Abstract
Background:
In the present clinical trial, we compared the efficacy and safety of the generic clopidogrel besylate (CB) with the innovator clopidogrel hydrogen sulfate (CHS) salt in patients eligible to receive clopidogrel.
Methods:
A prospective 2-arm, multicenter, open-label, phase 4 clinical trial. Consecutive patients (n = 1864) were screened and 1800 were enrolled in the trial and randomized to CHS or CB. Primary efficacy end point was the composite of myocardial infarction, stroke, or death from vascular causes, and primary safety end point was rate of bleeding events as defined by Bleeding Academic Research Consortium criteria.
Results:
At 12-month follow-up, no differences were observed between CB (n = 759) and CHS (n = 798) in primary efficacy and safety end points (age, sex, history of percutaneous coronary intervention adjusted odds ratio [OR], 0.70; 95% confidence interval [CI], 0.41-1.21 and OR, 0.81; 95% CI, 0.51-1.29, respectively) between CHS and CB. Analyses of efficacy and safety in subgroups that were defined according to the qualifying diagnosis revealed that there was no difference between CHS and CB.
Conclusion:
The efficacy and safety of CB administered for 12 months for the secondary prevention of atherothrombotic events are similar to that of CHS. (Salts of Clopidogrel: Investigation to ENsure Clinical Equivalence, SCIENCE trial; ClinicalTrials.gov Identifier:NCT02126982).
Keywords
Introduction
Clopidogrel is a widely used antiplatelet drug for the secondary prevention of atherothrombotic events, in combination with aspirin or as monotherapy. 1,2 Clopidogrel’s clinical benefit has been well established by a plethora of large clinical trials 1,3 –5 as well as in the real-world clinical practice. All this knowledge and experience have been obtained using the original product clopidogrel hydrogen sulfate (CHS; Plavix/Iscover). 1,3 –5 After expiration of its pharmaceutical patent in May 2012, clopidogrel has become generic, thus several commercial salt formulations have been developed and approved for use worldwide including Europe and the United States for the secondary prevention of atherothrombotic events. These include clopidogrel base, clopidogrel resinate, clopidogrel hydrochloride, and clopidogrel besylate (CB). 6,7 During the development of a new generic formulation, it is important to adhere to the manufacturing process and to use the same excipients as in the original salt in order to minimize stability problems. Specifically, during the development phase of an oral solid dosage form, several physiochemical as well as preformulation and formulation tests are carried out in order to achieve a high-quality generic product that is interchangeable with the original brand in terms of efficacy and safety. Accordingly, a generic product must show a dissolution profile in vitro, similar or overlapping that of the original product, in order to ensure a successful bioequivalence profile of the product. 8 However, according to the Biopharmaceutics Classification System, clopidogrel is a class II product, thus in vitro dissolution is not sufficient to prove that it is interchangeable with the original brand. 8,9 Thus, clopidogrel generic tablets should be subjected to bioequivalence study in comparison with the original formulation. Furthermore, it is of critical importance to demonstrate that these preparations have similar pharmacodynamic potency in inhibiting platelet reactivity and are also clinically bioequivalent to the original salt in terms of efficacy and safety. 7 In this regard, it should be emphasized that in contrast with other generic cardiovascular medications, such as statins or antihypertensives, for which minor differences in their biologic efficacy would be unlikely to have any discernible clinical effect, a minor decrement in the clopidogrel pharmacodynamic potency could result in severe ischemic thrombotic complications, especially in situations where therapeutic efficacy is critical, such as immediately after a percutaneous coronary intervention (PCI). 10 The necessity for evaluating the therapeutic efficacy of a generic clopidogrel salt is further supported by the substantial heterogeneity existing among various generic clopidogrel formulations, in terms of clopidogrel content, impurity profile, dissolution properties, and stability, even if these concern the same salt. 11
Today, a number of small and often underpowered studies comparing either pharmacodynamic or clinical end points in patients receiving generic versus brand name clopidogrel have been conducted. 12 –16 Among them, very few studies used hard outcomes and followed patients for more than 3 months, and many trials were conducted in healthy volunteers. 17 –20
