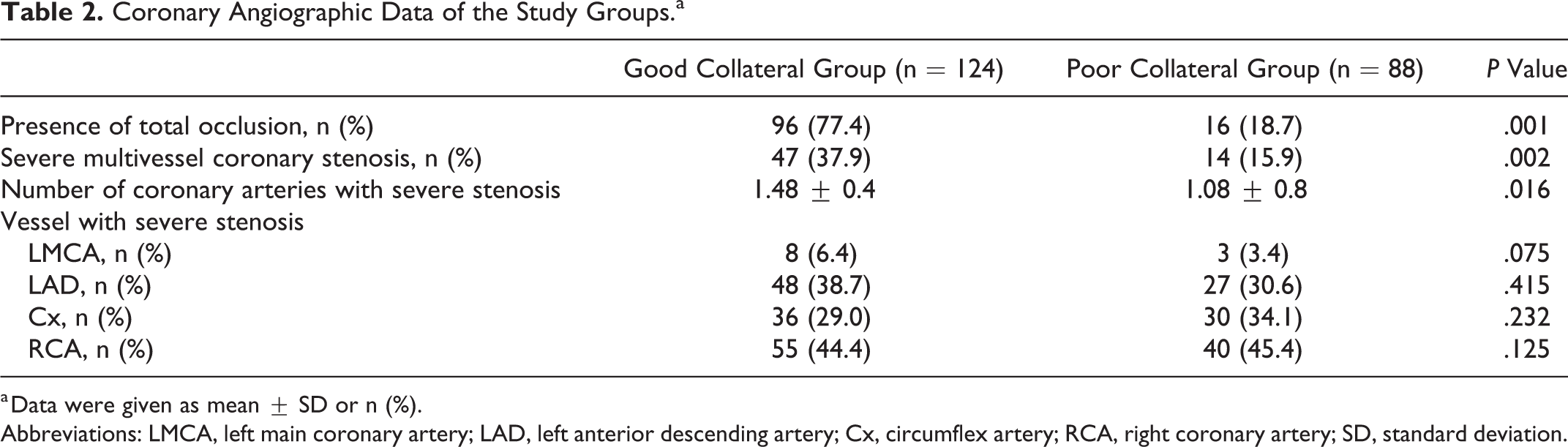
a Data were given as mean ± SD or n (%).
Abbreviations: LMCA, left main coronary artery; LAD, left anterior descending artery; Cx, circumflex artery; RCA, right coronary artery; SD, standard deviation.
Univariate and Multivariate Regression Analysis for Independent Predictor of GOOD Coronary Artery Development.
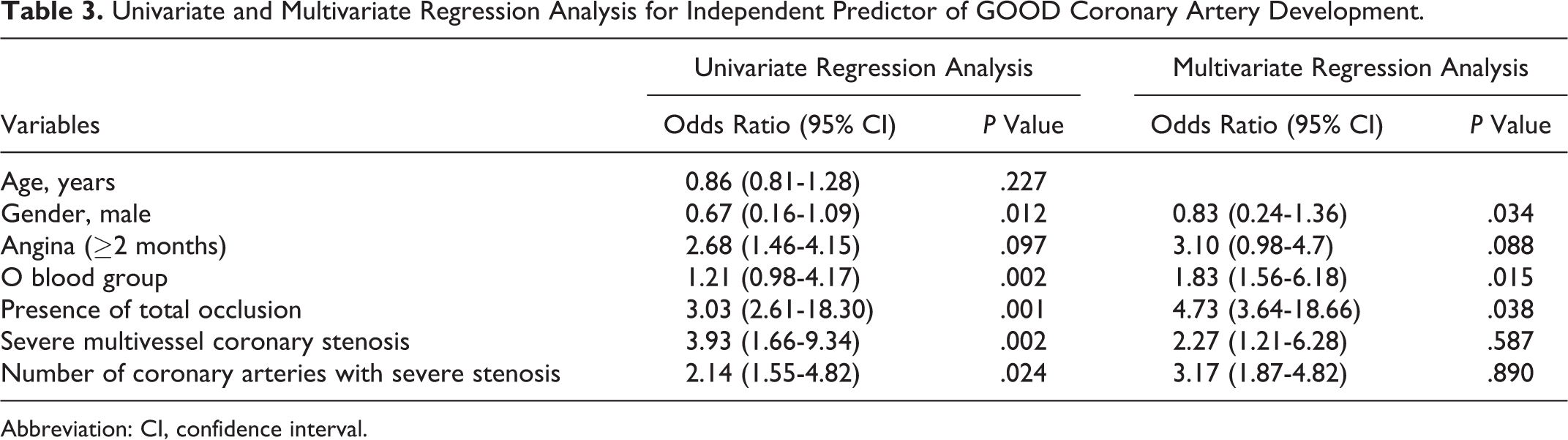
Abbreviation: CI, confidence interval.