Therefore, the aim of the present randomized phase 4 clinical trial named as Salts of Clopidogrel: Investigation to ENsure Clinical Equivalence (SCIENCE) was to compare the clinical efficacy and safety of the generic CB formulation with that of the original CHS, administered for 12 months in a relatively large number of patients eligible to receive clopidogrel. The interim analysis of this trial performed at 6 months of follow-up demonstrated that both clopidogrel salts were similar as regards the clinical efficacy and safety end points. 21
Methods
Trial Design
The SCIENCE was a prospective 2-arm, multicenter (9 centers), open-label, phase 4 clinical trial, designed to compare the short (6-month) and long (12-month) efficacy and safety of CB versus CHS in reducing atherothrombotic events in several types of patients eligible to receive clopidogrel. 21
Study Sample
Eligible patients were men and women >18 years old who were presented with one of the below-mentioned disorders, classified using the International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) codes. Patients with a stable coronary artery disease (CAD; I51.9) 22 being in treatment with 75 mg/day CHS for a minimum period of 2 months or patients who had the indication to undergo PCI (Z98.61) during randomization. Patients who had experienced an acute ischemic non-embolic stroke (I63) or a transient ischemic attack (TIA; Z86.73) during the previous 6 months or had a history of peripheral artery disease (PAD;I73.9) (I73.9) or carotid artery disease (I65.2) were also eligible for enrollment. Finally, patients with atrial fibrillation (AF; I48) who refused or were not eligible to use any oral anticoagulant were included in the study. The qualifying diagnosis of each disorder and the inclusion and exclusion criteria have been reported previously. 21
We screened 1864 consecutive patients in order to participate in the trial. 21 During screening, 4 patients were excluded because they did not meet the inclusion criteria, whereas 60 patients declined to participate since the procedure of participation had been explained. Thus, 1800 patients were randomized. Among them, 1766 patients (886 in the CHS and 880 in the CB group) received allocated intervention, whereas 34 patients (14 in the CHS and 20 in the CB group) were not treated because they underwent cardiac surgery (Figure 1).

CONSORT flow diagram of the Salts of Clopidogrel: Investigation to ENsure Clinical Equivalence (SCIENCE) Trial. CHS, clopidogrel hydrogen sulfate; CB, clopidogrel besylate; CONSORT, Consolidated Standards of Reporting Trials; FU, follow-up; ITT, intent-to-treat.
Randomization
After enrollment, patients with stable CAD, ischemic stroke or TIA, PAD, carotid artery disease, or AF were randomly assigned in a 1:1 ratio to receive 75 mg/day of either CHS or CB, by a central computerized system with hierarchical stratification according to the above qualifying diagnosis. Patients who underwent PCI were also randomly assigned in a 1:1 ratio to receive a loading dose of 600 mg clopidogrel as CHS or CB followed by 75 mg/day. Simple randomization was based on a computer-generated randomization list using Microsoft Excel software (Microsoft Hellas Co, Athens, Greece). The list was given to the study’s investigators who enrolled the patients and assigned them in one of the two groups. Medical personnel and patients were not blinded to treatment assignment during the whole study period. Therefore, outcomes were submitted to a 3-member adjudication committee constituted by the Atherothrombosis Research Centre of the University of Ioannina.
Follow-Up
The trial has been scheduled for 12 months, and outpatient visits were scheduled every month. Interim analysis at 6 months of follow-up was also performed. 21
End Points
The primary efficacy end point (classified according to ICD-10-CM) was the composite of myocardial infarction (MI) (I21), stroke (ischemic [I63] or hemorrhagic [I60]), or death from vascular causes for the entire follow-up period. Death from vascular causes was defined as death from cardiovascular (I46.2) or cerebrovascular (I60-I69) causes. The major secondary efficacy end point was the composite of MI, stroke, or death from any cause. Other secondary efficacy end points were MI, stroke (ischemic or hemorrhagic), death from vascular causes, death from cause other than vascular causes (except for cardiovascular and cerebrovascular causes), death from any cause, PCI (Z98.61), and stent thrombosis (T82.857A) during the follow-up period.
The primary safety end point was the rate of bleeding events as defined by Bleeding Academic Research Consortium (BARC) criteria. The secondary safety end points included permanent or temporary clopidogrel interruption for 1 to 5 days due to urticaria (L50), temporary clopidogrel interruption for 1 to 5 days, due to surgery, dental procedures, bleeding (BARC 1-3a) or personal reasons (for example financial difficulties). Other secondary end points were transient thrombocytopenia and initiation of anticoagulant therapy due to new AF.
Bioethics
The study protocol was approved by the ethics committee of the University Hospital of Ioannina and the study was conducted in accordance with the Declaration of Helsinki and consistent with the International Conference on Harmonised Tripartite Guideline for Good Clinical Practice. Written informed consent was obtained from each eligible patient before the screening procedure.
Statistical Analysis
The current analysis was designed to test whether either clopidogrel salt was equivalent to each other, within a 12-month follow-up period, as regards the primary end point, at 80% statistical power. To satisfy the equivalence hypothesis, the lower and upper bounds of the 2-sided 95% confidence interval (CI) for the odds ratios (ORs) of an outcome needed to fall between 0.90 and 1.25. This equivalence interval was derived from studying similar trials of clopidogrel salts in patients with CAD. Furthermore, a non-inferiority analysis of CB versus CHS was also applied setting the odds of an outcome less than 1.25 (statistical power = 83%). Data analysis was based on the intent-to-treat principle, whereas per-protocol analysis was also applied in order to compare the findings from the 2 approaches. Values of continuous variables were presented as mean and standard deviation, while categorical variables were expressed as counts and percentages. Logistic regression models, adjusted for age, sex, and history of PCI (due to lack of matching during randomization), were used to calculate ORs; 95% CI, and P values. Hosmer-Lemeshow test was used to evaluate models’ goodness of fit. Chi-square testing was used to compare rates of medication discontinuation and adverse events. Various patient characteristics × group interactions were also evaluated and when significant (at P < .05) were reported. Statistical analysis was performed using IBM SPSS software, version 21 (IBM Co, Minneapolis, Illinois).
Results
Among the 1766 patients who received allocated intervention, 1557 patients (759 in the CHS and 798 in the CB group) completed the 12-month follow-up, whereas 127 patients in CHS arm and 82 patients in CB arm lost to follow-up (Figure 1). Among them, 38 patients in CHS arm and 27 patients in CB arm presented with AF, switched to vitamin K antagonists, or to new oral anticoagulants within the first month of enrollment. Furthermore, 35 patients with stable CAD, 34 patients with stroke/TIA, 8 patients with PAD, and 12 patients with carotid artery disease in the CHS arm and 33 stable CAD, 13 stroke/TIA, 5 PAD, and 4 patients with carotid artery disease in the CB arm stopped clopidogrel and switched to aspirin monotherapy in the first 2 months after the recruitment, due to personal reasons (primarily financial difficulties). Baseline characteristics of the patients who completed the 12-month follow-up are shown in Table 1. Patients in the CHS group exhibited slightly lower mean age and higher ratio of male/female, which was taken into account in the analysis followed on. No differences between groups were observed in the qualifying diagnosis of patients, with the exception of the number of patients who underwent PCI, which was significantly higher in the CHS compared with the CB group (Table 1). No difference between groups was observed in the medical history as well as in major medications administered at baseline (Table 1). Details on medications administered in the trial participants have been published previously. 21
Baseline Characteristics of the Patients.

Abbreviations: CHS, clopidogrel hydrogen sulfate; CB, clopidogrel besylate; BMI, body mass index; CAD, coronary artery disease; PCI, percutaneous coronary intervention; TIA, transient ischemic attack; PAD, peripheral artery disease; CKD, chronic kidney disease; PPIs, proton pump inhibitors.