Univariate and Multivariate Regression Analysis for Independent Predictor for Poor Coronary Artery Development.

Abbreviation: CI, confidence interval; NLR, neutrophil to lymphocyte ratio.
Discussion
The main finding of our study is that the O blood group is associated with good CCC. The O blood group was a predictor of good collateral coronary circulation in patients with >90% stenosis at least in one major epicardial coronary vessel. To the best of our knowledge, no previous study in the literature has examined the relationship between blood groups and coronary collateral development.
Good CCC is important because it has protective effects on ventricular function and infarct size, and reduces mortality and morbidity of CAD. 12 –14 Therefore, factors affecting the coronary collateral development are of interest for clinical studies. Coronary collateral circulation formation is affected by many factors, such as severity and duration of coronary artery stenosis, shear stress, endothelial dysfunction, and endogenous mediators. 15,16 Additionally, studies showed that hypertension, obesity, DM, dyslipidemia, and cigarette smoking are also associated with coronary collateral development. 17 –20 Endogenous mediators, including apelin, NLR, mean corpuscular volume, monocyte count, high sensitive C reactive protein, adhesion molecules, and growth factors were shown to be related with coronary collateral development. 21 –24 Additionally, Liu et al showed that CCC development is associated with race and sex. 25 In our study, we found that factors associated with poor coronary collateral development were DM, hypercholesterolemia, NLR, and non-O blood group type. In contrast, predictors of good coronary collateral development were O blood group and total occluded coronary artery. These findings are consistent with the literature. In contrast with previous studies, we did not find a relationship between hematologic parameters such as monocyte count, MPV, RDW, and CCC development. Additionally, we did not find a relationship between smoking status and the development of CCC.
Clinical trials determined that blood group types are associated with CAD. 5,26–27 A recent meta-analysis confirmed that non-O blood groups appear to be an independent risk factor for CAD and myocardial infarction. 28 Non-O blood groups have been found to show a significant relationship with family history of CAD and hypercholesterolemia, and this might be a possible genetic interaction between blood groups and CAD. 6
Previous data showed that mortality and morbidity of CAD is higher in patients with non-O blood group type, which can be attributed to differences in platelet function and inflammation, but can also partially be due to significantly lower rates of good collaterals compared to the O blood group. 4 As collateralization is a protective mechanism after a cardiovascular event, O blood type patients are more likely to have better outcomes following coronary events than non-O blood types because the additional collateral vessels circulate oxygen-enriched blood to heart muscle to prevent further damage.
In our study, we found that coronary collateral development differs between non-O and O blood types. We think that the mechanism of reduced coronary collateralization in non-O blood groups remains unclear and is likely multifactorial. One possible mechanism may be the pathophysiology of coronary collateral development. It is clear that atherogenesis is one of the most crucial processes in good coronary collateral development. The cellular adhesion pathway is important in atherogenesis. Arteriogenesis involves activation of endothelial cells, degradation of the basal membrane, leukocyte invasion, vascular cell proliferation, neointima formation, changes to the extracellular matrix, and cytokine production. These factors are also in close relation with blood type groups. Recent genome-wide association studies have identified multiple ABO gene associations with circulating levels of a number of markers of endothelial function, including sP-selectin and sICAM-1. 7 –9,29 ABO glycosyltransferases may regulate endothelial markers. Because there are reports indicating a close relationship between endothelial dysfunction and CCC, the association between blood group types and coronary collateral development may be explained with this hypothesis.
Limitations
Our study has some limitations. First, the study sample might be considered small, which limits the reliability of our results. Second, we did not evaluate the effect of Rh groups on coronary collateral development.
Conclusion
In summary, we showed that there is a relationship between blood type groups and coronary collateral development. Non-O blood group was an independent risk factor for the poor coronary collateral development. Further studies are needed to clarify the pathophysiologic mechanism between blood groups and collateral development.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