Efficacy End Points
At 12 months of follow-up, the primary end point of MI, stroke, or death from vascular causes had occurred in 32 (4.2%) patients in the CHS group and 26 (3.2%) patients in the CB group (age, sex, history of PCI adjusted OR, 0.70; 95% CI, 0.41-1.21; Figure 2A and Table 2). As regards patients who underwent PCI, stratified analysis was applied showing no differences in the primary efficacy end point between the two clopidogrel salts; patients who underwent PCI (age, sex adjusted OR 1.14; 95% CI 0.39-3.34) and patients who did not undergo PCI (age, sex adjusted OR 0.56; 95% CI 0.21-1.64).

Kaplan-Meier plots of (A) the primary efficacy end point of the composite of myocardial infarction, stroke (ischemic or hemorrhagic), or death from vascular causes, (B) The major secondary efficacy end point of the composite of myocardial infarction, stroke, or death from any cause. CHS, clopidogrel hydrogen sulfate; CB, clopidogrel besylate.
Primary and Secondary Efficacy End Points at 12 Months.

Abbreviations: CHS, clopidogrel hydrogen sulfate; CB, clopidogrel besylate; MI, myocardial infarction; PCI, percutaneous coronary intervention.
aEffect size measures were age, sex, and history of PCI adjusted.
The major secondary efficacy end point of MI, stroke, or death from any cause occurred in 38 (5.0%) patients in the CHS group as compared with 36 (4.5%) patients in the CB group (age, sex, history of PCI adjusted OR, 0.85; 95% CI, 0.52-1.37; Figure 2B and Table 2). No significant differences between the two clopidogrel salts were observed when the analysis was stratified by history of PCI status (history of PCI, OR 0.86; 95% CI 0.39-1.92, no history of PCI, OR 1.28; 95% CI 0.51-3.57). No significant differences were also observed in the other secondary efficacy end points between the two patient groups (Table 2). Similar results in the primary and secondary efficacy end points were obtained when per-protocol analysis was applied (data not shown). The 34.4% of patients in the CHS group and the 32.1% in the CB group were receiving aspirin (Table 1). Our results revealed that the primary and major secondary efficacy end points in patients receiving either dual antiplatelet therapy or clopidogrel monotherapy were similar between CHS and CB (Table 3).
Efficacy and Safety End Points at 12 Months in Patients Receiving Clopidogrel Monotherapy or the Combination of Clopidogrel With Aspirin.
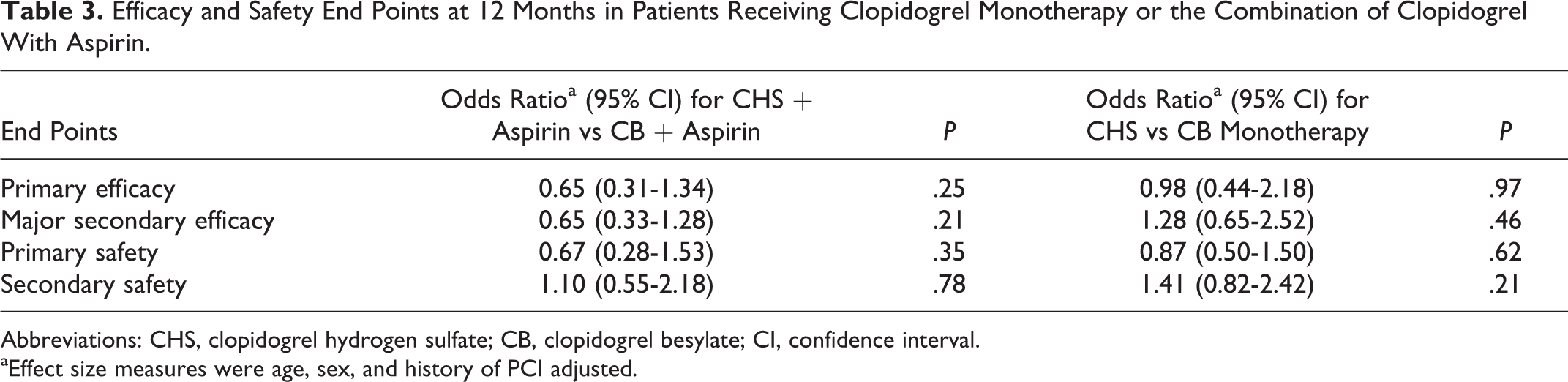
Abbreviations: CHS, clopidogrel hydrogen sulfate; CB, clopidogrel besylate; CI, confidence interval.
aEffect size measures were age, sex, and history of PCI adjusted.
Analyses of efficacy in subgroups that were defined according to the qualifying diagnosis revealed that there was no difference between CHS and CB groups in the primary or in the major secondary efficacy end point (Figure 3A and 3B). Similar results were observed in the other secondary efficacy end points (data not shown). It should be stated that due to small number of patients in the subgroups of PAD, carotid artery disease, and AF, subgroup analysis was performed by combining PAD and carotid artery disease subgroups, whereas this analysis was not performed in patients with AF. Finally, no significant interactions were observed between treatment group and patient age (P = .25) and sex (P = .52) on the studied efficacy end points.
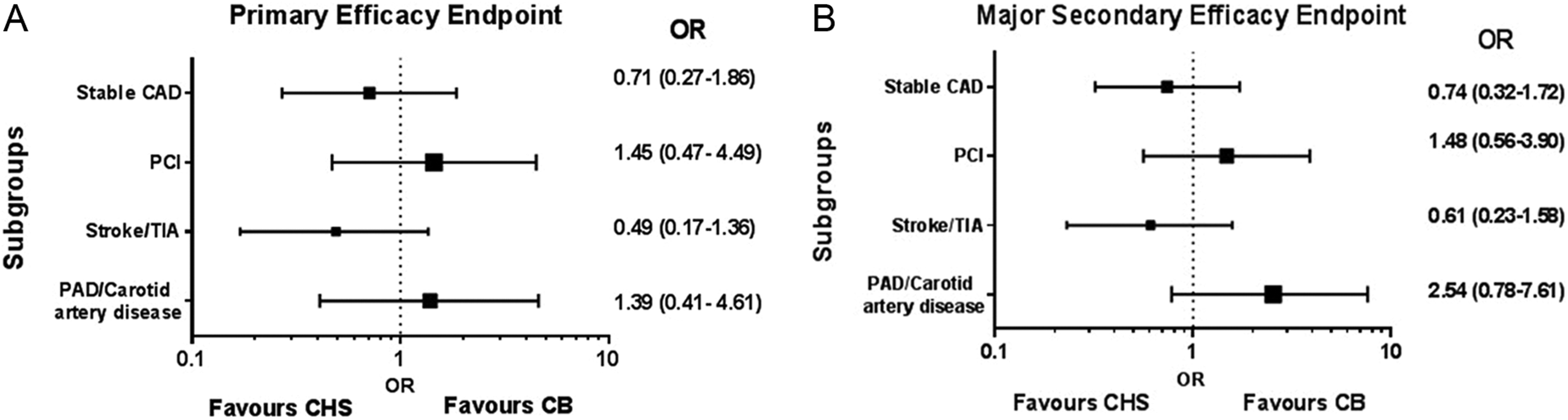
Forest plots in various patient subgroups of (A) primary efficacy end point of the composite of myocardial infarction, stroke (ischemic or hemorrhagic), or death from vascular causes. (B) Major secondary efficacy end point of the composite of myocardial infarction, stroke, or death from any cause. CHS, clopidogrel hydrogen sulfate; CB, clopidogrel besylate; CAD, coronary artery disease; PCI, percutaneous coronary intervention; TIA, Transient Ischaemic Attack; PAD, peripheral artery disease; OR, odds ratio.
Safety End Points
The primary safety end point of bleeding events according to BARC criteria occurred in 43 (5.6%) patients in the CHS group, as compared with 37 (4.6%) patients in the CB group (age, sex, history of PCI adjusted OR, 0.81; 95% CI, 0.51-1.29; Figure 4 and Table 4). Rates of BARC 1, 2, 3a, 3b, and 5b bleeding were similar between the 2 study groups, whereas 1 bleeding event according to BARC 3c occurred in the CHS group and no bleeding events according to BARC 4 and 5a were observed in either CHS or CB group (Table 4). Intracranial hemorrhage occurred in 2 patients in CHS group and in 1 in the CB group (OR, 0.41; 95% CI, 0.03-4.72), which were fatal. No difference in the secondary safety end points was also observed between the CHS and the CB groups (40 events; 5.2% and 53 events; 6.6%, respectively, age, sex, history of PCI adjusted OR, 1.27, 95% CI 0.82-1.96; Table 4). Similar results in the primary and secondary safety end points were obtained when per protocol analysis was applied (data not shown). The rates of primary and major secondary safety end points in patients receiving either dual antiplatelet therapy or clopidogrel monotherapy were similar between CHS and CB (Table 3). Analyses of safety in subgroups that were defined according to the qualifying diagnosis revealed that there was no difference between CHS and CB groups in the primary safety end point (Figure 5). Similar results were observed in the secondary efficacy end points (data not shown). It should be stated that due to small number of patients in the subgroups of PAD, carotid artery disease, and AF, subgroup analysis was performed by combining PAD and carotid artery disease subgroups, whereas this analysis was not performed in AF patients. Finally, no significant interactions were observed between treatment group and patient age (P = .98) and sex (P = .83) on the studied safety end points.
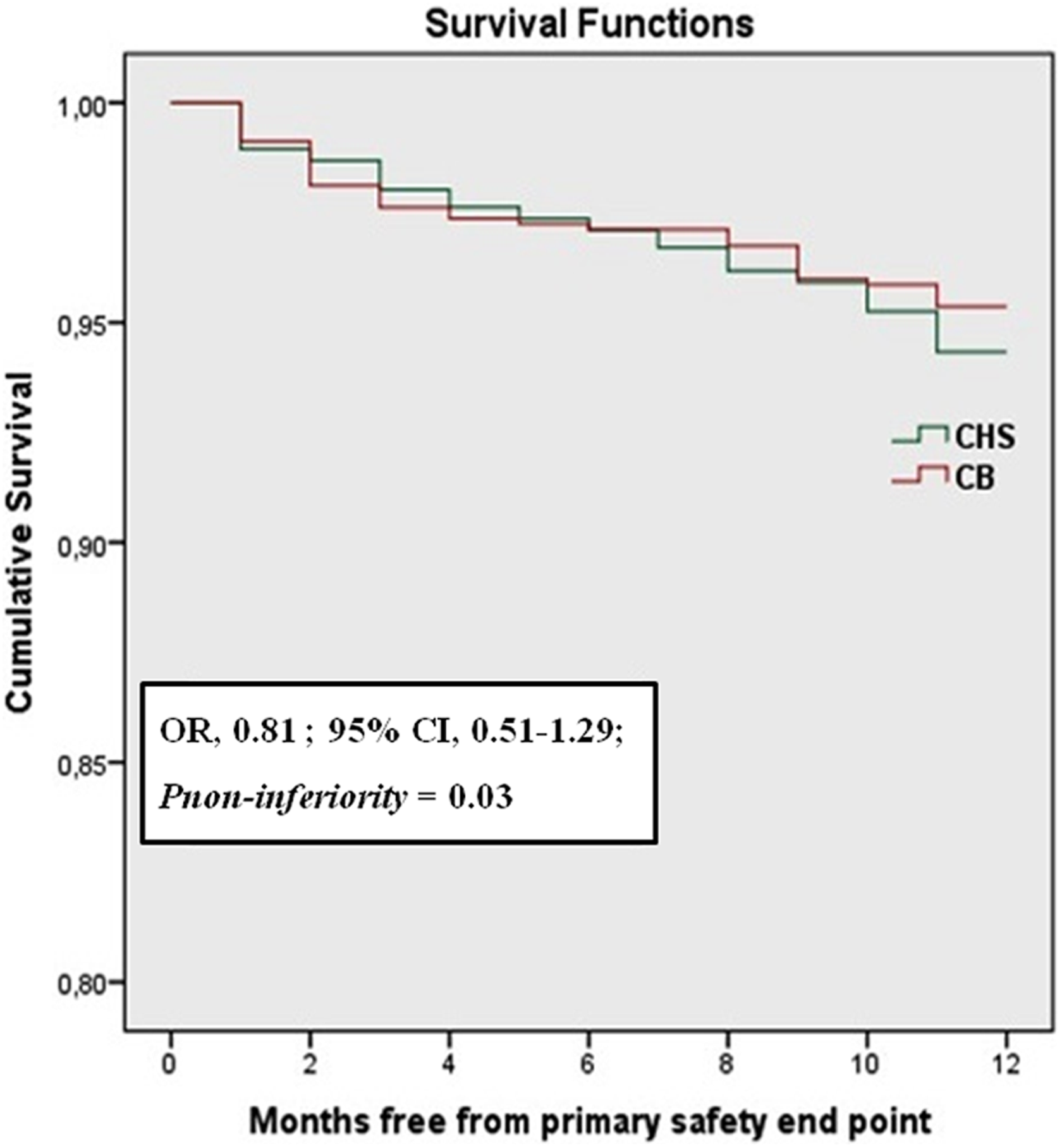
Kaplan-Meier curve of primary safety end point of the rate of bleeding events as defined by Bleeding Academic Research Consortium
Primary and Secondary Safety End Points at 12 Months.

Abbreviations: CHS, clopidogrel hydrogen sulfate; CB, clopidogrel besylate; BARC, Bleeding Academic Research Consortium; AF, atrial fibrillation; OR, odds ratio; PCI, percutaneous coronary intervention; CI, confidence interval.
aEffect size measures were age, sex, and history of PCI adjusted.

Forest plot in various patient subgroups of the primary safety end point. CHS, clopidogrel hydrogen sulfate; CB, clopidogrel besylate; CAD, coronary artery disease; PCI, percutaneous coronary intervention; PAD, peripheral artery disease; OR, odds ratio.
Discussion
The present clinical trial revealed that the clinical efficacy of CB salt administered for 12 months in several types of patients eligible to receive clopidogrel is similar to that of the innovator CHS salt. Furthermore, both clopidogrel formulations exhibited similar safety profile. The interim analysis of this trial performed at 6 months of follow-up also demonstrated that there were no differences in the clinical primary and secondary efficacy as well as safety end points between CB and CHS. 21 To our knowledge, this is the largest clinical trial performed to date in terms of population size, study duration, and subgroups of patients, aiming to compare the efficacy and safety of a generic clopidogrel salt with the innovator drug in the secondary prevention of atherothrombotic events.
As it has been reported, the number of observed events in the interim analysis of this trial was small, albeit similar to those reported for a 6-month period of follow-up. 23 Thus, we were not able to perform analysis of each patient subgroup participated in the study. This analysis was performed in the present study and revealed that CB and CHS exhibited similar efficacy and safety profile in each subgroup of patients, that is, in patients with stable CAD, ischemic stroke/TIA, PCI or PAD/carotid artery disease. Our results in the total population and especially in patients with stable CAD and in patients underwent PCI are consistent with previously published results by our group on 2 prospective pharmacodynamic studies comparing the antiplatelet effectiveness of the same CB product with that of original CHS formulation, in patients with a history of an ACS 24 and in patients with ACS undergoing PCI. 25 In both studies, CB was pharmacodynamically similar to CHS in all tests we used to evaluate the platelet functionality. 24,25 Furthermore, no differences in the number of patients exhibited high on-treatment platelet reactivity were observed between the 2 clopidogrel salts. 24,25 Although the duration of the above-mentioned pharmacodynamic studies did not exceed the 6 months, their results are relevant to the present study, since the platelet functionality did not change during the study period from 1 to 6 months. 24,25 Furthermore, previously published results by our group demonstrated that the platelet response to a 75 mg/day clopidogrel regimen is improved and stabilized at 1 month of therapy and then remains constant, even in clopidogrel non-responding patients. 26,27
The present data on the clinical efficacy and safety of the generic CB formulation are in line with previous results demonstrating that CB has pharmacokinetic and pharmacodynamic properties as well as tolerability similar to the innovator drug in healthy, medication-free volunteers. 17,18,28,29 Moreover, the intrinsic toxicity of the benzene sulfonic acid bound to clopidogrel to form CB salt 7 is low and proved to be safe. 30 Consistent with the above-mentioned data are the results of other pharmacodynamic and clinical studies comparing CB formulations with the innovator CHS salt. 14,31 Overall, CB is a pharmacodynamically and clinically effective generic clopidogrel salt that can be safely used for the prevention and treatment of atherothrombotic diseases. In this regard, it should however be stated that some studies have raised important concerns on the clinical and pharmacodynamic effectiveness of generic clopidogrel salt formulations, other than CB. 10,32 Importantly, a substantial heterogeneity in terms of mass uniformity, impurity profile, clopidogrel content, amount of impurities, dissolution properties, and stability was reported among various generic clopidogrel formulations, even if these concern the same salt. 11 These generic formulations were inferior, compared to the original drug product. 11 Despite this, the routine use of generic clopidogrels has rapidly gained momentum on a worldwide scale, primarily due to their low cost. Taking into account the above-mentioned data and considering the wide variability of platelet response to clopidogrel which significantly influences the drug’s pharmacodynamics and clinical effectiveness, 33 –36 we may suggest that studies comparing the pharmacodynamic and most importantly the clinical efficacy of various generic clopidogrel formulations available today would provide useful information to clinicians using these drugs in the daily clinical practice. Furthermore, it is of importance that any result arising from the use of a generic clopidogrel formulation should be referred to the specific product used and not generally to the generic clopidogrel.
Study Limitations
A limitation of the present clinical trial could be the significant differences in the age and sex between the CB and the CHS groups, at baseline examination. Furthermore significant difference between the 2 groups was observed in the number of patients who underwent PCI. Although a randomization procedure was applied by giving a list of random numbers that allocated patients to intervention groups, including PCI, it can’t guarantee the unexpected feature of the randomization nor prevent the knowledge of the allocations. This could lead to a major selection bias. Thus, all models were adjusted for these factors in order to account for potential bias in the results; nevertheless, no significant interactions were observed between the treatment groups and the above parameters. Another limitation of the present clinical trial could be heterogeneity in the number of patients receiving aspirin or drugs that could influence the efficacy of clopidogrel such as proton pump inhibitors. 37 However, our study groups were well balanced in terms of use of the above drugs (Table 1).
Conclusion
In conclusion, the SCIENCE trial demonstrates that the clinical efficacy and safety of CB in a relatively large cohort of patients eligible to receive clopidogrel are similar to that of the innovator CHS salt. This clinical efficacy is independent of the use of CB as monotherapy or in combination with aspirin. Consequently, the present study supports the routine use of the CB formulation for a period of 12 months for the prevention and treatment of atherothrombotic disorders.
Footnotes
Authors’ Note
The study was investigator initiated, and the sponsor had no involvement in the design and conduct of the study, in the collection, management, analysis, and interpretation of the data or in the preparation, review or approval of the manuscript. The corresponding author had full access to all the data in the study and had the final responsibility for the decision to submit for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by grants from ELPEN (Greece), manufacturer of the CB formulation (Clovelen®) used in the present study.
